Abstract
Background:
Huntington’s disease (HD) is a neurodegenerative disorder characterized by motor, cognitive, and psychiatric dysfunction caused by a mutant huntingtin protein. Compromised metabolic activity resulting from systemic administration of the mitochondrial toxin, 3-nitropropionic acid (3-NP), is known to mimic the pathology of HD and induce HD-like symptoms in rats. N-hexanoic-Tyr-Ile-(6)-amino hexanoic amide (PNB-0408), also known as Dihexa, has been shown to have neuroprotective and procognitive properties in animal models of Alzheimer’s and Parkinson’s diseases. Given the mechanism of action and success in other neurodegenerative diseases, we felt it an appropriate compound to investigate further for HD.
Objective:
The present study was designed to test if PNB-0408, an angiotensin IV analog, could attenuate 3-NP-induced HD-like symptoms in rats and serve as a potential therapeutic agent.
Methods:
Forty male Wistar rats were randomized into three groups consisting of a “vehicle” group, a “3-NP” group, and a “3-NP + PNB-0408” group. PNB-0408 was administered along with chronic exposure to 3-NP. Animal body weight, motor function, and cognitive abilities were measured for five weeks, before euthanasia and histopathological analysis.
Results:
Exposure to 3-NP decreased the amount of weight rats gained, impaired spatial learning and memory consolidation, and led to marked motor dysfunction. From our observations and analysis, PNB-0408 did not protect rats from the deficits induced by 3-NP neurotoxicity.
Conclusions:
Our findings suggest that PNB-0408 may not be an efficacious treatment strategy for preventing 3-NP-induced HD-like symptoms in a preclinical model. These data highlight the need for further research of this compound in alternate models and/or alternative approaches to managing this disorder.
Keywords
INTRODUCTION
Huntington’s disease (HD) is an incurable neurodegenerative disorder that is among high priority for researchers and physicians due to its midlife onset and rapid progression toward death 15 to 20 years from the time symptoms appear [1–3]. The genetic disorder is a triad of symptoms characterized by motor abnormalities, cognitive impairments, and psychiatric symptoms with the causative agent being a mutant huntingtin protein (mHtt) [4, 5]. MHtt is thought to have a toxic gain of function that leads to the development of HD, causing neuronal deterioration primarily in the medium-sized spiny neurons (MSNs) of the striatum and cerebral cortex pyramidal neurons that project to the striatum [6, 7]. This neuronal deterioration is thought to be related to mHtt protein interference of the mitochondrial electron transport chain which eventually leads to mitochondrial dysfunction and failure of adenosine triphosphate (ATP) production [8]. Currently, there are no treatments that can effectively alter the course of HD. Medications can reduce the severity of some psychiatric and motor symptoms, but none treat the cognitive dysfunction or slow the disease progression. Because of this, the discovery of an effective disease modifying treatment is essential.
N-hexanoic-Tyr-Ile-(6) aminohexanoic amide (PNB-0408), also known as Dihexa, is a small, orally active angiotensin IV (AngIV) peptidomimetic that can cross the blood-brain barrier and has been shown to have procognitive and antidementia properties in animal models of Alzheimer’s (AD) and Parkinson’s diseases (PD). It is demonstrated to stimulate neurogenesis and synaptogenesis, have neuroprotective activity, enhance dendritic sprouting, and induce stem cell proliferation and differentiation through the brain renin-angiotensin system and hepatocyte growth factor (HGF) pathway [9].
The high-affinity binding of PNB-0408 to the angiotensin Type 4 receptor subtype (AT4), which is found in high concentrations in certain brain structures including the striatum, is thought to regulate functions such as cerebral blood flow, neuroprotection, memory consolidation, and synaptogenesis [10–13]. It has been hypothesized that the AT4 receptor subtype coincides with the HGF/c-Met receptor system. HGF is a glycoprotein growth factor with documented roles in stem cell regulation, general proliferation, anti-apoptotic, mitogenic and morphogenic actions on many cell types [14]. This growth factor acts via the Type 1 tyrosine kinase receptor c-Met and is thought to be a regulator of neuronal stem cell expansion and differentiation which suggests the important involvement the HGF/c-Met system has on neural cell survival and growth [15, 16]. It is thought that AT4 receptor agonists, such as PNB-0408, coincide with the HGF/c-Met system by allosterically binding to the hinge region of HGF and facilitate activation of the c-Met receptor by promoting HGF dimerization [17].
It is well known that neurons in the brain have high energy demands that are fulfilled by the mitochondrial production of ATP through oxidative phosphorylation; however, an abnormality in mitochondrial function has been reported to have significant pathological impacts in cancer, aging, diabetes, and neurodegenerative diseases such as AD, PD, and HD [18]. 3-nitropropionic acid (3-NP) is a blood-brain barrier permeable environmental toxin that causes irreversible inhibition of mitochondrial complex II, succinate dehydrogenase which interrupts oxidative phosphorylation, and ultimately inhibits the production of ATP in neuronal brain cells [19]. Chronic administration of this mitochondrial toxin results in selective loss of MSNs in the striatum and has been reported extensively for its induction of HD-like symptoms in animal models. Studies have shown that 3-NP induced neurotoxicity reproduces a broad array of biochemical, behavioral, and histopathological alterations which are similar to those seen in HD [20, 21].
Although PNB-0408 is being explored as an efficacious drug design for the treatment of AD and PD, its neuroprotective, procognitive, and antidementia properties are yet to be explored in HD. Given the mechanism of action, we found it reasonable, applicable, and necessary to investigate the small AngIV analog in an HD animal model. We speculated that PNB-0408 might improve the behavioral and cognitive performance of 3-NP induced neurotoxic rats.
MATERIALS AND METHODS
Chemicals and peptide synthesis
3-nitropropionic acid (N5636-1G, purity≥97%) was purchased from Sigma-Aldrich (USA). N-hexanoic-Tyr-Ile-(6) amino-hexanoic amide (PNB-0408) was synthesized using 9-flourenylmethoxycarbomyl-based solid-phase peptide synthesis methods and purified by reverse-phase high-pressure liquid chromatography in the Sardinia laboratory [22]. Purity and structure were verified by liquid chromatography mass spectrometry.
