Abstract
Objective
Median sternotomy is a standard approach in cardiovascular surgery, and wire fixation is commonly used for sternal closure. However, postoperative intermittent stress on the sternum can lead to sternal breakdown, potentially resulting in deep sternal wound infection (DSWI). Sternal closure with rigid plate fixation was recently reported to be effective for preventing DSWI and promoting sternal healing. We investigated the effectiveness of a rigid plate fixation system in patients at a high risk of developing DSWI.
Methods
This retrospective observational study evaluated the incidence of DSWI and the progression of postoperative sternal fusion observed on computed tomography. Forty-eight patients at a high risk of DSWI who underwent sternal closure with a rigid plate fixation system between 2020 and 2023 were assessed.
Results
Among the 48 patients, 1 (2.1%) developed DSWI requiring surgical treatment. Sternal fusion improved over time, with significant progression observed during the follow-up period compared with the early postoperative period. Additionally, patients who did not show sternal fusion in the early postoperative period showed progressive fusion during follow-up.
Conclusions
The rigid plate fixation system prevents sternal displacement and may be beneficial in preventing DSWI by maintaining and promoting sternal fusion in high-risk patients.
Keywords
Introduction
Median sternotomy is a standard approach in cardiovascular surgery, with wire fixation commonly used for sternal closure. However, postoperative stress on the sternum can lead to sternal breakdown due to wire-related complications. This issue may result in mediastinal hematoma, sternal osteomyelitis, and an increased risk of deep sternal wound infection (DSWI). 1
DSWI, although rare, is a serious complication in cardiovascular surgery, with an incidence of 0.2% to 3.0% depending on patient characteristics.2–4 Risk factors for DSWI include severe obesity, insulin-dependent diabetes mellitus (DM), chronic obstructive pulmonary disease (COPD), hemodialysis, steroid or immunosuppressive drug therapy, reoperation, osteoporosis, and use of the bilateral internal thoracic arteries during coronary artery bypass surgery. 5 Sternal closure using a rigid plate fixation system (RPFS) has been reported to effectively reduce sternal stress, thereby preventing DSWI and promoting sternal healing. 6 A randomized trial demonstrated that rigid plate fixation improves sternal healing and reduces complications compared with wire cerclage.7,8
In this study, we evaluated the effectiveness of RPFS in patients undergoing sternal closure who were at high risk for developing DSWI.
Patients and methods
This retrospective observational study was approved by the Institutional Review Board at Kochi University (IRB2023-129; March 4, 2024), which waived the requirement for informed consent because of the retrospective nature of the study. The study was conducted in accordance with the Declaration of Helsinki.
The study included 48 patients who underwent heart surgery at Kochi University between January 2020 and December 2023, with sternal closure using both wire and an RPFS (SternaLock; Zimmer Biomet Inc., Warsaw, IN, USA). Data were obtained from hospital records.
The RPFS was employed to secure the sternum and prevent DSWI in patients at high risk for this complication. The criteria for RPFS use were a body mass index (BMI) of ≥30 kg/m2, DM requiring insulin administration, a requirement for hemodialysis, use of steroids or immunosuppressive drugs, severe COPD, reoperation, and surgeon’s discretion. These criteria align with Japan’s reimbursement guidelines for RPFS use in patients with a BMI of ≥30 kg/m2, insulin-dependent DM, hemodialysis, steroid or immunosuppressive therapy, severe COPD, use of bilateral internal thoracic arteries, and reoperation.
Endpoints
The primary endpoint was the incidence of DSWI. The incidence of DSWI was compared with predictions from JapanSCORE2. The secondary endpoint was the assessment of sternal fusion in patients who underwent postoperative computed tomography (CT). The sternal fusion score was based on the study by Allen et al. 9
Sternal closure using SternaLock
The surgical procedure involved median sternotomy followed by sternal closure using wires placed at three locations (two in the manubrium and one in the sternal body). The RPFS was used to secure the sternum once alignment was confirmed (Figure 1). Wires were used initially for sternal alignment, and the RPFS was employed for final fixation.
