Abstract
Here we describe a patient with genetically confirmed ATTR, a family history of the disease and histological confirmation following carpal tunnel release surgery but no other manifestations. The first major neurological or systemic manifestation was cauda equina syndrome with ATTR deposits contributing to lumbar spinal stenosis. Recent gene therapy trials showed improvement in the neuropathy in TTR amyloidosis. This case highlights the need for awareness of the heterogeneous neurological phenotype seen in ATTR to aid earlier diagnosis especially now that disease modifying therapies are available.
INTRODUCTION
Lumbar spinal stenosis is a relatively common condition in late middle age [1]. It is characterised by compression of nerves roots to the lower limbs, exacerbated by exercise and relieved with rest; disability due to pain, impaired mobility and sphincter disturbance accrues with time [2]. It is most often multifactorial; related to disc degeneration, spondylolisthesis and age-related degenerative processes affecting connective tissue. Amyloid deposits have been found in the ligamentum flavum of 25/26 lumbar canal stenosis cases, with co-localised immunohistochemical staining for transthyretin in 5/15 specimens studied [3]. In a larger orthopaedic cohort of 111 patients congo-red staining identified amyloid deposits in 42.6% with confirmation TTR-derived amyloid by Western blot or mass spectrometry in the majority of these: 39/44 [4]. The accrual of wild-type TTR amyloid deposits in the flexor retinaculum is accepted as the commonest cause of carpal tunnel syndrome in the elderly [5]. Alongside cardiomyopathy, orthopaedic problems are increasingly recognised and common age-related manifestations in WT-TTR related disease with histological evidence found in up to 25% of autopsied patients aged over 80 years [6].
One case of spinal stenosis in a 65 year old Swedish man who presented with arrhythmia secondary to ATTR cardiac amyloidosis (His88Arg) has been described. But decompressive surgery had no effect on his painful leg symptoms suggesting the contribution of a co-existent neuropathy [7]. No histologically confirmed cases of spinal stenosis due to mutant TTR have been reported.
Surgical decompression is the mainstay of treatment with clinical improvement in most cases [8]. However given the growing evidence of the contribution of amyloid deposition to this presentation we should be more aware of TTR as a potential cause of the ligamentum flavum hypertrophy. The recent successful gene silencing trials in transthyretin derived amyloidosis have highlighted the need for earlier diagnosis [9–11]. An increased awareness of the phenotypic heterogeneity of this multisystem disease including atypical neurological presentations such as spinal stenosis is needed.
Here we describe a patient with genetically confirmed ATTR, a family history of the disease and histological confirmation following carpal tunnel release surgery but no other manifestations. Finding ATTR deposits contributing to lumbar spinal stenosis highlights the heterogeneity of neurological manifestations of the disease.
CASE
A woman of German ancestry was found to be a carrier of a heterozygous I84S TTR mutation at the age of 42. Genetic testing was performed on the basis of a strong family history of systemic amyloidosis affecting her mother, maternal uncle and older brother with carpal tunnel syndrome, peripheral neuropathy and restrictive cardiomyopathy. From this point she was under co-ordinated 2 yearly reviews by the National Amyloidosis Centre (NAC) in the Royal Free Hospital and peripheral nerve and autonomic services in the National Hospital of Neurology and Neurosurgery. At age 44, tissue confirmation was made on flexor retinaculum from carpal tunnel release surgery. She had been suffering from bilateral worsening CTS for approximately 10 years but denied symptoms of peripheral neuropathy, autonomic dysfunction, cardiac failure or visual impairment. Systemic and neurological examinations were normal except for positive Tinnel’s at the left wrist. Nerve conduction studies (NCS) were normal other than evidence of bilateral CTS and borderline sural sensory nerve action potentials (8μV on the right). Normal thermal thresholds suggest lack of small fibre dysfunction. Autonomic screening tests and 24 hour BP monitoring were normal. ECOG performance status was 1, echocardiogram was normal but DPD scintigraphy (Perugini grade 1) and cardiac MRI were in keeping with early cardiac amyloidosis, NT pro-BNP = 16 pMol/L. She was commenced on Diflunisal 500 mg. There was no evidence of disease progression either subjectively or objectively over the next 4 years.
