Abstract
BACKGROUND:
Deep sternal wound infections (DSWI) following cardiothoracic surgery represent a life quality endangering sequelae and may lead to sternal osteomyelitis. Radical debridement followed by Negative Pressure Wound Therapy (NPWT) may achieve infection control, provide angiogenesis, and improve respiratory function. When stable wound conditions have been established a sustainable plastic surgical flap reconstruction should be undertaken.
OBJECTIVE:
This study analyses a method to simplify defect coverage with a single Latissimus Dorsi Myocutaneous Flap (LDMF).
METHODS:
Preparation of 20 LDMF in ten fresh frozen cadavers was conducted. Surgical steps to increase pedicle length were evaluated. The common surgical preparation of LDMF was compared with additional transection of the Circumflex Scapular Artery (CSA).
RESULTS:
Alteration of the surgical preparation of LDMF by sacrificing the CSA may provide highly valuable well-vascularized muscle tissue above the sensitive area of the Xiphisternum. All defects could be completely reconstructed with a single LDMF. The gain in length of flap tissue in the inferior third of the sternum was 3.86±0.9 cm (range 2.2 to 8 cm).
CONCLUSIONS:
By sacrificing the CSA in harvesting the LDMF a promising gain in length, perfusion and volume may be achieved to cover big sternal defects with a single flap.
Keywords
Introduction
In contemporary cardiothoracic surgery sternal infections are still a common complication [1–5]. Despite minimally invasive procedures nowadays common for heart valve reconstruction or replacement respectively via transfemoral-or transthoracic approaches (TAVI), there are still reasons for an open technique as in Coronary Artery Bypass Graft (CABG) [6–8]. The moribund patient population suffers from strongly reduced general condition and malnutrition [4, 9]. Further risk factors include diabetes [10–13], coronary heart disease (12), age of patient (13), smoking habits, chronically obstructive lung disease (COPD) [14], cortisone therapy [12], emergency surgery [15], long theatre time, obesity [16], time of ischemia [12], hypothermia [17], wound infection by multi resistant bacteria and fungi [14, 18].
Reduced blood supply to the sternum [12, 19] as a result of single or bilateral harvest of the internal mammary artery (IMA) may cause delayed or incomplete wound healing [20, 21]. Furthermore, there is evidence that bilateral loss of IMA with accompanying diabetes mellitus are pathognomonic for a high risk of postoperative wound healing problems [21–23]. Resulting deep sternal defects, particularly in the lower third with persistent or chronic wound healing deficiency therefore still represent a paramount problem in plastic surgery [24]. Most literature agrees on the necessity of a meticulous debridement of the infected tissue followed by NPWT [25] to achieve neo angiogenesis and improvement of respiratory function [26–29]. This may provide infection control prior to an either local or regional flap coverage [30]. Although various surgical approaches have been published, no gold standard for a sustainable single tissue replacement has been established so far. The most common flaps are the Latissimus Dorsi Myocutaneous Flap (LDMF), M. Pectoralis Major Flap (PMF), Transverse or Vertical Rectus Abdominis Myocutaneous Flap (TRAM/VRAM) and the Omentum Majus FLAP (OM) (30).
LDMF and PMF may up to some extent be used as a common tool to cover superior and medial defects of the sternum [24, 31]. For inferior defects most authors suggest TRAM/VRAM or OM reconstructions [32–34]. If the defect compromises the entire sternal area, all of the previously named options may be insufficient to provide a simple, stable and durable reconstruction. These obstacles also include the LDMF, which utilizes the largest muscle of the human body. Striving for reliable reconstructive tools, combinations of the before mentioned flaps are considerable, but may result in higher peri- and postoperative risk [35]. With progress in microsurgical techniques free tissue transfer was introduced to accost defect reconstruction following DSWI [36, 37]. However, with reference to the age group, perioperative risk factors and comorbidities, free flaps should be reserved for certain cases due to longer theatre time and multi-stage strategies such as arterial-venous loops for instance [38].
Respecting all these aspects to achieve a secure and stable methodical way to reconstruct these demanding defects, the latter question arose.
May a solitary flap for reconstruction of the whole sternal region be sufficient?
I. Tansini described a partial LDMF on behalf of breast reconstruction and established the method in a series of 17 patients for the first time as early as 1906 [39]. The LDMF features the Thoracodorsal Artery (TDA) as its dominant blood supply and secondary segmental vessels entering the flap from its medial paravertebral aspect. It is categorized as a Type V regarding the Nahai/Mathes classification of muscle flaps [40].
However, the LDMF as a reconstructive tool was somewhat neglected and forgotten until the mid 1970ties when Neven Olivari was confronted with the problem of a valuable reconstruction of radiodermatitis of the cranial thorax. He rediscovered the LDMF and started popularizing it again [41–43]. In 1978 Marco Godina transplanted the first free LDMF onto the lower leg [44]. Since then, the LDMF has been used as a versatile and reliable tool in local, regional or even microsurgical reconstructions due to its consistent anatomy and reliable blood supply.
The aim of the following study was to thoroughly examine whether the lower third of the sternal region may be sufficiently reconstructed with a single flap. In order to verify our clinical experience, we evaluated classic and altered preparation of LDMF. Therefore, ten fresh frozen human cadavers were object of our study.
