Abstract
Background
Cervical cancer (CC) is the fourth most common malignancy. The significant prognostic factors are tumor size and lympho-vascular space invasion. Considering that these are nonspecific factors, research has been aimed at finding a specific molecular marker related to a higher incidence of relapse and mortality in patients with CC.
Objective
Our study investigated the prognostic value of L1 cell adhesion molecule (L1CAM) expression in rare histological subtypes of cervical cancer (adenocarcinomas and adenosquamous cell carcinomas).
Methods
This is a single-institution retrospective study with 35 patients who underwent radical hysterectomy for early-stage cervical adenocarcinoma or adenosquamous cell carcinoma in 2007 through 2017. Paraffin sections of the tumor were used for L1CAM analysis by immunohistochemistry.
Results
L1CAM expression was positive in 15 (42.8%) of the 35 tumors. L1CAM expression did not differ significantly in regard to the stage of disease, tumor size, grading, or lymphovascular space invasion (LVSI) (p = 0.619, p = 0.341, p = 0.445, p = 0.999). Progression-free interval and overall survival did not differ between L1CAM-positive and L1CAM-negative groups (p = 0.704, p = 0.386, respectively).
Conclusions
In our study, L1CAM expression is not a negative prognostic factor associated with aggressive tumor behavior, recurrence risk and overall survival.
Keywords
Introduction
According to FIGO Cancer Report: Update 2021 Cervical cancer (CC) is the fourth most common malignancy. Following breast cancer, colorectum and lung cancer, CC is the fourth most common cause of death by malignancy in women worldwide. The majority of CC cases are of the squamous cell carcinoma (SCC) type, with other histological subtypes being less common.1–3
In 2020, the WHO updated the Female Genital Tumors classification (5th edition) and recognized that a proportion of cervical cancers are not associated with HPV infection, especially adenocarcinomas. Based on this statement adenocarcinomas were subdivided into HPV-associated, including usual type: villoglandular variant; mucinous type: mucinous not otherwise specified (NOS) adenocarcinoma, intestinal adenocarcinoma, signet-ring cell adenocarcinoma, and stratified mucin-producing adenocarcinoma, and HPV-independent tumors, including gastric type adenocarcinoma; clear cell adenocarcinoma; mesonephric adenocarcinoma; and endometrioid adenocarcinoma. 4
Adenocarcinomas compared with SCC, a more frequent incidence was reported in younger women, who are more commonly diagnosed in the early stages of the disease.1–3
Adenosquamous cell carcinoma is even less frequent, with aggressive behavior. It is a tumor of both glandular and squamous components, and it is reported to have a poorer prognosis since the glandular component is usually poorly differentiated.1–3,5
The significant prognostic factors for CC are tumor size, LVSI, extracervical spread of disease, and surgical methods.6,7 Research has been targeting a specific molecular marker linked to higher relapse and mortality rates in CC patients, instead of nonspecific factors. L1 cell adhesion molecule (L1CAM) is a transmembrane glycoprotein that mediates communication between cells and their extracellular matrix. It belongs to the immunoglobulin-like family of molecules. Increased expression of L1CAM in tumor cells promotes tumor angiogenesis, motility, and invasion through the epithelial-mesenchymal transition.8,9
L1CAM expression is a strong prognostic factor for early recurrence and metastasis in many malignancies (colorectal, kidney, pancreatic, and other carcinomas). These tumors are considered to have an unfavorable prognosis.10–12 Multiple studies have confirmed that L1CAM expression is a negative prognostic factor for more aggressive tumor behavior, especially in cases of endometrial cancer.13–16
In this study, we aimed to investigate the prognostic value of L1 cell adhesion molecule expression in rare histotypes of FIGO I and IIa stage cervical carcinoma (HPV-associated adenocarcinoma and adenosquamous cell carcinoma) as a negative biomarker.