At age 47, she developed a patch of numbness on the dorsum of her left foot and pain in her lower back which radiated to both anterior thighs and knees. Initially this came on after walking approximately one mile and would worsen if she continued walking but settle completely after 10–15 minutes rest. Over the next 12 months the sensory disturbance in her foot progressed proximally along the L5 dermatome, pain worsened in severity and exercise tolerance declined to less than 100 yards. She also described a tingling sensation in the same distribution and occasional feelings of her “legs giving way”. She had long-standing urge incontinence of urine but latterly developed a constant leak with urinary and faecal incontinence and worsening of all symptoms on bending and straining. There were no upper limb complaints or symptoms above the neck. Neurological examination now revealed decreased, but not absent, pinprick sensation from the left great toe and dorsum of the foot respecting the L5 dermatome and slight asymmetry of ankle and knee jerks with reinforcement required on the left. Pulses were present and straight leg raising test was limited to 40°. A clinical diagnosis of cauda equine syndrome was made.
An MRI spinal cord showed multiple lumbosacral disc bulges, most marked at L2/3, superimposed on constitutional narrowing of the lumbar vertebral canal due to ligamentum flavum hypertrophy and neurosurgical referral for spinal decompression was made (Fig. 1a). Visualisation of the stenosed segment during surgery allowed for estimation of the relative contribution from the L2/3 disc prolapse and ligamentum flavum hypertrophy as 30% and 70% respectively. Histological examination confirmed the presence of amyloid deposits, immunohistochemistry labelled these as transthyretin (Fig. 1b). Six weeks post-operatively she reported marked improvement in pain, exercise tolerance was greater than one mile, continence had improved and the area of decreased sensation was reduced to a few cm2 around the dorsum of the left great toe, the rest of the neurological examination was normal.
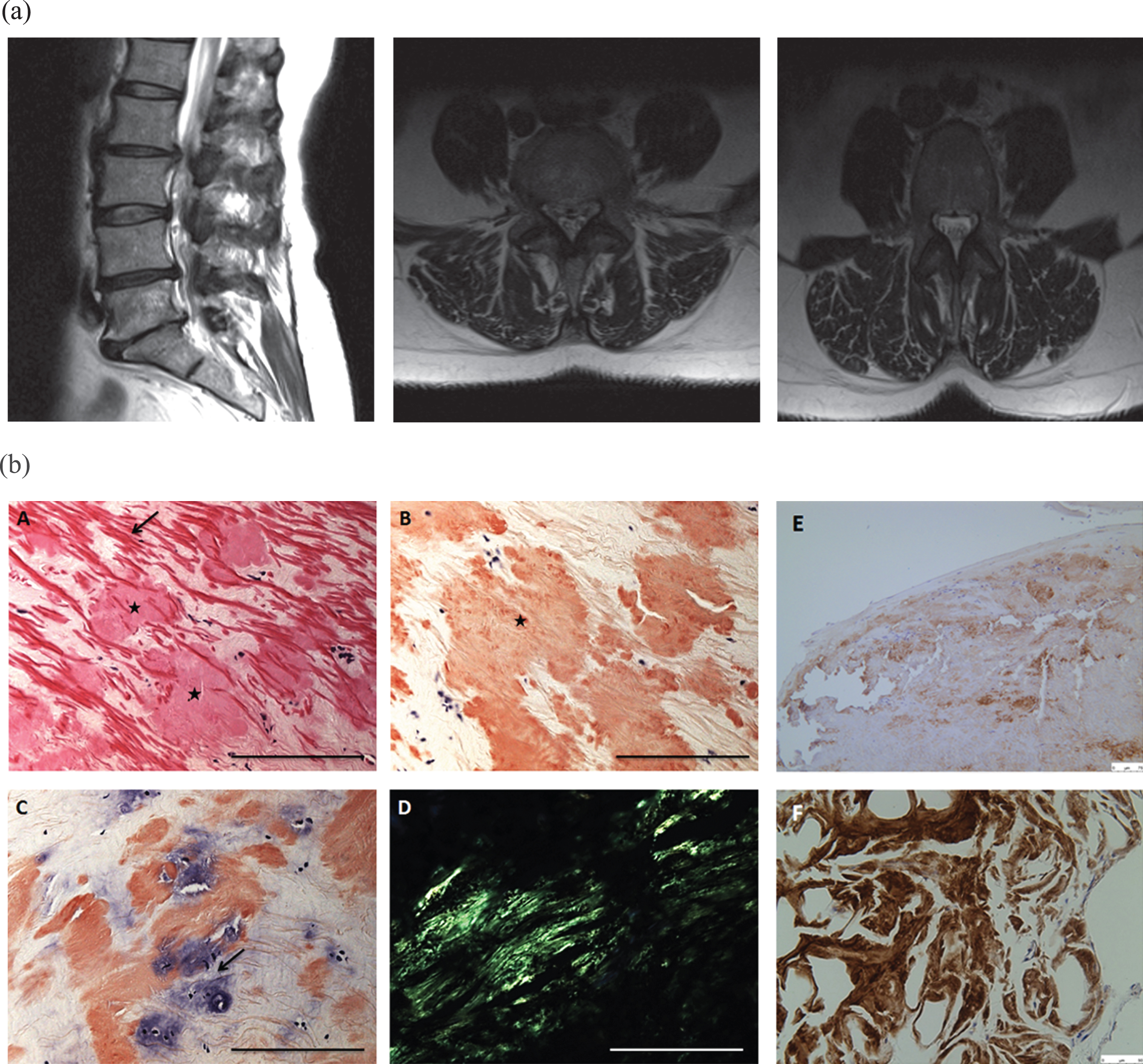