Methods
The study was conducted at the Anatomical Institute of the University of Halle/Germany and executed in accordance with the Helsinki Declaration. All corpses included entered the Departments of Anatomy through a donation program. Written informed consent was obtained during lifetime. The study was approved by the Institutional Review Board Committee of the University of Halle.
At the Anatomical Institute a total of ten fresh frozen human cadavers were dissected.
Surgical technique
We planned the reconstruction of the extended sternal defects with LDMF achieving full coverage in the vertical and horizontal plane. With the corpses positioned supine, the first part of the study consisted of creating sternal defects in a standardized fashion. Defects were generated as extensive as seen in complex clinical cases in vivo via resection of the entire adipose and cutaneous tissue overlying the sternum, including the xiphoid and manubrium as well as the adjoining sternocostal joints on both sides. The defects were measured in the vertical and horizontal plane in cm with a ruler (Fig. 1).
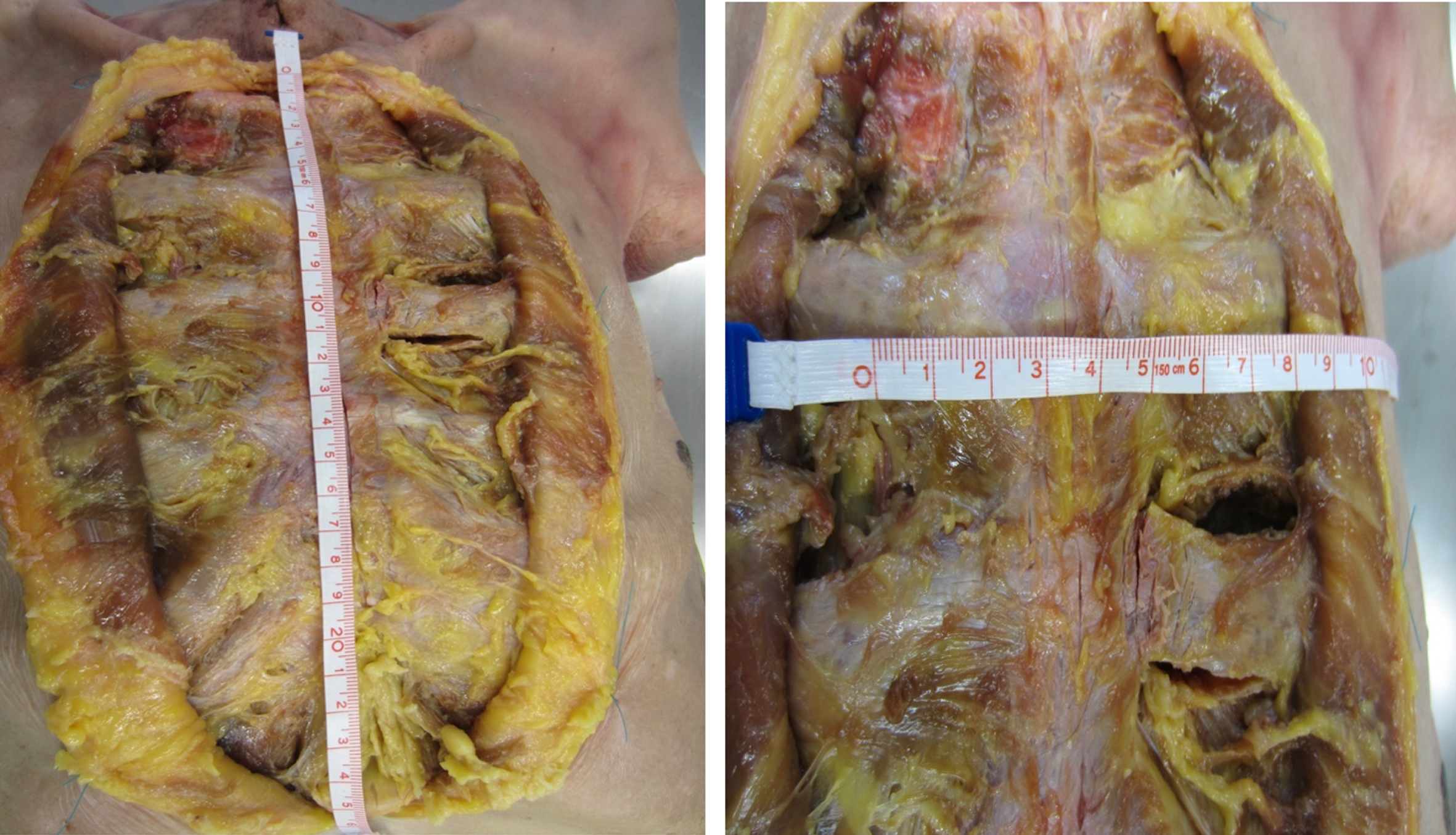
Horizontal and vertical dimension of sternal defect in cm.