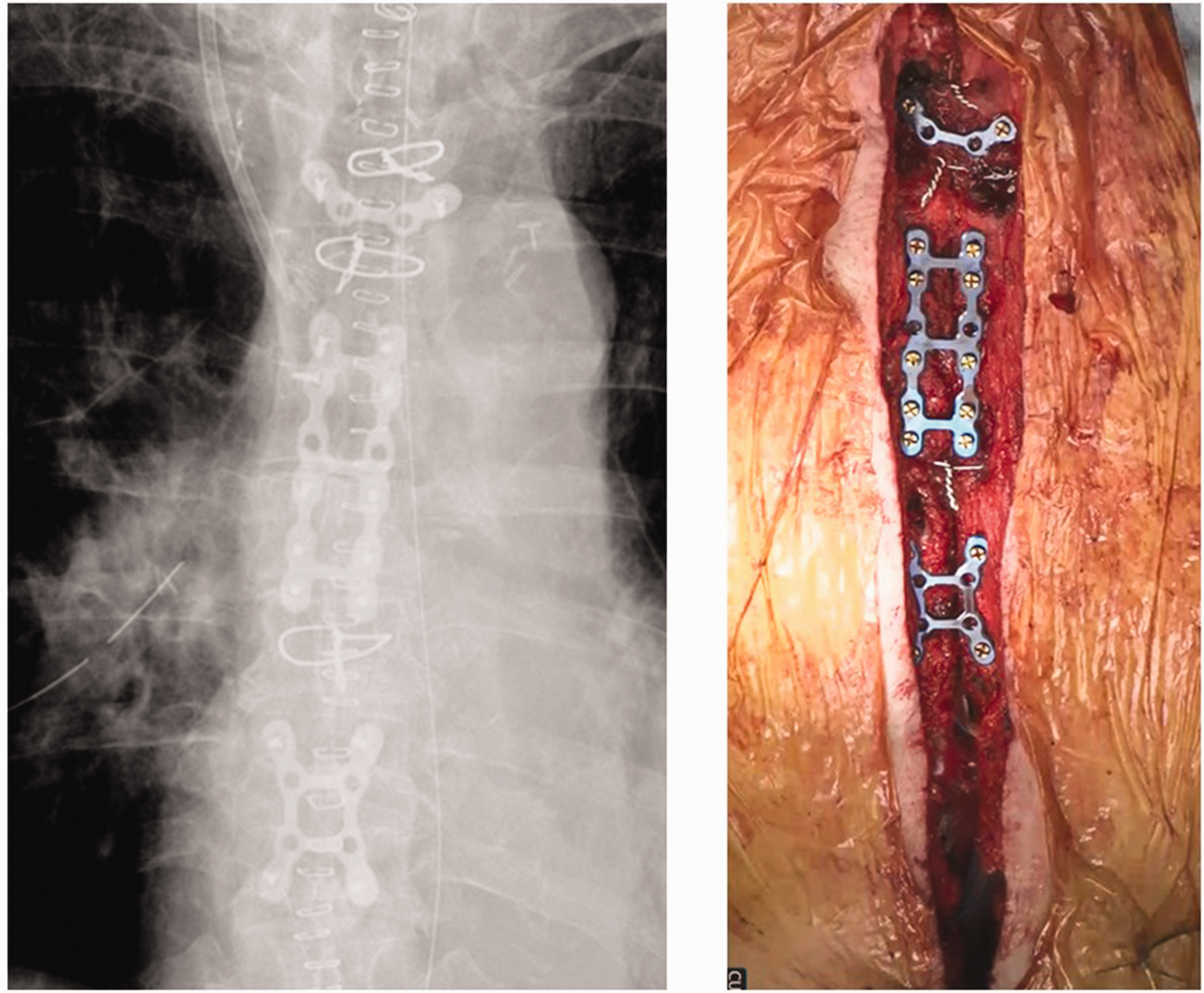
Method of sternal closure in high-risk patients. The sternum is closed by placing two wires around the manubrium and one wire around the sternal body. Following this, the sternum is secured using a rigid plate fixation system (SternaLock). A plate of appropriate size is selected based on the length of the sternum.