a MRI lumbosacral spine. Sagittal T1 reveal moderate background degenerative disc bulges at L2/3, L4/5 and L5/S1 superimposed on constitutional narrowing of the lumbar canal (A), in combination with ligamentum flavum thickening causing moderate stenosis of the vertebral canal. b Ligamentum flavum histology. A formalin-fixed, paraffin-embedded section from the ligamentum flavum biopsy (L2/3 level) shows one of the fragments of the ligament rich in elastic fibres (arrow) containing several amorphous eosinophilic proteinaceous deposits (stars) (A). These deposits stain strongly with the Congo Red dye (arrow) (B) and are seen to extend in to the cartilaginous intervertebral disc (arrow) that shows severe degenerative changes (C). The Congo Red-positive deposits show characteristic apple-green birefringence when visualised under polarised light confirming their amyloid nature (D).The deposits stain strongly with TTR-specific immunoflourescent antibody (E, F). Scale bar in A, B, C, E = 100μm and D, F = 50μm.
DISCUSSION
This is the first reported case of histologically confirmed mutant ATTR-associated spinal stenosis. Following the recent positive gene therapy trials in ATTR amyloidosis, we suggest a biopsy of the ligamentum flavum is taken during decompressive surgery in selected patients where a high suspicion of ATTR amyloidosis exists (those with a family history and/or a positive genetic test, those with a high clinical suspicion of ATTR amyloidosis and a positive genetic test but previous negative tissue biopsy). Early diagnosis of ATTR amyloidosis is likely to be increasingly important.
CONFLICTS OF INTEREST
Professor Julian Gillmore sit on Advisory Boards for Alnylam and Akce-Ionis.
The authors have no conflicts of interest.
Footnotes
ACKNOWLEDGMENTS COMPETING INTEREST AND FUNDING
M. M. R. is grateful to the Medical Research Council (MRC) and NINDS/ORD (1U54NS065712-01) for their support. This work was undertaken at University College London Hospitals/University College London, which received a proportion of funding from the Department of Health’s National Institute for Health Research Biomedical Research Centres funding scheme.
