Abstract
BACKGROUND:
Prostate cancer (PCa) is one of the most common malignancies in men. PCa is difficult to detect in its early stages, and most patients are diagnosed in the middle to late stages. At present, drug therapy for advanced PCa is still insufficient. Some patients develop drug resistance in the later stage of therapy, which leads to tumor recurrence, metastasis and even treatment failure. Therefore, it is crucial to find new and effective drugs to treat prostate cancer.
OBJECTIVE:
The aim of this study was to investigate the anti-cancer effect of salidroside, an active ingredient in a traditional Chinese herbal medicine, on PCa.
METHODS:
Two human PCa cell lines, PC3 and DU145, were cultured and treated with salidroside. Cell viability and proliferation ability were analyzed through CCK-8 and colony assays, and cell migration ability was detected by Transwell and Scratch assays. RT-PCR and WB were used to detected the expression levels of moleculars related to cell proliferation, apoptosis, migration, and AKT signaling pathway. Forthmore, we performed rescue experiments with agonist to verify the affected signaling pathway.
RESULTS:
Salidroside inhibited the proliferation, colony formation, and migration of PCa cells. Meanwhile, apoptosis of PCa cells was enhanced. Moreover, salidroside inhibited PI3K/AKT pathway in PCa cells. The treatment of AKT agonist 740Y-P abrogated the inhibitory effect of salidroside on the PI3K/AKT signaling pathway.
CONCLUSIONS:
Our study demonstrated that in PCa cells, salidroside inhibites proliferation and migration and promots apoptosis via inhibiting PI3K/AKT pathway.
Introduction
According to the latest cancer statistics, prostate cancer (PCa) has become the most common tumor among men in the United States, and the death caused by PCa were only fewer to that of lung cancer [1]. Epidemiological statistics showed that although the incid ence of prostate cancer in China is lower than that in European and American countries, the proportion of advanced prostate cancer cases is higher [2]. As a highly heterogeneous malignant tumor, the overall prognosis and survival of PCa patients also have great individualized differences [3]. Due to the extreme heterogeneity at the genetic and cellular biological levels, patients with advanced, recurrent and castration resistant prostate cancer (CRPC) are limited to non-surgical or palliative treatment, which generally results in poor efficacy and high mortality rates [4, 5]. The effective chemotherapeutic drugs against prostate cancer is still lacking. With the wide application of Chinese herbal medicine, the safety of its natural ingredients has attracted more and more attention [6]. Currently, the active ingredients from natural substances are considered as important resource for the development of anti-prostate cancer drugs [7]. Therefore, it has become a new trend to discover effective bio-active ingredients of herbal medicines for the treatment of prostate cancer [8].
Rhodiola, a perennial plant of crassulaceae grows in the alpine and pollution-free areas at an altitude of 800–2500 meters, which is used as a whole herb in the traditional Chinese medicinal materials. It is widely believed that Rhodiola can resist oxidation and improve the body immunity, especially has obvious anti-fatigue effect. Rhodiola mainly contains fats, waxes, sterols, phenolic compounds, flavonoids, organic acids, tannins, proteins, water-soluble components, etc. Flavonoid, especially Salidroside, were proved to be the main pharmacological active ingredients of Rhodiola. Salidroside is a polysaponin, mainly derived from the dried roots and rhizomes of Rhodiola. The molecular formula is C
In the present study, we elucidated the effect of salidroside on PCa cells and the underlying mechanism. The proliferation, colony formation and migration were suppressed in PCa cells upon salidroside treatment. Furthermore, salidroside promoted the apoptosis of PCa cells. Mechanically, we found that salidroside inhibited the phosphorylation of PI3K/AKT pathway. In summary, our findings illuminated that salidroside could suppress the proliferation and migration, enhance apoptosis of PCa cells via inhibition of PI3K/AKT pathway.
Methods and materials
Cell culture
PC3 cells were cultured in RPMI1640 (Gibco, C11875500BT 500 ml) complete medium containing 10% serum (Kangyuan, KY-01003) and 1% dual antibody (Biosharp, BL505A). DU145 cells were cultured in DMEM (Bio-Channel, BC-M-005-500 ml) complete medium containing 10% serum and 1% dual antibody.
The primer sequence used for RT-PCR detection
The primer sequence used for RT-PCR detection
PC3 (5000 cells/well) and DU145 (3000 cells/well) cells were seeded in a 96-well plate (Thermo, CAT. NO. 167008). Salidroside (dissolved in DMSO, the final concentration of DMSO was less than 0.1%) was added immediately after cell inoculation. Viability of cells were assayed at predetermined time points using the CCK8 kit (Bimake, B34304) according to the instructions.
