Abstract
Background
Duchenne muscular dystrophy (DMD) leads to dilated cardiomyopathy and heart failure during teenage years or young adulthood. Eteplirsen promotes dystrophin production through skipping of exon 51 of the DMD gene.
Objective
This analysis compared LVEF decline between eteplirsen-treated and control patients with exon 51 skip-amenable DMD.
Methods
Eteplirsen-treated patients from clinical trials were matched with control patients from natural history studies in a propensity score analysis. Risk of reaching LVEF thresholds (50%, 55%, and 60%) was evaluated using Cox proportional hazard models. Annual rate of LVEF decline was characterised using linear mixed effects models.
Results
Among 141 eteplirsen-treated and 103 control patients available for matching, the analysis included 122 eteplirsen-treated patients matched with 122 control patients (64 unique control patients). No eteplirsen-treated and 27 controls (22.1%) reached LVEF <50%; eteplirsen-treated patients had a lower risk of reaching <55% and <60% thresholds versus controls (hazard ratio = 0.22; 95% CI = 0.07–0.66; P < 0.01 and hazard ratio = 0.40; 95% CI = 0.22–0.76; P < 0.01, respectively). Annual rate of LVEF decline for eteplirsen-treated and controls was −0.66% (95% CI = –0.96 to −0.36, P < 0.01) and −1.38% (95% CI = –1.60 to −1.16; P < 0.01), respectively. Results were consistent with a sensitivity analysis matching each eteplirsen-treated patient once with a unique control patient and with several tests for potential bias.
Conclusions
In this retrospective study, eteplirsen-treated patients were observed to have a significantly lower risk of reaching LVEF thresholds indicative of cardiac function decline and attenuation of LVEF decline compared with matched controls.
Keywords
Introduction
Duchenne muscular dystrophy (DMD) is the most common neuromuscular disease in children, with an X-linked inheritance pattern and affecting about 1 in 3500–5000 male births worldwide. 1 DMD is caused by mutations in the DMD gene that result in the absence of functional dystrophin protein, which leads to a progressive decline in ambulatory, respiratory, and cardiac functions, development of dilated cardiomyopathy, and progression to terminal heart failure during the teenage years or young adulthood. 2
Eteplirsen is a phosphorodiamidate morpholino oligomer (PMO) approved for the treatment of patients with DMD with mutations amenable to exon 51 skipping, representing approximately 13% of the DMD population. 3 Eteplirsen restores the open reading frame of the gene encoding dystrophin by pre-mRNA exon-skipping. This enables translation of an internally shortened, yet functional, dystrophin protein in skeletal and cardiac muscles. 4 Previous studies have demonstrated that treating patients with DMD with eteplirsen was associated with significant delays in the loss of ambulatory ability and attenuation of decline in pulmonary functions compared with mutation-matched natural history (NH) control patients.5–7 Preclinical studies suggest PMOs have a protective effect on cardiac function.8,9 However, studies quantifying the potential of eteplirsen to attenuate the decline in cardiac function for patients with DMD who are amenable to exon 51 skipping are still lacking.
In this study, the risk of reaching left ventricular ejection fraction (LVEF) thresholds indicative of cardiac function decline, and annual rate of decline in cardiac function via LVEF over time were evaluated and compared between eteplirsen-treated and natural history control patients.
Methods
Ethical approval
The analysis was conducted in compliance with European and French legal and regulatory requirements, such as the Declaration of Helsinki and the General Data Protection Regulation. 10 The use of the French DMD Heart Registry was confirmed through Health Data Hub and Commission Nationale de l’Informatique et des Libertés registration.10,11 This analysis received an exemption in accordance with 45 CRF 46.104(d)(4) ‘Secondary Research Uses of Data or Specimens’ from the Pearl IRB Institutional Review Board (IRB ID 2023-0294). All data of French patients remained in France and were accessed only in France during the analysis.
Data sources
A broad set of data sources were considered to provide an adequate sample of patients with sufficient data for analysis, due to limited patients with exon 51 skip-amenable DMD and available cardiac function measures. Among observed cardiac function measures, LVEF was the most systematically available and clinically relevant measure across all data sets that could be compared directly. Requirements for clinical trial and NH data sources also included the availability of genotype information and patient baseline and medication characteristics related to cardiac function.
To estimate the effect of eteplirsen, cardiac outcomes of patients with exon 51 skip-amenable DMD treated with eteplirsen in clinical trials and controls from several observational and cohort studies were compared. The data for eteplirsen-treated patients were obtained from clinical trials performed at US-based clinical sites. The clinical trials included for the analysis were Study 201 (NCT01396239), 5 Study 202 (NCT01540409), 5 Study 203 (NCT02420379), 12 Study 204 (NCT02286947), 13 and Study 301 (NCT02255552) (Table S1). 7 The data for control patients were obtained from the Cooperative International Neuromuscular Research Group DMD Natural History Study (CINRG-DNHS; NCT00468832), 14 a Prospective Natural History Study of Progression of Subjects With Duchenne Muscular Dystrophy (PRO-DMD-01; NCT01753804), 15 and the French DMD Heart Registry (NCT03443115) (Table S2). 16 The study was reported in accordance with STROBE guidelines for cohort studies.17,18
Patient selection
Patients with DMD amenable to exon 51 skipping administered eteplirsen and/or standard of care treatment (long-term glucocorticoid use, relevant pulmonary, ambulatory, and cardiac management care) 19 were included in the study based on the following inclusion criteria: 1) availability of LVEF at baseline and ≥1 post-baseline visit and 2) complete information on patient's age and whether or not they were using concomitant medications, including cardiac medications (e.g., angiotensin-converting enzyme [ACE] inhibitors and/or angiotensin-receptor blockers [ARBs]) and corticosteroids at the baseline visit. Baseline visit was defined as the screening or initial treatment visit for eteplirsen-treated patients, and as the first visit with non-missing LVEF for NH control patients.
