Abstract
Objective
There is a lack of research evaluating the effect of tumor markers for prognosis in cervical adenocarcinoma. We aimed to develop and validate a preoperative tumor-marker-based model including clinicopathological factors to clarify the prognostic value of endocervical adenocarcinoma.
Methods
A total of 572 patients with cervical adenocarcinoma who were staged at the International Federation of Gynecology and Obstetrics (FIGO) IA–IIA were reviewed retrospectively. Preoperative serum carcinoembryonic antigen (CEA), carbohydrate antigen (CA)-125 and CA19-9 levels were measured. The survival and recurrence patterns were analyzed according to the tumor-marker-related stratification. The predictive values of biomarkers and clinical variables were assessed with Cox regression and competing risk models.
Results
Patients with elevated preoperative tumor markers had evidently poor overall survival and recurrence-free survival. The triple-elevated tumor marker (TETM) subgroup had the worst overall survival and progression-free survival than the triple-negative tumor marker (TNTM) subgroup and the single-elevated tumor marker (SETM) subgroup. The most important predictors for overall survival were elevated tumor markers, FIGO-stage, tumor differentiation, lymphovascular space invasion (LVSI) and lymph nodes metastasis. The most important predictors for recurrence-free survival were elevated tumor markers, FIGO-stage, tumor differentiation, LVSI and deep stromal invasion. Stratified analysis showed that elevated CA-125 and CA19-9 were significantly associated with postoperative distant metastasis. A decision curve analysis confirmed that a combination of tumor markers as predictors significantly outperformed the other common predictors used (FIGO-stage, intermediate and high-risk factors, tumor differentiation, lymph nodes).
Conclusions
Elevated preoperative serum CEA, CA-125, and CA19-9 levels exhibited poor overall survival and recurrence-free survival in cervical adenocarcinoma patients. Combined preoperative serum CA-125 and CA19-9 independently predicted distant metastasis in patients with endocervical adenocarcinoma.
Introduction
Cervical cancer is the fourth most common gynecologic cancer worldwide and a leading cause of cancer-related deaths in developing countries with an estimated 604,000 new cases and 342,000 deaths worldwide in the global estimates of incidence and mortality in 185 countries in 2020. 1 Squamous cell carcinoma (SCC) accounts for approximately 80% of all cervical cancers while adenocarcinoma approaches about 20%. The incidence of cervical adenocarcinoma has increased over the past three decades owing to the increased detection of the human papillomavirus (HPV) infections and the growing popularity of cervical cytologic screening methods in undeveloped areas.2,3 Previous studies have found that patients with cervical adenocarcinoma have larger tumor volumes, easier nodal metastasis, are more resistant to radiotherapy and chemotherapy and have a poorer prognosis, compared to SCC.4–6 For early-stage endocervical adenocarcinoma, the 5-year recurrence-free survival (RFS) and overall survival (OS) were reported to be approximately 80–85%.7,8 Similarly to the malignant behavior of adenocarcinoma in other solid malignancies, cervical adenocarcinoma presented a high distant metastasis rate even when the primary tumor volume was small. 9
Currently, there have been several classifications for adenocarcinoma in clinical management. The most frequent usual type of cervical adenocarcinoma is HPV-related and HPV-unrelated subtypes. However, some studies reported a considerably lower prevalence of HPV in adenocarcinoma, from approximately 62–80%, because the detection of HPV in adenocarcinoma encountered some technical difficulties due to the low copy numbers of HPV-DNA.10,11 Some findings suggested that the Sedlis criteria and high-risk factors used in adenocarcinoma may differ from those in SCC.9,12,13 The prognosis of such clinicopathological factors and classification was still unclear because the research was performed only on a small number of patients.7,14 Thus, a more effective risk-factor model should be proposed in such a refractory malignancy. In our clinical practice, we found that adenocarcinoma could exhibit some specific elevated tumor markers such as carcinoembryonic antigen (CEA), carbohydrate antigen (CA) 19-9 and CA125, which were not commonly detected in SCC. Many studies in some adenocarcinoma of the digestive and reproductive tracts revealed that the tumor markers had a strong correlation with prognosis and recurrence.15–17 However, scarce studies assessed the prognostic prediction of serum CEA, CA125, and CA19-9 levels in cervical adenocarcinoma patients. As the limitations mentioned above, we designed a tumor-marker-based classification stratified by serum CEA, CA125, and CA19-9 levels, sought to explore the prognosis and recurrence patterns of each stratification and attempted to identify the possible use of such a tumor-marker-based method to be a preoperative prognostic predictor of cervical adenocarcinoma.
Materials and methods
Patients
We retrospectively extracted the electronic medical records of 2107 cases with cervical adenocarcinoma, of which 579 patients with histologically confirmed adenocarcinoma and the FIGO stage (2009) IA2–IIA disease. All patients enrolled underwent standard abdominal radical hysterectomy (Querleu-Morrow-C2, QM-C2) according to the Querleu-Morrow classification. 18 Patients with stage IA1 were exclude because of the treatment of extra-fascial hysterectomy. Systematic lymph node (LN) dissection was performed (radical hysterectomy with bilateral pelvic lymphadenectomy and para-aortic lymphadenectomy sampling is the preferred treatment for FIGO stage IA2, IB1, IB2, and select IB3–IIA1 lesions) at the Department of Gynecologic Oncology, Fudan University Shanghai Cancer Center, China, between May 2006 and January 2014. Adjuvant radiotherapy following radical hysterectomy are performed for those who have one or more pathologic risk factors (e.g., positive lymph nodes, parametrial infiltration, positive surgical, margins, large tumor size, deep stromal invasion, and lymphovascular space invasion (LVSI)).