Patient characteristics, preoperative and perioperative data, and follow-up tests
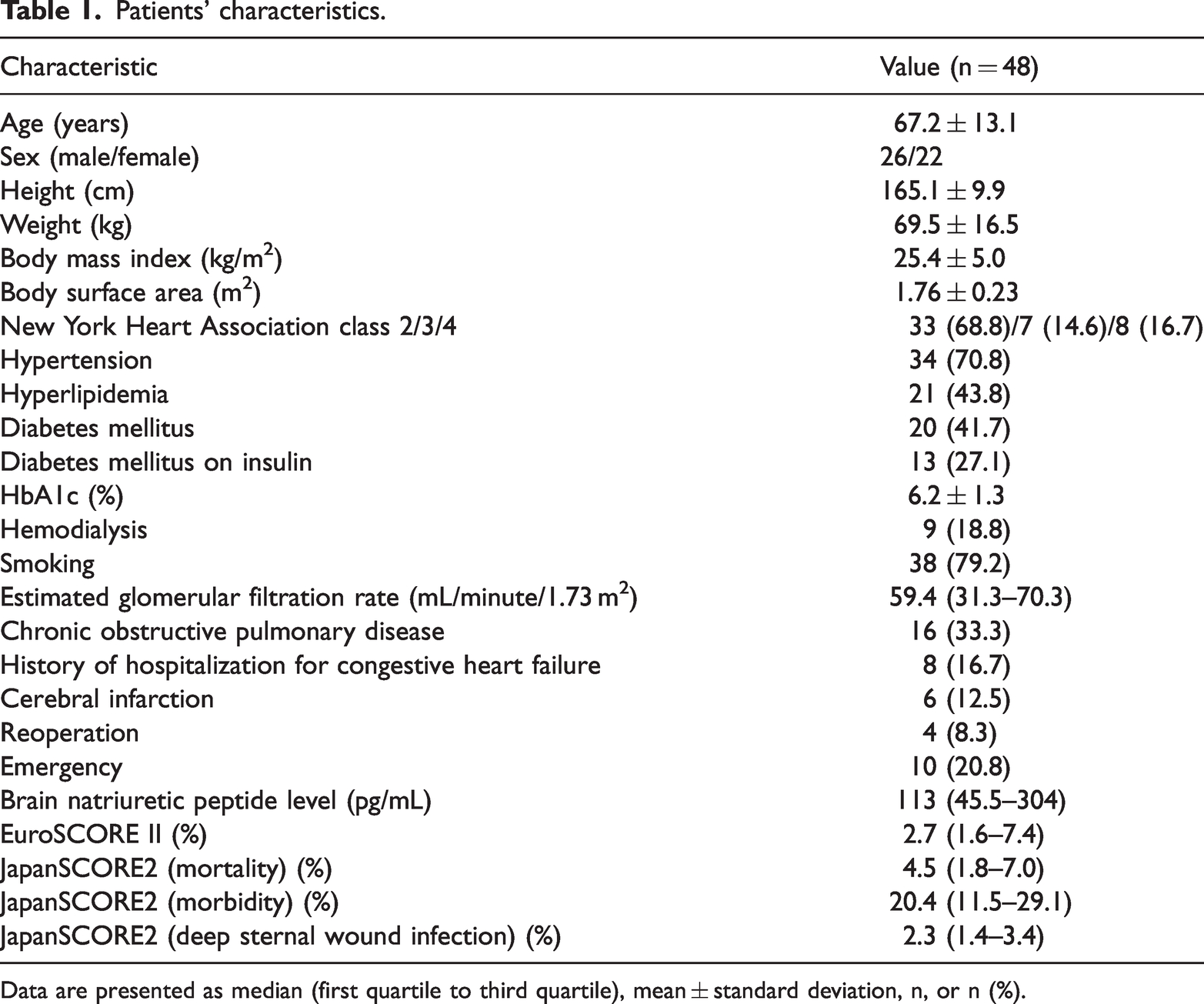
The patient characteristics are listed in Table 1. Clinical data included age, sex, weight, height, BMI, body surface area, New York Heart Association class, history of hospitalization for congestive heart failure, hypertension, hyperlipidemia, DM (with or without insulin use), cerebral infarction, smoking history, COPD, reoperation, hemodialysis requirement, emergency information, preoperative blood test results (estimated glomerular filtration rate, brain natriuretic peptide level, and HbA1c level), EuroSCORE II (preoperative and perioperative risk), and JapanSCORE2 rates (mortality, morbidity, and DSWI risk). 10
Patients’ characteristics.