Colony formation analysis
PC3 and DU145 cells were seeded into six-well plates at a density of 3000 cells/well for colony formation assays, and salidroside was added 24 h after cell inoculation. Fresh complete medium was replaced every 2 days and salidroside stimulation was maintained throughout the experiment. After 9-day’s culture, cells were fixed with 4% paraformaldehyde (Dingguo, Beijing) at room temperature for 15 min and stained with 0.1% crystal violet (CAS No. 548-62-9) solution at 37
Transwell assay
For migration analysis, PC3 or DU145 cells were inoculated into the upper chamber of the transwell chamber with 100
Scratch assay
About 5
RT-PCR
Cells were lysed with Trizol reagent, RNA was extracted with chloroform, and the obtained 1–2
Antibodies information used for western blot detection
Antibodies information used for western blot detection
Cells were lysed with RIPA lysis buffer containing protease inhibitors (65 mM Tris-HCl, 150 mM NaCl, 1 mM EDTA, 1% Nonidet P-40, 0.5% sodium deoxycholate, and 0.1% SDS). Total protein was extracted by high speed centrifugation at 4 degrees and the concentration of protein was quantified by BCA kit. Equal amounts of protein were separated by 6–10% SDS-PAGE and transferred to PVDF membranes. After membrane transformation, PVDF membrane was blocked at room temperature for 1 h, and specific primary antibody was added and incubated overnight at 4 degrees, and HRP conjugated secondary antibody was added and incubated for 1h the next day. Each step was washed 3 times with TBST. Data were developed and analyzed with a ChemiDoc MP imaging system (Bio-Rad). Corresponding antibodies are shown in the Table 2.
