Abstract
Background:
Improved neuromuscular and respiratory therapies have altered the natural history of Duchenne muscular dystrophy (DMD) such that the most common cause of mortality is progressive cardiomyopathy. Despite imaging evidence of progressive cardiomyopathy, troponin I (cTn) is not significantly elevated in asymptomatic DMD patients.
Results:
We describe eight boys with DMD evaluated for acute chest pain (ACP) and found to have acute cTn elevation with depressed left ventricular ejection fraction (LVEF). Of our eight patients, five presented with a primary complaint of ACP, while three presented with secondary myocardial injury in the context of systemic illness requiring hospitalization. Electrocardiograms showed diffuse ST changes and mean peak cTn level was 44±15.4 ng/mL (reference range <0.03 ng/mL). cTn levels normalized with only supportive care. Cardiac magnetic resonance imaging (CMR) was performed during the event on all but one patient, demonstrating increased late gadolinium enhancement (LGE) from 12.4±11.4% to 36.5±10.3% with associated deterioration of LVEF from 61±4.4% to 47.6±6.6% which remained depressed on follow-up CMR study (49.1±7.8%). All viral studies were negative. Additional investigations varied among patients, but no causative findings were demonstrated.
Conclusions:
ACP with cTn elevation occurs in DMD boys and may be indicative of cardiomyopathy progression as evidenced by acute left ventricular dysfunction and development or progression of myocardial fibrosis. This clinical presentation is under recognized. These events may represent an important pathophysiological mechanism in cardiomyopathy progression.
INTRODUCTION
Duchenne muscular dystrophy (DMD) is an X-linked neuromuscular disorder with an incidence of 10.7 to 27.8 per 100,000 [1]. Patients develop progressive skeletal and cardiac muscle weakness which leads to death during early adulthood. Improved neuromuscular and respiratory therapies have altered the natural history such that cardiomyopathy is now the leading cause of mortality.
The pathophysiological mechanisms resulting in the development of the associated cardiomyopathy are complex and incompletely understood. Unfortunately, heart failure signs and symptoms are frequently vague secondary to the accompanying skeletal muscle myopathy. Acute chest pain (ACP) is usually attributed to the musculoskeletal system and is frequently dismissed. Assessment of myocardial injury with cardiac Troponin (cTn) has been shown to be minimally elevated at baseline, and not predictive of disease progression [2]. With the ability to evaluate myocardial disease, non-invasive imaging utilizing cardiac magnetic resonance imaging (CMR) has improved our understanding of the DMD-associated cardiomyopathy [3–11].
We report a series of eight DMD patients who presented with ACP associated with transient elevations in cTn levels. Patients demonstrated a decline in LVEF as well as progressive fibrosis assessed by late gadolinium enhancement (LGE) by CMR. We hypothesize that these episodes of ACP may represent an under-recognized component of the DMD cardiomyopathy progression; consisting of myocardial injury, progression of myocardial fibrosis, and decline in cardiac function as assessed by LVEF.
RESULTS
Eight DMD patients with an average age of 15.0±4.5 years (range 9 to 23 years) presented with ACP and troponin elevation between January 2013 and February 2017. This report was approved by the Nationwide Children’s Hospital Institutional Review Board. Individual details regarding presentation, medications, and clinical summary are presented in Table 1. 8/8 patients were prescribed an angiotensin-converting enzyme (ACE) inhibitor or angiotensin receptor blocker (ARB) and 5/8 patients were taking a corticosteroid. The majority of patients presented with ACP, electrocardiographic changes, and elevation of cTn with no known antecedent stress. Three patients had a concurrent illness which included pneumonia, gastroenteritis, and sepsis respectively. The patient admitted for presumed sepsis required fluid resuscitation and positive pressure ventilation. His cTn was initially normal but abruptly elevated during his hospitalization at a time of clinical deterioration.