Materials and methods
Study group and follow-up
We have conducted a retrospective study at one institution, which involved 35 patients diagnosed with HPV-associated adenocarcinoma (21 patients, Figure 1) and adenosquamous cell carcinoma (14 patients, Figure 2) of the uterine cervix. All of these patients underwent radical surgery and follow-up at a tertiary cancer center between the years 2007 and 2017. All the included patients had preoperative stage FIGO IB and IIA (Fédération Internationale de Gynécologie et d'Obstétrique classification 2018) disease. 1
Adenocarcinoma formed by a tubular and cribriform component, hematoxylin-eosin staining (magnification 100x).

Adenosquamous carcinoma formed by solid squamous component and tubular adenomatous component, hematoxylin-eosin staining (magnification 200x).
This study was approved by the local Ethics Committee number 400/2020. Thirty-one patients were diagnosed with FIGO IB disease stage and four with FIGO IIa. The cohort grading distribution was as follows: 14 tumors were well differentiated, 16 were intermediately differentiated, and four were poorly differentiated. One case was of unknown grade. The majority of tumors measured more than 2 centimeters (n = 24), and LVSI occurred in 6 tumors (Table 1). The adjuvant treatment was administered only in case of positive vaginal resection margin, metastatic involvement of pelvic lymph nodes and bulky tumors more than 40 mm in diameter. No patient had a parametrial involvement. In total 9 patients with adenocarcinomas underwent adjuvant treatment (5 patients L1CAM positive and 4 patients L1CAM negative) and 8 patients with adenosquamous cell carcinoma (4 patients L1CAM positive and 4 patients L1CAM negative).
Patient characteristics (n = 35; the values represent the absolute frequencies and the relative frequencies in percentages in brackets, if not specified differently).

Range = (minimum; maximum).
p-value, Fisher's exact test.
p-value, Mann-Whitney test.
After surgery, patient follow-up consisted of clinical examination, blood sampling, and ultrasound examination. If suspected recurrence occurred, we added a different imaging method. The focus was confirmed by tru-cut biopsy.
Patients underwent follow-up visits every three months during the first two years, followed by visits every six months until the fifth year. After that, follow-up visits were conducted annually until the end of the study. Any instances of malignant tissue were classified as local or distant recurrences, provided they were confirmed through histological or radiological testing. Any cases where follow-up was not conducted were excluded from the database. The median total length of follow-up from surgery to the end of the study was 68 months, with a range of 1 to 143 months. Recurrence occurred in 6 patients, and three patients died.
Pathological processing
The immunohistochemical processing technique utilized for assessment was initially described by Bosse et al. 14 Formalin-fixed paraffin-embedded tissue blocks were cut into 4-μm slices and placed on superfrost microscope slides. Immunohistochemistry was then performed using a Ventana Discovery automated immunostaining system (Ventana Medical Systems, Tucson, AZ, USA). Briefly, after deparaffinization with an inorganic buffer and rehydration, antigen retrieval was achieved by incubating the slides in Cell Conditioning Solution 1 (CC1; Ventana Medical Systems). The primary L1 antibody (CD171) (BioLegend, Dedham, MA, USA) was then applied at a 1:40 dilution, followed by a 32-min incubation. Next, diaminobenzidine- and horseradish peroxidasecontaining detection kit was used (iView Universal DAB detection Kit; Ventana Medical Systems) and performed counterstaining with hematoxylin and bluing reagents. Immunohistochemical evaluation of L1CAM and scoring were performed by experienced pathologist dedicated for this study. Only unequivocal membranous staining was considered specific. A case was classified as L1CAM-positive if over 10% of tumor cells were scored Positive 6 (Figures 3 to 7).

Adenocarcinoma, negative expression of L1CAM on the cell membrane (brown staining in left upper corner), less than 10% of tumor cells (magnification 200x).

Adenocarcinoma, positive expression of L1CAM on the cell membrane (intensive brown staining), more than 10% (approximately 30%) of tumor cells (magnification 100x).

Adenocarcinoma, intensive positive expression of L1CAM on the cell membrane in almost all tumor cells (aproximately 90%)(magnification 100x).

Adenosquamous carcinoma, negative expression of L1CAM on the cell membrane, less than 10% of tumor cells (magnification 200x).