Data are presented as median (first quartile to third quartile), mean ± standard deviation, n, or n (%).
The sternal fusion score was based on CT evaluation, classified as follows: 0, nonunion; 1, indeterminate; 2, signs of minimal or early healing; 3, mild synthesis; 4, moderate synthesis; and 5, complete synthesis. Scoring was performed across five sections: the manubrium, upper sternal body, middle sternal body, lower sternal body, and xiphoid process (Figure 2).

Location and degree of sternal fusion as assessed by computed tomography. The left image depicts the areas of the sternum evaluated for fusion on computed tomography: (a) manubrium, (b) upper sternal body, (c) middle sternal body, (d) lower sternal body, and (e) xiphoid process. The right image illustrates the degree of sternal fusion. This image is adapted from the study by Allen et al. 9
Sternal fusion was evaluated at 1–14, 15–30, 31–90, 91–180, and >180 days postoperatively. For patients with sternal disarticulation, the distance was measured using VINCENT (Fujifilm Co., Ltd., Tokyo, Japan) to magnify the image.
All patient data were de-identified to protect patient confidentiality. This study adheres to the STROBE guidelines. 11
Statistical analysis
Continuous variables are expressed as median (first to third quartile) or mean (standard deviation), and categorical variables are expressed as frequency (percentage). All variables were analyzed using the unpaired t-test, Mann–Whitney U test, or Fisher’s exact test. For sternal fusion comparisons, unpaired t-tests were used at each time point because not all patients underwent CT scans at all intervals.
All analyses were conducted using SPSS software version 22.0 (IBM Corp., Armonk, NY, USA). A P-value of <0.05 was considered statistically significant.
Results
In this study, the RPFS was utilized in 48 patients. Among these 48 patients, 2 required re-fixation because of postoperative sternal dehiscence. The patient demographics are summarized in Table 1. The mean patient age was 67.2 ± 13.1 years, and the study population comprised 26 men and 22 women. The mean BMI was 25.4 ± 5.0 kg/m2. Hypertension was present in 34 (70.8%) patients and hyperlipidemia in 21 (43.8%) patients. DM was present in 20 (41.7%) patients, with 13 (27.1%) requiring insulin. The mean HbA1c value was 6.2% ± 1.3%, and the median estimated glomerular filtration rate was 59.4 mL/minute/1.73 m2 (range: 31.3–70.3). COPD was noted in 16 (33.3%) patients. Reoperation was performed in 4 (8.3%) patients. The EuroSCORE II rate was 2.7% (range: 1.6%–7.4%). The JapanSCORE2 estimates showed a mortality risk of 4.5% (range: 1.8%–7.0%), morbidity risk of 20.4% (range: 11.5%–29.1%), and DSWI risk of 2.3% (range: 1.4%–3.4%).
The primary risk factors were a BMI of ≥30 kg/m2, insulin-dependent DM, hemodialysis, COPD, steroid or immunosuppressive drug use, and reoperation. Among the 48 patients, 4 (8.3%) had 0 risk factors, 31 (64.6%) had 1 risk factor, 11 (22.9%) had 2 risk factors, and 2 (4.2%) had 3 risk factors. Although four patients had no DSWI risk factors, the RPFS was used because of intraoperative assessment of bone fragility and the high likelihood of sternal disarticulation if only wires were used.
Cardiopulmonary bypass was employed in 41 (85.4%) patients, with a mean duration of 193.7 ± 67.4 minutes. The mean aortic cross-clamping time was 138.5 ± 49.6 minutes. Concomitant operations were performed in 24 (50.0%) patients.
There were no cases of 30-day mortality; however, two (4.2%) in-hospital deaths occurred, attributed to sepsis and nonobstructive intestinal ischemia. DSWI developed in two (4.2%) patients. The first case involved a 66-year-old patient who underwent emergency total arch replacement for acute aortic dissection. This patient developed DSWI, which was treated with debridement and omentopexy. The patient had COPD associated with heavy smoking, identified as a risk factor for DSWI, with a JapanSCORE2 estimated risk rate of 6.3%. The second case of DSWI was diagnosed based on blood culture results, CT imaging, and clinical examinations and was successfully managed with antibiotic therapy.
