Abstract
Breast cancer is the most common malignancy in women worldwide. Administration of oncolytic viruses is one of the novel promising cancer therapy approaches. Replication of these viruses is usually limited to cancer cells that have interferon (IFN) signaling defects. However, Interferon signaling is not completely impaired in all cancer cells which may limit the benefits of virotherapy.
Identification of realistic IFN-mediated biomarkers to identify patients who most likely respond to virotherapy would be helpful.
In this study, eight patients-derived primary tumor cultures were infected with an ICP34.5 deleted oHSV, then the rate of infectivity, cell survival, and expression of the gene involved in IFN pathway were analyzed.
Data showed that mRNA expressions of Myeloid differentiation primary response protein (Myd88) is significantly higher in tumors whose primary cultures showed less cell death and resistance to oHSV infectivity (
Identifying such biomarker improves our ability to select the patients who do not exhibit resistance to virotherapy.
Keywords
Introduction
Breast cancer, the most frequent malignancy in women worldwide, is divided into 5 main subtypes based on the presence or absence of molecular markers for estrogen or progesterone receptors (ER/PR) and human epidermal growth factor (ERBB2; formerly HER2). According to these pathologic criteria, invasive ductal carcinoma (IDC) accounts for 50% to 75% of breast cancer cases, while invasive lobular carcinoma (ILC) accounts for 5% to 15% of cases. Mixed ductal/lobular carcinoma and other less common histology make up the remaining cases [1]. 70% to 80% of patients with early-stage, non-metastatic breast cancer can be cured, while advanced breast cancer with distant organ metastases is still regarded as incurable with currently available treatments [2].
Novel breast cancer treatment concepts emphasize the need for multidisciplinary management of this heterogeneous disease and treatment based on tumor biology as well as individualization of therapy. One of the most recent developments in immunotherapy is virotherapy by oncolytic virus administration, which can selectively replicate in and kill cancer cells while causing no harm to normal tissues [3]. The first effort in the design and construction of a viral genome to achieve a cancer cell-specific replicative virus was done in 1991 and resulted in a HSV-1 with a mutation in thymidine kinase (TK) for the treatment of brain tumors [4]. Several mutants are currently underway in clinical trials for a variety of cancers, e.g. T-VEC (Talimogen laherparepvec, IMLYGIC, formerly OncoVEX
As it appears that oncolytic virotherapy efficacy could be cell line context-dependent, and cancer cells characteristics, microenvironment, and cell signaling play a critical role in response to OVs [6]. One possible candidate for such a mechanism in vivo is the interferon responses. Following viral infection, a class of cytokines named interferon (IFNs) is produced and signals the infected and non-infected cells to prevent proliferation and downregulate metabolism, to preclude viral replication [7, 8]. Type I IFN (IFN
Material and methods
Sample collection and patient-derived primary tumor cell culture
Twenty tumor samples and the marginal zone from the same non-diabetic luminal A and B breast cancer female patients (aged
A part of the tissue was preserved in RNA later (Cat No. 1234b, Invitrogen) at
Virus propagation and titration and ex vivo culture infection
oHSV-RR were propagated in VERO (African green monkey kidney cell) (Cat no. C101, National Cell Bank of Iran) in DMEM supplemented with 10% FBS at 37
For confirming that the virus’s ability to infect patient tumor cells is varied, oHSV-RR was added to 8 different primary tissue cultures derived from 8 patients at a multiplicity of infection of 1 (MOI 1) in a serum-free medium. Two hours after infection, oHSV-RR inoculum was discarded. Cells were washed with PBS and fresh media containing 2% FBS was replaced. At 48 h post-infection, tumor cells in a 24-well plate were observed by a CETI inverted fluorescent microscope (Medline Scientific, UK). Changes in morphology were quantified as cytopathic effect (CPE) foci while the red fluorescent dye intensity and virally infected area of every well across the whole plate area were calculated by ImageJ software (Version 1.52a, National Institutes of Health, Maryland, US).