Study endpoints
In this study, LVEF by echocardiography was used as a measure of cardiac function and the endpoints included time to reach various threshold values of LVEF. Specifically, a 50% threshold was considered an indicator of abnormal cardiac systolic function,20,21 and two earlier thresholds (i.e., LVEF <55% and <60%) were included to better illustrate the decline in cardiac function. Though these latter two thresholds are not commonly used to define the thresholds of the abnormal range of cardiac function, they are generally not reached in healthy paediatric populations as the normal values for ejection fraction are usually between 56%–78%. 22 Continuous LVEF was used to characterise the trajectory of cardiac function decline over time. All studies from which data were drawn allowed for practitioners’ choice to derive LVEF, including the Simpson method or a visual assessment.
Statistical analyses
This study included several analytical and validation steps to assess the potential for differences in cardiac outcomes between the eteplirsen-treated and matched NH control patients. Propensity score (PS) matching was conducted to mitigate the effect of differences in the underlying population resulting from the use of multiple data sources with different study designs and to align the observed baseline characteristics and prognostic factors that may have had a potential confounding influence on LVEF decline. To assess the potential differences between the matched cohorts, time-to-event and trajectory analyses were performed.
A PS matching approach was used to establish balanced cohorts by aligning the time of eteplirsen initiation for the eteplirsen-treated patients with a point in the lifespan of the NH control patients when their characteristics were the most comparable, while allowing for sufficient availability of follow-up data for the analyses. All baseline characteristics used in earlier research were considered and only those available across data sets were used for PS matching. Similar to the factors discussed in the latest research by Landfeldt et al., 23 baseline characteristics used in this analysis included baseline age, LVEF, and exposure to cardiac medication, including prophylactic ACE inhibitor and/or ARB use prior to the first LVEF <50% event (to address the potential risk of reverse causation whereby the early use of these medications could have delayed the advent of cardiac dysfunction), and stable corticosteroid use. To identify patients with enough length of exposure to corticosteroids and reduce the risk of a confounding effect, stable corticosteroid use was defined as any type of corticosteroid medication, including prednisone, prednisolone, or deflazacort, recorded in ≥30% of visits over ≥18 months, based on clinical expert input.
The PS was calculated for the baseline visit of eteplirsen-treated patients and for all candidate visits of NH control patients with comparable characteristics and sufficient follow-up data available for analysis. Each eteplirsen-treated patient (at their baseline visit) was matched with a control patient with the closest PS at a qualifying visit. The qualifying visit that had the closest PS to a baseline visit of a treated patient was matched, and this matched visit was defined as the post-matching baseline visit of the control patient. To manage the trade-off between closeness of the matched pair and maximal use of available data, a calliper approach was used, which imposes a limit on the maximum allowable distance between the PS of any matched pair. Eteplirsen-treated patients without a control patient with a close enough PS were excluded. Limited replacement was allowed, whereby each control patient could be matched a maximum of two times, and a calliper of 1.6 was empirically set to balance the closeness of the match (i.e., to achieve P-values >0.2 in the t-test comparisons for matching characteristics to assess whether the matched samples were balanced on the PS matching variables) and the maximal use of available data (retaining more than 80% of eteplirsen-treated patients). To provide a more rigorous assessment of the data, a sensitivity analysis was conducted using an alternative PS matching approach in which each eteplirsen-treated patient was matched with a unique control patient with the closest PS at a qualifying visit (i.e., each NH control patient was matched only once; Supplementary Appendix). 24
In the time-to-event-analysis, Kaplan-Meier curves were used to describe LVEF decline thresholds over time among eteplirsen-treated and matched NH control patients. The median age to the event was reported, and when the median age was not directly available, a parametric log-logistic accelerated failure time model with the lowest Akaike Information Criterion/Bayesian Information Criterion value was fitted to the Kaplan-Meier curves to provide an alternative estimate of the median age to reach LVEF thresholds.
With adjustments for baseline characteristics and prognostic factors, Cox proportional hazard models were used to estimate the hazard ratio (HR) of eteplirsen treatment and evaluate the impact of eteplirsen on the risk of reaching LVEF decline thresholds. The baseline characteristics and prognostic factors adjusted for in the models included baseline age and LVEF, prophylactic ACE inhibitor/ARB use prior to the first LVEF <50% event, and stable corticosteroid use.
In the trajectory analyses, the LVEF annual rate of decline was characterised using a linear mixed effects models (LMEM) framework, which evaluated the impact of eteplirsen treatment on the annual rate of LVEF change compared with matched NH control patients. The LMEM regressed LVEF on age, eteplirsen treatment, and interaction term. The coefficient of age was estimated to derive the average annual rate of LVEF decline among NH control patients. The coefficient of the interaction term between age and treatment was estimated to identify the difference in the average annual rate of LVEF decline between the eteplirsen-treated and matched NH control patients. Other control variables in the LMEMs included baseline age and LVEF, prophylactic ACE inhibitor/ARB use prior to the first LVEF <50% event, an indicator of stable corticosteroid use, and an indicator of age >18 and its interaction term with age (to account for the potentially slower decline of LVEF after 18 years of age). Further, a linear extrapolation was performed using model-estimated annual decline in LVEF starting from the average LVEF observed in patients at 9 years of age (i.e., the average baseline age of eteplirsen-treated patients observed in the data).
Three validation analyses were conducted to evaluate potential remaining sources of bias in this non-randomised cohort study: 1) an assessment of the risk of geographic imbalance between the treated and control patients, 2) an E-value analysis to evaluate the risk that any uncontrolled confounder could be the source of an observed treatment effect, according to the recommendation of assessing the threat of bias via quantitative bias analysis by Gray et al., 25 and 3) a restricted mean survival time analysis to evaluate the significance of any observed treatment effect within different age segments (Supplemental Methods). All statistical analyses were performed using the R programming software (version 3.6.2). 26
Results
Study population and patient characteristics
A total of 141 eteplirsen-treated patients and 103 NH control patients with DMD amenable to exon 51 skipping were available for matching (Figure 1). One hundred twenty-two eteplirsen-treated patients were matched with 122 NH controls (with data from 64 unique NH control patients, as each control patient could be matched with up to two eteplirsen-treated patients) (Table 1; Supplemental Methods). Among the matched cohort, the mean age at baseline was 9.1 ± 3.1 and 9.6 ± 5.3 years and the mean LVEF was 67.4% ± 5.3% and 67.7% ± 5.9% in the eteplirsen-treated and NH control patients, respectively. Mean (±SD) duration of follow-up was 26.5 ± 9.7 months and 69.3 ± 71.1 months among the 122 eteplirsen-treated and 122 NH control matched patients, respectively (Table 2).