Reoperation was necessary in six (12.5%) patients. Among these, two patients experienced sternal dehiscence following surgery, both of whom initially had their sternum closed with wires only. The first patient was a 71-year-old man who had undergone total arch replacement and aortic valvuloplasty, and he had a BMI of 31.4 kg/m2. Twelve days postoperatively, he required sternal re-fixation with the RPFS for sternum closure. The second patient was a 78-year-old man who required a two-vessel bypass for stenosis of the left coronary main trunk following ascending aortic replacement for acute aortic dissection; he also had COPD as a risk factor for DSWI. Of the remaining four patients, two developed delayed tamponade and required drainage via the left intercostal chest approach, one developed postoperative hemorrhagic shock and underwent hemostasis via repeat median sternotomy, and one developed DSWI and was treated with debridement and omentopexy.
Figure 3 illustrates the progression of sternal fusion as evaluated by CT over time. During the first 1 to 14 days postoperatively, the sternal fusion scores were 1.41 ± 0.64 for the manubrium, 1.38 ± 0.55 for the upper portion of the sternal body, 1.35 ± 0.54 for the middle portion of the sternal body, 1.19 ± 0.7 for the lower portion of the sternal body, and 1.08 ± 0.68 for the xiphoid process. Sternal fusion improved progressively at all sites, and by 180 days, the scores had increased to 3.25 ± 1.14 for the manubrium, 3.75 ± 0.87 for the upper portion of the sternal body, 3.83 ± 0.83 for the middle portion of the sternal body, 3.67 ± 0.98 for the lower portion of the sternal body, and 3.33 ± 0.98 for the xiphoid process (all P < 0.001).
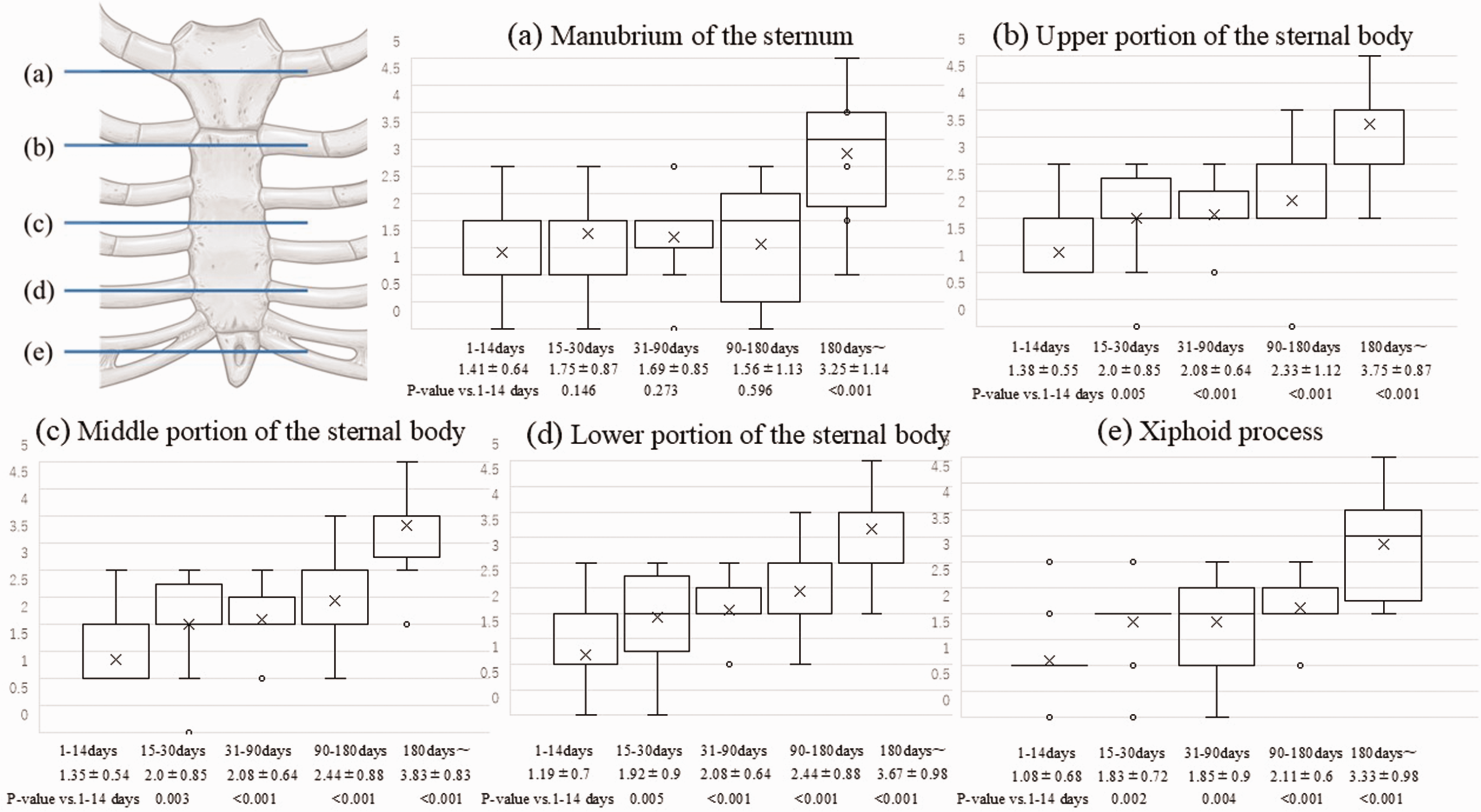