Adenosquamous carcinoma, positive expression of L1CAM on the cell membrane, more than 10% (between 10-20%) of tumor cells (magnification 100x).
Statistical processing
Quantitative variables are presented as medians and ranges (minimums and maximums); qualitative variables are presented as absolute values and proportions. The comparison of qualitative variables between groups was carried out by Fisher's exact test and in quantitative variables by Mann-Whitney test. Progression-free survival and overall survival were analysed using Kaplan-Meier curves and log-rank test. The significance level was set to 0.05 and the statistical analysis was performed using R software (version 4.2.3) with maximum available data, if missing data occur, it is stated in the table or in the title of the figure.
Results
Of the whole cohort of 35 patients with HPV-associated adenocarcinoma and adenosquamous cell carcinoma of the uterine cervix, nearly half of the group were L1CAM positive (15 cases, 42.8%).
L1CAM expression in relation to PFS and OS
The median length of follow-up was 68 months (range 1–143 months). During this time, recurrence occurred in 6 cases. Two cases were L1CAM positive, and four patients were L1CAM negative. In total, 3 patients died (2 patients were L1CAM positive and 1 patient was L1CAM negative) Regardless of the small group of patients, there was no difference in L1CAM expression. Regarding progression-free survival and overall survival, our data did not show a significant difference in L1CAM expression either (log-rank test, p = 0.704; p = 0.386, respectively; Figures 1 and 2).
L1CAM expression in relation to risk pathological characteristics
In our retrospective analysis, we did not notice a significant difference in the degree of tumor differentiation, the presence of substantial LVSI, or the FIGO stage (Fisher's exact test, p = 0.445, p > 0.999, p = 0.619, respectively). Although most tumors measured more than 2 centimeters, we did not find a statistically significant association of tumor size and L1CAM expression (Mann-Whitney test, p = 0.341) (Figures 8 and 9).