Statistical analysis
All data statistics in this study were in the form of mean value
Salidroside suppresses the viability and colony formation of PCa cells. (A) The chemical formula of salidroside. (B, C) The relative cell viability of PC3 (B) and DU145 (C) cells treated with different concentrations (0, 30, 60, 90, 120 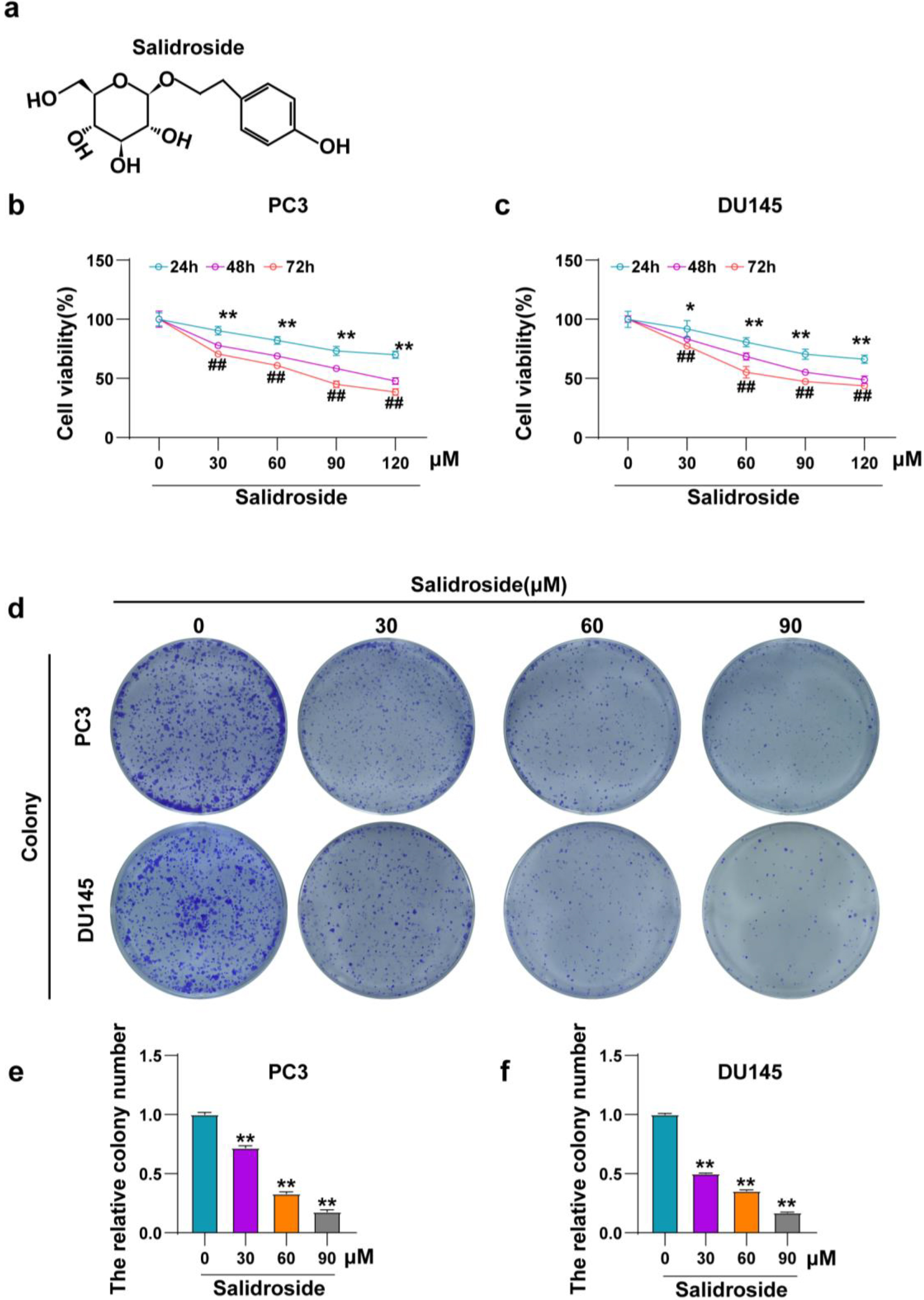
cotinued.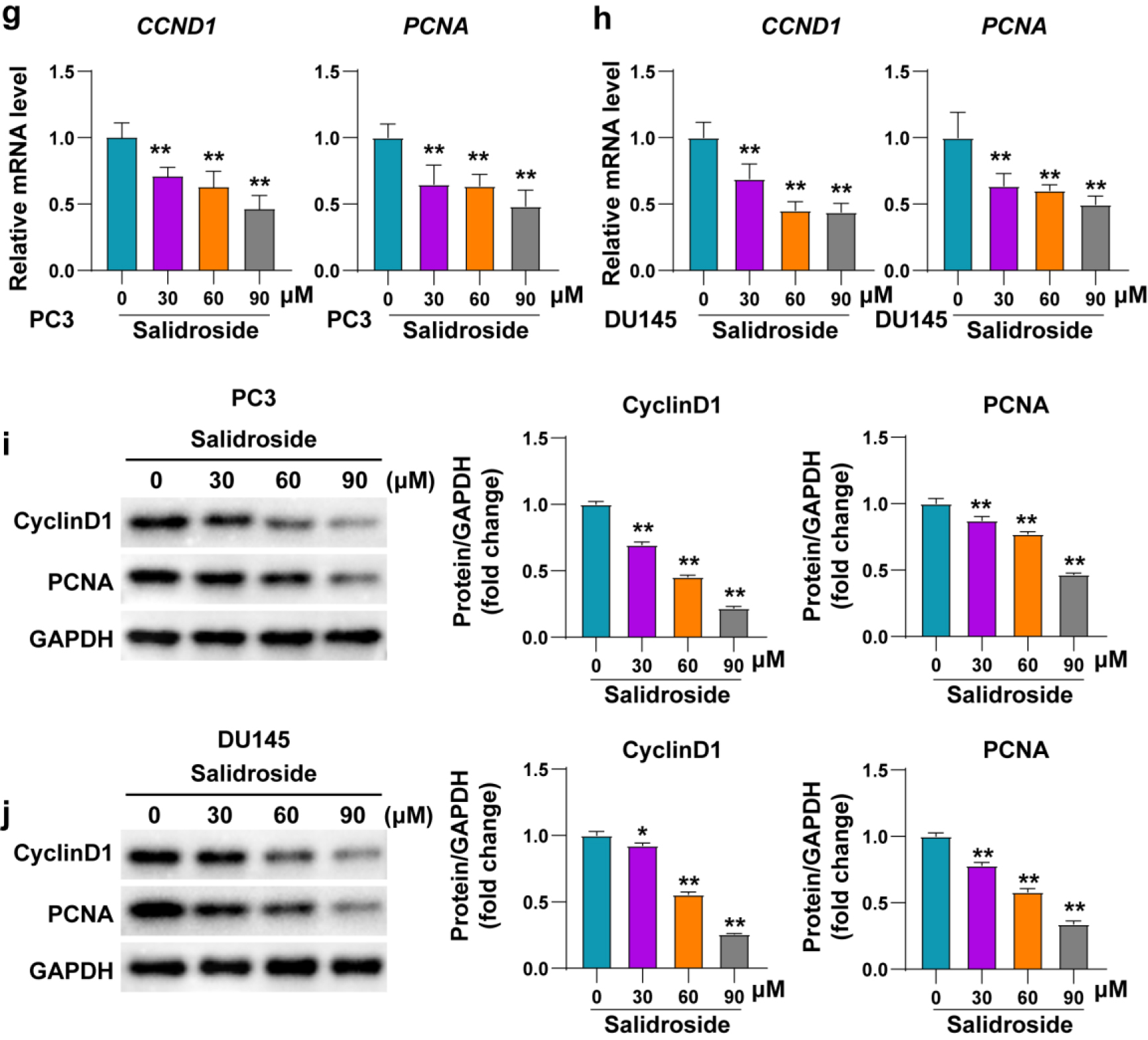
Salidroside suppresses the viability and colony formation of PCa cells in vitro
As described in the previous studies, the chemical formula of salidroside is shown in the figure (Fig. 1A). To investigate the effect of salidroside on PCa, two different PCa cell lines, PC3 and DU145, were cultured in medium containing different concentrations (30, 60, 90, 120