Degree of fusion at various sternal sites as assessed by computed tomography. The upper left diagram indicates the areas evaluated on computed tomography: (a) manubrium, (b) upper sternal body, (c) middle sternal body, (d) lower sternal body, and (e) xiphoid process. The accompanying graphs illustrate the progression of sternal fusion over time at each site, with findings ranging from 1–14 days to >180 days postoperatively.
In the first 1 to 14 days, the percentage of locations with a score of 0 (indicating nonunion) was 5.5% for the manubrium, 0.0% for the upper and middle portions of the sternal body, 13.5% for the lower portion of the sternal body, and 16.2% for the xiphoid process. The progression of the mean sternal dehiscence distance for sites with a score of 0 was as follows: 3.1 ± 2.8 mm (range: 0.8–11 mm) at 1 to 14 days, 1.4 ± 1.7 mm (range: 0–4.2 mm) at 31 to 90 days, 0.8 ± 1.2 mm (range: 0–2.2 mm) at 90 to 180 days, and 1.0 ± 1.1 mm (range: 0.0–2.4 mm) after 180 days. None of the patients evaluated by CT exhibited advanced sternal disarticulation (Figure 4).
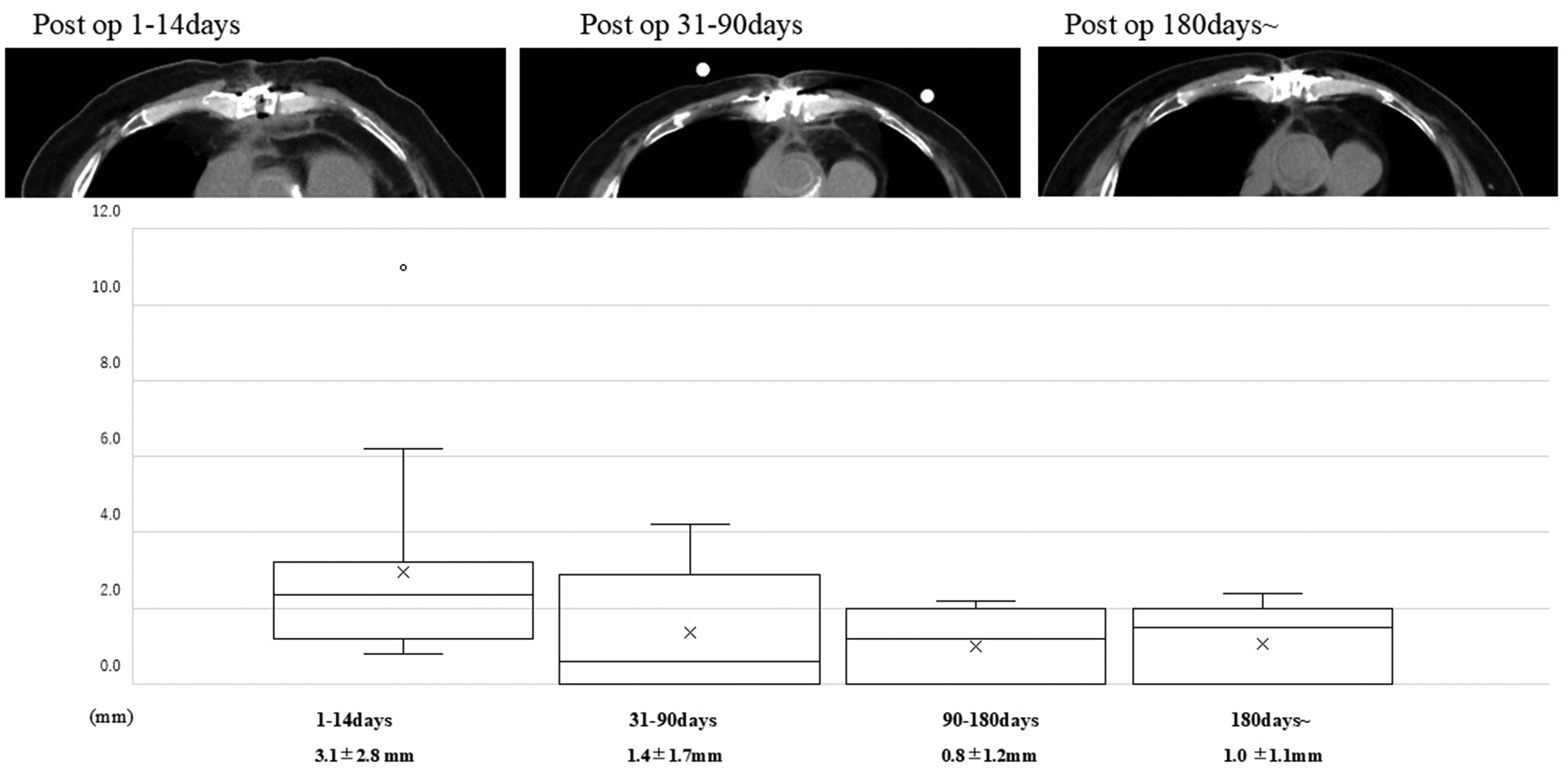
Changes in sternal fusion in cases of immediate postoperative sternal disarticulation. The upper images display the degree of sternal fusion as seen on computed tomography. Even in cases where the sternum did not fuse early postoperatively, fusion was observed over time. The graph below depicts the changes in disarticulation distance in cases of early postoperative sternal disarticulation.
Discussion
This study examined the efficacy of RPFS in patients at high risk for DSWI. DSWI requiring debridement and omentopexy was observed in one (2.4%) patient, a rate consistent with the predicted DSWI risk according to JapanSCORE2. Sternal bone fusion progressed over time across all evaluated regions, even when early postoperative sternal fusion had not occurred (Figure 4). Importantly, there were no instances of advanced sternal dehiscence.