The corpses were then repositioned and stabilized in a lateral decubitus position. The LDMF was harvested from the corresponding side using surgical 2.3 x-loupes according to the initial technique published by Neven Olivari in 1976 [41]. Scapular tip, iliac crest and the dorsal axillary line served as landmarks. The size of the skin island was designed with reference to the sternal defect. It is designed at the margin from the upper to the middle third of the LDMF at the axillary rim, running obliquely in a 45 degree angle dorsally and inferiorly towards the spine. In order to identify the frontal margin of the flap we made the incision along the pectoral rim and exposed the supplying vessel TDA. It enters the LDMF usually 2 cm in front and above the scapular tip. The preparation of the vascular pedicle was continued towards the axilla, approaching the CSA, the Subscapular and Axillary Artery (Fig. 2). The vascular junction/intersection of the CSA and the TDA is securely found 10 to 12 cm superior to the inferior angle of the scapula. The entire flap was then harvested and the tendon transected at its humeral insertion. The vessels were marked with a loop. The LDMF was now tunneled underneath the pectoralis major muscle (PM) and the tendon sutured to the origin of the PM to prevent kinking of the thoracodorsal vessels. With regards to a fully accomplished coverage of the sternal defect, the amount of muscle tissue overlapping the Xiphoid was then measured in cm with a ruler as shown in Fig. 3.
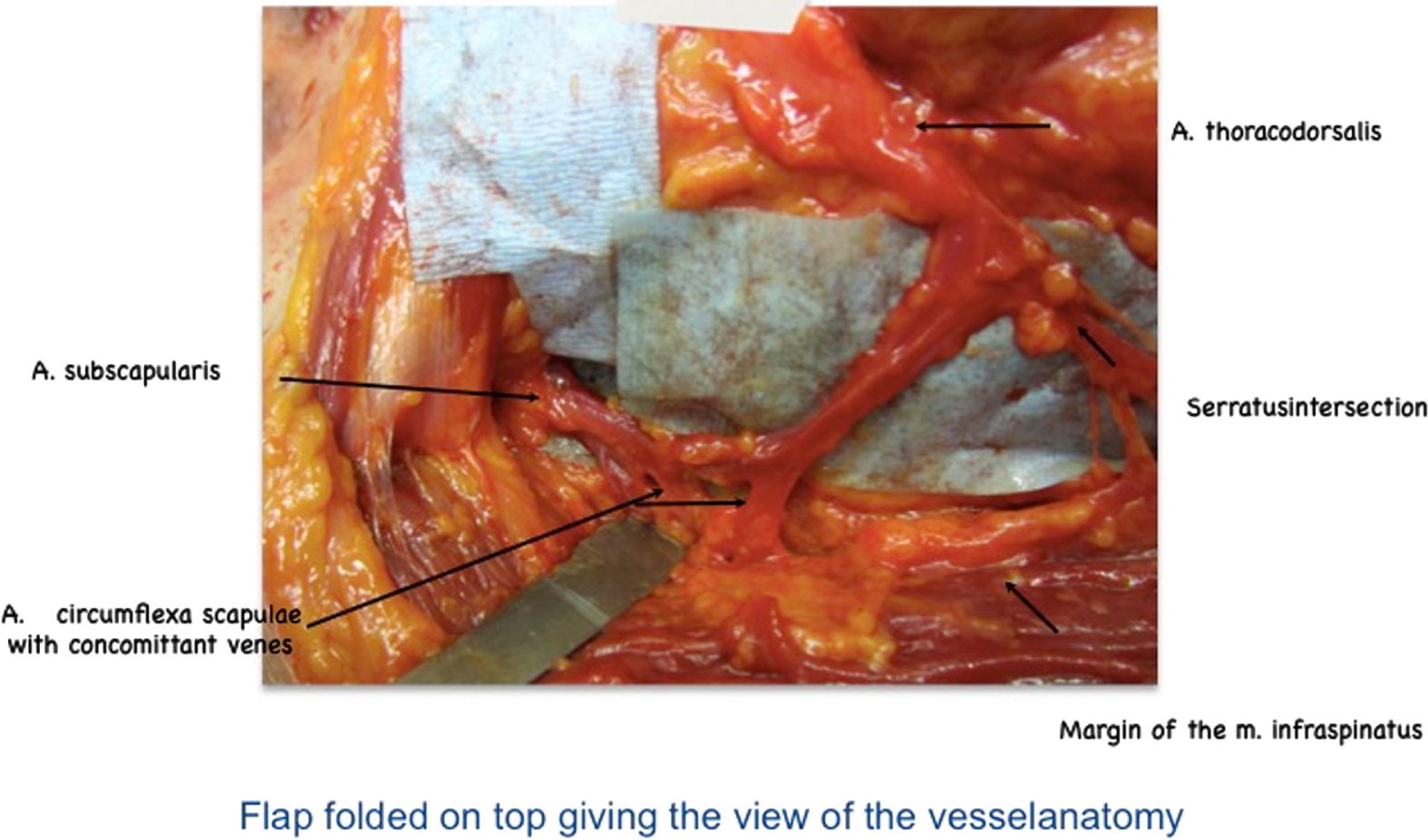
The CSA deriving from the A. subscapularis in relation to the vessel anatomy of the LDMF (representative picture of the corresponding anatomical study).
Vertical dimension of the reconstructed sternum before and after ligation of the CSA in cm (representative picture of the corresponding anatomical study).
Now the CSA and its concomitant veins were clipped and transected to transpose the LDMF more caudally. Measurements in accordance to the predefined reference points were obtained again and the resulting differences overlapping the Xiphisternum evaluated.