Cell death measurement by PI staining flow cytometry
The isolated tumor cells from primary tissue cultures were infected with oHSV-RR at MOI 1, then incubated for 48 h beside the non-infected control. All samples were detached with trypsin-0.53 mM EDTA and harvested at 400
RNA extraction and RT-qPCR
The tumor cells derived from the aforementioned 8 primary cultures onto a 24-well plate were overlaid with TRIzol (Cat no. 15596026 ThermoFisher Scientific, USA). Total RNA was isolated following the manufacture’s protocol and treated with RNase-free Dnase. The enzyme was inactivated at 80
Reverse transcription was primed by oligo (dT) primers from 1
All qPCR assays were conducted in triplicated reactions on Step one plus
The mRNA level of test was normalized to reference genes (geometric mean of GAPDH and RPL13A). Primer pairs as shown in Table 1 were designed with the IDT (Integrated DNA Technologies, Inc. USA) algorithm and Allele ID software V7.70 (Primer Biosoft) and checked for the secondary structure consideration and scoring criteria by Gene Runner V 6.5.52x64 Beta, oligo 7 software V 7.56 (Molecular Biology Insights, Inc.) and integrated DNA technologies (IDT) online tool. Finally, specific products were in-silico checked by NCBI database primer-blast and validated in vitro by gel electrophoresis of qPCR products to generate a single-size product on 2% gel and analysis of their melting curve in all experiments. The primers specific for target genes span exon-exon junctions or multi-exons separated by a large intron in qPCR. To determine the annealing temperature of the primers, gradient quantitative q-PCR reactions were performed and for PCR efficiency calculation by REST software version 2.0.13 (Qiagen), a standard curve in 5 serial dilutions (500 ng/
Sequence of primers designated for qPCR
Sequence of primers designated for qPCR
Statistical tests were performed using R software version 4.2.1 (2022-06-23 ucrt) and SPSS version 26.