All patients had provided written consent for their data to be used for research purposes and the study was conducted following the Declaration of Helsinki. Data on clinicopathological parameters, including age at diagnosis, preoperative serum CEA, CA125, and CA19-9 levels, FIGO stage, postoperative pathological findings (tumor differentiation, LVSI status, tumor size, depth of stromal invasion (DSI) and LN status), adjuvant treatment, date and type of recurrence, and date of death or last follow-up, were collected. The study was approved by the Ethics Committee of Fudan University Shanghai Cancer Center, Shanghai, China.
Tumor-marker-associated subgroups
Serum CEA, CA-125, and CA19-9 levels were measured using radioimmunoassay kits from Abbott Laboratories (Chicago, IL, USA). The preoperative level of serum markers was determined within 1 week before resection. The normal upper limits of serum tumor markers were adopted as follows: CEA (5 ng/mL), CA19-9 (27 U/mL), and CA-125 (35 U/mL). All cases fell into four possible subgroups, each of which was subclassified based on the tumor marker levels. Patients with normal CEA, CA-125, and CA19-9 were referred to the triple-negative tumor marker (TNTM) subgroup. Patients with only one elevated tumor marker were classified as single-elevated tumor marker (SETM) subgroup. The double-elevated tumor marker (DETM) was defined as patients with two tumor markers elevated. The triple-elevated tumor marker (TETM) was defined as patients with elevated CEA, CA19-9, and CA-125.
Follow-up and recurrence type
This study followed the Society of Gynecologic Oncology's recommendations for post-treatment surveillance. 19 The recommended surveillance was based on the patient's risk of recurrence and personal preferences. During the follow-up visits, recurrence was confirmed based on findings of biopsy and/or imaging-based examinations such as computed tomography (CT), magnetic resonance imaging, or positron emission tomography (PET). For recurrent cases, locoregional recurrence (LR) was defined as isolated pelvic recurrence, including vaginal recurrence with or without pelvic LN recurrence. Distant metastasis (DM) was defined as distant site relapse at either organs or LN area. Multiple-site recurrence was defined as recurrence both inside and outside the pelvis, and this type of recurrence was also classified as DM for analysis.
Statistical analyses
Patients’ demographic and clinicopathological characteristics are reported as frequencies. Univariable survival curves were created using the Kaplan–Meier method. Between-group comparisons were performed using the log-rank test. Cox proportional hazards regression was used for univariate and multivariate analyses. Variables with P-values <0.10 in the univariate analysis were included in the multivariate analyses. For all statistical tests, two-tailed P-values <0.05 were considered significant. A prognostic nomogram was generated based on multivariable analysis results. Harrel's concordance index (C-index) was used for each prognostic factor. 20 Clinical use and net benefit were estimated with decision curve analysis. The final model was also assessed in different models to evaluate the prognosis, including model 1 (FIGO-stage and lymph node status), model 2 (Sedlis criteria and high-risk factors), model 3 (tumor marker-associated stratification), and model 4 (multi-model combination). Data were analyzed with R version 4.0.5.
Results
Patient characteristics
A total of 579 eligible patients were screened in this study, and all patients had one or more confirmed tumor marker results. Only 496 patients who had definite CEA, CA19-9, and CA-125 results were stratified into four subgroups, including 154 (31%) in the TNTM group, 170 (34%) in the SETM group, 128 (26%) the DETM group, and 44 (9%) in the TETM group. The patients’ median age was 45 (range: 23∼72 years). A total of 188 (37.7%) showed elevated CEA levels; 36% of all cases had elevated CA19-9 levels; and 166 patients (28%) had elevated CA-125 levels. Clinicopathological characteristics were statistically different among the four groups, including tumor differentiation (P = 0.033), LVSI (P = 0.041), LN metastasis (P < 0.001), LR (P < 0.001), and DM (P < 0.001). The DETM and TETM groups had more cases with poor differentiation than cases in TNTM and SETM (29.7% and 40.9% vs. 18.4% and 24.8%, respectively). The DETM and TETM subgroups had more patients with positive LVSI (40.6% and 43.2%, respectively) and LN (39.8% and 52.3%, respectively) metastasis than TNTM and SETM subgroups (LVSI: 26.6% and 37.6%, respectively; LN: 13.0% and 37.6%, respectively). Patients in the TETM subgroup had a higher rate of LR (29.5%) and DM (41.9%) than the other three subgroups. As shown in Table 1, no statistically significant difference was found in age, FIGO stage, tumor size, DSI, surgical margin and adjuvant radiotherapy (ART) among the four subgroups.
Baseline characteristic of each subgroup.
