Abstract
Background
Fatigue in myasthenia gravis (MG) is common and difficult to manage. Unlike myasthenic weakness it is not amenable to drug therapies.
Objective
Our primary aim was to investigate whether a combination of physical and psychological therapy would help address symptoms of fatigue in MG patients, who have stable disease but residual problematic fatigue. Our secondary aim was to quantitate fatigue by applying different scores and to ascertain which would be most relevant to apply in MG.
Methods
We recruited 10 MG patients with stable disease and who suffer from fatigue. Nine of these 10 patients participated in a 10-week program that involved physical and psychological intervention. We quantified their fatigue using the modified fatigue impact scale (MFIS), the visual analogue fatigue scale (VAFS) and the fatigue severity scale (FSS) at the start of the study, at various intervals during the program and 3 months later.
Results
During the program, there was a small improvement in the physical and psychosocial
subscale of the MFIS. There was a significant improvement (
Conclusions
This is a small pilot study, which utilized a combined approach with physical and psychological therapy, and showed some benefit in improving fatigue in patients with MG. The improvement was small and unsustained. Because of the small patient cohort, one cannot derive any firm conclusions and a larger study is required to investigate this further.
Keywords
INTRODUCTION
Primary fatigue in myasthenia gravis is distinct from muscle fatigability and weakness [1, 2]. More than 80% of MG patients will experience fatigue. It is estimated that half of MG patients will describe physical and cognitive fatigue [3–5] at some stage. Fatigue can be severely disabling, interfering with patients’ ability to pursue activities of daily living, limiting their work, family and social lives and negatively impacting their quality of life. Secondary fatigue stems from external factors such as mood changes, anxiety resulting from the diagnosis of MG, sleep disturbance, pain and discomfort, effects of medication especially analgesia, and weight gain particularly secondary to corticosteroid treatment. Ascertaining which factors lead to secondary fatigue, their temporal evolution, and dissecting them out from primary fatigue can be difficult.
Commonly, patients perceive their MG to be particularly active because they are disabled by fatigue and expect their medication to be escalated. However, fatigue does not respond to increases in dosages of pyridostigmine or immunosuppression. Some patients become dependent on pyridostigmine and feel that this helps with their fatigue (without altering their muscle strength, however) and in some cases refuse to be weaned off pyridostigmine since they feel “better” but not necessarily stronger on the drug.
The aim of this pilot study was to investigate whether intervention with physical therapy and psychology could help patients manage fatigue better. We quantitated fatigue using different scales in an attempt to identify which was most relevant to apply in MG.
MATERIALS AND METHODS
Funding for this pilot study was granted through MyAware. We obtained ethical approval in July 2016 (REC reference 16/WA/0197; IRAS project ID 202039). We screened our database (630 MG patients). We excluded patients who had comorbidities that would cause significant fatigue for example cancer, significant cardiac disease and obstructive sleep apnoea. We approached 25 MG patients who attend the myasthenia clinic in Glasgow, with stable disease and who are disabled by symptoms of fatigue. We explained to these patients the rationale behind the study and the details of the program, which required to engage in an exercise program and psychological intervention in a group setting. The program ran weekly for 10 weeks, between March and May 2017, between 1800 and 2000. This posed limitations to patients in employment, or who had family commitments after working hours. From the 25, we recruited 10 patients, who agreed to commit to the 10-week program. We restricted our cohort to this number because any larger would have made it difficult to manage the group sessions. Unfortunately, one patient (patient 6) abandoned the study after attending for the first session of the program.
At clinic, prior to initiation of the 10-week program, the MG nurse assessed the status of the 10 MG patients using the MG composite score [6]. At this same visit, patients filled in questionnaires including the MG-ADL [7], MG-QOL15r [8, 9], Modified fatigue impact scale (MFIS) [10, 11], the fatigue severity scale (FSS) [11], the visual analog fatigue scale (VAFS) [12] and the Hospital anxiety and depression scale (HADS) [13]. We refer to this time-point as “baseline”.