The ratio of PI positive cells was measured in each patient-derived primary tissue culture. An arbitrary cut off of 10% survival compared to the untreated corresponding sample was used to define sensitive tumors. Unpaired
The predictive power of genes was tested by area under the receiver operating characteristic (ROC) curve analysis (AUC, 95% confidence interval). The best cut off for discrimination between resistant and sensitive samples was also determined by ROC curve analysis.
The primary tumor cell derived from patient’s tissue are able to successfully adhere (a) after 5 h and proliferate (b) after 24 h.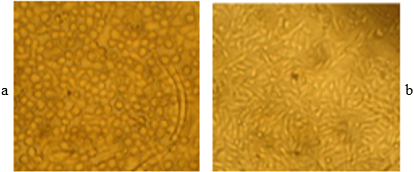
Patient-derived tissue culture of luminal samples
24 breast tumor samples (consist of 20 fresh and 4 cryopreserved samples) with the biological size of 1.2 grams (standard deviation
Different sensitivity of tumor cells derived various patients to oHSV-RR infection. A sensitive primary tissue culture by light vision (a) and UV vision (c). A resistant primary tissue culture by light vision (b) and UV vision (d). The mean score of red color of the sensitive group is significantly higher than the resistant group (graph e) (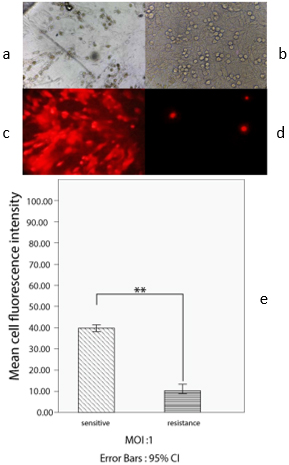
To assess the sensitivity of patient-derived primary tissue culture to oHSV-RR, the ability of the virus to make CPE and the red fluorescent dye intensity of BleCherry were microscopically monitored 48 h post-infection. The comparison of mean scores for red fluorescent intensity as analyzed by ImageJ software reveals that oHSV1-RR infection and the virally infected area of every well across the whole tumor cell culture area in primary tissue culture derived from four patients’ numbers (sensitive group) is significantly higher than four other patients (resistant group) (
The correlation between dead cell ratio in flow cytometry and BleCherry expression in fluorescent microscopic examination of tissue cultures
Flow cytometry data analysis by FlowJo-V10 software was concordant with the microscopic observation data showing different sensitivity to oHSV-RR. 48 h post-infection, PI positive population of oHSV-RR infected at MOI 1 was subtracted from PI positive population of non-infected control cells (Fig. 3a and b). Data has shown that the mean percentage of PI-stained cells of primary cultures derived from four sensitive patients is 6 times higher than the other four resistant patients (Unpaired