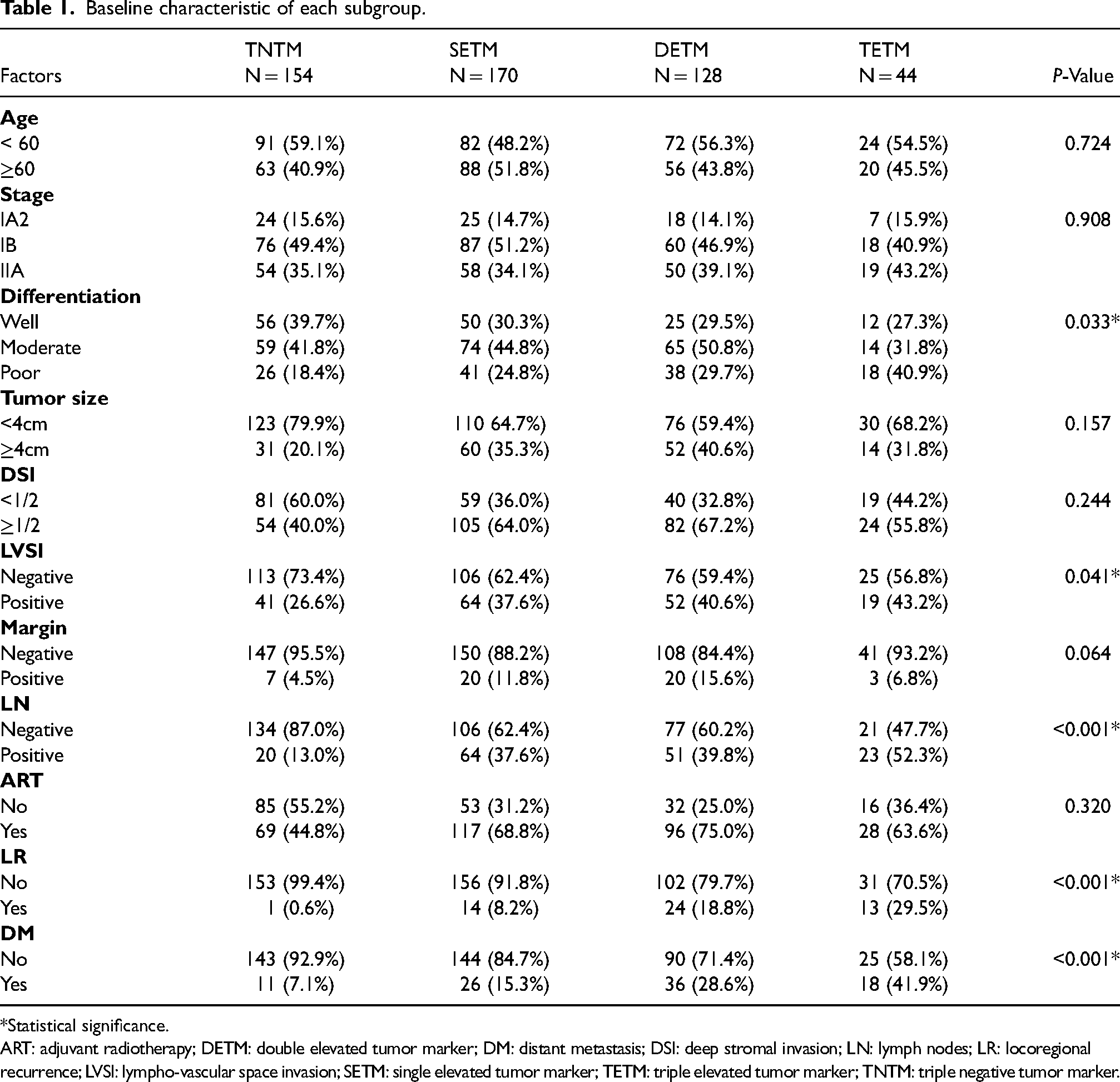
*Statistical significance.
ART: adjuvant radiotherapy; DETM: double elevated tumor marker; DM: distant metastasis; DSI: deep stromal invasion; LN: lymph nodes; LR: locoregional recurrence; LVSI: lympho-vascular space invasion; SETM: single elevated tumor marker; TETM: triple elevated tumor marker; TNTM: triple negative tumor marker.
Survival of tumor-marker-related stratification
During the follow-up period, the median follow-up time of the cohort was 59.1 (range 1.9–146.6) months. The 5-year OS and RFS rate of the overall population in this study was 81.3% and 70.8%, respectively. The 5-year OS rates in TNTM, SETM, DETM, and TETM subgroups were 92.5%, 83.6%, 63.6%, and 40.8%, respectively (P < 0.001, Figure 1(a)). The 5-year RFS rates in TNTM, SETM, DETM, and TETM subgroups were 88.5%, 80.0%, 60.3%, and 34.7%, respectively (P < 0.001, Figure 1(b)). During the follow-up period, 61 patients were identified with LR disease, and the 5-year LR rate was 11.4%. DM was identified in 86 patients, and the 5-year DM rate was 17.8%. The 5-year LR rates in TNTM, SETM, DETM, and TETM subgroups were 3.0%, 6.5%, 15.1%, and 26.6%, respectively (P < 0.001, Figure 1(c)). Similarly, the 5-year DM rates in TNTM, SETM, DETM, and TETM subgroups were 8.5%, 13.5%, 24.6%, and 38.7%, respectively (P < 0.001, Figure 1(d)).