Detailed clinical presentation and study summary for DMD acute chest pain presentation case series
Abbreviations: CMR = Cardiac Magnetic Resonance Imaging at B = Baseline, A = ACP Presentation, S = Short and L = Long term follow-up; CTA = Computed Tomographic Angiography; Cath = Cardiac Catheterization; ECG = Electrocardiogram; LGE = Late Gadolinium Enhancement; LVEF = Left Ventricular Ejection Fraction; PT = Patient; ng/mL=nanogram per milliliter; Yrs = Years. *Cases either baseline or event CMR was not available so an LVEF change is not reported. **There is no baseline CMR to compare so ACP event LGE % is reported as change from baseline to ACP presentation
Electrocardiograms were available for review at baseline on 8/8 patients and demonstrated normal ST segments. All patients developed diffuse ST segment elevation but did not demonstrate an ST-Elevation Myocardial Infarction (STEMI) pattern consistent with coronary ischemia. ST segment elevation normalized following symptom resolution in all patients (Fig. 1). Individual cTn serum levels (Fig. 2) were markedly elevated with a mean peak troponin level of 44.1±15.4 ng/mL (range 31–62 ng/mL, normal reference range <0.03 ng/mL). In all but one patient cTn levels were elevated on admission and normalized within 4 days following the initial troponin level. One patient who presented with sepsis had late onset cTn level elevation and had persistent elevation of troponin >200 hours.
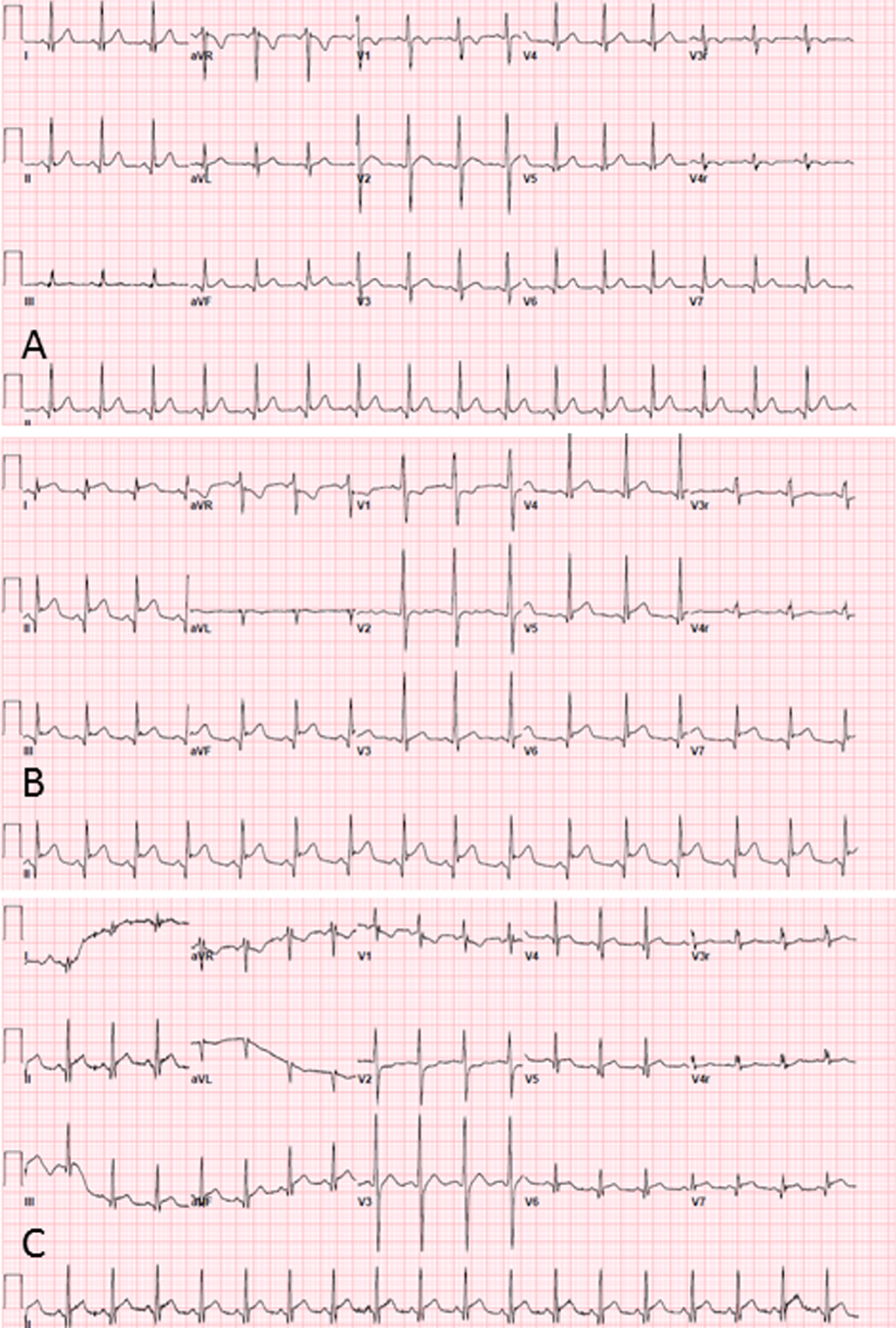
Serial Electrocardiogram Findings: (A) at baseline with no ST segment elevation (B) chest pain with significant ST segment elevation (C) follow-up with normalization of ST segment elevation.