Animals
Forty male Wistar rats, six weeks old, weighing 150–240 g at the onset of the study, were used in this experiment. They were procured from Charles River Laboratories and maintained in the vivarium lab in the Robinson Science Center located on the campus of Whitworth University. All handling and experimenting were performed in this vivarium. The rats were in a temperature-regulated room and on a 12-h light/dark cycle. The research was conducted over a period of 38 days, and for the duration of the research, the rats had free access to food and water ad libitum. The experimental protocol was carried out per the Guide for the Care and Use of Laboratory Animals as adopted and promulgated by the U.S. National Institutes of Health and was approved by the Institutional Animal Care and Use Committee of Whitworth University.
Experimental design
The entire treatment protocol lasted 38 days, as shown in Fig. 1. The duration of the study and 3-NP dosage was exploratory and was determined once 3-NP administration induced observable impairment seen in behavioral testing. We based our power calculations on the outcome measures of weight change, beam walking, hanging wire, and Morris Water Maze test performance. Using data from published work by other groups, differences in group means were selected to represent meaningful and relevant changes induced by 3-NP and therapeutic molecules that have been shown to ameliorate the effects of 3-NP, specifically changes in body weight and the beam walking test [23]. Additionally, we considered data published on PNB-0408 and selected differences in means that were significant in PD and AD animal models in tests such as the hanging wire and Morris Water Maze [9, 22]. The results of our calculations supported the need for 11–12 rats per group. Allowing for a 10–15% mortality rate in the groups exposed to 3-NP, we randomly assigned rats into three groups as follows:
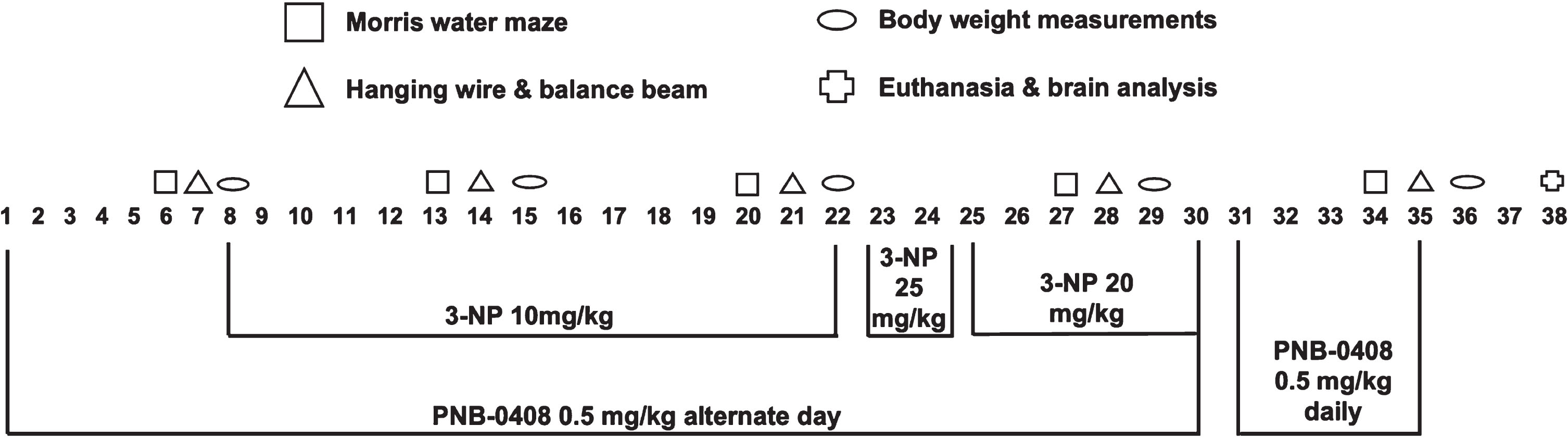
Experimental procedure and treatment protocol schedule. 40 male Wistar rats were randomly divided into three groups (N = 12–14 rats/group). One group received vehicle injections of saline and DMSO, another group received injections of 3-nitropropionic acid (3-NP), and the third group received injections of both 3-NP and PNB-0408. The dosage and injection timeline are presented above. Each week, animals were assessed for various physical, behavioral, and cognitive parameters. On day 38, five rats from each group were euthanized, and brain samples were collected for histopathological analysis.
“Vehicle” (12 rats): Saline (vehicle for 3-NP) and DMSO (vehicle for PNB-0408) (i.p.) “3-NP” (14 rats): 3-NP (10 then 25 then 20 mg/kg, i.p.) and DMSO (i.p.) “3-NP + PNB-0408” (14 rats): 3-NP (10, then 25, then 20 mg/kg, i.p) and PNB-0408 (0.5 mg/kg, i.p.)
3-NP was dissolved in normal saline and administered intraperitoneally (i.p.) at a dose of 10 mg/kg b.w./day from the 8th to the 22nd day of the treatment protocol [24]. After no evident impairment was observed through motor function and cognitive testing, the dosage was increased to 25 mg/kg b.w./day for the 23rd and 24th day [25, 26]. Following the second injection at this dose, we observed one mortality and, because of this, decreased the dose to 20 mg/kg b.w./day for injections from the 25th to the 30th day [27]. PNB-0408 was dissolved in DMSO and administered at a dose of 0.5 mg/kg b.w./day through i.p. route from the first day to the 30th on alternate days of the treatment protocol [9]. It was then administered at the same dose daily from the 31st to the 35th day [17, 22]. The dosage of PNB-0408 was selected based on the maximal and most efficacious intraperitoneal dosage used by other groups [17, 22]. PNB-0408 was given one hour before 3-NP treatment on days when rats were subject to both injections. Each week, animals were assessed for various physical, behavioral, and cognitive parameters described below in the testing protocol. On day 38, five rats from each group were euthanized, and brain samples were collected for histopathological analysis. All rats were decapitated under deep anesthesia using ketamine hydrochloride (50 mg/kg, i.p.) and xylazine (10 mg/kg, i.m.).
Measurement of body weights
Animals were weighed every third day throughout the treatment protocol to ensure accurate drug administration. Body weights were also explicitly recorded on the 8th and the 36th day of the study to calculate the percent change in body weight.
Behavioral testing
The animals were moved to testing rooms within the vivarium, where they were given at least 30 min to acclimate before the start of the behavioral testing.