The program was delivered by the myasthenia nurse (CC), a neuromuscular physiotherapist (MD) and a neuropsychologist (DK). The time-points “week 0, week 1 etc” refer to the time-period for this program. During the program, we asked patients to score their levels of fatigue every fortnight using the MFIS, FSS and VAFS. At the end of the 10-week program, patients completed the MG-QOL15r, HADS and the fatigue-related questionnaires. We refer to this time-point as end-of-program. Three months after the program (August 2017), we reassessed patients in clinic using the MG composite, MG-ADL, MG-QOL15r, MFIS, VAFS, FSS and HADS, and we asked them specifically whether they had pursued any life-style changes after completing the program. We refer to this time-point as “end-of-study”.
Details of the 10-week program have been included in the Supplement Table 1. It aimed at introducing safe and effective physical activity for patients, encouraging them to integrate appropriate levels of exercise into their lives depending upon their individual fitness levels and motivational experience. The program encouraged patients to discuss, in a group setting, the emotions they commonly experience as a result of but not necessarily directly arising from MG — fatigue, pain, sleep deprivation, anger and frustration — and to educate them on managing these positively. The program integrated the characteristics of the shared decision making paradigm, where the patient is the “source of control” and the program was patient-centered [14]. The group worked on the concept that both patient experience and professional knowledge are fundamental in health behaviour change [15]. The program also gave opportunity to discuss their MG symptoms and educate on how to manage these effectively.
The delivery of each topic was conducted in presentation format followed by discussion on how these health behavior changes could be incorporated into individual circumstances ensuring patients could take forward these concepts at their own pace. The second part of the session was practical. The physiotherapist demonstrated simple aerobic exercise techniques, resistance and flexibility work with a view to improving well-being, energy levels and fatigue, and encouraging patients to eventually integrate exercise in their daily routine. She did not invoke incremental changes to the exercise component in terms of duration and endurance but each patient would undertake as much exercise as they could on any given day taking account of their energy levels. Teaching patients to “tune — in” to their energy and fatigue experience was the primary focus. After each week, the physical aspect of the program was modified slightly to reflect the direction within which the group was progressing. i.e. if patients complained of tightness more time would be allocated to stretching to reflect the overall needs of the participants.
The physiotherapist also worked on breathing and relaxation methods to help patients
develop strategies for sleep and rest. During the 10-week intervention, patients were
encouraged to develop skills for progressing their own personalized activity program by
working on: Frequency
— encouraging patients to undertake some form of activity on most days of the week
even if it was only for a few minutes working on the premise of doing more than they
would have done before. Duration —
learning how to assess personal levels of fatigue and knowing how much activity could
be undertaken without experiencing increased fatigue levels the following
day. Intensity — developing an
understanding of the different levels of intensity and how to assess if they were
doing too much, learning how to integrate submaximal intensity into their activity
rather than the “all or nothing” approach that some participants had
developed.
Week 9 of the program included family members, friends and significant others, since patient support outwith the hospital setting is important to help facilitate change in health behavior. Shared decision making also takes account of significant others, and this provided the group with an opportunity to reflect on the program and share their journey.
All patients were provided with blank diaries (provided by MyAware). These diaries allowed MG patients to rate their day during the 10 weeks: 1 star - a lot of MG symptoms, 2 star - many MG symptoms, 3 star - some MG symptoms, 4 star - minor MG symptoms, 5 star - no MG symptoms. They were also asked to insert comments about symptoms and their activities. During the program, time was allocated for patients to discuss their diaries and address how their symptoms could be potentially managed.
Statistical analyses were performed using PRISM 7.0 and EXCEL.