Different dead tumor cell ratio of various patients following oHSV-RR infection by Flow cytometry. The PI-positive population of oHSV-RR infected subtracted from non-infected control culture in a sensitive tumor (a) in a resistant tumor (b). Unpaired 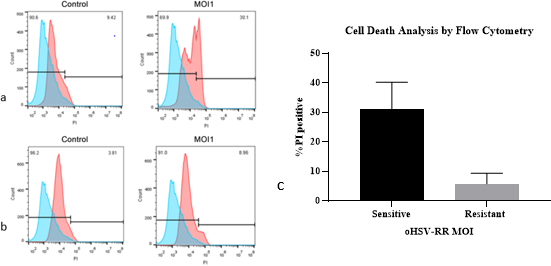
Among the studied interferon pathway genes only the expression of Myeloid differentiation primary response protein (MyD88) was distinctively elevated in original biopsies of patients whose primary tissue cultures were resistant to oHSV-RR (
The higher expression of MyD88 in tissue whose primary tissue cultures were resistant to oHSV-RR Two-Sample 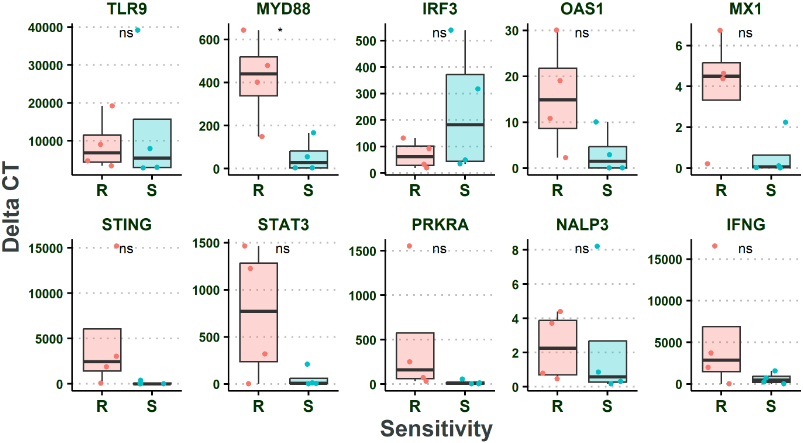
No significant change in gene expression during culturing of tumor cells. Two Sample 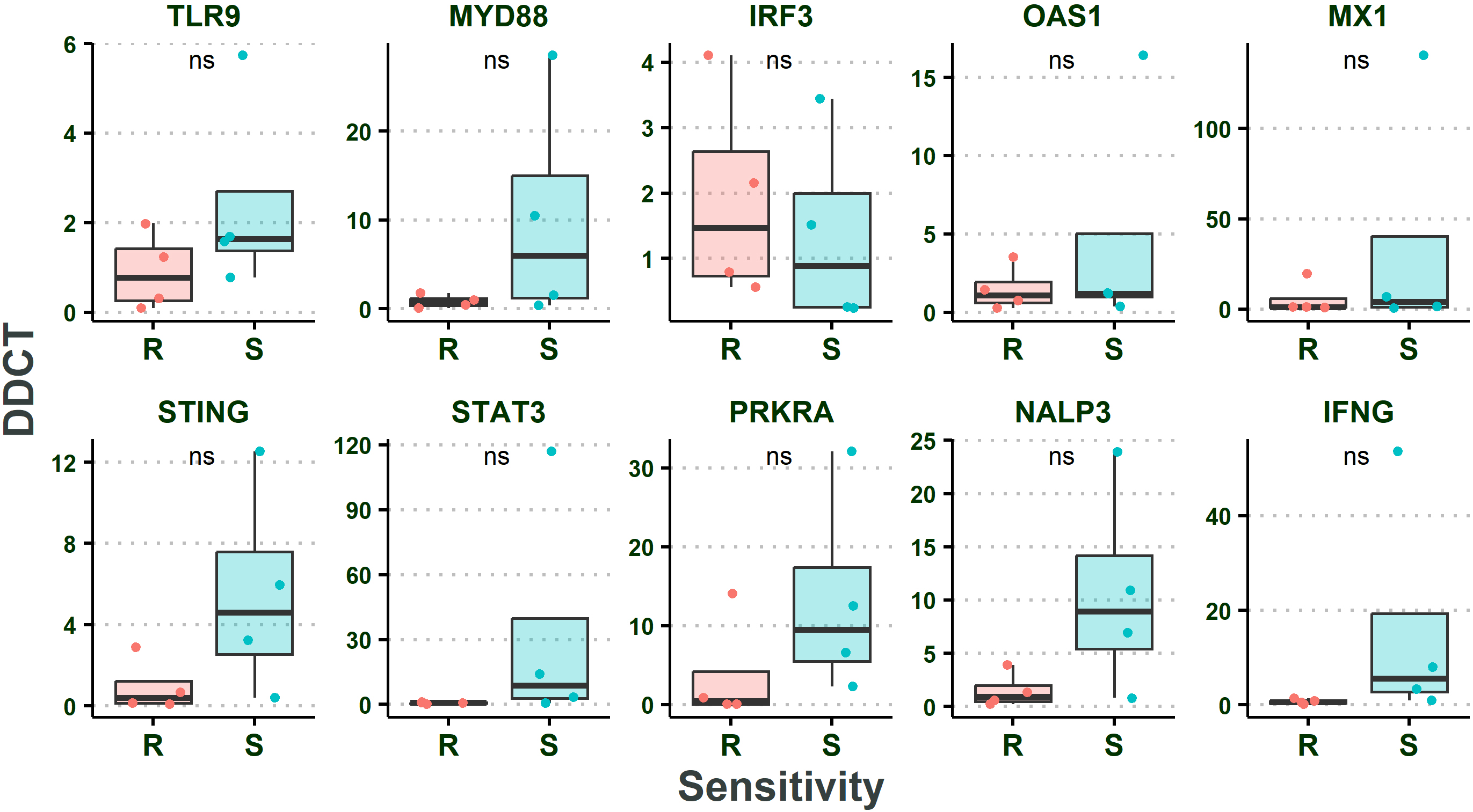
Besides, there was no significant difference between the expression levels of the studied genes (normalized to GAPDH and RPL13A housekeeping genes) in the non-infected primary tissue culture compared to the same gene in the tissue biopsy samples indicating that the expression of these genes were not affected by culturing (Two Sample
There was also no significant change in the expression level of the targeted genes post infection in patient-derived primary tissue culture that showed resistance to ‘oHSV-RR compared to the primary tissue cultures in the sensitive group (Two Sample
No significant change in resistant primary tissue cultures gene expression compared to sensitive group. Two-Sample 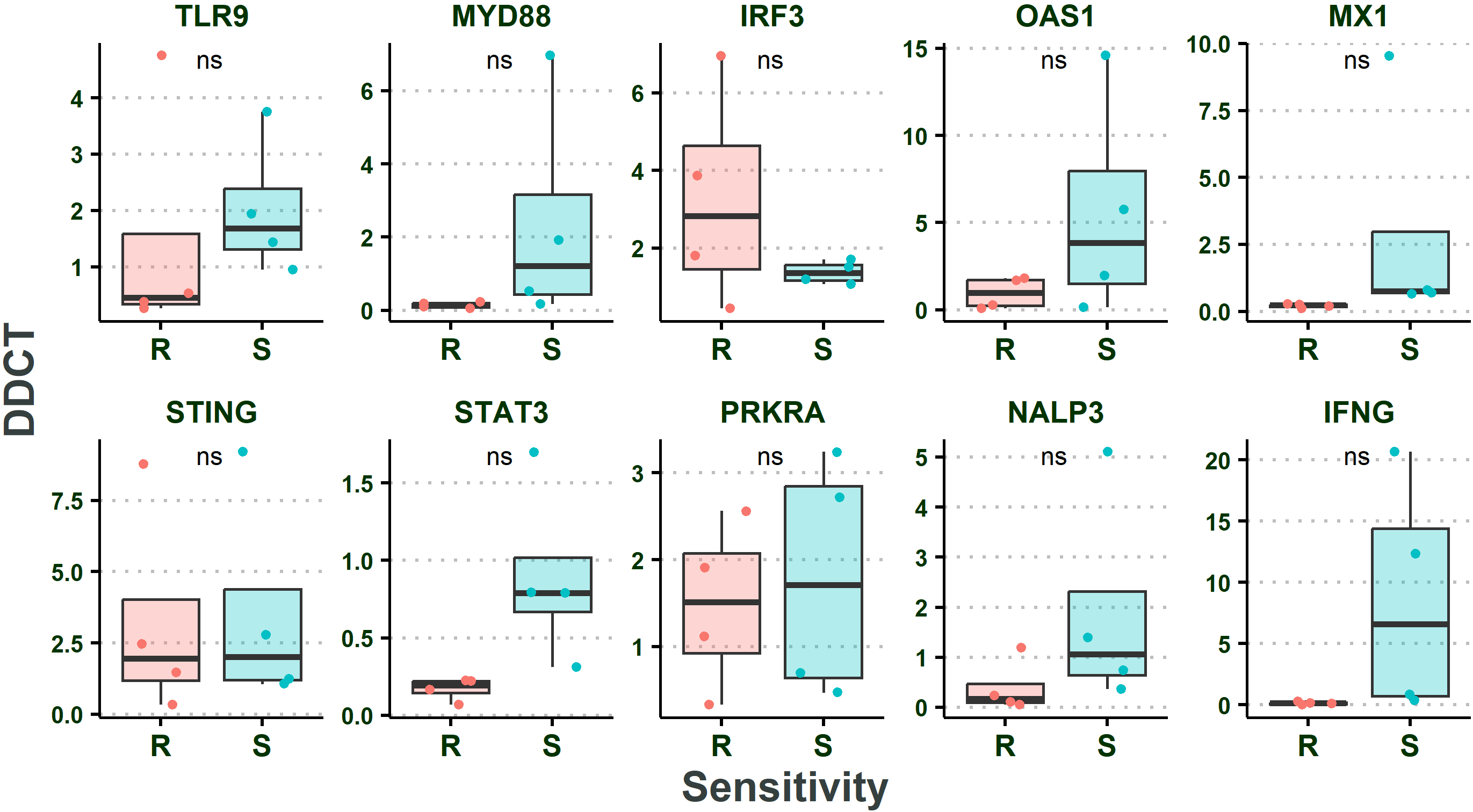
Our data show a significant difference in MyD88 gene expression in breast cancer biopsy between patients whose tumors primary cultures exhibit resistance or sensitivity to oHSV-RR infection. Indeed, our results show the variability of breast cancer patients in responding to virotherapy and proposes MyD88 as a biomarker to predict whether patients may benefit from virotherapy. Two Sample