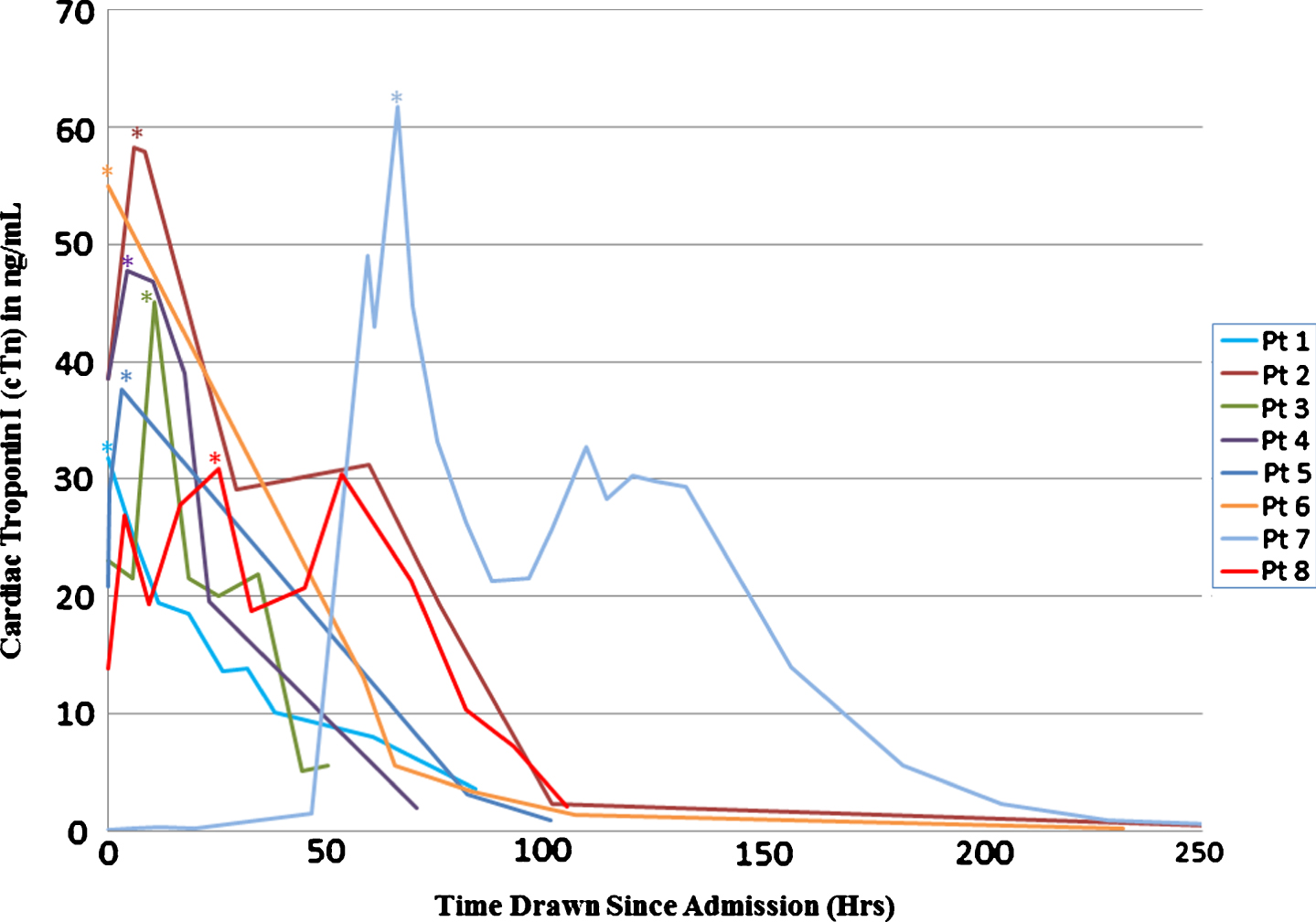
Serial Troponin I Findings: Individual troponin findings following presentation with chest pain. Peak troponin I is denoted by*.