Morris water maze
The Morris Water Maze was used to evaluate cognitive changes in the animals, specifically spatial learning and memory, as the test’s success depends on normal hippocampal function. The water maze consisted of a black circular tank filled to a depth of 26 cm with 26–28°C water. A black circular platform (diameter: 12 cm; height: 24 cm) was placed 30 cm from the wall and submerged 2 cm below the water surface. The maze was operationally sectioned into four equal quadrants: NW, NE, SW, and SE. For each rat, the platform’s location was randomly assigned to one of the quadrants and remained fixed throughout training and testing. Entry points were at the quadrant corners (i.e., N, S, E, and W) and were pseudo-randomly assigned such that each trial began at a different entry point than the preceding trial. Trials were conducted on days 6, 13, 20, 27, and 34, with four trials per day. Before the first trial on day six, there were four training days. On the first training day, the animals were placed on the platform for 30 s before the first trial. Training and testing trials commenced with the placement of the rat facing the wall of the maze at one of the assigned entry points. The rat was allowed a maximum of 120 s to locate the platform. Once the animal located the platform, it was permitted a 30-s rest period on the platform. The time it took for the animal to locate the platform was recorded. If the rat did not find the platform within the time limit, the rat was placed on the platform for the 30-s rest period. The next trial began immediately after the rest period concluded. After each daily set of trials, the animal was towel-dried, placed under a heating lamp for five minutes, and then returned to its home cage [17, 29].
Beam walking test
The narrow beam walking test was used to evaluate the motor coordination of the rats. A wooden beam with 2.3 cm×120 cm measurements was elevated to a height of 50 cm from the ground. To prevent injury in the case of a fall, the ground was padded with a foam cushion. During the test, the animals were placed at one end of the beam and allowed to cross to the other, where a platform and black box served as an enticing safety destination. If the animals could not cross the beam or fell from the beam, the trial ended. The time to cross the beam and the distance covered by the animal was measured. The test was performed on days 7, 14, 21, 28, and 35 with four trials per day. Between each trial, the rat was allowed a 70-second rest period in the black box on the destination platform. Rats were trained on the beam for three days before the first test on day 7 [30]. Rats were excluded from analysis if they displayed abnormal behavior and did not cooperate with the test as evidenced by a refusal to cross the beam (3 rats).
Hanging wire test
The hanging wire test was used to indirectly assess motor function through grip strength measurements. Animals were allowed to hang by their forelimbs from a steel wire with a diameter of 2 mm and a length of 80 cm elevated to a height of a 50 cm from the ground, padded with a foam cushion. The latency to the grip loss was measured, and the cutoff time was kept at 90 s. Animals performed three trials on each testing day, and the greatest latency to fall time was recorded. The test was performed on day 7, 14, 21, 28, and 35 [31]. Rats were excluded from analysis if they displayed abnormal behavior and did not cooperate with the test as evidenced by a refusal to hang on the wire with forelimbs (2 rats).
Histopathological analysis
Brain tissue was extracted and analyzed from five animals of each group to further characterize the neuroprotective properties of PNB-0408 against 3-NP induced neurodegeneration. The animals were anesthetized using ketamine hydrochloride (50 mg/kg, i.p), xylazine (10 mg/kg, i.m), and decapitated. Brain samples were extracted immediately and placed in O.C.T. medium within a cryostat block. The block was flash-frozen in 100% ethanol at –95°C for 5 min and then stored in a –80°C freezer. The samples were then sliced coronally into five μm thick slices with the help of a cryostat (Thermo Scientific HM 525NX). The slices were stained with hematoxylin and eosin (H & E) to observe the striatum and hippocampus (CA1 and CA3). The sections were observed with a light microscope [23]. Results were quantitated by categorization into the one of the following three groups: major histopathologic findings, minor histopathologic findings, or normal histologic findings. Major histopathologic findings are defined by marked deviations from normal morphologic and cellular presentation, including observable lesioning, evident necrosis, inflammatory response, and widespread cell death within the lesioned brain tissue. Minor histopathologic findings are defined by any deviation from normal morphologic and cellular presentation without observable lesioning, including minute foci of necrosis and any evidence of cell death or damage. Normal histologic findings are defined by expected morphologic and cellular presentation for the given brain region, including normal vasculature, no evident necrosis or inflammatory response, and no evidence of cell death or damage.
Testing protocol
For each behavioral test, rats were moved to testing rooms within the vivarium where they were allowed a minimum of 20 min to acclimate to the new environment before each test began. All behavioral and histological testing was performed and analyzed while blind to the treatment group.
Statistical analysis
Figures were created and data were analyzed using Prism (GraphPad Software Inc., San Diego, CA). All values are expressed as the mean±S.D., except for the data points in the line graphs seen in Figs. 3, 4A, and 5A, where points are plotted as the mean±S.E.M. for graph clarity, and Fig. 2, where percent change is reported as mean with 95% confidence interval. Data from each test were evaluated using one-way ANOVA followed by Tukey’s multiple comparisons, except for an analysis of intragroup performance in the Morris Water Maze where a paired t-test was used as noted in the results section. Additionally, simple linear regression analysis was performed to visualize and compare groups’ change in latencies to find the platform in the Morris Water Maze over the course of the treatment schedule (Fig. 3). The slope of the regression line was used as a measure of each groups ability to learn, with a negative slope representing successful abilities to learn and store spatial memories. Each slope is presented as the 95% confidence interval (Fig. 3).