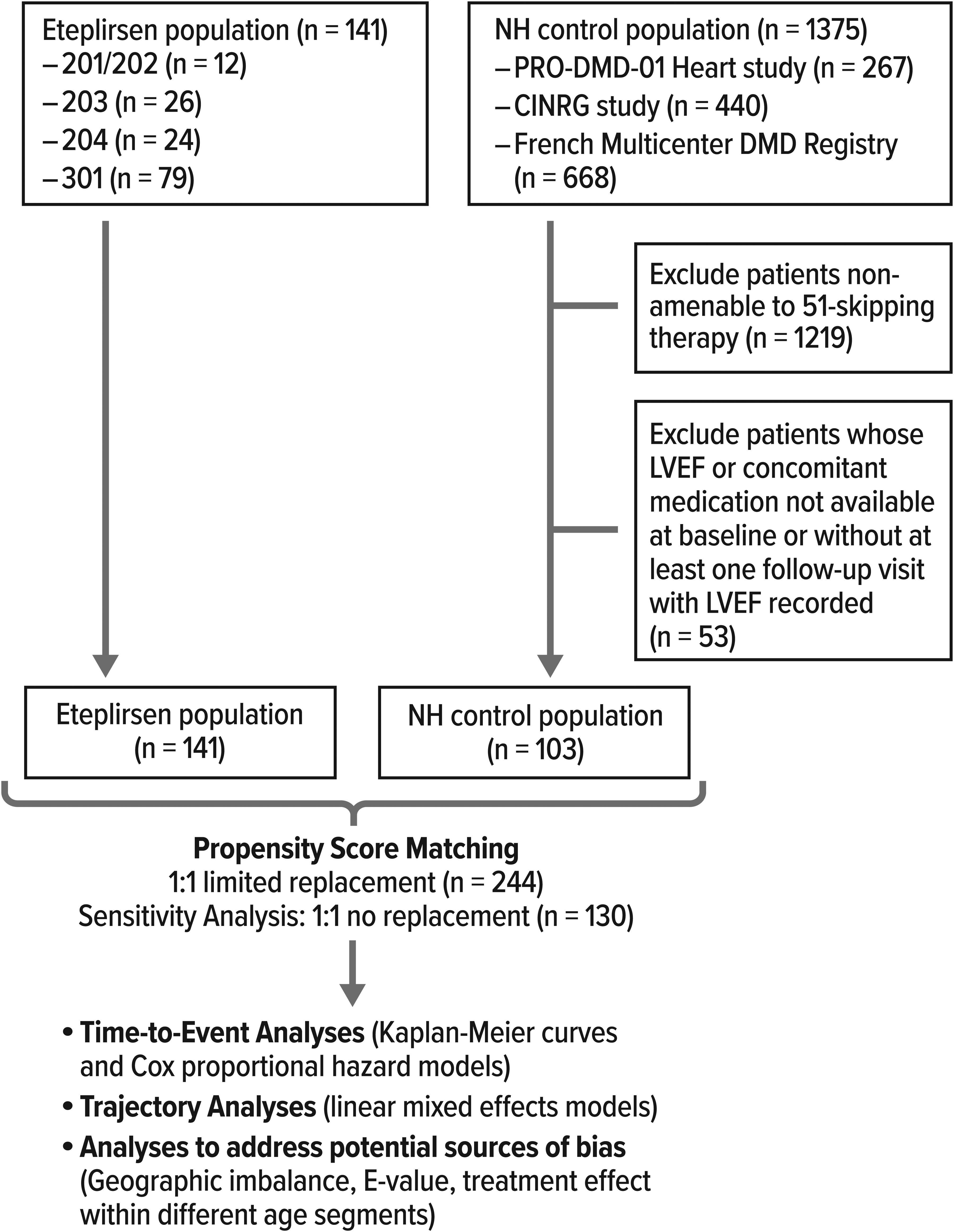
Diagram of sample selection and analysis. CINRG-DNHS, Cooperative International Neuromuscular Research Group DMD Natural History Study; DMD, Duchenne muscular dystrophy; NH, natural history; LMEM, linear mixed effects models; LVEF, left ventricular ejection fraction; PRO-DMD-01, Prospective Natural History Study of Progression of Subjects with Duchenne Muscular Dystrophy.
Baseline and matched characteristics.