In addition, we measured the circumference of the LDMF, which can be geometrically considered as a triangle, determined by its side lengths cranially, inferiorly and pectorally.
Finally, the flap volume was measured in total and muscle tissue separately with the water displacement method. Both flap volumes as well as the circumference were set in correlation to gender, side, and range of motion of the LDMF.
The data collected focused on the gain of arc of rotation, the overlapping of muscle tissue inferiorly with regards to the side of harvest in cm before and after dissection of CSA and its concomitant veins. The data was statistically evaluated with two variables set in correlation with the Pearson test. The results were defined as significant with a p-value < 0.05. The results were furthermore shown as mean values (MV) and in standard deviation (SD). Statistical analyses were performed using SPSS (IBM Corp. Released 2016. IBM SPSS Statistics for Macintosh, Version 24.0. Armonk NY: IBM Corp.) and R (version 3.5.1; The R Foundation for Statistical Computing, Vienna, Austria).
Results
A total of 10 fresh frozen human cadavers (2 males, 8 females) were investigated. The mean age was 86.3 years, ranging from 74 to 93 years. Sternal defects were 8.5 cm x 19.8 cm (ranging from 6 to 12 cm horizontally, 18 to 21 cm vertically).
Circumference of the LDMF
The side lengths cranially, inferiorly, and thoracically were summoned with 85.5±5.9 cm in total (left 84.9 cm, right 86.1 cm) almost without any major side difference. A raise of circumference subsequently led to both an increase in muscular (p < 0.002) and total flap volume p < 0.011).
Volume of the MLDF
Figure 4 shows the muscular flap volume 198.8±67.3 ml (range 140 to 370 ml) and the total flap volume 333.8±117.3 ml (range 170 to 590 ml). Total flap volume of the left side was 329±114.3 ml (range 140 to 590 ml) and 349.5±109.2 ml (range 170 to 590 ml) of the right side. The muscular volume of the left side measured 195±66.9 ml (range 100 to 370 ml), of the right side 212.5±65.6 ml (range 140 to 370 ml). No significant differences between the side of harvest and the corresponding volumes could be stated with reference to the length benefit. Increase in flap circumference and adjusting rise in either complete volume (p < 0.011) and muscular volume alone (p < 0.002) proved evident and significant.
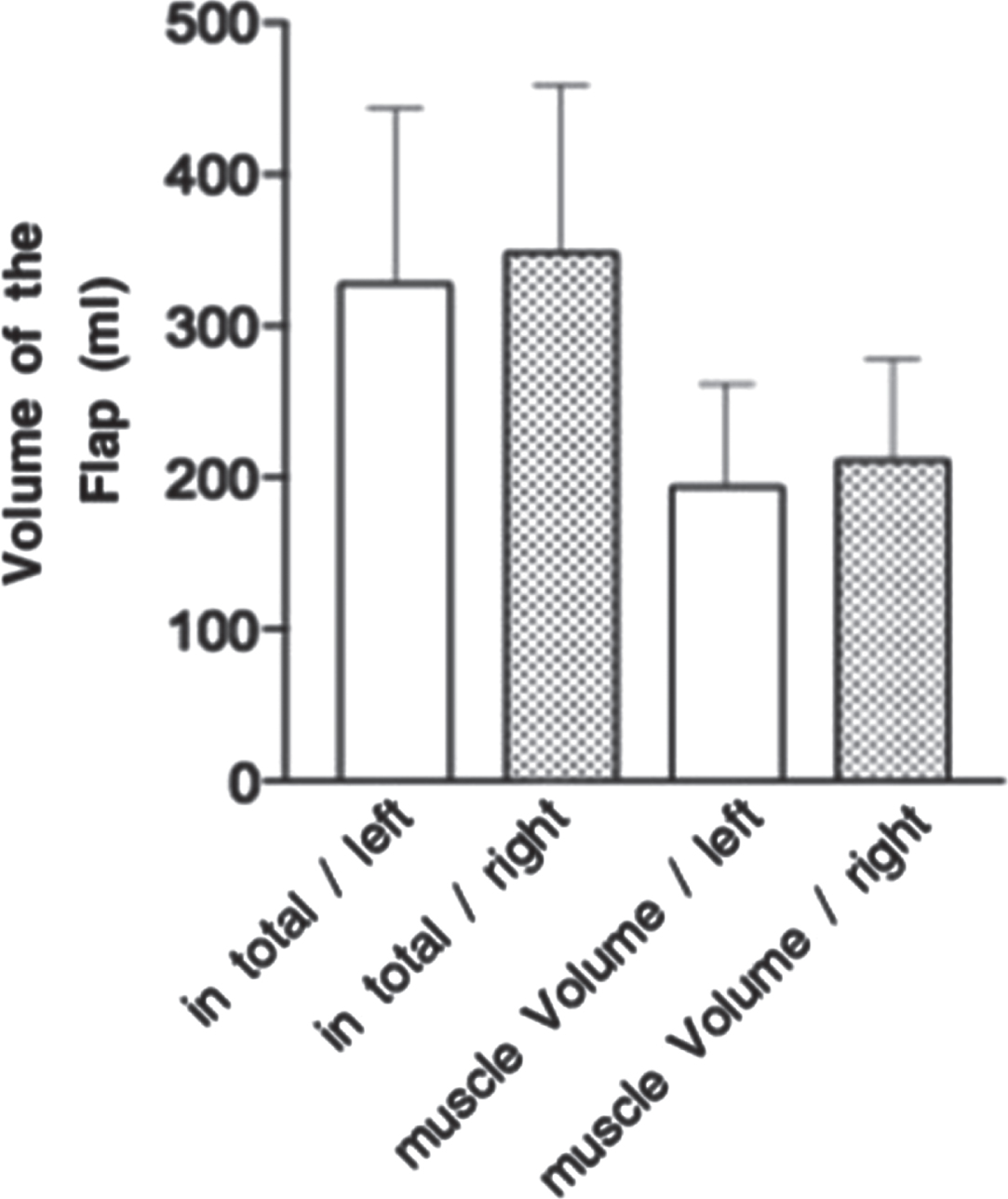
Muscular and total flap volume with reference to the side of harvest.