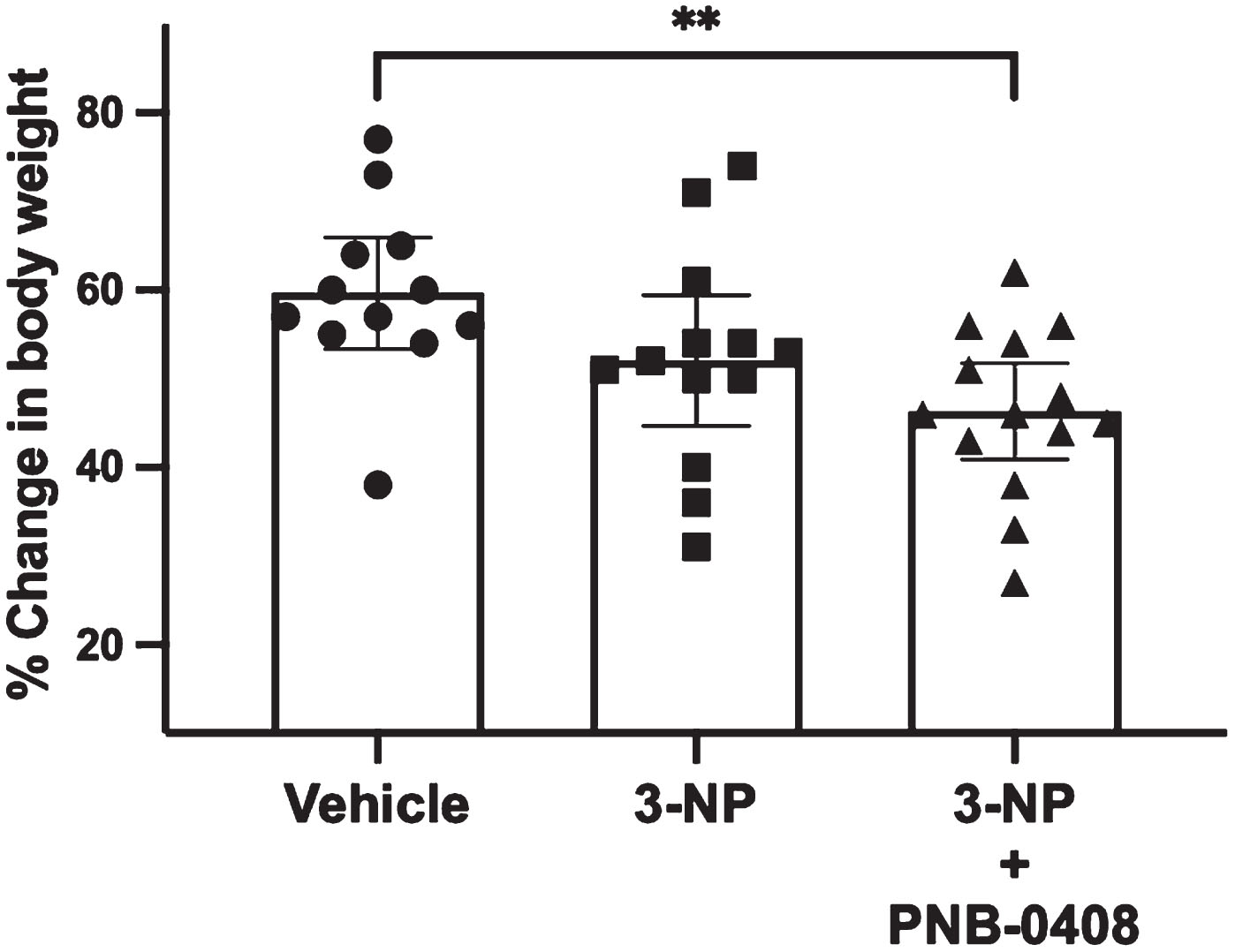
Effect of 3-NP exposure and PNB-0408 treatment on change in body weight. Each animal’s percent change in weight from day eight to day 36 was calculated. Results are presented as a scatter dot plot with each bar representing the group mean with 95% CI (N = 12–14 rats/group). P values were obtained from one-way ANOVA followed by Tukey’s test comparing all three groups (vehicle vs. 3-NP, p = 0.1857; vehicle vs. 3-NP + PNB-0408, p = 0.0078; 3-NP vs. 3-NP + PNB-0408, p = 0.3486).
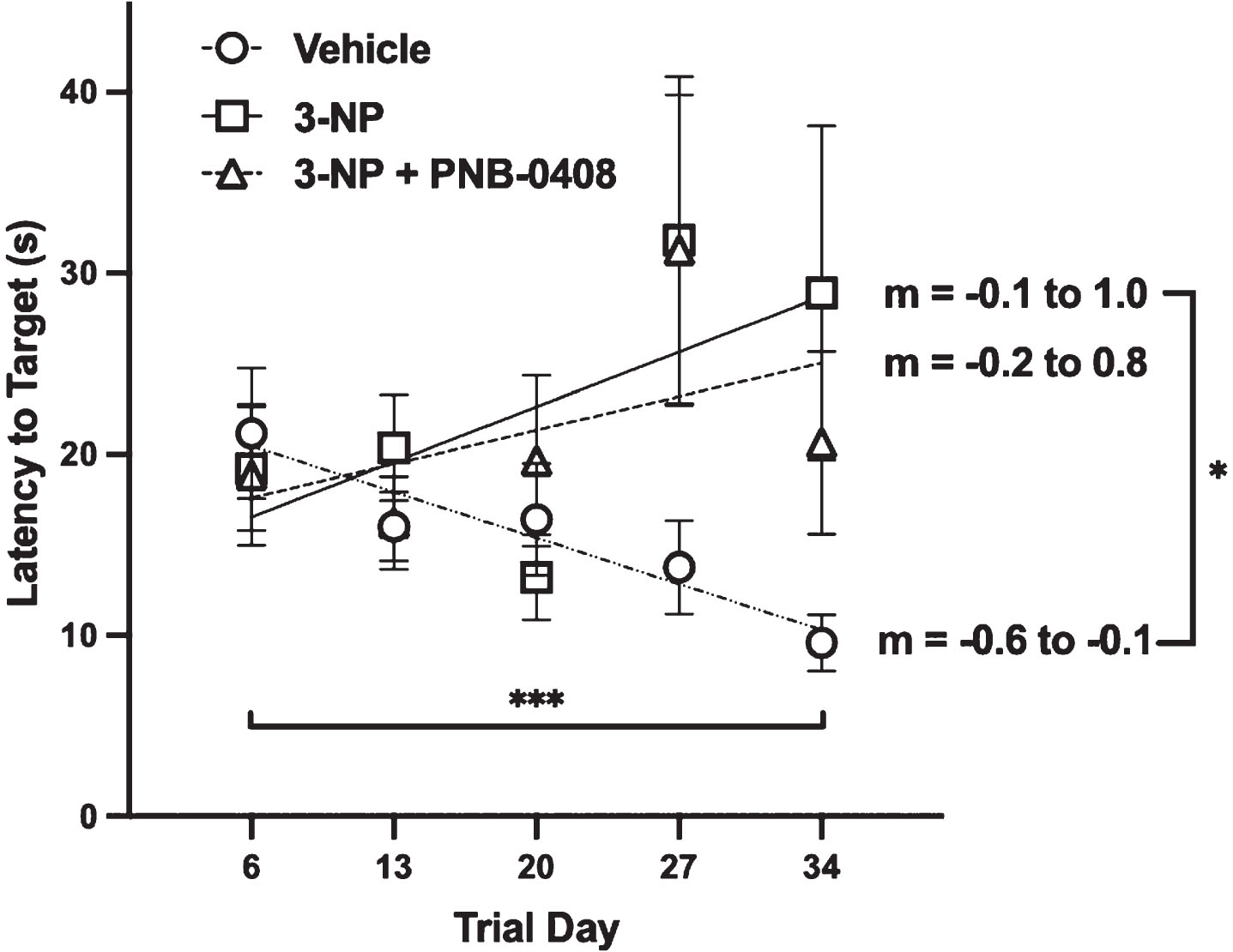
Effect of 3-NP exposure and PNB-0408 treatment on MWM performance. Spatial long-term memory and cognitive function were analyzed using the MWM. Group latencies to find the submerged platform are presented as mean±S.E.M. (N = 12–14 rats/group). Regression lines are plotted along with slopes listed (m = 95% CI) as a measure of each group’s ability to learn over the course of the treatment schedule. Linear regression analysis with slope comparison using one-way ANOVA with Tukey’s multiple comparisons test revealed that the slope (m) of the vehicle group was significantly different from the 3-NP group, *p = 0.0423, but not the 3-NP + PNB-0408 group, p = 0.1351. A paired t-test comparing each group’s performance from trials on day 6 to trials on day 34 was employed as another measure of cognitive changes within each group. The vehicle group decreased their latency to the platform, ***p = 0.0006, while the 3-NP and 3-NP + PNB-0408 group latencies increased slightly from start to finish (p = 0.4123; p = 0.7941, respectively).