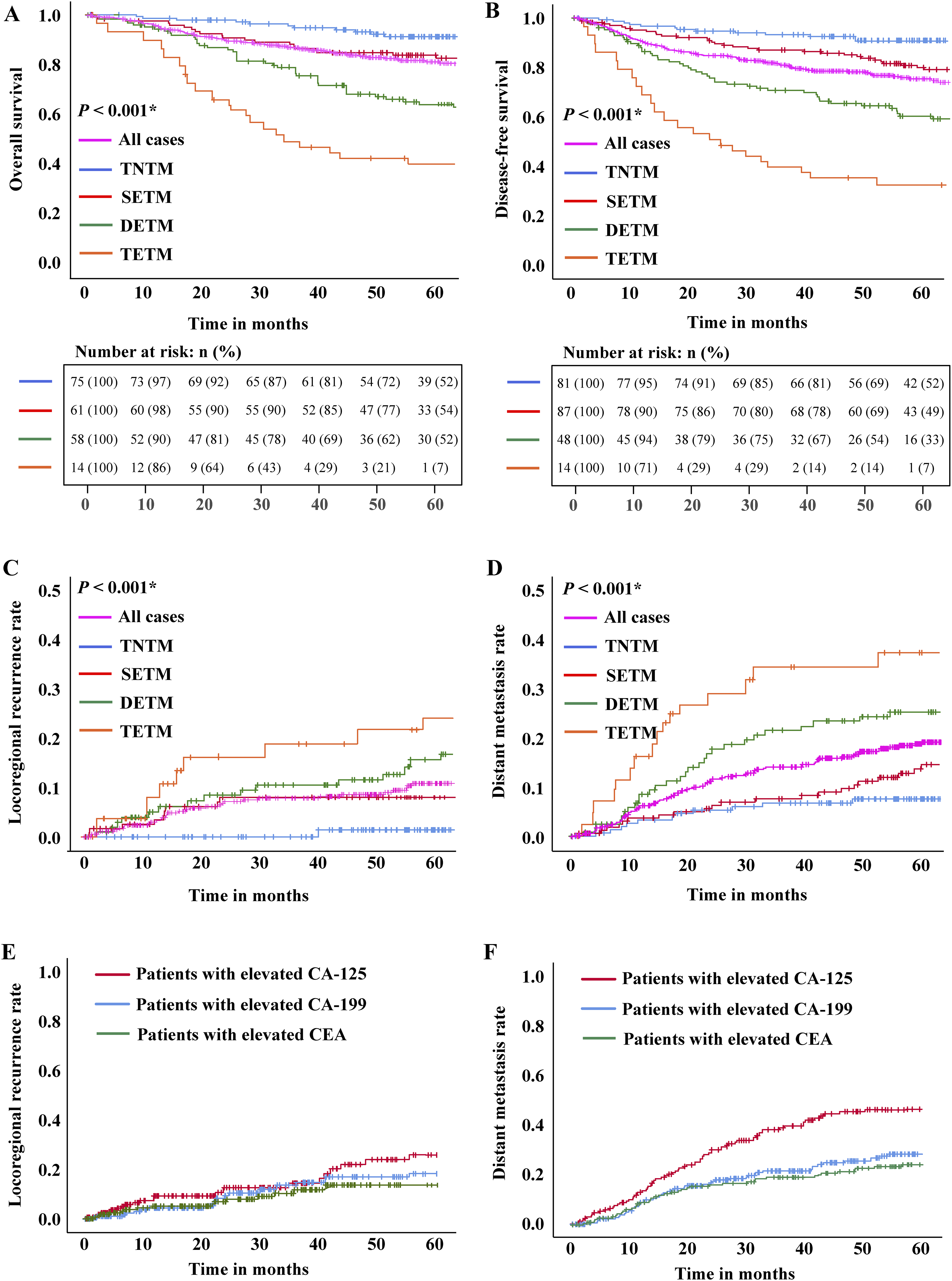
(a) The overall survival (OS) rates of 496 cases who had complete CEA, CA19-9 and CA-125 data and were stratified into 4 subgroups by tumor markers. (b) The recurrence-free survival (RFS) rates of 496 cases. (c) Cumulative rates of locoregional recurrence for tumor marker-related subgroups by the Kaplan–Meier method. (d) Cumulative rates of distant metastasis for tumor marker-related subgroups by the Kaplan–Meier method. (e) Cumulative rates of locoregional recurrence for different elevated tumor markers by the Kaplan–Meier method. (f) Cumulative rates of distant metastasis for different elevated tumor markers by the Kaplan–Meier method.
Using Kaplan–Meier analysis, the 5-year LR rates of patients with elevated CEA, CA19-9, and CA-125 were 13.1%, 18.7%, and 23.4%, respectively (Figure 1(e)). The 5-year DM rates were 23.9%, 30.7%, and 46.5% in patients with elevated CEA, CA19-9, and CA-125, respectively (Figure 1(f)).
Prognostic factors for survival and recurrence
In the univariate analysis, the factors associated with the poor OS were tumor-marker-based subgroups, FIGO-stage, tumor differentiation, tumor size, DSI, LVSI, LN status, and ART. The multivariate analysis for OS identified that DETM (hazard ratio (HR) = 3.215; 95% confidence interval (CI) 1.525∼6.775; P < 0.001), TETM (HR = 6.681; 95% CI 3.021∼14.775; P < 0.001), stage at IIA (HR = 2.095; 95% CI 1.083∼4.465; P = 0.032), tumor differentiation (HR = 2.391 and 4.300; 95% CI 1.184∼4.829 and 2.127∼8.694; P < 0.001), LVSI (HR = 1.904; 95% CI 1.208∼3.002; P = 0.006), and LN metastasis (HR = 1.735; 95% CI 1.064∼2.830; P = 0.027) were independent predictors. In the univariate analysis, the factors associated with a poor RFS were tumor-marker-based subgroups, FIGO-stage, tumor differentiation, DSI, LVSI, LN, and ART. The multivariate analysis for RFS revealed that tumor-marker-based subgroups (HR = 2.079, 4.565, and 6.923; 95% CI 1.074∼4.027, 2.407∼8.657, and 3.418∼14.021, respectively), stage at IIA (HR = 1.479; 95% CI 1.047∼2.585; P = 0.044), tumor differentiation (HR =2.288 and 3.471; 95% CI 1.322∼3.960 and 1.978∼6.091; P < 0.001), DSI (HR = 1.808; 95% CI 1.149∼2.849; P = 0.011), and LVSI (HR = 1.866; 95% CI 1.283∼2.713; P = 0.001) were independent prognostic factors (Table 2).