Receiver operating characteristic (ROC) of MyD88. The value of area under curve (AUC), with 95% confidence interval for diagnostic experiment. AUC 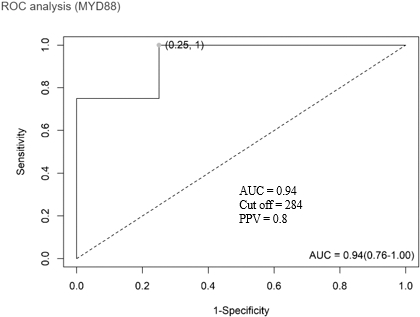
We have studied the fold increase ratio of targeted genes expression in these twenty fresh tumor and four cryopreserved samples. The ROC curve indicates that a patient with the expression level ratio of Myd88 to reference genes (geometric mean of GAPDH and RPL13A) of more than the cut off 283.9 is most likely resistant to oHSV-RR virotherapy (sensitivity 100%, specificity 75%), as shown in Fig. 7. Applying this cut off on the 16 not-cultured samples indicated that MyD88 expression in one tumor sample is highly more than cut off (766.6), while 2 others are very close this cut off. As a result, 81 percent’s (13 out of 16) of patients of this study, may benefit from virotherapy and 6–19 percent probably may not. It is noteworthy that both the culture process and viral treatment of the sensitive samples mildly increased the expression of Myd88, however this increase is not comparable with hundred folds difference between the original biopsies of two groups. This may further support the hypothesis that the mechanism of inducing resistance to oHSV-1 is related to high primary level of MyD88 in the tumor cells (Figs 5 and 6).
Type I IFN signaling pathway is a crucial aspect of innate immunity that modifies the intracellular environment by activating a network of ISGs encoding proteins with antiviral, anti-proliferative, and immunomodulatory effects and signals the presence of a viral pathogen to the adaptive immune response [21]. MyD88 is an intermediary molecule that is recruited by toll-like receptors (TLRs). HSV-1 nucleic acid is detected by TLR9 and downstream protein MyD88 is recruited via the toll/IL-1 receptor (TIR) domain. TLR9-MyD88 acts as a primary DNA sensing mechanism and initiates a downstream signaling cascade terminated by NF- kB-mediated rapid