RESULTS
Weight measurements
In the present study, body weight was measured and reported as the percent change in weight between the 8th and 36th day of the treatment schedule to evaluate 3-NP’s effect on weight gain and assess if PNB-0408 could ameliorate any changes. As seen in Fig. 2, while all groups saw an increase in body weight, the 3-NP and 3-NP + PNB-0408-treated groups gained less weight than the control group (mean difference = –7.59%, p = 0.1857; mean difference = –13.31%, p = 0.0078, respectively). Although the PNB-0408-treated group gained less weight than the 3-NP group, the difference was minimal and not significant (mean difference = –5.72%, p = 0.3486).
Morris Water Maze
As shown in Fig. 3, we observed that the vehicle group significantly decreased their latency to find the swim platform from the 6th day to the 34th day (Δlatency = –11.59 s, p = 0.0006). The latencies to find the platform in the 3-NP group slightly increased, although with considerable variability, from the first to the last trial day (Δlatency = 9.71 s, p = 0.4123). The 3-NP + PNB-0408 group also saw a slight increase in latency with considerable variability in finding the platform from start to finish (Δlatency = 1.78 s, p = 0.7941). P values were calculated using a paired t-test comparing intragroup performance on the 6th day to the 34th day. After simple linear regression analysis, we then compared the slope for the 3-NP group and the 3-NP + PNB-0408 group to that of the vehicle group (difference in slope = 0.80, p = 0.0432; difference in slope = 0.63, p = 0.1351, respectively). Additionally, we compared the regression slopes of the 3-NP group to that of the 3-NP + PNB-0408-treated group (difference in slope = 0.17, p = 0.8557).
Beam walking test
The present study found that the time to cross the beam in the 3-NP and 3-NP + PNB-0408 groups was significantly increased on day 28 and day 36 of the treatment protocol compared to a decrease in time to cross the beam seen in the vehicle group (Fig. 4B, C). We compared the average time to cross the beam of the three groups on day 28 (vehicle vs. 3-NP mean difference = –14.93 s, p = 0.0140; vehicle vs. 3-NP + PNB-0408 mean difference=–17.53 s, p = 0.0030; 3-NP vs. 3-NP + PNB-0408 mean difference = –2.60 s, p = 0.8530) and day 35 (vehicle vs. 3-NP mean difference = –3.92 s, p = 0.0004; vehicle vs. 3-NP + PNB-0408 mean difference = –3.35 s, p = 0.0023; 3-NP vs. 3-NP + PNB-0408 mean difference = 0.57 s, p = 0.8085). We also observed that the distance covered on the beam in the 3-NP and 3-NP + PNB-0408 groups was significantly decreased on the 28th day compared to the vehicle group, where every rat was able to cover the entire 120 cm beam distance (Fig. 4D, E). We compared each group’s average distance covered on the beam (vehicle vs. 3-NP mean difference = 37.50 cm, p = 0.0446; vehicle vs. 3-NP + PNB-0408 mean difference = 51.61 cm, p = 0.0036; 3-NP vs. 3-NP + PNB-0408 mean difference = 14.11 cm, p = 0.5969). On the other four trial days, most rats were able to cover the total 120 cm beam distance (Fig. 4D, F). Three rats, one from the 3-NP group and two from the 3-NP + PNB-0408 group, were excluded from the analysis of the final trial day due to anxious behavior and unwillingness to cross the beam.