COX regression of OS and DFS.
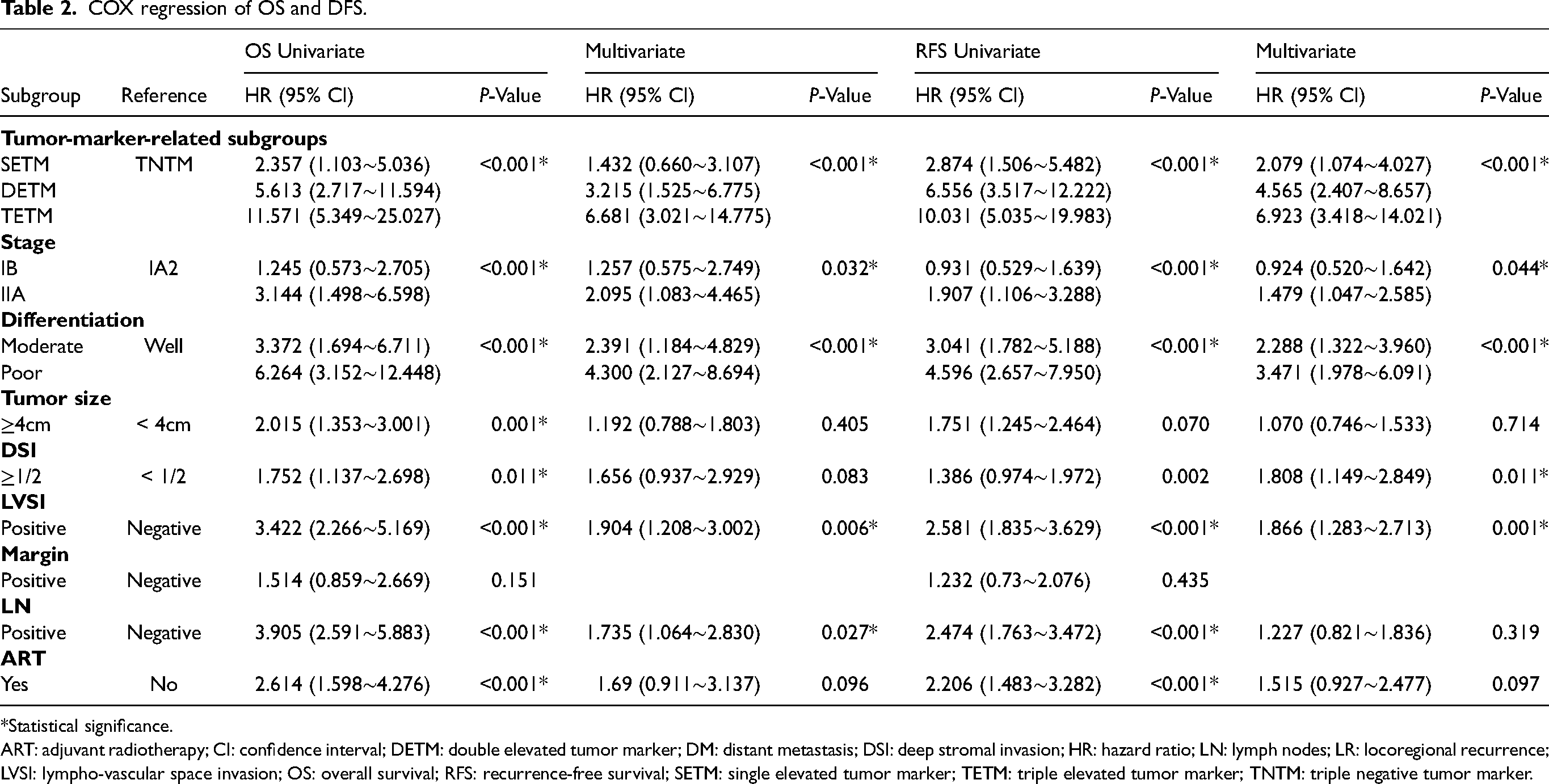
*Statistical significance.
ART: adjuvant radiotherapy; CI: confidence interval; DETM: double elevated tumor marker; DM: distant metastasis; DSI: deep stromal invasion; HR: hazard ratio; LN: lymph nodes; LR: locoregional recurrence; LVSI: lympho-vascular space invasion; OS: overall survival; RFS: recurrence-free survival; SETM: single elevated tumor marker; TETM: triple elevated tumor marker; TNTM: triple negative tumor marker.
Using the competing risk strategies, we found that patients with elevated tumor markers were more likely to have recurrent disease. Multivariate analysis showed that there were no independent prognostic factors identified in the risk of tumor-marker-associated LR. However, we found that the elevated serum CA-125 and CA19-9 were the independent factors for DM occurrence (P < 0.001), and patients with elevated CA-125 and CA19-9 were more likely to have DM disease (P = 0.048). Especially for patients with elevated CA-125 levels, the risk of DM occurrence was 4.66 times higher than those with normal CA-125 levels (HR = 4.66; 95% CI 3.33∼6.96; P < 0.001) (Table 3).
Recurrence patterns related to different elevated tumor marker.