The vertical dimension of the LDMF showed a length of 22±1.4 cm (range 20 to 24.4 cm). All harvested flaps reached the inferior defect area of the exposed Xiphisternum. Inferior overlapping of muscle tissue with an average of 2.3 cm ranging from 0 to 5.3 cm could be recorded. After ligation of the CSA a covering flap length of 25.9±1.6 cm (range 22 to 28 cm) was measured. A length benefit considering the full coverage of the Sternum in the vertical plane of 3.86±0.8 cm (range, 2–8 cm) was achieved. The beneficial length gain in tissue overlapping the Xiphisternum could therefore been stated with 3.86 cm and is illustrated in Fig. 5. The 95% confidence-interval was (3.3 cm: 4.6 cm) with a p-value of<0.0001. A rise in both muscular volumes alone as well as in total flap volume resulted in an overlapping length gain inferiorly.
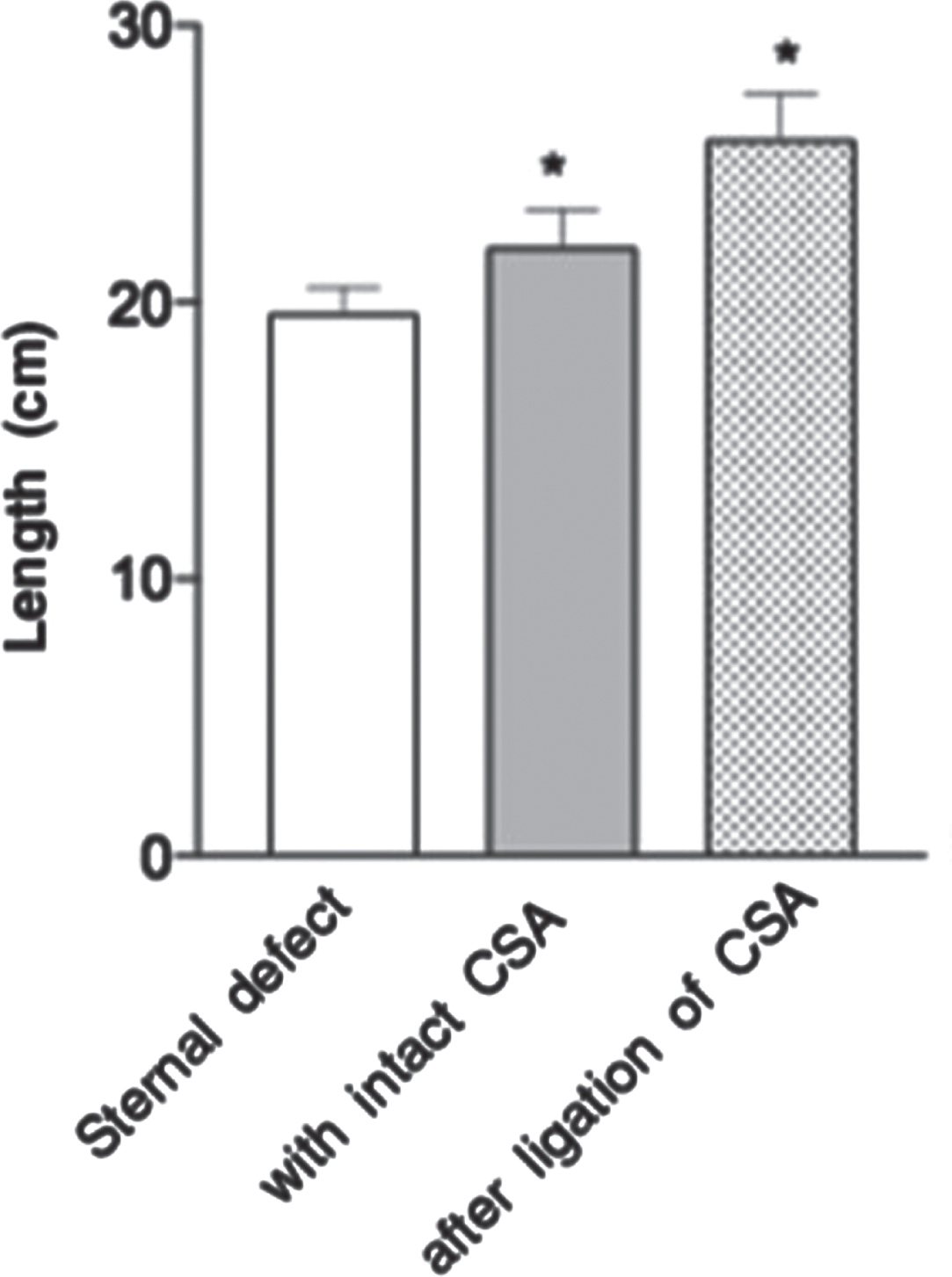
Vertical dimension of the sternal defect, the reconstructed dimension before and after the ligation of the CSA; MW+SD; * = statistically significant towards the vertical length of the sternal defect (p < 0,0001).