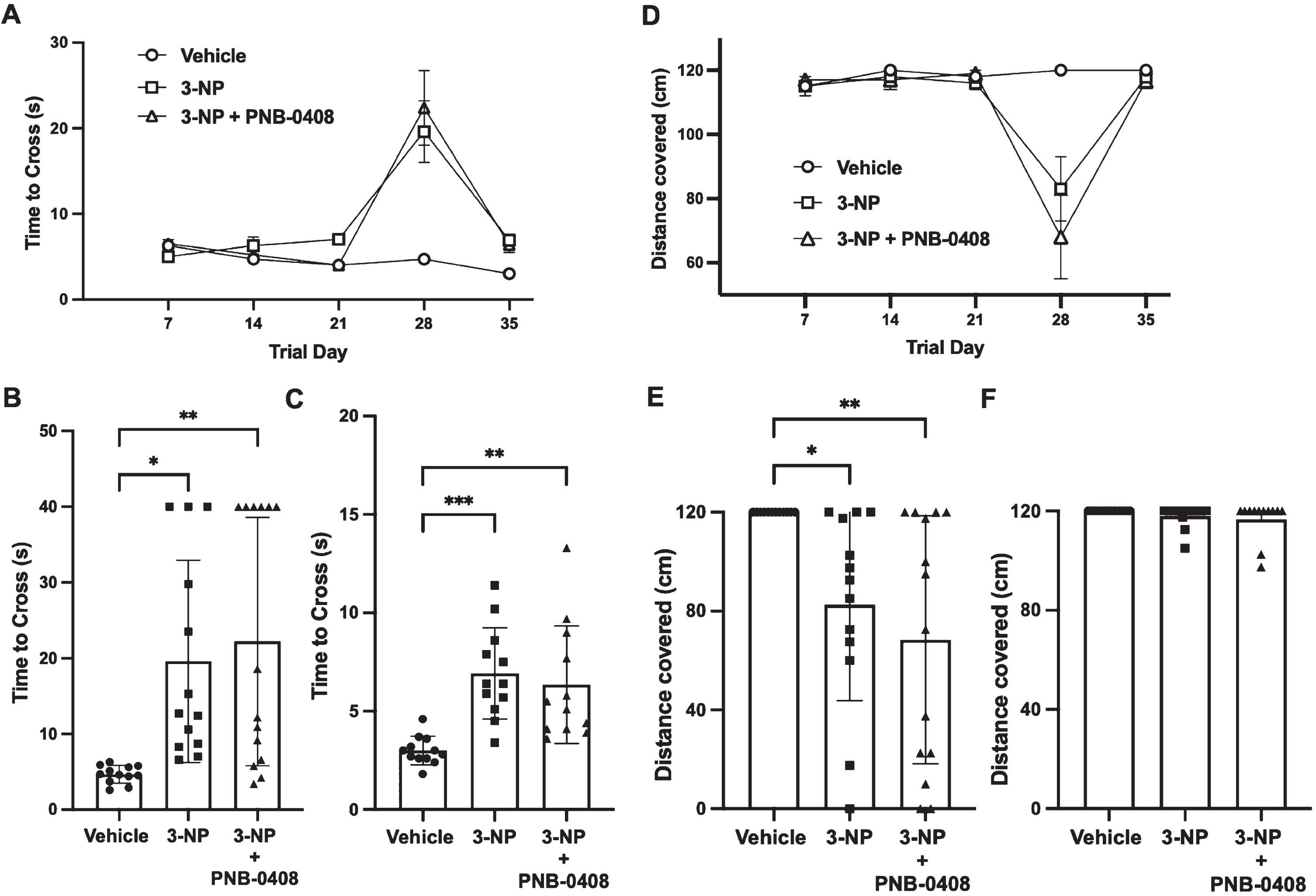
Effect of 3-NP exposure and PNB-0408 treatment on beam walking test performance. Motor coordination and function were analyzed by examining each rat’s ability to cross a narrow beam, measured by time (A, B, C) and distance (D, E, F). A) Group times to cross the beam are presented as mean±S.E.M. for each of the five trial days (N = 12–14 rats/group). Scatter dot plots reveal the time elapsed to cross the beam on days 28 (B) and 35 (C), where each bar represents the mean±S.D. (N = 12–14 rats/group). P values were obtained from one-way ANOVA followed by Tukey’s test comparing all three groups (B, *p = 0.0140, **p = 0.0030, p = 0.8530; C, ***p = 0.0004, **p = 0.0023, p = 0.8085). D) Group distances covered on the 120 cm beam are presented as mean±S.E.M. for each of the five trial days (N = 12–14 rats/group). Scatter dot plots reveal the distance covered on days 28 (E) and 35 (F), where each bar represents the mean±S.D. (N = 12–14 rats/group). P values were obtained using the same method as stated above (E, *p = 0.0446, **p = 0.0036, p = 0.5969).
Hanging wire test
The current study found that the time to fall from the wire in the 3-NP and 3-NP + PNB-0408 groups was decreased on the 28th and 35th day of the treatment protocol (Fig. 5). The vehicle group performed similarly throughout the experiment with >80% of rats hanging onto the wire for the maximum 90 s allowed on each trial day. We compared the average time to fall from the wire in the three groups on day 28 (vehicle vs. 3-NP mean difference = 32.38 s, p = 0.0140; vehicle vs. 3-NP + PNB-0408 mean difference = 35.79 s, p = 0.0054; 3-NP vs. 3-NP + PNB-0408 mean difference = 3.40 s, p = 0.9374) and day 35 (vehicle vs. 3-NP mean difference = 27.50 s, p = 0.0619; vehicle vs. 3-NP + PNB-0408 mean difference = 30.97 s, p = 0.0023; 3-NP vs. 3-NP + PNB-0408 mean difference = 3.47, p = 0.9401) (Fig. 5B, C).