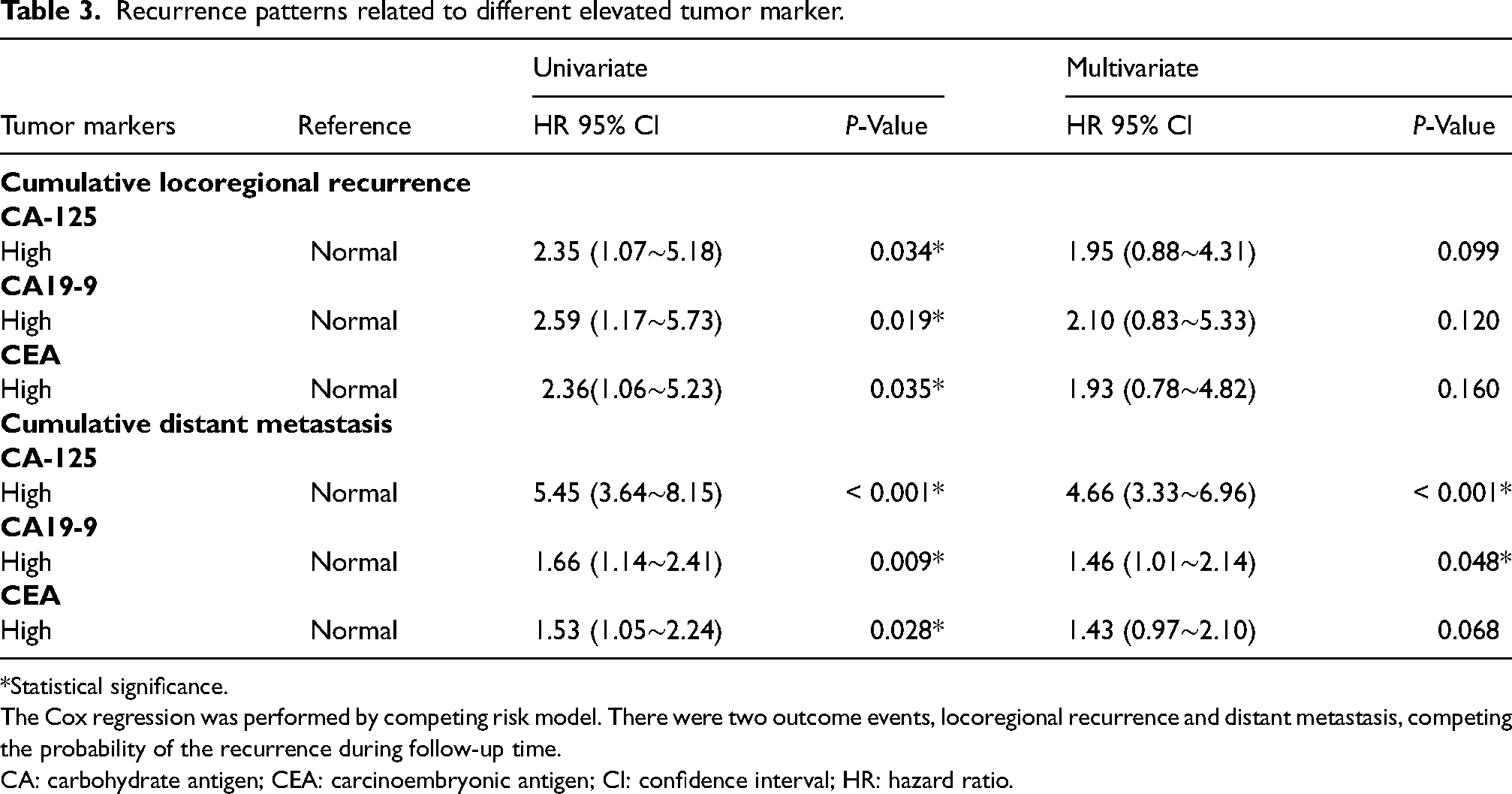
*Statistical significance.
The Cox regression was performed by competing risk model. There were two outcome events, locoregional recurrence and distant metastasis, competing the probability of the recurrence during follow-up time.
CA: carbohydrate antigen; CEA: carcinoembryonic antigen; CI: confidence interval; HR: hazard ratio.
Comparison between different predicted factors
The clinical-marker-based survival risk score was developed from clinical variables that were commonly used for prognosis after multivariate Cox regression. We found that the tumor-marker-based model was the most predictive of both 5-year OS and RFS among the five predictive models. The weights and scores associated with the five variables are shown in nomograms for OS and RFS (Figure 2(a) and (b)) and were tested by C-index (Supplementary Table 1). The tumor-marker-based model achieved a high value of C-index for both OS (C-index = 0.71) and RFS (C-index = 0.72). The multi-factors combination (Model 4) achieved the strongest prognostic effect for both OS and RFS (C-index = 0.81 and 0.76, respectively) (Supplementary Table 1). Decision curve analysis was used to facilitate the comparison between different prediction models. The decision curve analysis showed that the tumor-marker-based subgroups provided a larger net benefit across the range of death and recurrence risk compared with the common use of predictive factors (FIGO-stage, intermediate and high risk, tumor differentiation). The multi-factors combination (model) provides the largest net benefit across a large range of 5-year death risk and recurrence risk (Figure 2(c) and (d)).