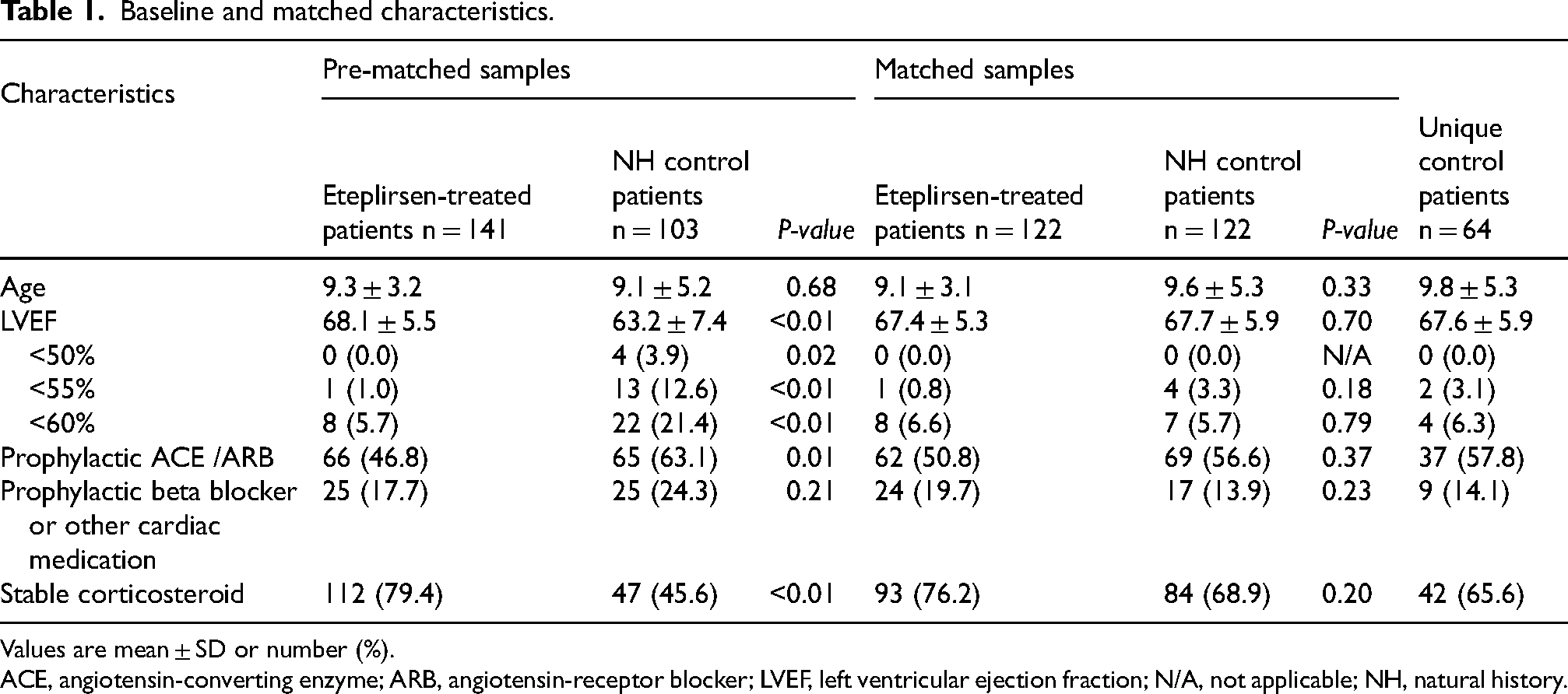
Values are mean ± SD or number (%).
ACE, angiotensin-converting enzyme; ARB, angiotensin-receptor blocker; LVEF, left ventricular ejection fraction; N/A, not applicable; NH, natural history.
Follow-up characteristics.
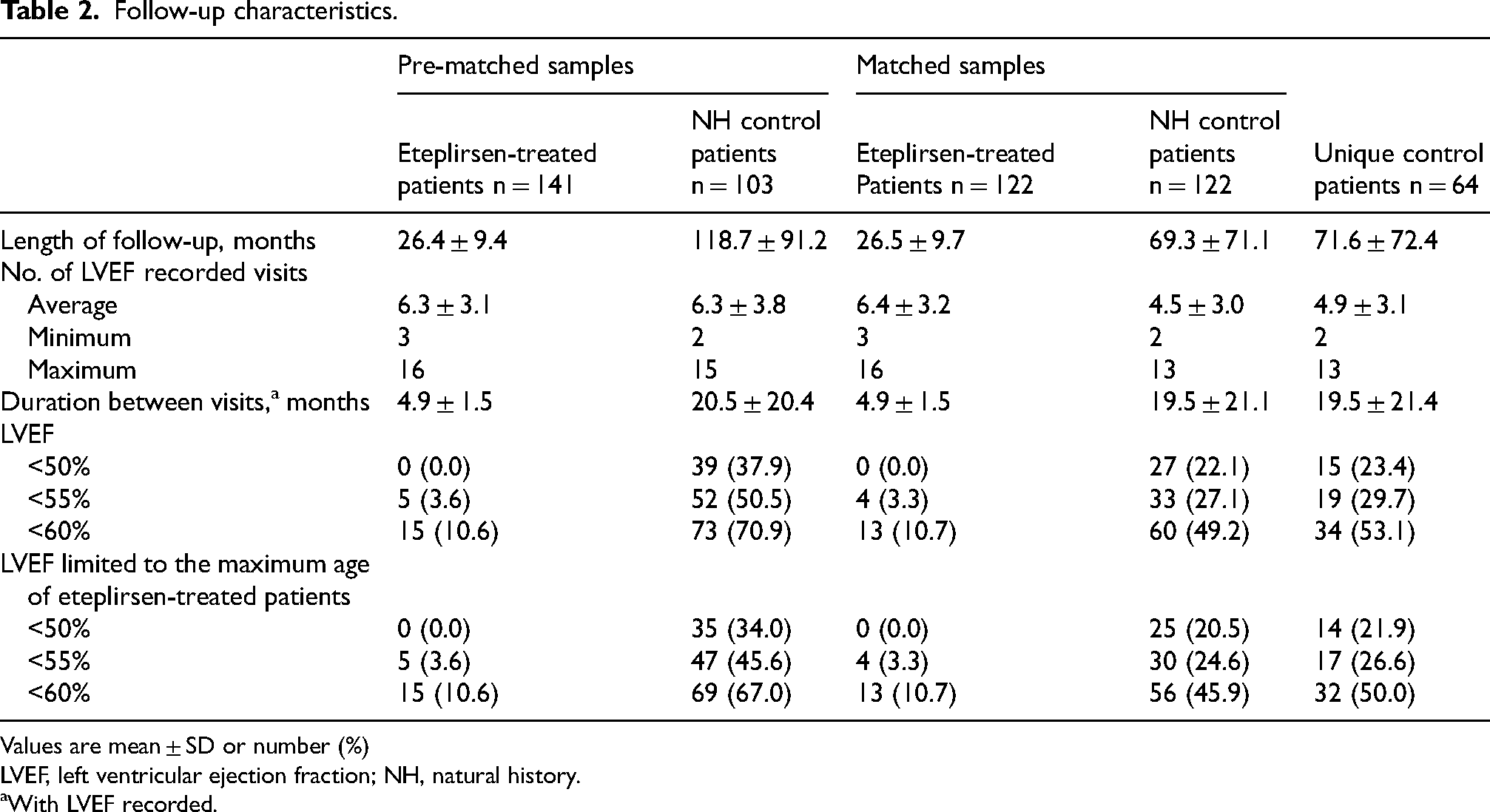
Values are mean ± SD or number (%)
LVEF, left ventricular ejection fraction; NH, natural history.
With LVEF recorded.
Nineteen eteplirsen-treated patients and 39 unique NH control patients were excluded in the matching process. Characteristics for these excluded patients are in Table S3.