inflammation that is characterized by increased expression of a number of interleukins and cytokines and subsequently traffics to lysosome-related organelles (LRO) to induce type I IFNs. MyD88 is also involved in the formation of discrete protein complexes to activate interferon regulatory factor 3 (IRF3) and IRF7 for the expression of IFN
Myd88 is employed by almost all TLRs and activated against a broad class of pathogen-associated molecular patterns (PAMP) including RNA, DNA, and lipopolysaccharide (LPS) and the critical role of this key protein to control the pathogen infection is reported previously [26].
ROC of Mx1, STING, and PRKRA. The value of area under curve (AUC), with 95% confidence interval for diagnostic experiment. AUC 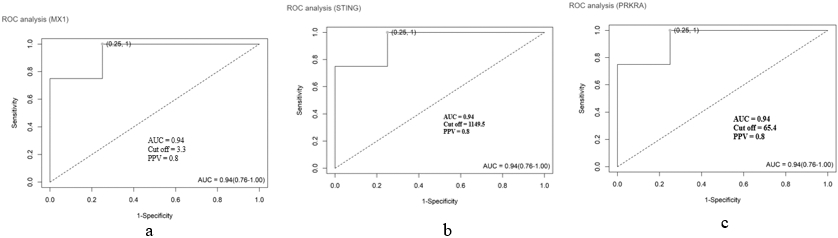
Numerous findings confirm that the different genes involved in the IFN pathway mediate the resistance to virotherapy to various types of oncolytic viruses. It seems that these inconsistent outcomes depend on the type of cell, oncolytic virus, root of administration, and other factors. In 2012, Grdzelishvili et al. analyzed the expression of 33 genes associated with type I IFN pathways, including IRF3, PRKRA, MxA, and OAS, in a panel of 11 human PDA (Pancreatic Ductal Adenocarcinoma) cell lines in response to different viruses. The type I IFN signaling status of PDA cells was shown to be heterogeneous, and MxA and OAS are suggested as potential biomarkers for resistance to VSV in highly resistant PDA cells. However, HSV and vaccinia virus’s potential to infect and kill PDA cells (unlike VSV) did not depend on the type I IFN signaling [27]. Wenmin Fu et al. in 2021 [28] also found that Mx2 protein mediates tumor resistance to oHSV-1 infection in human glioblastoma cell line A172 which intrinsically accumulated this protein at the highest levels. Likewise, it was found that MyD88 mRNA levels in A172 cells fell as a result of Mx2 depletion and Mx2 knocking down. IFN production, IRF3 activity, and NF- kB signaling decrease subsequently, which to increased virus yield in A172 cells.