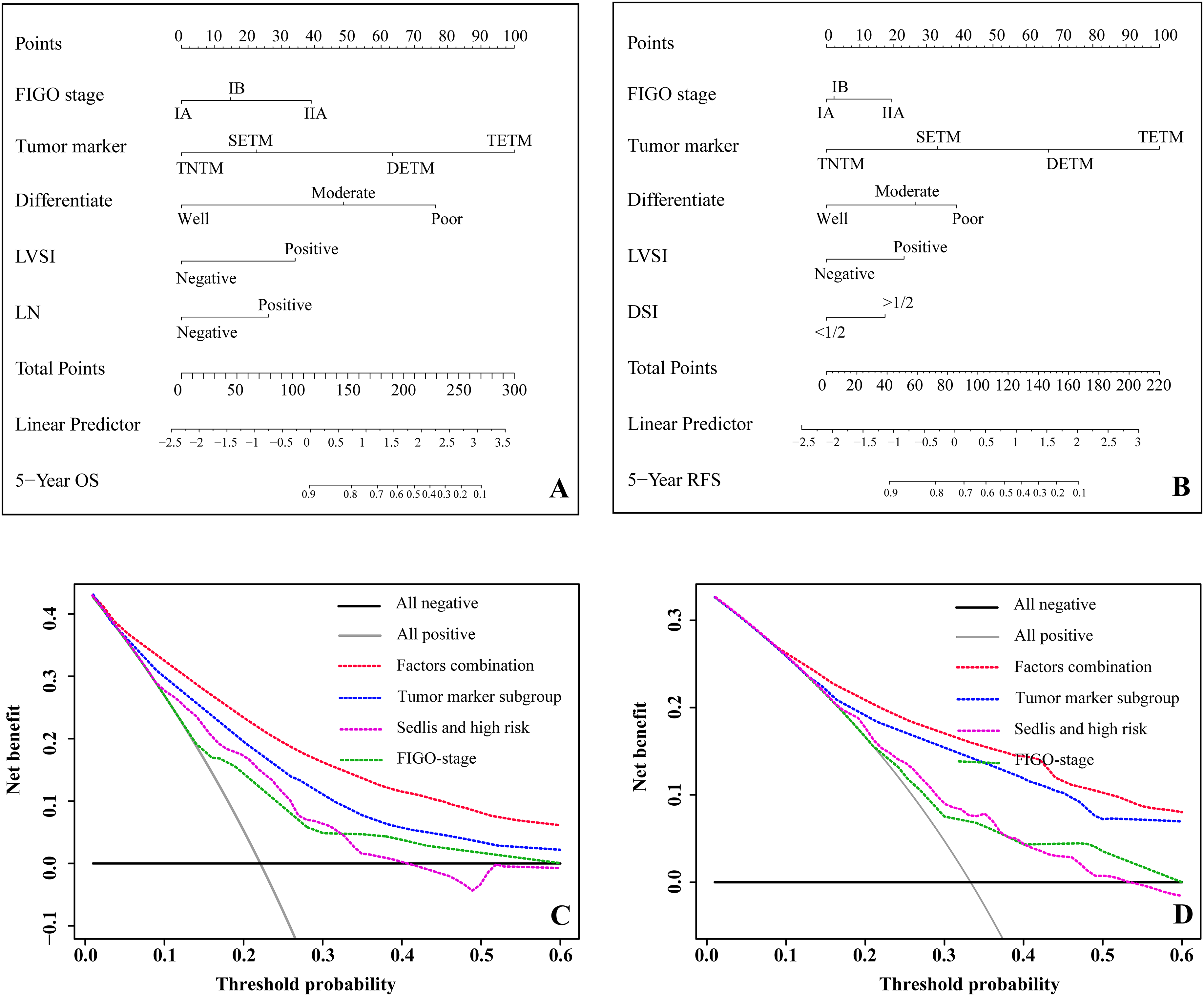
(a) Nomogram of 5-year OS for all prognostic factor combinations selected by multivariate Cox regression. (b) Nomogram of 5-year RFS for all prognostic factor combinations selected by multivariate Cox regression. (c) Decision curve analysis for the death risk prediction models. The x-axis was the threshold probability of death. The intermediate and high-risk factors included the deep stromal invasion (DSI), lymphovascular space invasion (LVSI), lymph nodes (LN) and tumor size. (d) Decision curve analysis for the recurrence risk prediction models. The x-axis was the threshold probability of recurrence.
Discussion
There are a lot of challenges in diagnosing, treating, and monitoring women with cervical adenocarcinoma. The prediction for the prognosis of the disease and the extent of recurrence is particularly difficult for this entity. Because of the increasing incidence and persistent poor prognosis of cervical adenocarcinoma, it is warranted that the pursuit for effective prognostic tumor biomarkers is urgent and evident. Unlike cervical squamous cell carcinoma (SCC), which has specific risk factors (Sedlis Criteria), there is no consensus on definite risk factors for both recurrence and OS. Although previous studies have revealed that FIGO stage, lymphovascular space invasion, and lymph node metastasis were significantly associated with PFS and OS in patients with adenocarcinoma, there is still a lack of evidence on pretreatment biomarker monitoring and predictors for recurrence after treatment. 21 To overcome the shortage, the current study aimed to identify the robust tumor markers as prognosis predictors of cervical adenocarcinoma and concluded that elevated preoperative CA-125, CA19-9, and CEA might have significant prognostic value. Furthermore, elevated serum CA-125 and CA19-9 were the independent effect factors for DM, which defines a group of patients at high risk for remote recurrence and mortality.
Some tumor-associated antigens measured in serum from patients with malignancy were found to be related to the clinical course of the disease. Tumor biomarkers, such as CA-125, offer a novel solution for the evaluation of tumor burden of epithelial ovarian and endometrial tumors during the treatment. 22 For SCC, the serum expression level of SCC antigen (SCC-Ag) is found to be an effective way to evaluate before and during the treatment of SCC. 23 Similarly, CA19-9 and CEA were found to be specifically correlated to the clinical stage in gastrointestinal tumors. 24 According to a study by Fujii et al., 25 compared to the normal cervical mucus, the cervical mucus samples from cervical adenocarcinomas showed a higher ratio of (CEA + CA19-9)/CA-125, suggesting that CA-125, CA19-9, and CEA might have roles in cervical adenocarcinomas. Our study collected clinicopathological information of cervical adenocarcinoma patients and analyzed the association of pre-operative serum tumor markers (CA-125, CA19-9, and CEA) with prognostic value, to further explore the predictive risk factors. Results from our study may help offer evidence for the use of pre-operative serum tumor markers for prognostic assessment in the surveillance of cervical adenocarcinomas.