LVEF and myocardial fibrosis was assessed by CMR and reported as percent of late gadolinium enhancement (LGE) to normal myocardium (Fig. 3) [12–14]. LVEF assessment by echocardiogram was used when CMR was not available. 6/8 patients had baseline CMR studies available for review. The average time between the attainment of the baseline LVEF by CMR to the ACP event was 12.8±11.8 months (range 2–35 months). All 6 demonstrated a normal LVEF with 4/6 exhibiting evidence of myocardial fibrosis by LGE. The two patients without a baseline CMR study had a normal LVEF by echocardiogram. Short term post-event follow-up was available for 5/8 patients at a duration of 1.4±0.7 months (range 0.5–2.5months). Long-term follow-up was available for 3/8 patients at a duration of 21.1±16 months (range 6–38 months).
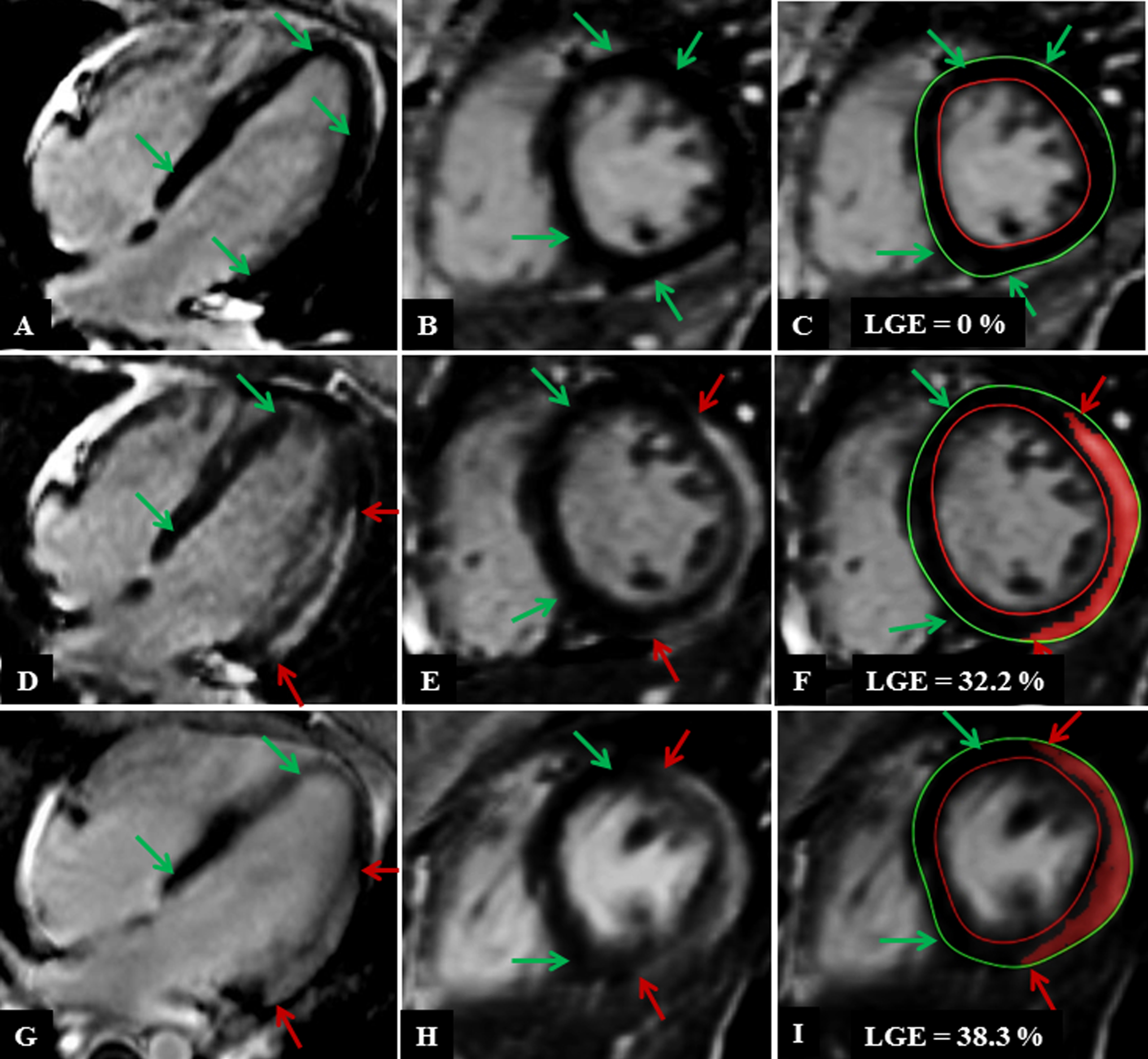
CMR LGE findings in 4 Chamber and Short Axis Image for patient 6: (A-C) at baseline, (D-F) during the acute phase of event (G-I) follow-up. Normal myocardium is denoted by green arrows and late gadolinium enhanced myocardium is denoted by the red arrows. (C, F, I) shows quantification of the LGE at (C) baseline with no LGE, (F) during episode of chest pain (32.2%) and (I) on routine outpatient follow-up (38.3%).