Kaplan–Meier curves displaying the estimated probability of nonrecurrence by L1CAM positivity (34 out of 35 patients included; one L1CAM positive patient was excluded from the survival analysis due to an error in data entry; for patients without recurrence, time to last follow-up visit or time to death was considered). No significant difference was found in survival without recurrence between L1CAM-positive and L1CAM-negative patients (log-rank test, p = 0.704).
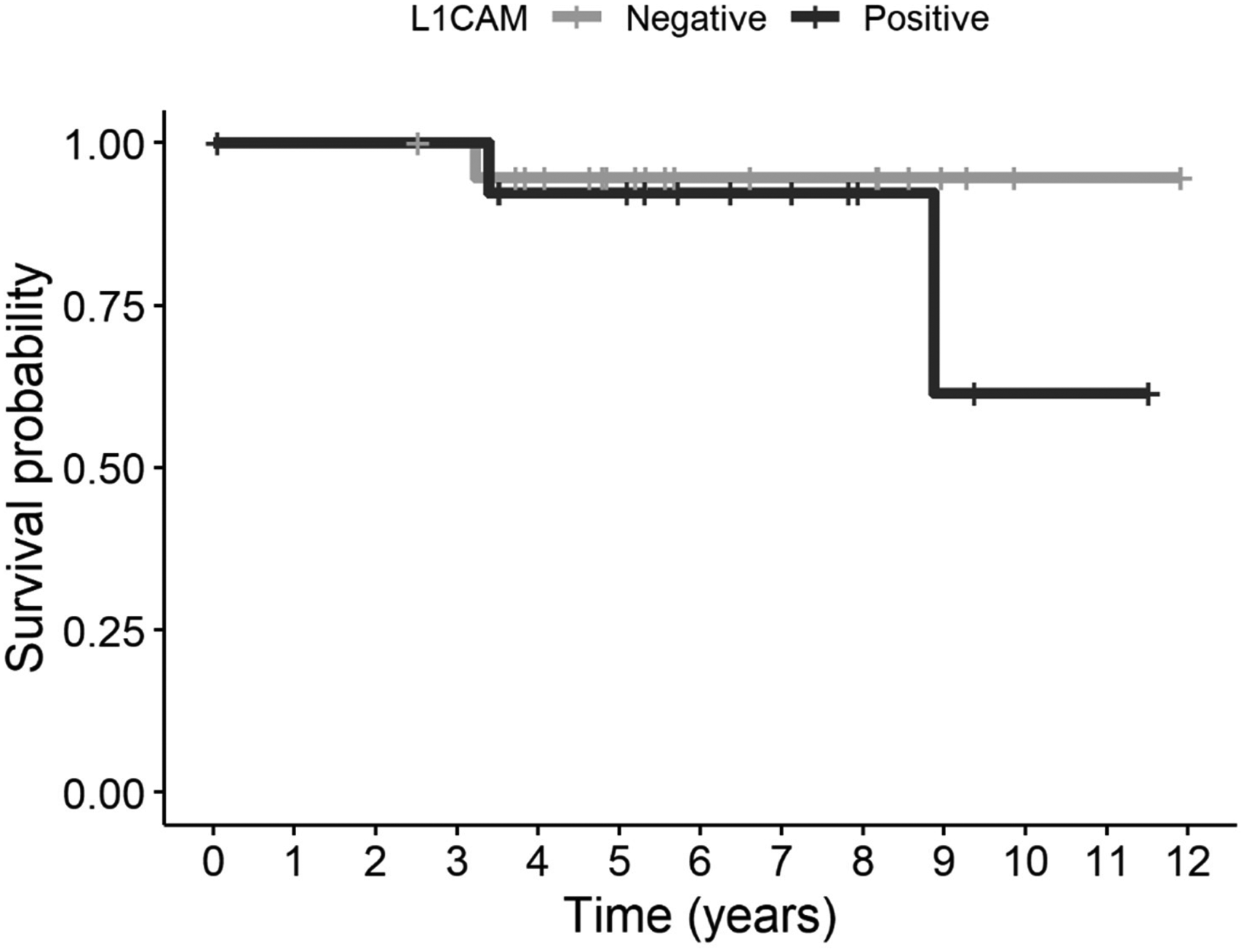
Kaplan-Meier curves displaying the estimated probability of survival by L1CAM status (34 out of 35 patients included; one L1CAM positive patient was excluded from the survival analysis due to an error in data entry; time to death or time to last follow-up visit was considered). No significant difference was found in survival between L1CAM-positive and L1CAM-negative patients (log-rank test, p = 0.386).
Discussion
In our single-institution retrospective study, we investigated L1 cell adhesion molecule expression in rare histotypes of FIGO I and IIa stage cervical carcinoma (HPV-associated adenocarcinoma and adenosquamous cell carcinoma). Several studies have identified L1CAM expression as a negative prognostic factor for different types of tumors.10–12 In oncologic gynecology, L1CAM expression has also been identified as a negative biomarker in endometrial carcinoma.13–16 Whether or not the presence of this marker has the same role in CC is still unclear. Only a few studies have investigated this topic.17–19
Regardless of the histotype, the results of all previous studies show that L1CAM expression in cervical cancer patients is associated with more aggressive tumor behavior, more frequent recurrence of disease, and generally worse disease-free survival (DFS). All these studies analyzed both squamous cell carcinomas and adenocarcinomas. However, some studies suggest that L1CAM expression varies among different tumor histotypes (with higher levels of expression in glandular cell tumors than in squamous cell carcinomas). Hence, our research focused on rare cervical cancers with glandular components, specifically HPV-associated adenocarcinoma and adenosquamous cell carcinoma.