Except for Myd88, our finding does not support the significant difference in other targeted genes like Mx1 (
Taken together, we have, for the first time, provided the experimental evidence of an IFN pathway-depended gene related to resistance to virotherapy with an oHSV-1 in breast cancer patient-derived tissue culture which more closely resembles the patient tissue in vivo than the established breast cell lines used in previous studies. Our data show that the mRNA level of target genes in patient-derived primary cultures did not significantly differ from their biopsy tissues (
Patient-derived culture in 3D model is a more promising approach which can mimic the complexities of tumor more than monolayer culture system. The tumor microenvironment is complex and the core (center) of tumor is less oxygenated and more acidic. It seems that 3D models can mimic these condition and affect the HSV-1 replication and induce resistance to virotherapy, consequently [31]. In silico analysis recently has shown the networks of differentially expression genes as potential hypoxia biomarkers which may affect the oncolytic virus replication too (Shayan et al., in-press). mRNA profiling of such experimental models through RNASeq and combination of OVs with IFNs modulators can identify more (IFN-dependent/independent) biomarkers involved in resistance to virotherapy and promote our understanding for personalized medicine. Besides, knocking down/out MyD88 in resistant cell lines may also provide more evidence to support usefulness of this gene as a marker for oncolytic virotherapy. Evaluating the correlation between MyD88 expression in blood and tumor samples could provide a non-invasive biomarker in the future studies. If such a correlation exist, blood sample will be a better choice to look for an expression cut-off to predict the virotherapy resistance.
Breast cancer is not a homogenous disease but a diverse set of diseases with heterogeneity in protein expression. This heterogeneity alters patients’ response to virotherapy and proves the need for biomarkers to predict how each patient will react to the therapy.
A deeper understanding of the IFN pathway dependent/independent key players to determine the viral sensitivity of the patients’ tumor cells to OV is vital for the selection of only those patients who are more likely to benefit from OV treatment to decrease the costs and side effects of treatment. The markers inducing OV resistance give us new insights regarding the effectiveness of combinatorial therapy approaches to overrule those resistance genes and construct a new generation of gene-manipulated OVs that override the genes involved in virotherapy resistance too.
Author contribiution
Zahra Nejatipour: conception, interpretation and analysis of data, preparation of the manuscript, revision for important intelectual content.
Mahdieh Shokrollahi Barough: interpretation of data.
Ladan Teimouri Toulabi: revision for important intelectual content, supervision.
Ramin Sarrami Forooshani: revision for important intelectual content.
Mohammad Farahmand: analysis of data.
Alireza Biglari: Correspond Author, Supervision, preparation of the manuscript.
Kayhan Azadmanesh: Correspond Author, Supervision, conception, interpretation and analysis of data, preparation of the manuscript, revision for important intelectual content.
Funding
Zanjan University of Medical Sciences.
Avalability of data and materials
All dataset could be accessed. But any transfer ot the patients’ samples needs authorization by the national ethical board of the ministry of health.