Salidroside promoted the apoptosis of PCa cells. (A, B) The mRNA expression of BCL2 and BAX in PC3 (A) and DU145 (B) cells treated with 0, 30, 60 and 90 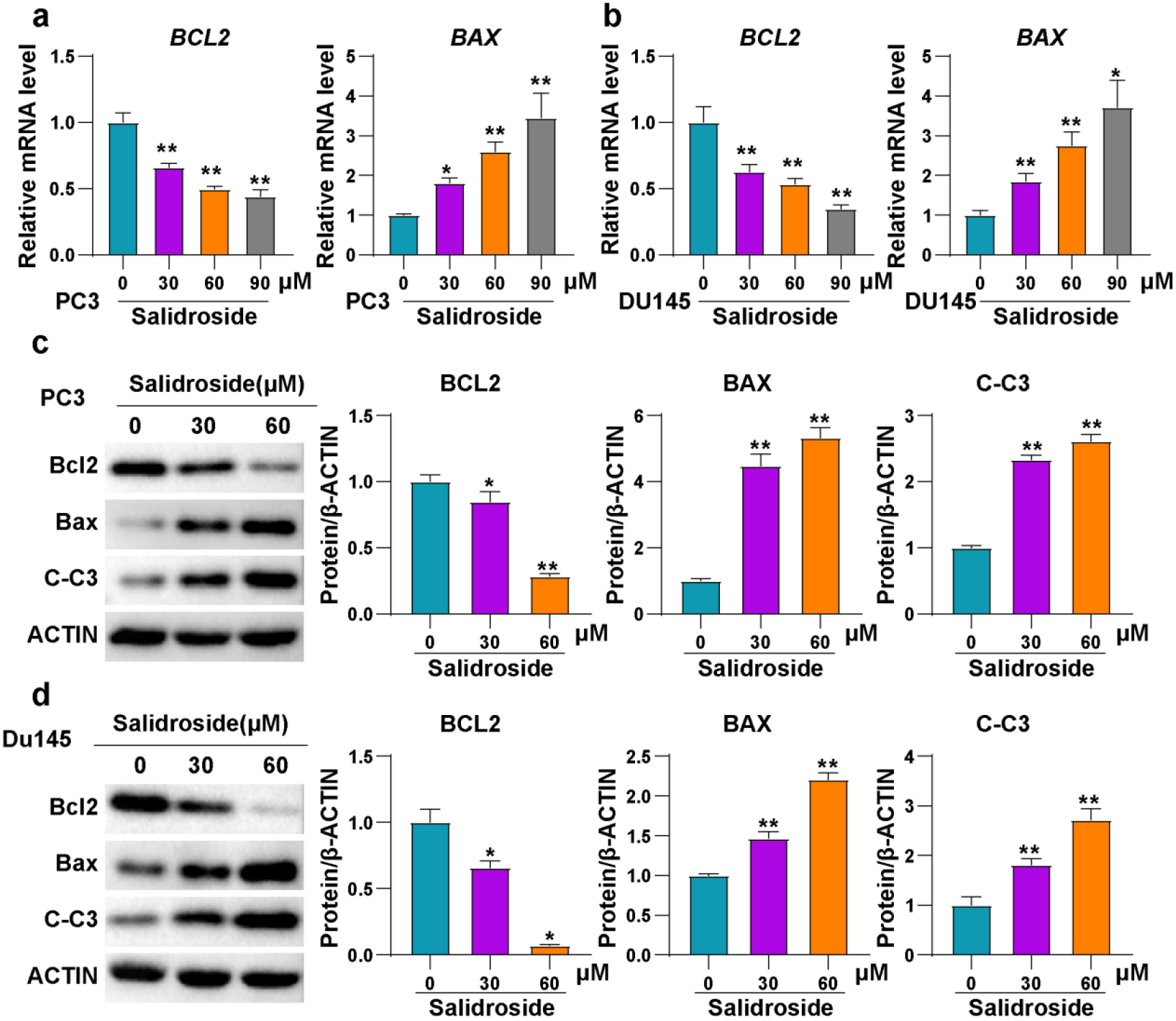
Promoting cell apoptosis is one of the most important mechanisms of most antitumor drugs. To investigate the effect of salidroside on apoptosis of PCa cells, mRNA was extracted from PC3 and DU145 cells treated with three different concentrations (30, 60, 90
Salidroside inhibited the migration of PCa cells. (A, B) Representative images and quantitative statistics of migration for PC3 (A) and DU145 (B) cells treated with 0, 30, 60 and 90 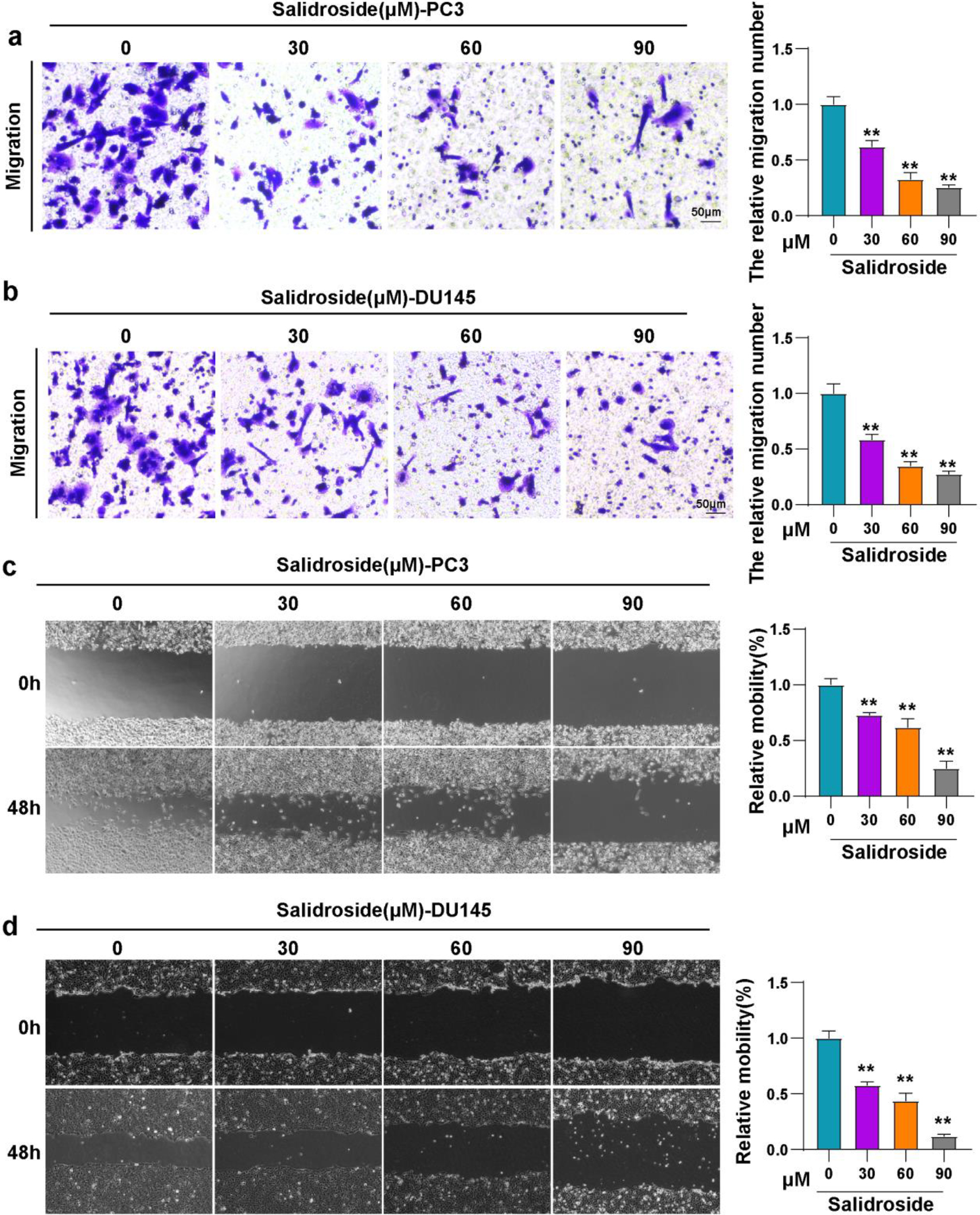
cotinued.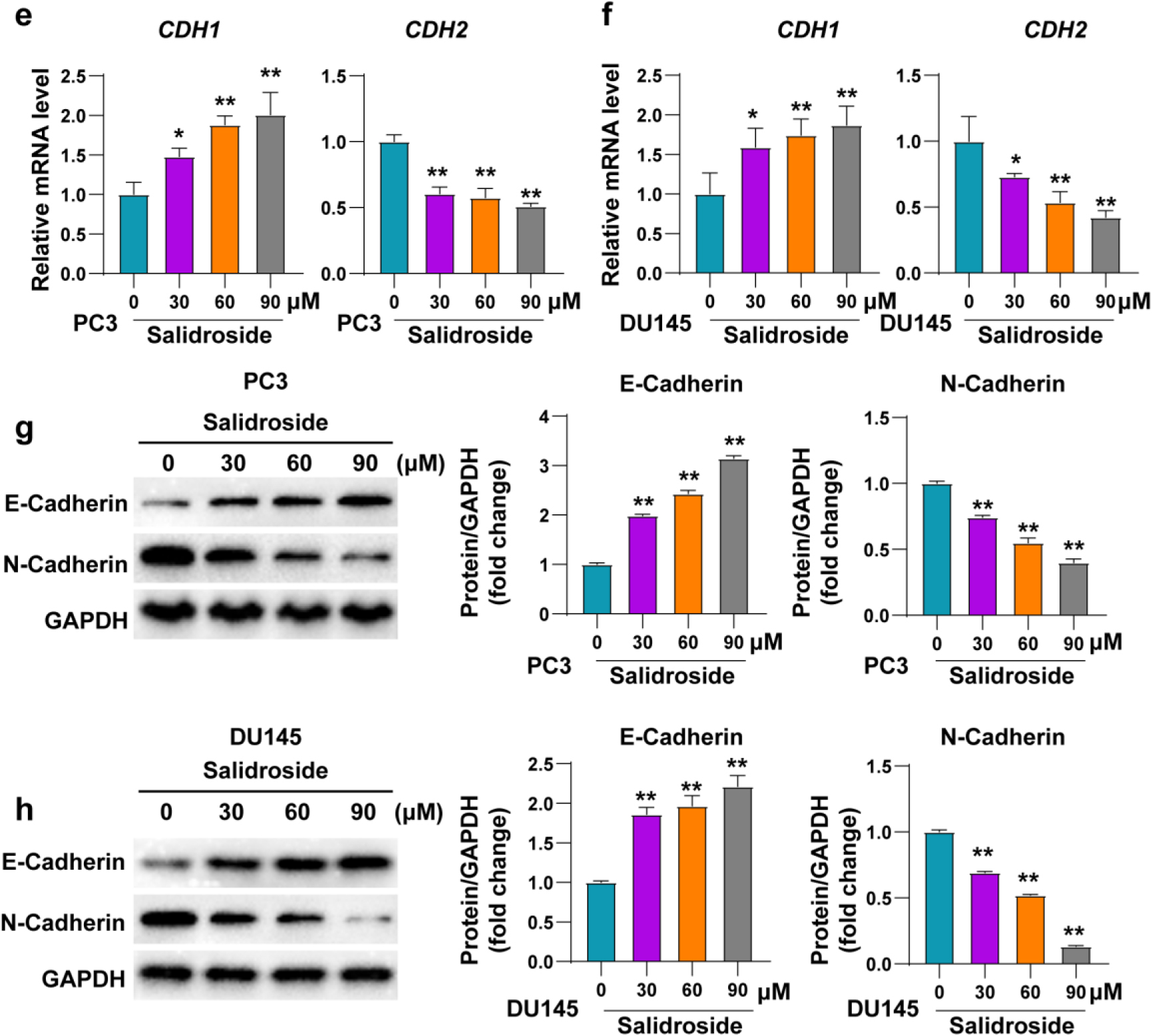
To further investigate the effect of salidroside on other tumor phenotypes of PCa, transwell and scratch assay were used to evaluated the migration of PC3 and DU145 cells. Results showed that the migration cell numbers of both PCa cell lines were reduced upon the treatment of salidroside, the degree of reduction enhanced with the increase of salidroside concentration (Fig. 3A and B). Consistently, the migration rates of PCa cells treated with salidroside were also decreased compared with control cells. This effect of salidroside exhibited dose-dependent (Fig. 3C and D). In addition, the mRNA and protein levels of migration-related molecules (CDH1, CDH2) were consistent with the results of the migration experiments (Fig. 3E–H). Altogether, our results indicated that salidroside could inhibit the migration of PCa cells.