The t-test for main variables of interest indicated a well-matched sample for baseline age (P = 0.33), baseline LVEF (P = 0.70), prophylactic ACE/ARB use (P = 0.37), and stable corticosteroid use (P = 0.20; Table 1). The PS density plot also indicated a similar distribution for the matched eteplirsen-treated and NH control patients (Figure S1). Samples remained well-matched in the sensitivity analysis (Table S4; Figure S2). Patient baseline characteristics are presented in Table S5.
Time-to-event analysis
Patient characteristics during the follow-up visits for the pre-matched and matched patients are presented in Table 2 and by data source in Table S6. In the pre-matched samples, no eteplirsen-treated patients reached LVEF <50% and the proportion of patients reaching the <55% and <60% thresholds was significantly lower for eteplirsen-treated patients compared with NH control patients. In the matched cohort (i.e., 122 eteplirsen-treated and 122 NH control patients), no eteplirsen-treated patients reached LVEF <50%, whereas 27 NH control patients (22.1%) reached this threshold (Table 2). Four eteplirsen-treated patients (3.3%) and 33 NH control patients (27.1%) reached LVEF <55%. Finally, 13 eteplirsen-treated patients (10.7%) and 60 NH control patients (49.2%) reached LVEF <60%. The sensitivity analysis with PS matching approach with a unique control patient yielded similar time-to-event analysis results (Table S7).
In eteplirsen-treated patients, the median age was not reached for the <50% or <55% thresholds and was 20.1 years for the LVEF <60% threshold. The median age to reach the LVEF <50%, < 55%, and <60% thresholds in matched NH control patients was 19.9, 17.3, and 14.6 years, respectively (Figure 2). The estimated median age to LVEF <55% and <60% based on a parametric fit was 27.6 and 20.7 years for eteplirsen-treated patients and 19.2 and 14.9 years for NH control patients, suggesting an estimated additional time of 8.4 and 5.8 years to reach these LVEF thresholds among eteplirsen-treated patients (Table S8). The sensitivity analysis yielded similar median ages to reach LVEF thresholds (Figure S3 and Table S8).
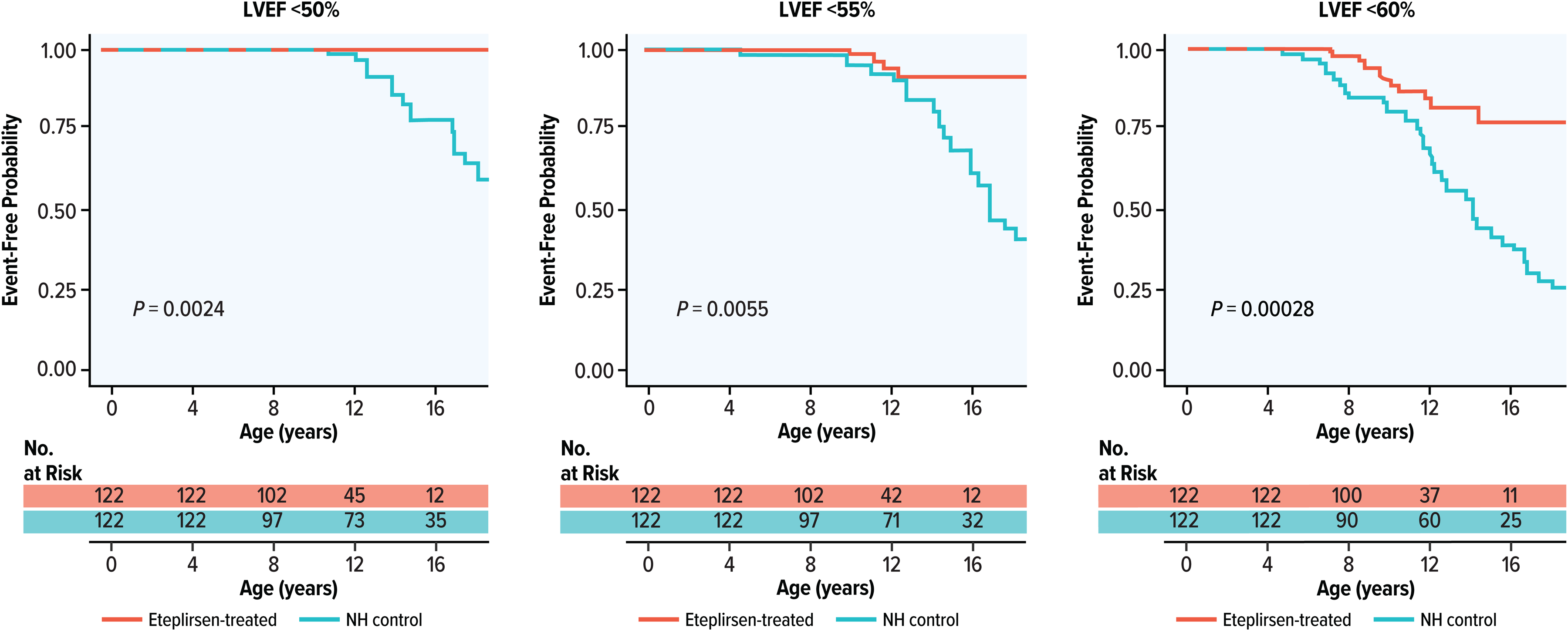
Kaplan-Meier curve for LVEF thresholds. KM, Kaplan-Meier; LVEF, left ventricular ejection fraction; NH, natural history.
Since no eteplirsen-treated patient reached the LVEF <50% threshold, the Cox model for the LVEF <50% threshold did not converge and could not be developed (Figure 2). Based on the Cox models with adjustments for patient baseline characteristics (Table 3 and Table S9
Multivariable Cox proportional hazard models.