Sternal instability and dehiscence are key contributors to the development of DSWI, underscoring the importance of meticulous sternal alignment and closure. 12 The American Association for Thoracic Surgery guidelines recommend rigid sternal fixation with plates or bands (Class IIb, Level B Evidence) to mitigate the risk of sternal dehiscence. 13 In an in vitro comparison by Pai et al., 6 wire fixation was found to result in significant lateral migration, particularly toward the lower sternum, with the widest separation occurring at the xiphoid process. Conversely, rigid metal plates did not exhibit greater lateral separation than did wire fixation. Consistent with these findings, our study showed that both the early sternal fusion and disarticulation rates were worse in the lower part of the sternum. However, the degree of sternal fusion improved progressively over time in all regions. Additionally, cases of early postoperative nonfusion did not advance to dehiscence, and some non-fused cases in the early postoperative period showed signs of fusion later, resembling the fracture repair process. It is well-established in orthopedics that limiting relative motion between broken bone segments promotes faster bone healing. 14
Wire closure of the sternum may lead to pseudoarticular formation and sternal cutting. 15 By contrast, plates are secured with screws at multiple points. The SternaLock system, for example, utilizes a locking mechanism that stabilizes the plate and screw, providing angular stability. Unlike wires, plates do not need to be crimped to the periosteum, preserving blood flow to the periosteum and facilitating bone fusion. 16 Pai et al. 6 further demonstrated that plate fixation significantly reduces lateral migration and offers stronger resistance to lateral pull forces compared with wire fixation. These findings suggest that plates are more effective in promoting early sternal fusion because of their ability to maintain stable fixation.