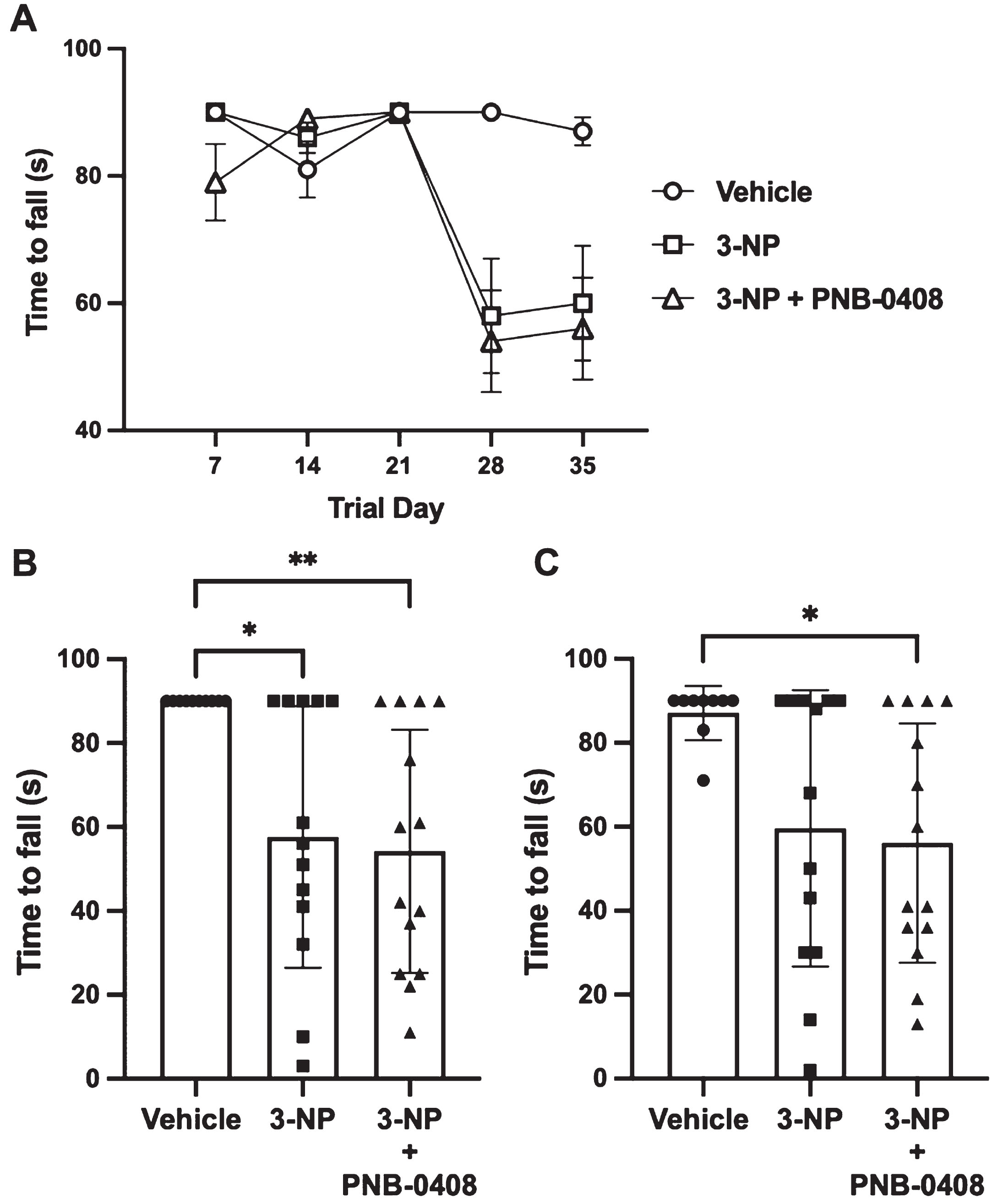
Effect of 3-NP exposure and PNB-0408 treatment on hanging wire test performance. Grip strength was measured using the hanging wire test to assess the motor function of each rat. A) Group latencies to fall are presented as mean±S.E.M. for each of the five trial days (N = 12–14 rats/group). Scatter dot plots reveal times to fall on days 28 (B) and 35 (C), where each bar represents the mean±S.D. (N = 12–14 rats/group). P values were obtained from one-way ANOVA followed by Tukey’s test comparing all three groups (B, *p = 0.0140, **p = 0.0054, p = 0.9374; C, vehicle vs. 3-NP, p = 0.0619, *p = 0.0023, 3NP vs. 3-NP + PNB-0408, p = 0.9401).
Histopathological analysis
We observed large variability in striatal and hippocampal damage in rats exposed to 3-NP (Supplementary Figures 1–3). Of the five rats analyzed in the 3-NP group, one displayed severe bilateral lesioning, necrosis, perivascular edema, and cell shrinkage in the striatum, along with variable localization of necrosis and complete absence of viable pyramidal neurons in both the CA1 and CA3 regions of the hippocampus. One rat of the five analyzed in the 3-NP + PNB-0408 group, displayed a bilateral minute focus of striatal necrosis with no visible hippocampal damage. Another rat in this group presented with variably selective pyramidal cell loss in both the CA1 and CA3 regions while the striatum appeared otherwise normal. The remaining animals in each group showed little to no evidence of structural or cellular damage observable with H & E staining. All five rats in the vehicle group displayed normal histology for each brain region (Table 1).
Histologic Results
N = 5 for each group. Hematoxylin and eosin staining of the striatum and CA1/CA3 regions of the hippocampus was performed on five rats from each group. Major histopathologic findings are defined by marked deviations from normal morphologic and cellular presentation, including observable lesioning, evident necrosis, inflammatory response, and widespread cell death within the lesioned brain tissue. Minor histopathologic findings are defined by any deviation from normal morphologic and cellular presentation without observable lesioning, including minute foci of necrosis and any evidence of cell death or damage. Normal histologic findings are defined by expected morphologic and cellular presentation for the given brain region, including normal vasculature, no evident necrosis or inflammatory response, and no evidence of cell death or damage.
DISCUSSION
The present study aimed to investigate the potential neuroprotective effects of the angiotensin IV analog, PNB-0408, on rats with 3-NP-induced neurotoxicity. 3-NP is a toxic chemical that disrupts brain metabolism, damages hypothalamic neurons, and causes bilateral striatal injuries, which can lead to decreased appetite [32–34]. In a study characterizing differences in R6/2 transgenic mice, a Huntington’s disease animal model, and wild-type mice, body weight decreased significantly at ten weeks of age in the R6/2 transgenic mice [35]. Similar studies using 3-NP in rat models demonstrated that 3-NP exposure inhibited weight gain [23, 24]. From our observations, rats treated with 3-NP did not gain as much weight as the control group; however, this difference was not statistically significant. The group administered 3-NP and PNB-0408 gained the least weight during the experiment, suggesting that PNB-0408 was unsuccessful in preventing alterations in body weight caused by 3-NP neurotoxicity.
Cognitive impairment is a common symptom of HD and is thought to be linked to reduced acetylcholine levels and increased acetylcholinesterase activity in the brain [36]. Similarly, systemic 3-NP administration has been shown to increase acetylcholinesterase activity and cause CA1 and CA3 neuronal damage in the hippocampus, resulting in cognitive impairment representative of HD [23, 28]. A previous study found that rats exposed to 3-NP displayed cognitive impairment as they required a significantly longer time to find the escape platform in the Morris Water Maze test compared to controls, demonstrating deficits in spatial learning and memory [24]. In accordance with this previous literature, we observed that rats administered 3-NP did not decrease their latency to find the platform in the Morris Water Maze Test throughout the treatment protocol, which exemplifies impaired memory and learning abilities compared to the control group, whose times consistently decreased with each testing day (Fig. 3). In a study exploring PNB-0408’s potential to treat AD, researchers used the Morris Water Maze and found that it significantly improved memory and learning in rats with scopolamine-induced amnesia by shortening swim latency to find the platform; however, in the present study, PNB-0408, at the same dose, displayed no evident signs of attenuating cognitive impairment caused by 3-NP neurotoxicity [9, 17]. On average, rats administered PNB-0408 in the presence of 3-NP and rats solely administered 3-NP performed worse in the Morris Water Test than the control group.