RESULTS
We recruited 10 MG patients into the study (5 male and 5 female), with an age range of 32 to 76 years. The mean duration of their MG was 11.6 years (median 9.0, SD 9.2). Table 1a gives further demographics of the cohort, including their worst Myasthenia Gravis foundation of America (MGFA) classification of disease [16] and their post-intervention MGFA status, their current medication and what symptoms they report. None of the patients experienced any significant relapses during the study period and no changes were made to their medication. Table 1b includes other details including their body mass index, their smoking and sleeping patterns, other unrelated symptoms, use of opiates (50% of the cohort) and employment status. Unfortunately, one patient (patient 6) abandoned the study after the first session of the program. We have included his data in the demographics Tables 1a and 1b but have omitted any remaining results pertaining to him from all other analyses. Four patients missed one or two sessions of the 10-week program.
Demographics, Myasthenia status and treatment
AChR+, acetylcholine receptor antibody positive; Ab negative, seronegative; MM, minimal
manifestations; Pyrido, pyridostigmine; MMF, mycophenolate mofetil.
Demographics, Myasthenia status and treatment
AChR+, acetylcholine receptor antibody positive; Ab negative, seronegative; MM, minimal manifestations; Pyrido, pyridostigmine; MMF, mycophenolate mofetil.
Demographics, sleep, exercise, opiate use and employment status
BMI, body mass index.
The mean MG composite score at baseline was 9.9 (median = 8.0; SD 4.8). Post-study the mean MG composite score was 9.9 (median 9; SD 5.5). The mean MG-ADL at baseline was 8.0 (median 9.0, SD 2.5) with mean MG-ADL at end of study of 8.5 (median 9.0, SD 3.4). The mean MG-QOL15r at baseline was 37.8 (median 39; SD = 10.9). At end of program, MG-QOL15r was 31.2 (median 34; SD = 7.6). at the end of the study (3 months after completion of the program), the mean MG-QOL15r was 31.3 with a median of 33.0, SD of 8.9.
HADS
Scores of between 0 and 7 are considered normal, 8–10 is borderline and above 11 is