Salidroside inhibited PI3K/AKT pathway in PCa cells. (A, B) Representative images and quantitative statistics of Western blot of PI3K and AKT in PC3 (A) and DU145 (B) cells treated with 0, 30 and 60 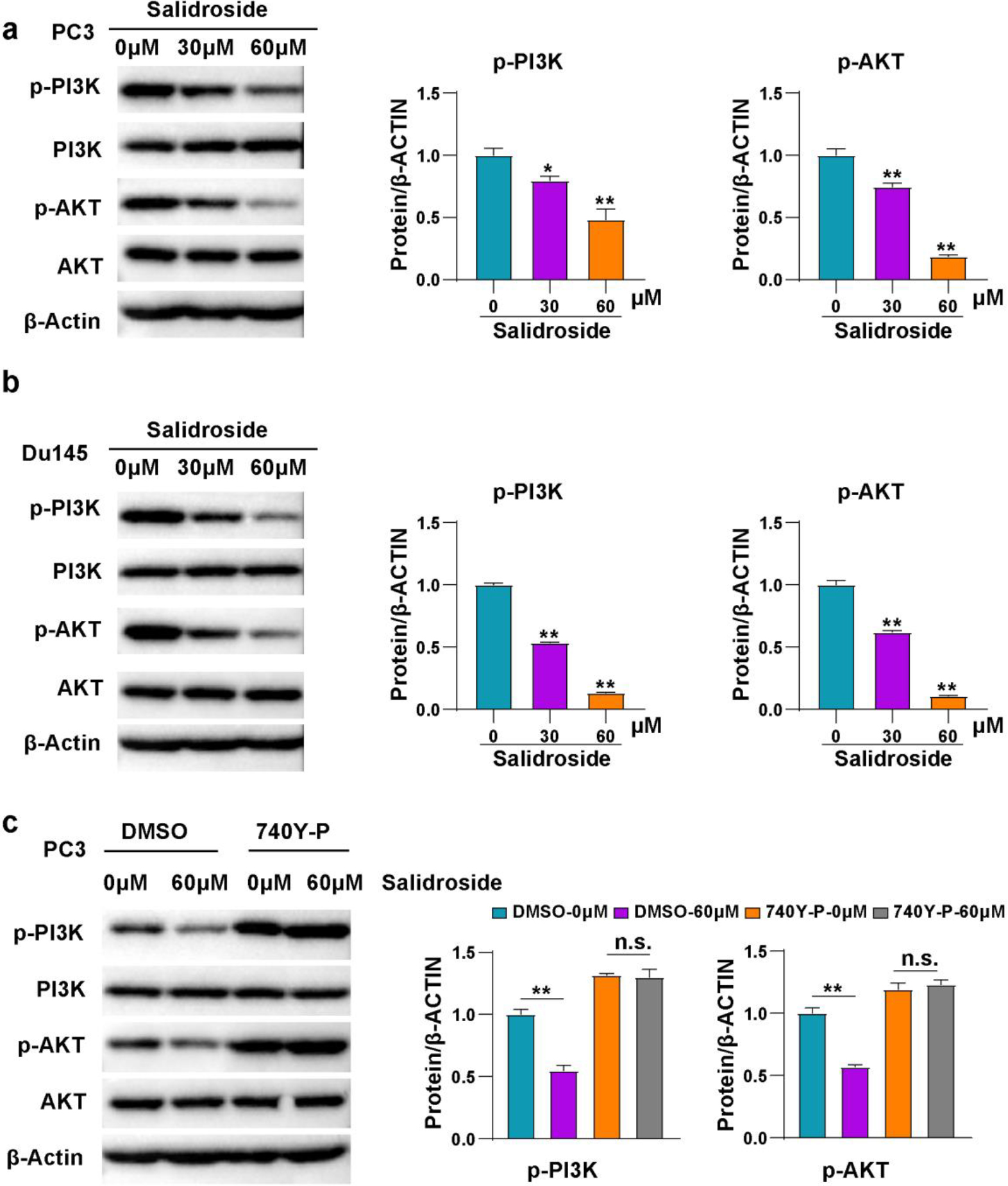
Given that salidroside exhibited anti-tumor effect on PCa cells, we further investigated its underlying mechanism. It has been reported that salidroside suppresses Phosphatidylinositol 3-kinase (PI3K)/AKT pathway in myocardial injury and gastric cancer cells [9, 20]. We determined the total and phosphorylated protein level of PI3K and AKT. These results exhibited that salidroside treatment down-regulated the phosphorylation levels of PI3K and AKT, and the higher the concentration of salidroside, the lower the phosphorylation levels (Fig. 4A and B). To verify the relationship of salidroside and PI3K/AKT pathway, a PI3K agonist 740Y-P was applied in the experiments. The results showed that 740Y-P could eliminate the inhibition of PI3K and AKT induced by salidroside (Fig. 4C). Collectively, the above results indicated that salidroside inhibits PI3K/AKT pathway in PCa cells.