Although previous studies have revealed that an elevated value of serum CA-125 (> 35 U/mL) was found in more than 70% of cervical adenocarcinomas, 26 there were limited data concerning the role of CA-125 in cervical adenocarcinoma. Moreover, pretreatment CA-125, CA19-9, and CEA were found to be positively correlated with clinicopathological characteristics in cervical adenocarcinomas. 27 Similar findings in our study revealed that elevated CA-125, CA19-9, and CEA were significantly associated with tumor differentiation, LVSI, lymph nodes metastasis, and recurrence. Survival analysis demonstrated that elevated preoperative tumor marker patients have a shorter RFS and OS, compared with normal tumor markers, indicating a poor prognosis.
Different studies used multiple tumor markers to identify the optimal prediction model for prognosis. Zhu et al. 22 found the combination of both CA-125 and CA19-9 was an independent predictor of the prognosis of ovarian clear cell carcinoma. In the current study, we combined three tumor markers (CA-125, CA19-9, and CEA) and analyzed the prognostic predictors by dividing CA-125, CA19-9, and CEA into four subgroups. Among the subgroups, TETM patients had the worst RFS and OS. Multivariate analysis also verified that a combination of elevated CA-125, CA19-9, and CEA simultaneously was an independent predictor for poor DFS and OS of cervical adenocarcinoma. Huang et al. 28 studied the preoperative serum CA-125 and CEA and revealed that combined elevated preoperative serum CEA and CA125 levels independently predicted the prognosis of early-stage cervical adenocarcinoma.
It is intriguing in our study that patients with elevated serum CA-125 and CA19-9 were related to a higher risk of DM than LR occurrence. The tumor-marker-based survival model shared the highest C-index for both OS and RFS, compared to other clinical models. Decision curve analyses were further used to verify the predictive benefit of tumor-marker-related subgroups. Most studies on tumor markers only analyzed the effect of tumor markers on recurrence and survival, neglecting the potential predictors for specific recurrence patterns. Levy et al. 29 evaluated the survival of patients with epithelial ovarian cancer recurrence isolated to the retroperitoneal lymph nodes and found that CA-125 was significantly lower in the retroperitoneal recurrence group at diagnosis, end of treatment, and recurrence, compared to the intraperitoneal recurrence group. For patients with gastric cancer, CEA level was associated with earlier detection of recurrent peritoneal dissemination and a poor prognosis. 30 Such combined analysis of these markers is therefore considered to be helpful to accurately evaluate the recurrent sites and prognosis for solid tumor patients.
Our study included a large sample size of cervical adenocarcinoma patients and was initially designed to evaluate serum tumor markers on their diagnostic performance for recurrence detection and survival. Interpretation of the study is limited in that it is a single-institution retrospective study. Results from our study should always be validated in a future dataset or, preferably, a prospective study. A partial relationship between recurrence sites and the elevation of tumor markers might have confusion bias. However, the fact that the multivariate analysis of subgroups could reduce the bias in our study achieved statistical significance and substantiates the strength of the effect measures under study. Future studies should be designed to detect specific tumor markers before and after treatment, and even before relapse, achieving the purpose of whole-course surveillances.
Preoperative serum CA-125, CA19-9, and CEA testing appeared related to multiple clinicopathological characteristics and were predictive for survival and detection of tumor relapse in cervical adenocarcinoma patients. The combination of all tumor markers has evident superiority over either one, or different markers could be associated with disease recurrence patterns, which may provide additional treatment options. Prospective studies should be designed to validate the utility of the tumor markers in the management of cervical adenocarcinoma.
Supplemental Material
sj-docx-1-jbm-10.1177_03936155231206839 - Supplemental material for Identifying tumor markers-stratified subtypes (CA-125/CA19-9/carcinoembryonic antigen) in cervical adenocarcinoma
Supplemental material, sj-docx-1-jbm-10.1177_03936155231206839 for Identifying tumor markers-stratified subtypes (CA-125/CA19-9/carcinoembryonic antigen) in cervical adenocarcinoma by Zongkai Zhang, Yin Li, Ying Wu, Rui Bi, Xiaohua Wu, Guihao Ke and Jun Zhu in The International Journal of Biological Markers
Footnotes
Acknowledgments
We thank all the faculty members in our department and all the patients, and their families involved in the current study.
Author contributions
Conception and design: Zongkai Zhang, Guihao Ke, Xiaohua Wu, and Jun Zhu; collection and assembly of data: Zongkai Zhang, Guihao Ke, and Jun Zhu; data analysis and interpretation: Zongkai Zhang and Jun Zhu; pathological slides reviewing: Rui Bi; manuscript writing: Zongkai Zhang, Guihao Ke, and Jun Zhu; and final approval of manuscript: Zongkai Zhang, Rui Bi, Xiaohua Wu, Guihao Ke, and Jun Zhu. Zongkai Zhang and Yin Li contributed equally to this work.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China, (grant number Youth Fund no. 81902640).
Data availability
The datasets supporting the conclusions of this article are included within the article. Any request for data and material may be sent to the corresponding author.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