Nishimura et al. 5 reported significantly lower rates of sternal dislocation in patients undergoing fixation with the SternaLock plating system than in those receiving conventional wire fixation. Additionally, Madjarov et al. 17 found that longitudinal rigid sternal fixation was superior to conventional wire cerclage closure at 3 months postoperatively.
In this study, two patients required sternal re-fixation. Both patients initially had their sternum closed with wires alone but subsequently underwent re-fixation with the RPFS because of sternal dehiscence. One case involved a 71-year-old man with a BMI of 31.4 kg/m2, whose predicted DSWI incidence according to JapanSCORE2 was 4.3%. He required sternal re-fixation on postoperative day 12. The other patient was a 78-year-old man with COPD, a significant risk factor for DSWI, and his predicted DSWI incidence was 7.9% according to JapanSCORE2. He underwent sternal re-fixation on postoperative day 14. Both patients were discharged without DSWI following re-fixation with the RPFS.
Obesity is a well-documented independent risk factor that increases the likelihood of DSWI by up to 2.6 times. 18 Several hypotheses explain the relationship between obesity and DSWI. One theory suggests that a larger chest wall perimeter increases tension on the sternal wound, rendering it unstable and more susceptible to infection. 19 Smoking-related coughing can also stress sternal wires, potentially leading to wire breakage, sternal fracture, and wound dehiscence. 19 Moreover, smoking reduces local blood flow, leading to decreased skin circulation and tissue hypoxia, which impairs wound healing. 20 These factors contribute to COPD being a significant risk factor for sternal avulsion, 21 and patients with COPD are at a high risk of developing DSWI.22,23
Obesity and COPD both contribute to sternal instability due to external pressure. Use of the RPFS may be particularly effective in patients with these risk factors because it can counteract the lateral pressure caused by external forces.
Limitations
This study has several limitations. First, although it focused on patients at high risk of DSWI, the number of included patients was insufficient to comprehensively assess DSWI, which is a low-incidence complication. Second, no comparative study was conducted between groups treated with and without the RPFS, making it difficult to determine the impact of the RPFS on sternal fusion and disarticulation rates. Further statistical studies are needed to identify the factors that most strongly influence DSWI and to evaluate the efficacy of the RPFS.
Additionally, in this study, the RPFS was not used alone for sternal closure but in combination with wires. Therefore, the results do not reflect the outcomes of using the RPFS alone. The combination approach was used because wires are effective for aligning the sternum during surgery, with the RPFS providing additional security for final closure.
Future studies should explore methods and techniques for closing the sternum using only the RPFS. However, this study suggests that the RPFS is effective in promoting sternal fusion, even in patients with early postoperative nonunion, and in preventing the progression of sternal dehiscence.
CT evaluation also has limitations. Although it can assess sternal dehiscence, this does not necessarily indicate DSWI. A comprehensive assessment, including physical findings and other clinical data, is essential. Nonetheless, sternal dehiscence is a critical factor in DSWI development, and CT is crucial for its evaluation.
Conclusion
The RPFS effectively prevents sternal displacement and promotes sternal fusion. This system also supports fusion in cases of early postoperative nonunion and prevents the progression of sternal dehiscence. Therefore, the RPFS may be a valuable tool in preventing DSWI in high-risk patients undergoing median sternotomy.
Footnotes
Acknowledgement
The authors would like to thank Enago for the English language review.
Authors’ contributions
The authors confirm their contributions to the paper as follows: Study conception and design: HN, YM, and KO; data collection: HN; analysis and interpretation of results: HN; drafting of the manuscript: HN, KY, NE, and RS. All authors reviewed the results and approved the final version of the manuscript.
Data availability
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The authors declare that there are no conflicts of interest.
Funding
This study did not receive any specific funding.
Institutional review board approval and consent
The institutional review board at Kochi University approved this retrospective observational study (IRB2023-129; March 04, 2024) and waived the requirement for informed consent owing to the retrospective nature of the study. The study was performed in accordance with the Declaration of Helsinki.