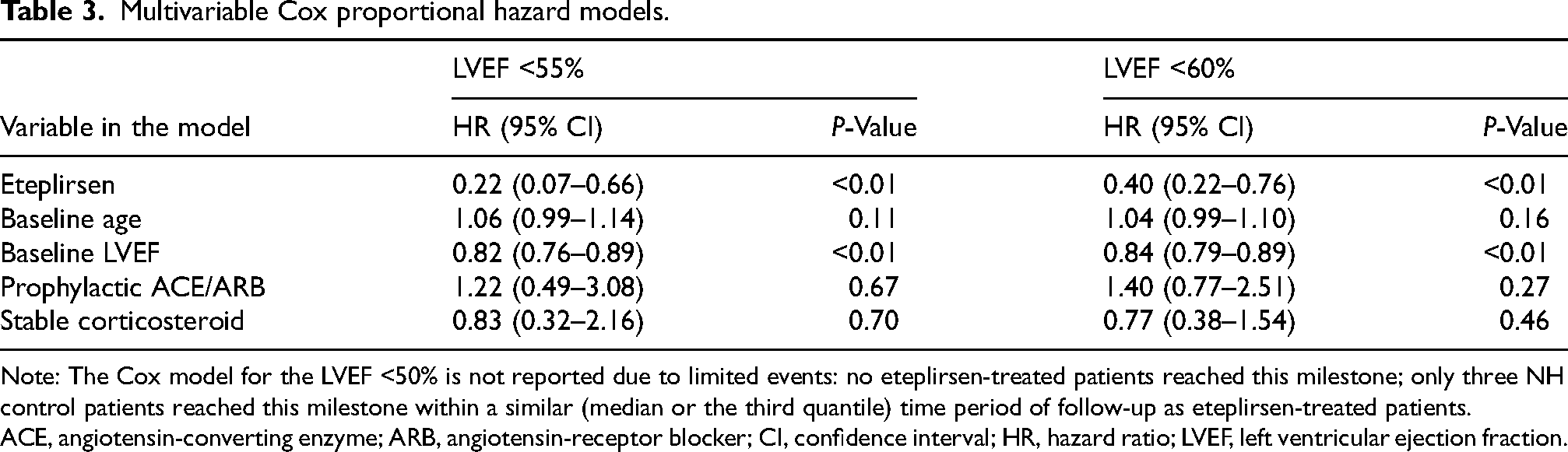
Note: The Cox model for the LVEF <50% is not reported due to limited events: no eteplirsen-treated patients reached this milestone; only three NH control patients reached this milestone within a similar (median or the third quantile) time period of follow-up as eteplirsen-treated patients.
ACE, angiotensin-converting enzyme; ARB, angiotensin-receptor blocker; CI, confidence interval; HR, hazard ratio; LVEF, left ventricular ejection fraction.
Trajectory analysis
The estimated rate of LVEF decline in the eteplirsen-treated and the NH control patients was −0.66 (95% CI = –0.96 to −0.36; P < 0.01) and −1.38 (95% CI = –1.60 to −1.16; P < 0.01) percentage points per year, respectively (Table S11). The rate of LVEF decline in eteplirsen-treated patients was derived based on the coefficients of age (–1.38; P < 0.1) and the coefficient of the interaction term between age and eteplirsen treatment (0.72 [95% CI = 0.41–1.03; P < 0.01]). The coefficients of the interaction term indicated the difference in LVEF decline rates between eteplirsen-treated and NH control patients. The control variables included in the multivariate model and their coefficients are presented in Table S11. A scatter plot for LVEF over time and the projected LVEF decline over time for a hypothetical patient with and without treatment started at age 9 are both displayed in Figure 3. Sensitivity analysis results were similar (Table S11 and Figure S4).
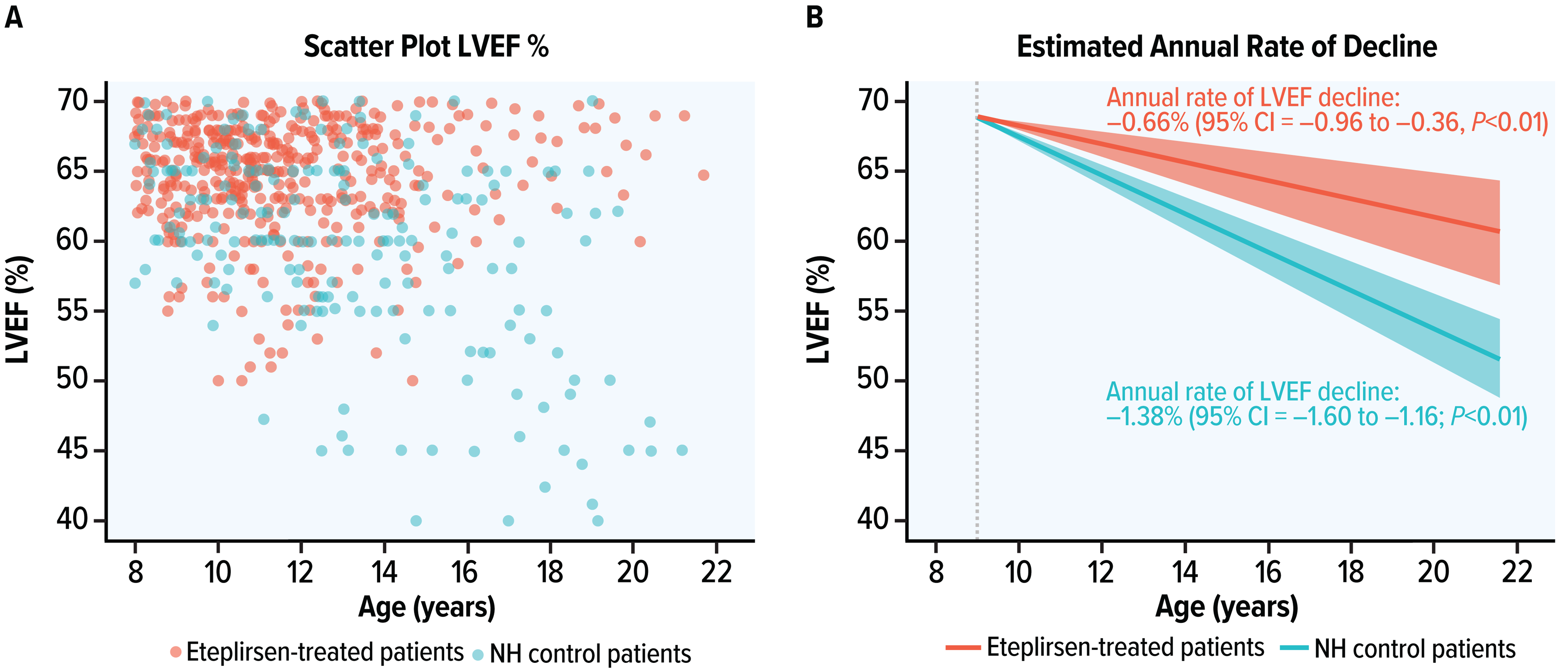
Scatter plot and estimated rate of LVEF decline for a hypothetical patient with and without treatment starting at age 9 in the LMEM trajectory analysis. Left panel: scatter plot, and right panel: decline extrapolation in the LMEM. LMEM, linear mixed effects model; LVEF, left ventricular ejection fraction; NH, natural history.