During the ACP episode, all patients experienced a decline in LVEF from baseline LVEF (61.8±4.6% to 49.0±6.6%; representing a decline of 12.8%). This decline was maintained on both short term (average LVEF of 48.4±9.0%; range 37–57) and long-term follow-up studies (47.3±7.8%; range 41–56) as shown in Table 2 and Fig. 4. In contrast, the right ventricular EF remained normal throughout the observation period (Table 2). The 2 patients that demonstrated no evidence of fibrosis by LGE at baseline developed significant myocardial fibrosis during the ACP episode (Table 1). The two patients who had a baseline echocardiogram were also noted to have significant myocardial fibrosis by CMR at the time of chest pain presentation. 6/8 patients who had baseline CMR studies demonstrated progression in the degree of fibrosis during the acute phase of the episode from a baseline of 12.4±11.4% (range 0% –29%) to 36.5±10.3% (range 26% –57%). The fibrosis persisted in all patients on both short and long-term follow-up studies with an average LGE of 39.3±9.3% (range 32% –53%) and 42±13.1% (range 33% –57%) respectively as shown in Table 2 and Fig. 5.
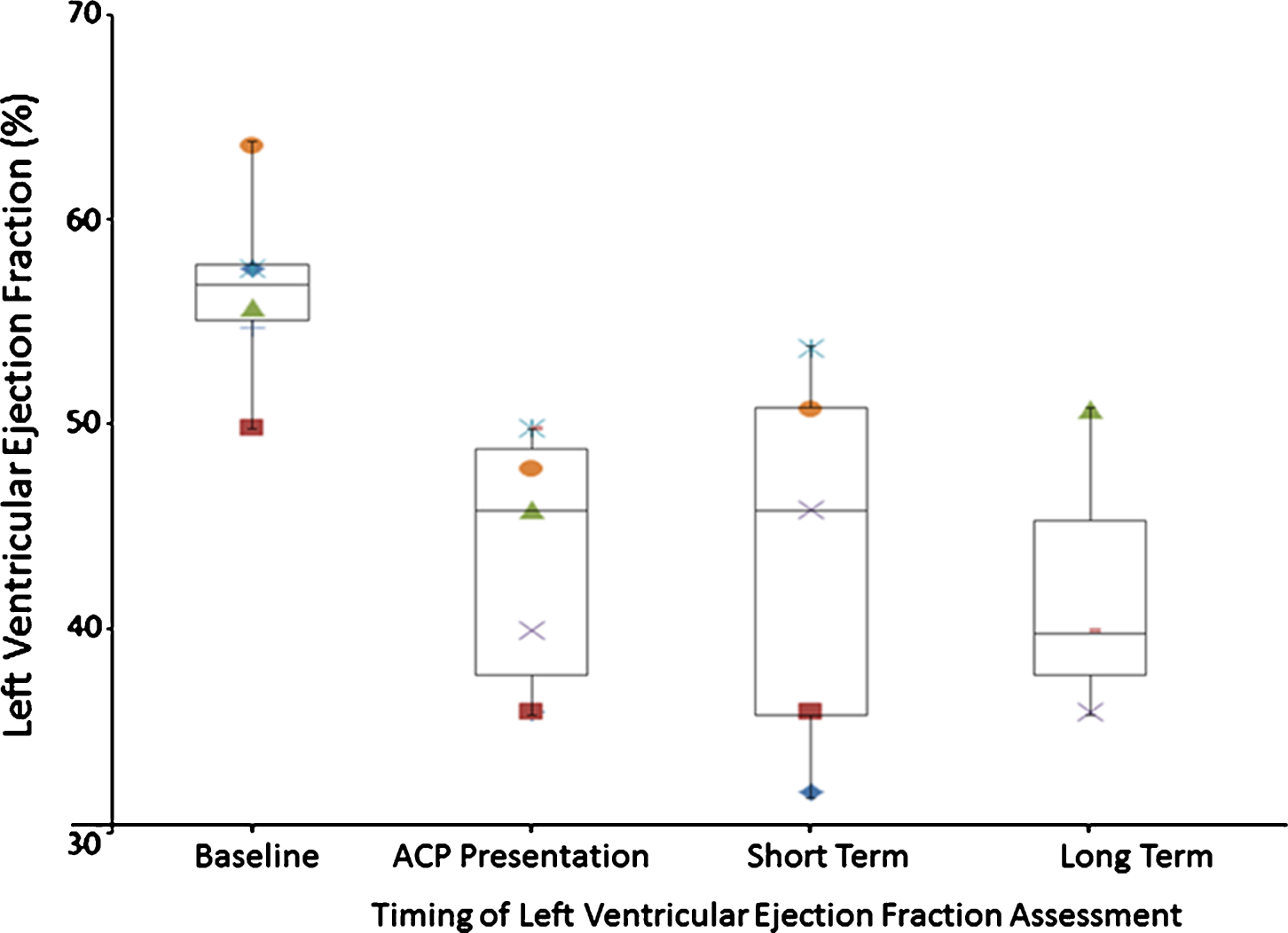
Box and Whisker Plot of Serial LVEF Assessment by CMR: Individual left ventricular ejection fraction evaluations at baseline (all normal, n = 6), during acute episode (n = 7) and progressive decrease in LVEF on short (n = 5) and long-term (n = 3) follow-up.
CMR and troponin i findings at baseline, chest pain event and follow-up in DMD patients