Chorea is a characteristic motor feature of HD [37]. Systemic administration of 3-NP reproduces late-phase symptoms of HD, like hypo and hyperkinetic motor impairments [38]. Prior studies concluded that rats exposed to 3-NP experience significant decreases in motor coordination, balance, and grip strength as evidenced by increased times to cross a narrow beam, decreased total distance covered on the beam, and decreased latency to fall from a hanging wire [23, 24]. In our study, the administration of 3-NP produced significant impairment in motor function, which was visualized by the narrow beam walking and hang wire tests. It is predicted that striatal lesions and degeneration of MSNs in the striatum could be a possible reason for motor impairment in the 3-NP-induced neurotoxic rats [39]. The group exposed to 3-NP and treated with PNB-0408 performed similarly to the group solely exposed to 3-NP on the balance beam, suggesting that PNB-0408 was unable to ameliorate motor dysfunction and balance irregularities caused by 3-NP. Additionally, while previous work discovered PNB-0408’s ability to attenuate impaired grip strength on the hanging wire test in substantia nigra lesioned rat models of PD, the present study found that PNB-0408 at the same dose did not display similar benefits in 3-NP rat models of HD [9]. Rats exposed to 3-NP and treated with PNB-0408 performed similarly to the 3-NP group.
It has been reported that individual animals differ in their striatal susceptibility and behavioral response to systemic 3-NP toxicity [38, 41]. Nishino et al. (1995) found that two-thirds of their rats showed no detectable striatal lesions or changes in behavior. In our study, we also found limitations to the use of 3-NP as a method to induce neurodegenerative HD-like symptoms reliably and consistently in Wistar rats. Of the rats subject to 3-NP administration, roughly 35% did not display any observable motor function or cognitive impairment in behavioral testing. The rats that did show signs of impairment varied considerably from each other. One rat died following the second injection of 3-NP at 25 mg/kg. Three others showed severe impairment with hind limb immobility, decreased body weight, and severely decreased ability to hang on the wire or walk on the balance beam. Others showed mild signs of impairment, as measured by their performance in behavioral testing.
Histopathological analysis of the striatum and hippocampus revealed marked variability in neuronal and structural damage as well. Previous literature showed chronic administration of 3-NP induced bilateral striatal lesioning and pyramidal neuronal cell loss in the CA1 and CA3 regions of the hippocampus [42, 43]. In our study, similar to Nishino et al. (1995), we did not observe consistent lesioning. Of the five rats that were examined from the 3-NP group, we found one rat that displayed evidence of severe lesioning and necrosis while the others in that group appeared to have otherwise normal histology. Other groups, including Alexi et al. (1998), have also reported histologically inconsistent striatal lesioning in rats. Comparable results were observed in the group administered 3-NP and PNB-0408, where only about 65% of the rats appeared vulnerable and displayed some form of behavioral and cognitive impairment and the others remained resilient. Histopathological analysis revealed two rats in this group, of the five analyzed, that had signs of minor striatal or hippocampal damage while the remaining animals presented with normal histological features.
It is well documented that PNB-0408 and angiotensin IV analogs possess neuroprotective and neurorestorative properties shown to attenuate neurodegenerative symptoms seen in disorders such as AD and PD [9]. Here, we attempted to test PNB-0408’s neuroprotective effects in an HD animal model. From our observations, the administration of PNB-0408 did not protect against motor deficits, weight changes, or cognitive impairment induced by 3-NP neurotoxicity. While we used the highest dose tested by other group’s dose-response studies, it is possible that it was not enough to overcome the neurotoxicity of 3-NP. Additionally, since 3-NP has no influence on the huntingtin gene or protein and only mimics HD-like symptoms, it is conceivable that an alternative animal model, specifically a transgenic HD model, may have yielded different results. Given the genetic basis of HD, it is logical that a genetic model may recapitulate the disease more accurately; however, a toxic model was used here for a number of practical reasons (cost, available animal care facility, and rat model). Despite the limitations, we felt it was an appropriate model to support the goals of this proof-of-concept study which sought to analyze PNB-0408’s ability to prevent HD-like symptoms, representing its neuroprotective activity, or inactivity, in the same brain regions and cell types affected in HD. It is also possible that the timing of injections influenced the observed outcomes. On days when both PNB-0408 and 3-NP were administered, PNB-0408 was given 1 h before 3-NP. This scheduling aimed to focus more specifically on PNB-0408’s protective properties. However, the small molecule may have more potent restorative effects that this injection schedule could have concealed. Injecting PNB-0408 after 3-NP may have revealed different outcomes. Furthermore, PNB-0408 and 3-NP have never been used simultaneously in an animal model before, and although unlikely, it remains unknown if any interactions exist between the two compounds that could have confounded the study results. Lastly, due to budget constraints, molecular studies with the potential capability to clarify the underlying mechanisms of the observed results in this animal model were not performed and is therefore a study limitation. Future studies could focus on testing PNB-0408 in an HD transgenic animal model to avoid the variable vulnerability to 3-NP impairment our group observed or any mediating interactions between 3-NP and PNB-0408 that may exist.
While PNB-0408’s antidementia, neuroprotective, and procognitive potential is still relevant to neurodegenerative disorders, our study suggests that it may not be an efficacious treatment strategy for preventing 3-NP-induced HD-like symptoms in a preclinical model. We feel these data are relevant to the HD field and highlight the need for further research of this compound in alternate models that more closely recapitulate HD pathology and/or alternative approaches to managing this disorder.
Footnotes
ACKNOWLEDGMENTS
The authors are grateful to Dr. Alisha Epps for assisting with animal colony maintenance and behavioral testing, Dr. Grant Casady for statistical analysis guidance, and Dr. Marjorie Grafe for aiding in histopathological analysis.
FUNDING
This work was made possible through the financial support of the Huntington’s Disease Society of America, awarded to R. Wells, and the Whitworth University Biology Department, awarded to M. Sardinia.
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
DATA AVAILABILITY
The authors declare that all the animal group data supporting the findings of this study are contained within the paper. Individual animal data are available on request from the corresponding author.