considered as a potential clinical case. The mean anxiety score at screening was 11.3
(median 13.0; SD 5.6). This dropped to a mean of 8.4 at the end of the program (median 10,
SD 5.3;
MFIS
The physical subscale score ranges from 0 to 36. Figure 1a and Table 2 show a trend for improvement in most patients,
albeit small, during the 10-week program (mean of 30.6 at baseline dropping to 25.9 at
end-of-program) but the mean score increased back to almost baseline at end-of-study. This
did not reach significance when analysed against baseline on ANOVA. The cognitive subscale
ranges from 0 to 40 and was relatively unchanged during the program and at endpoint (Fig. 1b and Table 2). The psychosocial subscale ranges from 0 to
8. There was a definite though small improvement during the 10-week program (Fig. 1c, Table 2) although still not reaching statistical
significance (
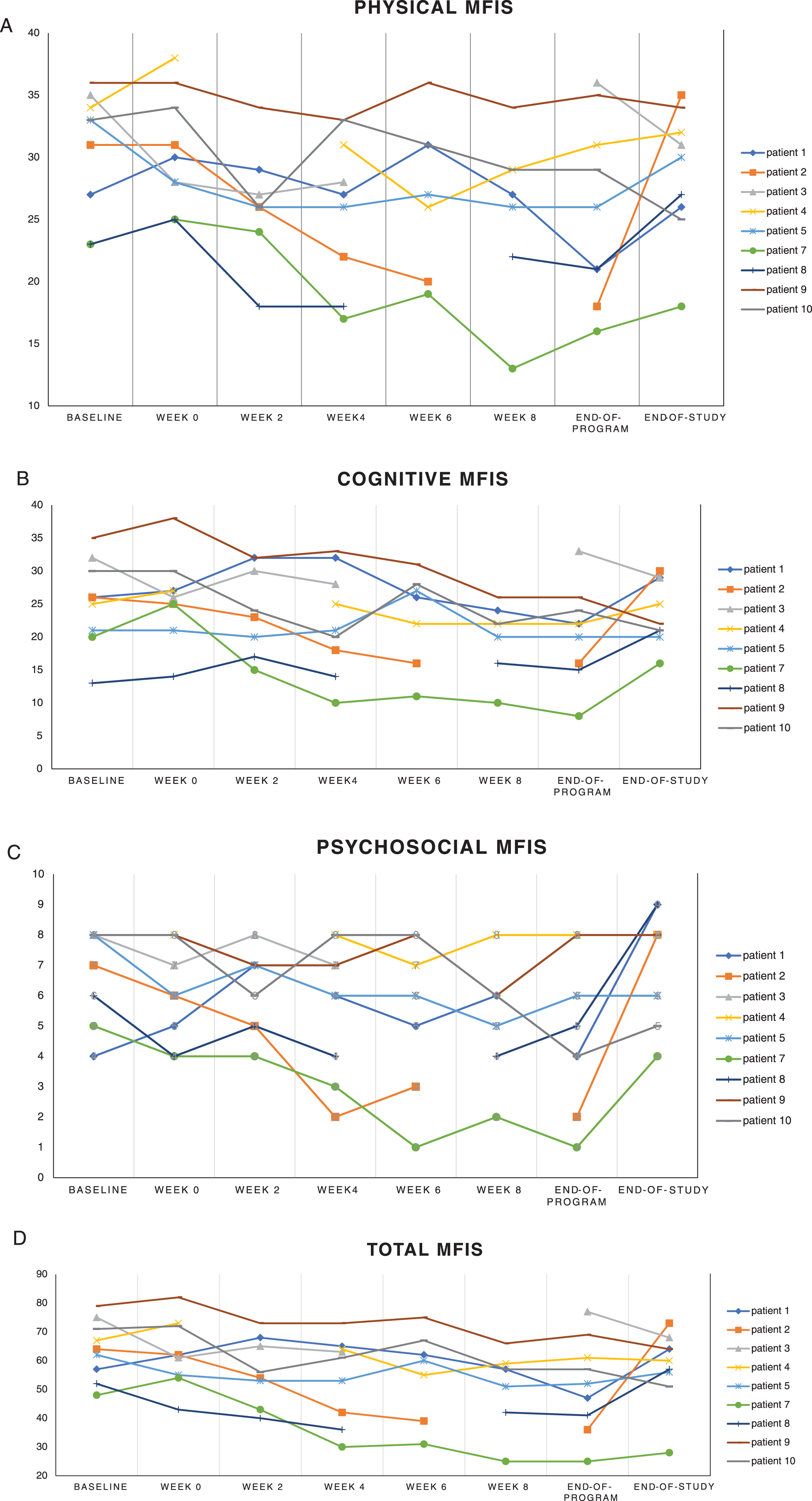
Total and breakdown of the MFIS, VAFS and FSS across the total study
MFIS, modified fatigue impact scale; VAFS, visual analog fatigue score; FSS, fatigue severity scale.
This scored patients’ fatigue on a number line from 0 (worst) to 10 (normal). Figure 2 shows individual patients’
plots. Table 2 shows an
improvement from a mean of 1.7 rising to 4.9 at end-of-program and reaching statistical
significance on ANOVA (
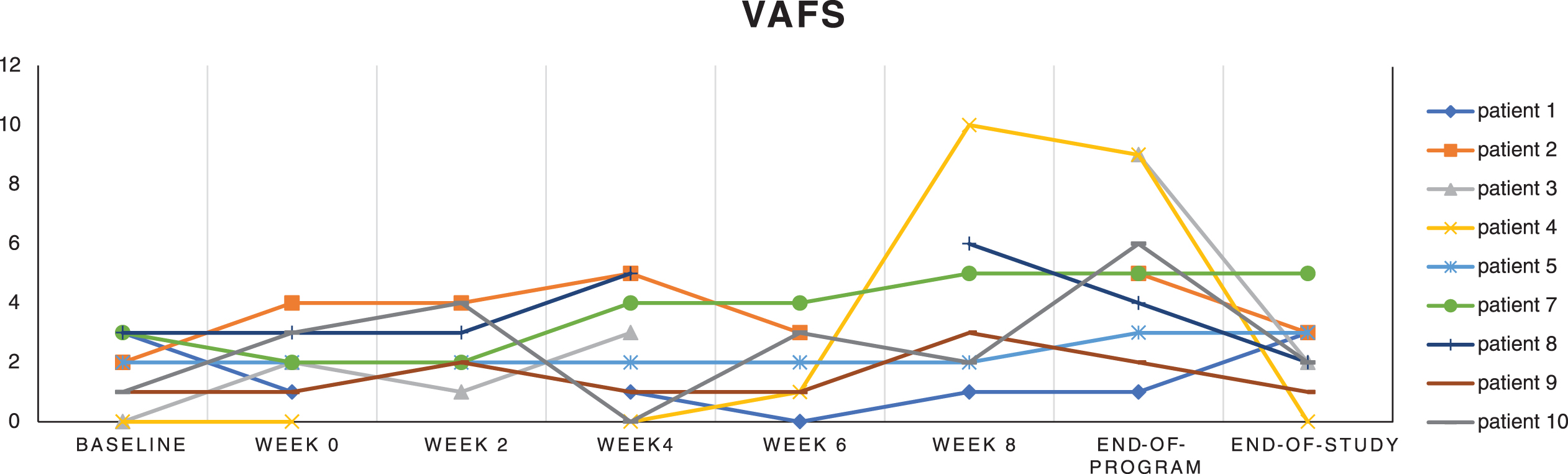
Plots for individual patients (patients 1–5 and 7–10) for the visual analog fatigue scale (VAFS) across various time-points of the study.
FSS
The range is 0 to 63 with the highest score indicating least energy. The patients scored themselves highly on FSS throughout the study with a suggestion of a small improvement at week 6 and 10 but not reaching significance. (Table 2).
Correlations
At baseline, the MG-QOL15r correlated with VAFS (
Diaries
All patients filled in these diaries except for patient 6. Table 3 shows the breakdown of the star days across each patient. There was a total of 41 1-star days, 185 2-star days, 258 3-star days, 110 4-star days and 7 5-star days. We were surprised that patient 3 reported so many 1-star days since we consider him to have mild disease. He is on a high dose of pyridostigmine although we feel that