Abbreviations: ACP = Acute Chest Pain, CMR = Cardiac Magnetic Resonance Imaging, DMD = Duchenne Muscular Dystrophy, LGE = Late Gadolinium Enhancement, LVEF = Left Ventricular Ejection Fraction, LVM = Left Ventricular Mass, % = Percent, LGE % LVM = Percent of Late Gadolinium Enhanced Myocardium of Total Left Ventricular Mass, N = Number of subjects, ng/mL = nanogram per milliliter, RVEF = Right Ventricular, Ejection Fraction, N/A = Not Available; φ= 2 patients could not be quantified due to not having baseline CMR, *3 patients could not be quantified due to not having follow-up CMR.
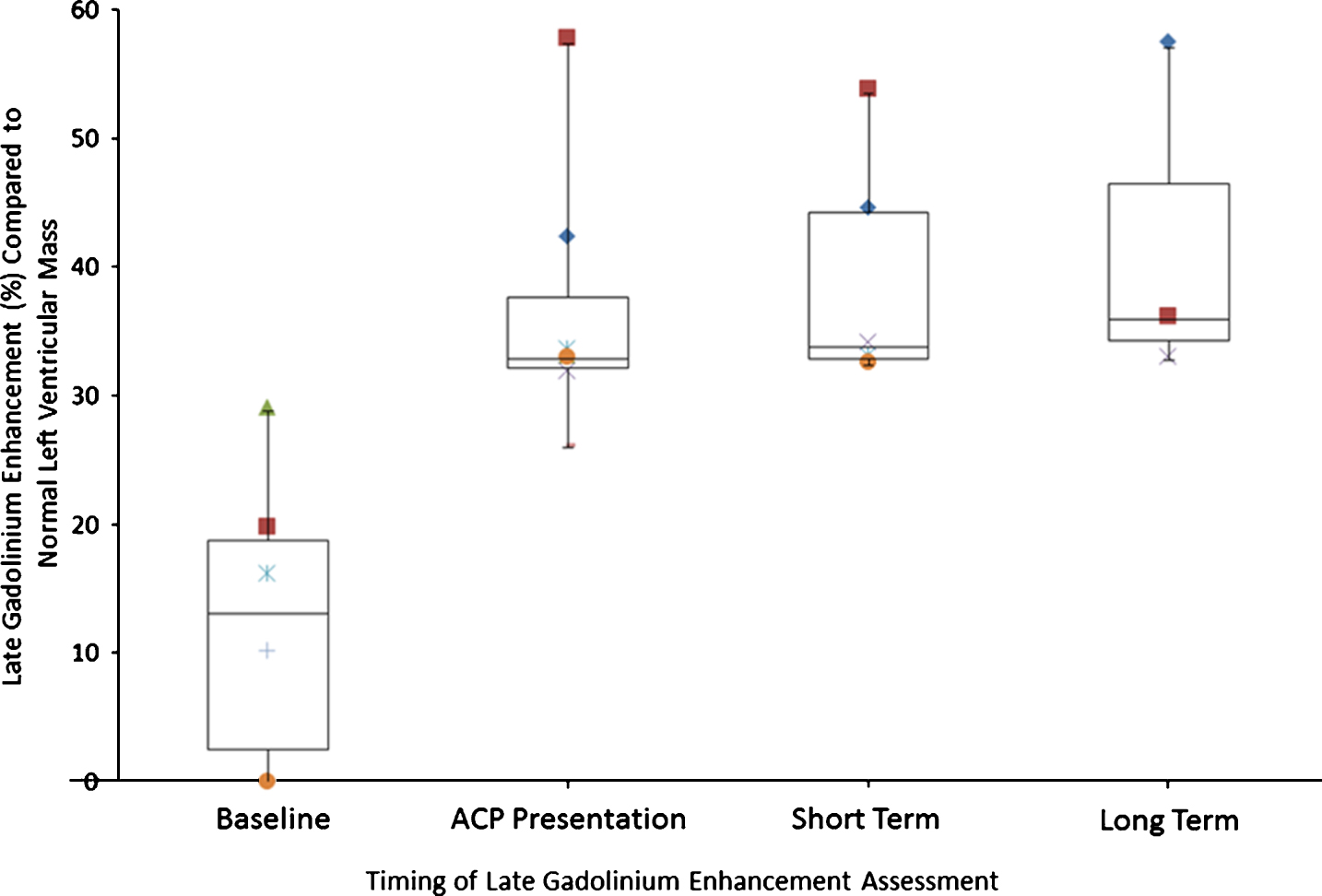
Box and Whisker Plot of Serial LGE Assessment by CMR: Progression of LGE in all patients with serial CMR from baseline (n = 6), during acute episode (n = 7) and on short (n = 5) and long-term (n = 3) follow-up.
4/8 patients underwent coronary artery evaluation (2/4 cardiac CT and 2/4 cardiac catheterization). No coronary abnormalities were noted in any patient. (Table 1). Since the differential diagnosis included myocarditis, viral studies were obtained by nasopharyngeal swab or blood polymerase change reaction (PCR) and were negative in all patients. Patients received supportive care with no cardiac specific treatment.
DISCUSSION
We report the largest case series to date of DMD patients presenting with ACP, markedly elevated troponin levels, and the development of progressive left ventricular dysfunction and myocardial fibrosis. Previously, isolated cases have been reported in the literature [15–18]. Our findings suggest that dystrophin-deficient cardiac myocytes may be subject to repeated episodic injury that accumulates with age, similar to the process believed to occur in DMD skeletal muscle [19]. This finding may provide important insight into the natural history of DMD-associated cardiomyopathy. ACP in DMD patients, particularly the young DMD patient, is often attributed to musculoskeletal pain. As such, it is unusual for the DMD patient who presents to a pediatric urgent care or emergency room to undergo evaluation for cardiac disease. This has resulted in a paucity of natural history data surrounding these events. We suspect the incidence of such events is grossly underappreciated.
Although we cannot be certain that these episodes of chest pain and elevation of cTn indicate progression of DMD-associated cardiomyopathy in the absence of other clinical signs such as infarction or coronary artery disease, the development of myocardial dysfunction and fibrosis since their baseline studies are highly suggestive of an association. On EKG, the ST changes we observed were non-specific and diffuse, and did not demonstrate a ST-Elevation Myocardial Infarction (STEMI) pattern consistent with coronary ischemia though they do represent corroboratory evidence of myocardial injury. The four patients who underwent coronary artery catheterization or angiography on CT scan showed no coronary involvement, confirming the nonspecific nature of the ECG findings. Given the short follow-up of our patients, the impact of these events on the overall progression of their cardiomyopathy is uncertain and will require more longitudinal studies.