Schrevel et al., in 2017, presented results from the to-date most extensive cohort of 372 patients with cervical carcinoma. There were 21% L1CAM-positive cases (n = 80), 266 squamous cell carcinomas, and 106 adenocarcinomas and adenosquamous cell carcinomas. Over the 88-month observation period, they found a link between higher expression of L1CAM and poorer DFS (p = 0.017). Compared with squamous cell carcinoma, a significantly higher frequency of L1CAM-positive samples was noted in adenocarcinomas (37%) and adenosquamous carcinoma (33%) (p < 0.001). 17 A similar result was published recently by Romanova et al. 20
Carvalho and colleagues published in 2020 a retrospective study of 71 patients with cervical cancer. The study included 45 squamous cell carcinomas and 26 adenocarcinomas. Among them, 20 (28.2%) were L1CAM-positive. This study confirmed worse DFS in L1CAM-positive patients. The average DFS in L1CAM-positive patients was shorter than that in L1CAM-negative patients (137.1 months vs 184.8 months, respectively; p = 0.032). The median follow-up was 66.2 months and the patient overal survival did not differ significantly (p = 0.220). In addition, this study showed significantly higher expression of L1CAM in adenocarcinomas in comparison with squamous cell tumors (50% vs 15.6%, p = 0.002). 18
In 2021, Elfeky and colleagues presented the findings of a study involving 62 patients with CC (44 with squamous cell carcinomas and 18 with adenocarcinomas). The study confirmed increased expression of L1CAM in 24 patients (38.7%). The results revealed a significant link between higher patient mortality and L1CAM levels, with mortality rates being 19.5 times higher in the L1CAM-positive group. However, when it comes to the histotype of the tumor, there was no variation observed. 19
In our retrospective study on 35 patients with FIGO stage I and IIa cervical carcinomas, 15 cases (42.8%) were L1CAM positive. Our cohort includes only rare histotypes: HPV-associated adenosquamous cell carcinomas (n = 14) and adenocarcinomas (n = 21). In defense of the small sample size, we want to emphasize that these are uncommon types of cervical tumors.
During the follow-up period (which lasted a median of 68 months), we encountered 6 cases of relapse. We found no variation in the expression of L1CAM. Our data revealed that there was no significant difference in the expression of L1CAM in relation to either progression-free survival or overall survival (as indicated by the log-rank test, with p-values of 0.704 and 0.386, respectively).
According to Carvalho et al., there is a significant link between tumor size and L1CAM expression. They found that 40% of patients who tested positive for L1CAM were diagnosed with FIGO stage IB3, compared with only 9.8% of L1CAM-negative patients (p = 0.005). 18 However, Elfeky et al. found no correlation between L1CAM expression and tumor differentiation or LVSI. 19 Similarly, Schrevel et al.'s study did not reveal any statistical differences in LVSI, disease stage, or tumor size. 17
The results of our study differ from those of the previous two studies. Regarding L1CAM expression, we did not confirm a difference in tumor differentiation, LVSI, or disease stage (p = 0.445, p > 0.999, p = 0.619 respectively). However, it is worth noting that the majority of tumors in our cohort measured more than 2 centimeters, and regarding tumor size, we also did not observe a statistically significant association of tumor size and L1CAM expression (p = 0.341).
Our study boasts a significant benefit – a diverse set of patients treated at a specialized medical facility, undergoing an uniform surgical procedure, and receiving consistent post-treatment monitoring. What‘s more, our research focuses only on infrequent types of cancer, specifically HPV-related adenosquamous cell carcinoma and adenocarcinoma.
However, our study had limitations due to its retrospective nature and the small number of recurrence instances. This is a common issue in cancer research when dealing with limited malignant potential, as there are only a few disease-specific events to base survival rates on during follow-up.
Regarding L1CAM expression and disease-free survival, overall survival, and relapse of disease, our data are not consistent with the results of previous studies on cervical cancer patients. In our cohort, we only focused on rare cervical carcinomas (adenocarcinomas and adenosquamous cell carcinomas) and its L1CAM expression. However, we were unable to confirm the assumption that higher L1CAM expression is linked to a higher risk of metastasis or early disease recurrence.
Footnotes
Acknowledgments
This study was supported by grant MH CZ - DRO (FNOs/2020).
Author contributions
For every author, his or her contribution to the manuscript needs to be provided using the following categories:
Conception: JK
Interpretation or analysis of data: AK
Preparation of the manuscript: JK, MR, VZ
Revision for important intellectual content: JK, AK, OS
Supervision: JK, OS
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grant MH CZ - DRO (FNOs/2020).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The datasets used and/or analyzed in the current study are available from the corresponding author upon reasonable request.