this should have been weaned off but he feels dependent on the drug. Table 2 in Supplement gives diary extracts for 9 patients. Patients spoke of total exhaustion, pain in muscles and joints and muscle twitching. They commented about their sleep. They mentioned their activities throughout the day including rest periods and periods of bedrest when they were having a bad day.
Patient self-reported diary days during 10 week program
Change in life-style
At the end of the study, we asked patients whether they changed their life-styles or the way they manage their MG symptoms, stress, pain and their sleep patterns. Tables 4a and 4b summarize their answers. Since the study, 4 patients have started to exercise, and another patient has enrolled in a gym although has not really been exercising since. 50% of patients are making dietary changes and increasing their level of hydration throughout the day. Two patients reported some improvement in sleep pattern, including one patient who has made significant changes and has abandoned her i-pad at night and is getting unbroken sleep. None of them reported a change in energy levels. Four patients have learnt to recognise how to manage their MG symptoms and fatigue better since the study. 50% are managing stress better using the techniques discussed during the program and one patient is learning how to manage her anxiety about going out of the house on her own with variability from day to day and attempting to socialize. Two patients have made changes to their social life. Only one patient reported feeling better overall.
Change in life-style since end of program and at end of study
Change in managing symptoms since end of program and at end of study
DISCUSSION
Fatigue is a disabling symptom across various neuromuscular disorders [17], which significantly and negatively influences patients’ quality of life, impacts their employment status and their family and social lives. It contributes heavily to disease burden and is difficult to quantify. Fatigue is also problematic in MG, even after patients’ active MG symptoms have been managed and they have achieved a satisfactory status such as minimal manifestations.