Traditional biomarkers of myocardial injury such as cTn have not been found to be significantly elevated when evaluated in asymptomatic DMD patients [2]. These data support our hypothesis that progression of DMD-associated cardiomyopathy results at least in part from episodic myocardial injury rather than exclusively from continuous ongoing injury. It is unclear whether this injury occurs spontaneously as primary cardiomyopathy progression or if an external trigger such as viral infection, physiological stress or other intercurrent illness is required. Certainly, inflammatory cytokines are important not only in the pathophysiology of sepsis-induced cardiomyopathy but also chronic progressive cardiomyopathy [20]. In support of an inflammatory etiology in our patients, we note that three of them were diagnosed with antecedent infection (gastroenteritis, pneumonia, sepsis) and two others had symptoms consistent with infection (stomach cramping, vomiting). Whether or not inflammatory cytokines are involved in the pathophysiology of acute cardiomyopathy flare-ups in DMD patients we reported here, and indeed whether such episodes are triggered by acute infections, is unknown. It may be worthwhile testing for elevated cytokines in such patients in future studies.
Studies utilizing CMR in DMD demonstrate a characteristic appearance of the myocardium with subepicardial gadolinium enhancement [11, 22]. Interestingly, this LGE pattern is reminiscent of LGE patterns seen in viral myocarditis [23]. The two processes may share common mechanistic pathways. The LGE pattern in our cohort was not consistent with ischemic cardiomyopathy, as the sub-endocardium was spared in all patients. Furthermore, in the patients who had an ischemic coronary evaluation as previously mentioned (CT angiography or coronary catheterization); none demonstrated evidence of an obstructive coronary lesion.
The lack of positive viral titers or laboratory evidence of acute inflammation argues against acute viral myocarditis, at least from the traditional viruses available in current clinical viral panels. As such, no patient received treatment for myocarditis with immune globin. We acknowledge, however, that a viral etiology could have gone undetected as literature exists which supports the fact that the dystrophin deficient myocardium may be more susceptible to viral infection [24].
Normal left ventricular systolic function as assessed by LVEF was documented in all patients prior to their acute event, and all patients demonstrated left ventricular dysfunction at presentation. This decline in LVEF persisted on short and long-term follow-up studies. This observation suggests that the dystrophin deficient myocardium may be vulnerable to ongoing sub-clinical injury prior to the onset of overt ventricular dysfunction. Our findings support current recommendations for both early and ongoing cardiac surveillance as well as a complete cardiac evaluation during an episode of acute chest pain in the DMD patient [18, 25].
Further formal investigation and a deeper understanding of the etiology of these events may provide a window to DMD-associated cardiomyopathy disease progression. We speculate that cardiomyopathy progression occurs as a result of a series of acute insults resulting in episodic myocardial damage. This suggests a step wise model of disease progression as opposed to a linear one. Relatively silent but recurrent events may lead to cumulative injury. Given that a subset of our cohort presented during a concurrent illness, the myocardium in DMD may be more vulnerable to metabolic crises [26]. A deeper understanding of these events may provide opportunities for meaningful treatments possibly altering the trajectory of the disease.
Regarding treatment, patients received only supportive cardiac care during the acute phase of the illness. Treatment with immunosuppressive agents or intravenous gamma globulin was considered but not pursued given the lack of understanding of the pathophysiology of the process and the potential for side effects associated with these therapies. All patients at the time of presentation were on an angiotensin converting enzyme inhibitor or an angiotensin receptor blocker. These drugs are currently recommended as first line therapy for the DMD patient with the onset of left ventricular dysfunction or by the age of 10 years [25]. Given the presence of progressive fibrosis and the decline in ventricular function there may be benefit to consider other heart failure medications such as mineralocorticoid receptor antagonists and/or a beta blocker, though no studies have been performed to identify the utility of these agents in this setting.
In conclusion, this case series raises critical clinical and mechanistic insight considerations for the patient with DMD. Acute cardiac injury should be considered in a DMD patient who presents with significant chest pain even at a young age. Based on our observations we would recommend at a minimum an electrocardiogram and evaluation of cTn. Abnormalities in either should prompt cardiac consultation. It is important to note that the assessment of coronary artery disease by coronary CT or cardiac catheterization should be reserved only for patients who present with evidence of STEMI pattern on ECG or other supportive diagnostic findings. Viral studies beyond clinically available PCR panels to include more sophisticated virome and host transcriptomics may need to be considered in the future [27, 28]. Finally, a deeper understanding of these events will hopefully lead to the development of therapies to slow disease progression.
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
Footnotes
ACKNOWLEDGMENTS
Authors would like to thank Parent Project Muscular Dystrophy for organizing and sponsoring the Acute Chest Pain in Duchenne Muscular Dystrophy Meeting in Columbus, Ohio in May 2016. The authors would like to thank all attendees for their important participation and intellectual contributions to this work.