Analyses to address potential sources of bias
The results did not suggest significant difference in the rate of LVEF decline by age among different regional groups (i.e., United States, Europe/Australia, and other; coefficient of the interaction term between age and region was 0.18; P = 0.58).
The E-value of the point estimate in the Cox model for time required to reach the first LVEF <55% threshold was 8.66 (95% CI upper bound = 2.38; Figure S5 and Table S9). This represents the minimum strength of association needed of an unmeasured confounder with both receiving eteplirsen and reaching LVEF <55% to nullify the observed treatment effect. Therefore, this association would need to be 8.66 times stronger than the estimated effect of eteplirsen treatment on time to reach LVEF <55% for the effect of eteplirsen to be deemed insignificant. For LVEF <60%, the estimated E-value was 4.40 (95% CI upper bound = 1.98).
The restricted mean survival time analysis results suggested that the eteplirsen-treated patients had a significantly lower risk of reaching LVEF thresholds at ≥16 years of age compared with the matched NH control patients (Table S12). Sensitivity analysis results were consistent with these findings (Table S12).
Discussion
In this study, eteplirsen treatment was associated with a reduced risk of reaching various LVEF thresholds, a longer time to reach LVEF thresholds, and a slower annual rate of LVEF decline in patients with exon 51 skip-amenable DMD compared with NH controls, after matching patient characteristics such as baseline age and LVEF, prophylactic use of ACE inhibitors or ARBs, and stable corticosteroid use.
Cardiomyopathy-associated heart failure with a reduced LVEF is a limiting factor for survival of patients with DMD, 2 and declining LVEF is associated with long-term severe cardiovascular events, including hospitalisation for heart failure and cardiovascular disease–related mortality.27,28 LVEF is one of the most robust prognostic parameters for patients with dilated cardiomyopathy of any cause. 29 Therefore, delaying the onset of LVEF thresholds could be clinically meaningful and help improve long-term outcomes of patients with DMD.
To our knowledge, the only approved non-investigational treatments that have proven beneficial to prevent LVEF decline in patients with DMD are ACE inhibitors, aldosterone receptor antagonists, and glucocorticoids.30–32 This study is the first to demonstrate significant change in measured cardiac function with an innovative therapy that aims to increase dystrophin expression, suggesting clinically meaningful multi-year delays in reaching cardiomyopathy milestones. Consistent with our results, previous clinical studies have indicated a beneficial effect of eteplirsen on skeletal muscle, including stabilisation of respiratory and ambulatory function compared with matched controls5–7; however, a comparative analysis of the effect of eteplirsen on cardiac function has not previously been conducted.
The results of this study suggest a positive association between eteplirsen and significantly lower risk of cardiac function decline as measured by LVEF. These results are consistent with preclinical studies in DMD mouse models using high doses of PMOs, which demonstrated low level cardiac muscle targeting following this treatment along with a functional benefit.8,9 For example, potential therapeutic levels of dystrophin were achieved in cardiac muscle, but at greater dosages than in skeletal muscles. 8 Additionally, PMO treatment induced effective target exon skipping and dystrophin expression in all muscles in a dose-dependent manner during 1 year of treatment, and improved cardiac function as demonstrated by haemodynamic analysis (e.g., cardiac output, ejection fraction). 9
This study compared pooled data from multiple clinical trials and NH studies and provides, for the first time, an evaluation of the effect of eteplirsen on LVEF. The use of non-randomised data sources was required as the effect of eteplirsen on cardiac outcomes has not been included in prior randomised studies. PS matching and adjusting for baseline characteristics and prognostic factors in Cox and LMEM were used to limit the influence of potential biases incurred by these data sources. All results consistently indicated a significant effect of eteplirsen on LVEF decline in patients with DMD, including the post-matching time-to-event and trajectory analysis with adjustment for key cardiac prognostic factors, such as baseline age and LVEF, and concomitant usage of ACE inhibitors, ARBs, and stable corticosteroids. In addition, quantitative bias analyses using the E-value estimation indicated that a very large amount of unmeasured confounding would be required to nullify observed findings. Other validation analyses for potential sources of bias also suggest the estimated effects of the treatment were robust. It is noteworthy that these indirect comparison statistical analyses are increasingly recognised by the Food and Drug Administration and European Medicines Agency as reasonable approaches for estimating the clinical benefit of treatments for rare diseases in a real-world setting. 33 This is particularly true when the clinical effect is considered highly significant, as was the case in this study, with the observed differences in LVEF decline, especially below the 50% threshold.