Fatigue is often multifactorial. Primary fatigue has a physical component and arises from muscle weakness and fatigability [4], which is heightened when MG symptoms are active. Secondary fatigue is multifactorial. In this study we did not endeavour to dissect between the two. The majority of our cohort were receiving prednisolone for their MG. Corticosteroids often result in weight gain, making exercise more challenging for an individual with muscle weakness. They may cause changes in sleep patterns and insomnia [18]. Weight gain can result in obstructive sleep apnoea [19]. Some patients develop severe mood disturbance. They become anxious and sometimes depressed about their physical limitations, the variability and unpredictability of MG and the changes in their appearance [19] resulting from corticosteroid therapy. They tend to exercise less and become severely deconditioned. This can result in musculoskeletal pain, which in turn discourages them to move or stretch in fear of causing more pain. They socialise less, which in turn negatively impacts their mood and well-being. Although MG is not a disease affecting the central nervous system, there is also a component of cognitive fatigue [5]. Patients complain of “brain fog”. There may be various molecules, including cytokines [20], which contribute to this. The challenge is how to manage physical and cognitive fatigue once the NMJ has recovered.
In this pilot study, we applied various fatigue-related scores. We specifically chose these scales to capture the complete landscape of how the patient perceived their fatigue and to ensure we could detect meaningful change in fatigue levels as the patients progressed through the 10-week intervention phase. By utilising different scales, this enabled triangulation during analyses. We applied the MFIS because it comprises physical, cognitive and psychosocial components, which are all relevant to MG and which are domains that we specifically wanted to study. The MFIS has been applied in previous studies [21]. We felt that the items of the FSS bore significant overlap with those from the MG-QOL15 and were surprised that it was not found to be a useful outcome measure. The FSS has also been applied in studies with myotonic dystrophy patients [22, 23]. Lastly, we chose the VAFS, because it is simple to use both in the clinic and in research. We found that the MFIS and the VAFS were the most useful scales for this cohort and were sensitive to change during the 10-week program.
Our findings indicate that patients’ fatigue levels showed a small improvement during the 10-week program, with a combination of physical and psychological input, albeit the changes were small, not reaching statistical significance and unsustained. It was disappointing that 3 months after program completion, most patients’ fatigue-related scores had reverted to baseline. In retrospect, we should have reassessed these patients earlier. However, some have made some changes in their life-style, reported they were managing stress better and were utilising the exercise and breathing techniques that had been demonstrated and discussed during the program.
There are various limitations to this study. The cohort was very small but this was deliberate to allow a group-therapy environment. The patient cohort was heterogeneous comprising of patients with different MG status and receiving different immunotherapies. It was challenging obtaining patient commitment. The program took place between 1800 and 2000. It is possible that this later schedule meant that patients were more tired although the cohort never raised this as an issue. There was an element of selection bias: we selected patients who we knew suffered from significant fatigue. We specifically delayed the program so that it commenced in March 2017, to ensure that the weather conditions were favourable, to avoid any holiday-periods and to give patients sufficient notice to plan. Despite this we had drop-outs prior to the recruitment phase, prior to the program initiation, as well as a drop-out during the program. One other limitation, was the duration of their disease, which was very long for some. It was felt that some patients were entrenched in their behaviours and were therefore less receptive to the management strategies proposed during the program. We purposefully chose to avoid selecting patients whose disease duration was less than 2 years since they were more likely to be undergoing changes in medication (and one of the study criteria was that medication should remain unchanged throughout the study which we achieved). A further limitation was that we did not perform a cognitive screen at baseline specifically to assess their executive functions. During the program, it was felt that many had problems with coping mechanisms, there were psychological comorbidities in the cohort and many had difficulties with conceptualizing. In our opinion, this is a distinct problem to cognitive fatigue. Furthermore, we did not ask them about their educational attainments and their psychosocial backgrounds, which are important factors.
Our patient cohort scored highly on the MG-QOL15r despite their MG not being active. They also scored highly on HADS, and it became apparent during the program that most were anxious and some were depressed. It was interesting that the MG-QOL15r correlated with various other scores related to fatigue. It reinforces that the MG-QOL15r is a tool that is sensitive to the physical component of MG as well as the psychosocial changes that patients experience secondary to MG.