DSWI with or without sternal osteomyelitis following open cardiothoracic surgery still imposes a challenging situation. For the elderly patient it may constitute a life-threatening condition due to accompanying comorbidities [2–5]. The Guidelines on Myocardial Revascularization propose a skeletonized artery for an open thoracic procedure. This procedure implements on the one hand an increased risk of vessel damage, but on the other hand an improved blood supply to the heart, elongated bypass distance and therefore a long-term patient‘s benefit [47]. CABG in general with one or both sided harvested IMA [45] is responsible for an impaired blood supply to the sternum. This procedure in combination with the comorbidity Diabetes mellitus contributes furthermore to wound healing deficiency [16, 46], resulting in sternum osteomyelitis [19].
An ideal reconstruction of sternal defects following salvage of osteomyelitis should preferably provide enough flap volume, sufficient blood supply of the transferred tissue and a fast and stable recovery [2, 11]. Flap reconstruction should furthermore be subject to the requirements regarding the size of defect, the patient’s general health condition, theatre time and the surgeon’s skills. Multiple reconstructive algorithms have been established to shorten hospitalization [48, 49]. A single gold standard has not yet been defined [50]. The literature agrees upon the fact that sufficient coverage of complete sternal defects can only be achieved with a combination of the most versatile flaps. These include the LDMF and PMF for the upper two thirds of the sternum in combination with TRAM/VRAM [35, 51] or the OM with supporting skin graft [2, 52] for the lower two thirds [53]. In 2007 Davison described anatomical reasons being responsible for an insufficient wound healing after sternal reconstruction with a single flap. Due to the blood supply, there are certain restrictions for any of the working horse flaps [24, 35]. TRAM/VRAM for instance may be limited to the middle and lower third of the sternum due to the blood supply by the deep superior epigastric artery (DSEA) [34, 54]. PMF and LDMF are nourished proximally by the thoracoacromial artery (TA) and TDA respectively. This accounts for a reduced blood supply in the most distal parts due to the regional random pattern blood distribution, which once again may result in a poorer wound healing [22, 56]. Furthermore, the latter often may not even reach the bony xiphoid process reliably [22, 57].
Clemens et al. compared the outcomes of PMF versus right sided TRAM flap for this indication and experienced a flap morbidity for both flap reconstructions of 34% [35].
The PMF flap in particular showed wound dehiscences or breakdown in the lower part and seromas at the donor site [33]. TRAM/VRAM and OM flaps require a two cavity procedure and may be susceptible to an elevated postoperative complication rate [58]. The TRAM may show a skin breakdown at the donor site due to cutting off the blood supply from the overlying skin and may result in abdominal hernias [24, 30]. On the other hand, infection might spread from the abdomen to the thorax or vice versa [59]. Clemens therefore advocated a combination of both PMF and TRAM for defects including the whole of the sternum(35). Aesthetic limitations may be encountered furthermore by mesh graft scaring, which is mandatory following the OM flap [60].
By sacrificing the CSA and therefore enlarging the arc of rotation of the LDMF, a stable reconstruction with a single sustainable flap may be achieved. Our anatomical preparations demonstrate that the described pedicle modification provides an enduring and secure length and volume benefit especially in the inferior sternum even in extensive sternal defects.