These results should be interpreted considering certain limitations as a number of biases are inherent in non-randomised, observational studies such as this one. A possible source of selection bias at study entry was the exclusion of some patients and the repetitive use of patient history for some control patients (who were matched twice) due to the use of PS matching. Results suggest, however, that the treatment effect of eteplirsen could have been larger if some of the excluded patients were included. This is because excluded eteplirsen-treated patients tended to have higher LVEF (less severe disease) at baseline and excluded NH control patients tended to have lower LVEF at baseline (more severe disease). This study was also subject to confounding as it encompasses data sources from multiple studies that may vary in underlying populations, follow-up periods, study inclusion criteria, and standard of care. Specifically, the median birth year of the control cohort from the PRO-DMD-01 study and French DMD Heart Registry (birth year data are not available for CINRG DNHS) was 5 years earlier than that of the eteplirsen-treated cohort (2001 for the control cohort vs 2006 for the eteplirsen-treated cohort), which may lead to differences in the standard of care received that could potentially influence the observed outcomes. All statistical tools available were used to limit the influence of such biases, including PS matching and regression adjustments for baseline characteristics and prognostic factors, and analyses such as E-value to quantify such biases. Results of all these statistical analyses suggest that such biases were not sufficient to impact the observed treatment effect. Additionally, in a recent study, the mean age of reaching LVEF <50% among patients with DMD treated with glucocorticoids was 17.6 years, suggesting similar disease progression between the control cohorts included in this analysis and other patient populations treated with glucocorticoids. 34 This analysis may have suffered from detection bias as eteplirsen-treated patients had a greater frequency of healthcare visits during clinical trials relative to NH control patients, but detection of LVEF events at an earlier stage in eteplirsen-treated patients compared with NH control patients who had less frequent healthcare visits would be expected to occur in a conservative direction against finding a treatment effect. Also, LVEF measurement by echocardiogram may have varied in the studies from which data were drawn as this was left to practitioner discretion. LVEF measurement requires some subjective evaluation which could introduce variation in the measurement. Nonetheless, LVEF has been a primary outcome considered in other studies of cardiac function in DMD21,23 and was the only marker of cardiac outcomes broadly available among the data sources for this study; thus, LVEF was selected as the outcome of interest in this study. Although concomitant use of cardiac medications and corticosteroids was balanced in the post-match samples of treated and control patients, dose and regimen data were not consistently available across data sources and were not included as controls in the analysis. Results of the E-value analysis indicate that an imbalance of individual dose or regimen as potential uncontrolled confounders would have to be extreme to invalidate the treatment effect estimation. Finally, although beyond the scope of this study, since no cardiac muscle biopsies in humans treated with eteplirsen are currently available, we cannot rule out a secondary cardiac benefit of dystrophin expression in respiratory skeletal musculature.
Conclusions
In conclusion, patients with exon 51 skip-amenable DMD, eteplirsen treatment was associated with a significant attenuation of the rate of LVEF decline, an absence of patients reaching LVEF <50%, a reduced risk of reaching other LVEF decline thresholds, and a longer time to reach LVEF thresholds after accounting for baseline age and LVEF, prophylactic use of ACE inhibitors or ARBs, and stable corticosteroid use.
Supplemental Material
sj-docx-1-jnd-10.1177_22143602251366721 - Supplemental material for Association between exon-skipping therapy with eteplirsen and cardiac outcomes in Duchenne muscular dystrophy
Supplemental material, sj-docx-1-jnd-10.1177_22143602251366721 for Association between exon-skipping therapy with eteplirsen and cardiac outcomes in Duchenne muscular dystrophy by Joel Iff, Isabelle Desguerre, Yunjuan Liu, Francois Sarkozy, Edward Tuttle, Francesco Muntoni, Craig M McDonald, Marie-Christine Nougues, Helge Amthor, Yi Zhong, Karim Wahbi and in Journal of Neuromuscular Diseases
Footnotes
Acknowledgements
We acknowledge the assistance of Didrik Prohorenko and Fangzhou Wei with data analysis. Both are employees of Analysis Group, Inc., a consulting company that has provided paid consulting services to Sarepta Therapeutics, Inc. Medical writing assistance was provided by Loraine Georgy, PhD, MWC, an employee of Analysis Group, Inc., and Angela Cimmino, PharmD, an employee of Kay Square Scientific. Initial editorial support was provided by Eloquent Scientific Services. Medical writing assistance and editorial support through submission were provided by Symbiotix, part of Havas Health & You. Sarepta Therapeutics, Inc., funded the development and conduct of this study and manuscript, as well as funded the above-mentioned organisations for their assistance.
Ethical considerations
The analysis was conducted in compliance with European and French legal and regulatory requirements, such as the Declaration of Helsinki and the General Data Protection Regulation. The use of the French DMD Heart Registry was confirmed through Health Data Hub and Commission Nationale de l’Informatique et des Libertés registration. This analysis received an exemption in accordance with 45 CRF 46.104(d)(4) ‘Secondary Research Uses of Data or Specimens’ from the Pearl IRB Institutional Review Board (IRB ID 2023-0294). All data of French patients remained in France and were accessed only in France during the analysis.
Author contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Sarepta Therapeutics, Inc., Massachusetts, United States.
Declaration of conflicting interest
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Yunjuan Liu, Edward Tuttle, and Yi Zhong are employees of Analysis Group, Inc., a consulting company that has provided paid consulting services to Sarepta Therapeutics, Inc., which funded the development and conduct of this study and manuscript. Francesco Muntoni has received grants and speaker and consultancy honoraria from Sarepta Therapeutics, Roche, PTC Therapeutics, Dyne Therapeutics, and Pfizer. François Sarkozy, the founder and president of FSNB Health & Care, has received consultancy honoraria from Sarepta Therapeutics. Joel Iff is an employee of Sarepta Therapeutics, Inc., and owns Sarepta stock. Helge Amthor has received speaker and consultancy honoraria from Sarepta Therapeutics, Roche, Pfizer, and SQY Therapeutics. Craig M. McDonald has received consulting fees and payment or honoraria from Astellas, Edgewise Therapeutics, Italfarmaco, Pfizer, Catalyst, PTC Therapeutics, Sarepta Therapeutics, Inc., Roche, Santhera Pharmaceuticals, and Capricor. Karim Wahbi has received grants and speaker and consultancy honoraria from Sarepta Therapeutics, PTC Therapeutics, and Pfizer. Isabelle Desguerre and Marie-Christine Nougues have no conflicts of interest to declare. All companies listed are or have been involved in the development of products for the treatment of Duchenne muscular dystrophy or provide other healthcare services. Sarepta Therapeutics, Inc. is the sponsor of the clinical trials from which the data for eteplirsen-treated patients were obtained in this study.
Data availability
For clinical trial data, individual de-identified patient data will not be available in a publicly accessible repository to protect the interests of the patients in accordance with the policies of Sarepta Therapeutics, Inc., and in line with the general data protection regulation. Additional data underlying this article were provided by CINRG DNHS, PRO-DMD-01, and the French DMD Heart Registry under license. Data requests may be directed to these organisations that have collected and curated patient data. These organisations will consider data requests according to their own data-sharing policies and governance.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