We were surprised to read the content of their diaries, particularly how much discomfort, pain, stiffness and breathlessness they reported. Patients took regular periods of rest, including bedrest during the day and many reported having disturbed sleep at night. In contrast, we were pleasantly surprised to find that some patients undertook significant activities even when or after they reported many MG symptoms, some of which seemed bona fide MG symptoms. Our opinion is that some misinterpreted symptoms stemming from MG. One patient during the program described how she had a “MG crisis” in the days between the program sessions but received no medical intervention and had no changes made to her medication. She was probably describing a panic attack. Patients expressed in their diaries and informally during the program a sense of relief that their symptoms were in common to other MG patients. They were happy to work in a group setting and some perceived the program as an opportunity to socialize. We realised that some patients were socially isolated, possibly as a result of their MG, which would naturally negatively impact an individual’s wellbeing. In the penultimate session of the 10-week program, family members were invited to attend to obtain their perspective of the disease. An understanding of MG is important for family members too, to allow the correct adaptations for patients in their home and social settings. Patients valued the support of their family members and were pleased to be able to include their significant others in describing what they had been doing throughout the program.
Fatigue is also common in other neuromuscular disorders. Fatigue, pain, and balance problems are common symptoms in patients with myotonic dystrophy type 1 (DM1) and fascioscapulohumeral muscular dystrophy (FSHD) [24, 25]. Hypersomnolence is a common feature in DM1 patients [26]. Managing fatigue with a combination of physical and psychological therapy is not a new concept. The OPTIMISTIC trial [27] aimed to study the effect of cognitive behavioural therapy (CBT) and graded exercise therapy in patients with DM1. Some of the outcomes of this study (as yet unpublished) included measuring cognitive and physical outcomes as well as quality of life. Various studies in FSHD have explored the safety and efficacy of different exercise programs including high intensity training, home-based exercise programs and aerobic exercise which led to functional benefits without compromise of muscle tissue [28–31]. Furthermore, CBT helped patients with FSHD and reduced the severity of fatigue.
There is no consensus on exercise in MG but simple aerobic exercise is deemed safe and beneficial [32–34]. Tai chi may be suitable for MG patients because it entails slow stretching, allowing even deconditioned individuals to adapt to various movements and improves balance [34]. It provides a building block for other forms of exercise such as pilates, yoga or gym work. When the patients commenced the 10-week program, there was a negative bias towards physical therapy and exercise. All had previous experience, and deemed physical therapy to be ineffective. This may have been due to the therapist having little experience of MG or the patient feeling overly fatigued post-exercise and not wanting to repeat this negative outcome. In this study we concentrated on simple stretching techniques for patients to pursue out in the community, also as a stepping stone to motivate them to pursue light exercise. Although we did not quantitate this, patients commented that they were able to perform their stretches with increased ease, in comparison to their performance at outset of the 10-week program.
Prescribing exercise can be difficult. Whilst there is published literature on recommended activity levels for the general population, physical therapists have expertise in adapting these recommendations for people with a variety of different disorders. The group we studied was heterogeneous with regards to their fitness levels, physical abilities and age, all factors with an impact on an individual’s ability to engage in the program. Each participant was undertaking activity within the class situation, as well as working at their own level, developing their own bespoke knowledge of fitness with the ultimate aim that this program could help them, in the longer term, maintain better physical activity. Working on the premise of doing more than you did before and explaining the recovery process helps patients understand that measuring improvement is best undertaken over a time-period, rather than within a single exercise episode.
In conclusion, this pilot study showed small improvements in fatigue levels when applying combined physical and psychological therapies in MG patients. The MFIS and VAFS are useful fatigue outcome scales which are relevant to MG patients and are easy to apply. However, a larger-scale study is required to investigate and confirm these observations further.
CONFLICTS OF INTEREST
The authors have no conflict of interest to declare.