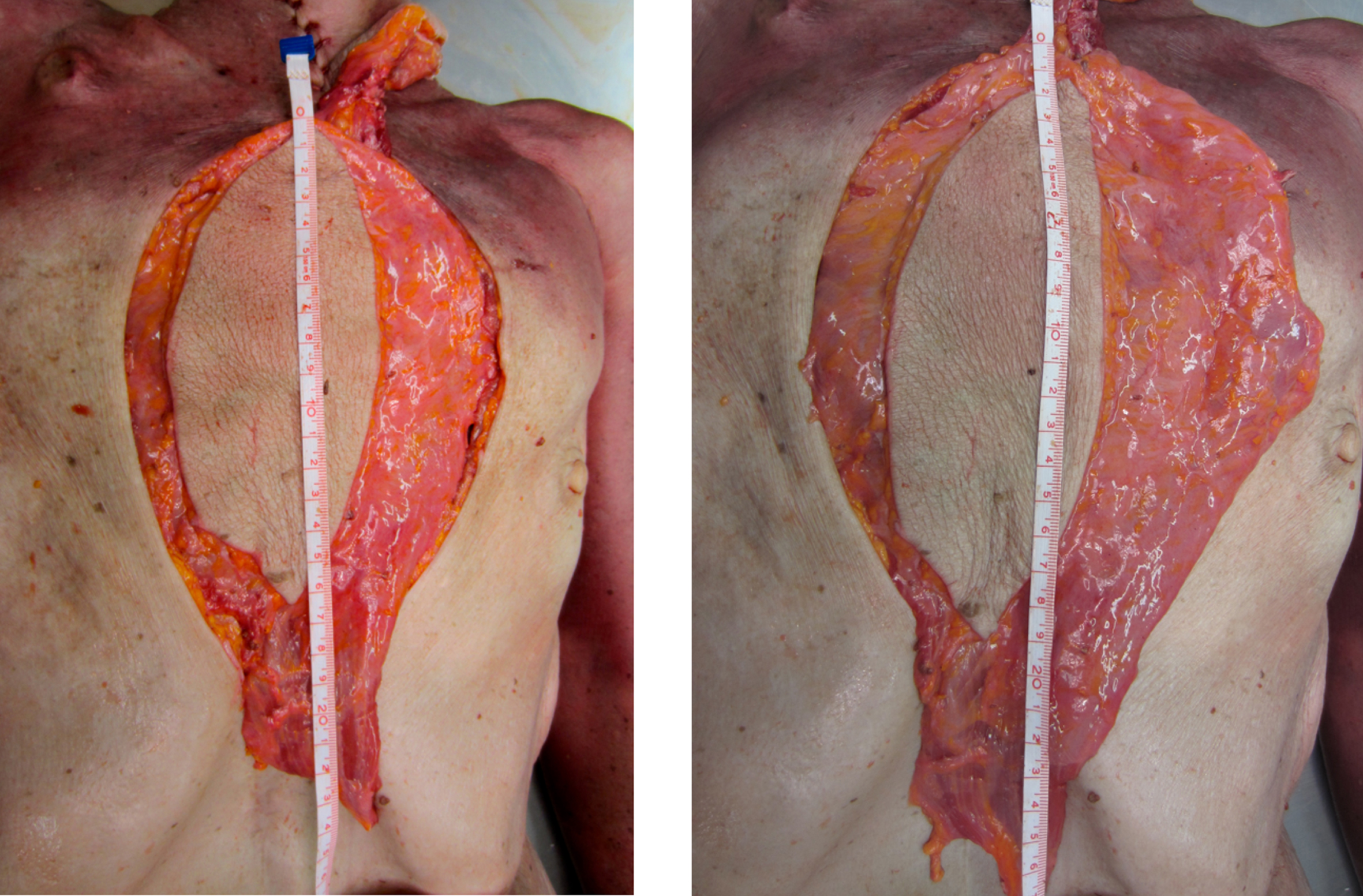
A significant gain of length with a greater muscle volume could be proven statistically. In cachectic patients both preparations of the LDMF may reach the Xiphisternum; but only after ligation of the CSA a mean beneficial gain in length of appropriately 4 cm due to the improved flap’s arc of rotation may be achieved. Flap volume is easily provided by this method, potentially avoiding wound healing deficiencies. Therefore, the modified surgical approach may accomplish a distinct extension of the original harvest of LDMF described and may also enable the surgeon to reconstruct the area above the Xiphisternum with high quality muscular tissue. Furthermore, does the flap volume correlate with the circumference of the flap significantly, which can be taken into consideration for designing the flap. Free flap coverage of deep sternal defects on the other hand has not yet been routinely established. The fear of hypotension during theatre time and the resulting indication for the application of vasopressors have not shown to affect the long-term outcome of flap survival. The use of ephedrine or other vasoactive substances show even a decreased risk of venous thrombosis [61, 62]. Microsurgical anastomoses show a higher complication rate due to lifelong anticoagulation after bypass or heart valve replacement surgery [63]. These procedures may be reserved for special indications, where the remaining tissue is often of poorer quality and not valuable for excessive movement due to intensive scaring and reduced blood supply. In addition, do microsurgical procedures in general demand a sufficient recipient vessel at the chronic wound site. As a result of brachytherapy, there might be the need of vessel loop adjustment and supply [38]. Special indications for extended LMDF coverage comprise of radio ulcers and/or defects after tumor resections. The gain in pedicle length simplifies flap reach to an extent, that even defects of the contralateral breast region may be reconstructed as shown in Fig. 6. The ligation of the CSA does not account for a loss of perfusion to the scapula and its adjoining muscles. Sufficient blood supply is maintained via the suprascapular artery (SSA), the dorsal scapular artery (DSA) and their arcades or anastomoses respectively [64]. A compromise in blood supply of the shoulder region will therefore not be expected. In 2006 Rubino published his results of an improved blood flow in an extended pedicle in general and especially in the lower and distal parts of the harvested flap following the inversion phenomenon [65]. An improved perfusion of the transferred enlarged muscle tissue volume of the LMDF over the xiphoid may be expected due to these studies. As published by Kehrer et al. flap design and perfusion are the keys of success in reconstruction. In their study pelvic defects were reconstructed by posterior thigh flaps, showing similar good results for an improved blood supply to the most distal parts based on the descending branch of the Inferior Gluteal Artery [66].
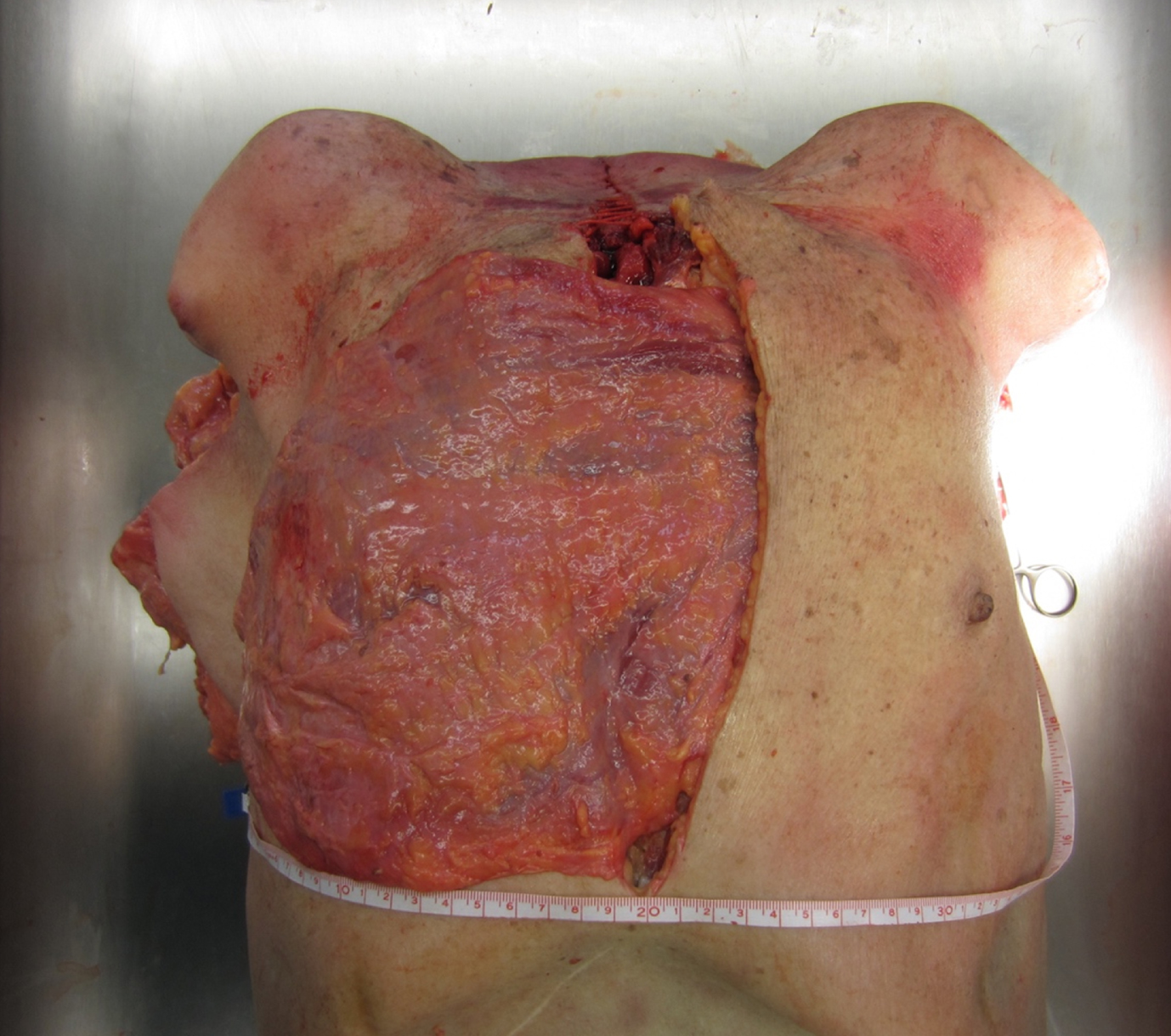
Possible reconstruction of the contralateral thorax due to the increase in arc of rotation (representative picture of the corresponding anatomical study).
Our study has several limitations. One is the limited number of body donors dissected. I n vivo measurements regarding the blood supply have not been performed, but could be subject to further intraoperative ultrasonography measurements before and after ligation of the CSA [67]. A disadvantage worth mentioning is the fact, that a secondary harvest of a free scapular or parascapular flap will not be available anymore after clipping the CSA. Larger prospective clinical studies could contribute towards higher evidence of our results. We would like to highlight that clipping and transecting the CSA and its concomitant veins is an easy procedure that may not only enable reconstructive surgeons to transpose the LDMF more caudally and potentially add blood flow to the flap tissue for a solid reconstructive solution for sternal defects, but as well result in a shortage of intensive care unit stay, hospitalization in general and thus reduction of costs at the same time.
Modified harvesting of the LDMF by sacrificing the CSA may supply more flap volume and length for the reconstruction of entire sternal region defects. This operational method can be a versatile and pristine tool in the armentarium of every plastic surgeon for achieving a stable and aesthetically acceptable coverage of the sternum with a single well-vascularized flap.
Contributions
All authors stated above meet the required conditions for authorship:
1) substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data;
2) drafting the article or revising it critically for important intellectual content;
3) final approval of the version to be published;
4) agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Financial disclosure
There are no financial disclosures of any author. None of the authors has a financial interest in any of the products, devices, or drugs mentioned in this manuscript.
Grants
None.