Discussion
Results calculated using cancer estimates from GLOBOCAN 2020 and population estimates from the United Nations showed prostate cancer is estimated to cause 125,646 new cases and 56,239 deaths in China, 2022, while the US is expected to have 216,900 new cases and 34,611 deaths [21]. Most patients in China are already in the middle and advanced stages of prostate cancer when diagnosed due to the lack of widespread early diagnosis [2]. Although the survival time of patients has greatly improved with advances in diagnosis and treatment, the 5-year overall survival rate of some patients with more advanced malignancies remains poor [22]. The 5-year relative survival rate for local PCa is nearly 100%, compared with 30% for advanced metastatic prostate cancer (mPCa) [23]. Many advanced prostate cancers initially respond to androgen ablation therapy, but then develop an aggressive androgen-independent phenotype that is resistant to conventional therapy and metastases to lymph nodes and bone [24, 25, 26]. Multiple drug resistance in prostate cancer is the main cause of tumor recurrence, metastasis and even treatment failure [27]. It is the most common and difficult problem to overcome in the clinical treatment of prostate cancer. As China is enters an aging society, the incidence and death of prostate cancer may peak in the next ten years. Therefore, it is urgent to explore the treatment methods for advanced prostate cancer.
Glucose and lipids are essential nutrients that provide energy for cell growth and division. However, in cancer cells, these metabolic pathways become dysregulated, leading to increased glucose uptake and lipid synthesis. It has been shown that alterations in glucose and lipid metabolism play a critical role in prostate cancer development and progression [28, 29, 30]. For example, the expression of a key enzyme involved in lipid synthesis, fatty acid synthase (FASN), was significantly higher in prostate cancer tissues and inhibition of FASN activity reduced prostate cancer cell proliferation and migration [31]. Cancer cells often require high levels of glucose for their rapid growth and proliferation. Thus, increased glucose uptake can also promote prostate cancer growth. Prostate cancer cells with high levels of glucose transporter 1 (GLUT1) expression had a higher rate of cell proliferation compared to cells with low GLUT1 expression [32].
Extracting effective anticancer components from traditional Chinese medicinal herbs and applying them in cancer treatment has attracted extensive attention in recent years [33]. Some compounds derived from Chinese Herbal Medicine have shown anticancer properties and can inhibit the development, proliferation, angiogenesis, and metastasis of human cancer. Resveratrol, curcumin and berberine, for example, have been used in many clinical trials to treat many types of cancer [34]. In most cases, traditional Chinese medicine is used as an adjuvant treatment for cancer [35, 36]. With the increasing application of traditional Chinese medicine in cancer treatment, researchers are increasingly studying the molecular mechanism of the action of traditional Chinese medicine in cancer treatment. Gapter et al., (2005) has reported that pachytearic acid induced apoptosis of PC cells by inducing caspase-9/-3 activity and increasing proportion of Bax/Bcl-2 protein [37]. Dietary isothiocyanate form papaya promotes apoptosis of PC cells by activating the caspase-8 and -9 pathways and the phosphorylation of the anti-apoptotic protein Bcl-xL [38]. 6-Shogaol extract form dry Ginger inhibits the growth of PC cells by inhibiting nuclear factor kappa B (NF-
Salidroside, a bioactive compound extracted from Rhodiola rosea L., has been reported to possess various pharmacological properties, including anti-inflammatory, antioxidant, neuroprotective, and cardioprotective effects. Several studies have suggested that salidroside can regulates PI3K activity depending on the enviroment and cell type [40, 41], and salidroside has been reported that alleviates diet-induced glucose and lipid metabolism disorders in mice by regulating the activity of AMPK [42]. AMPK can further regulate the proliferation and migration of a variety of tumors including prostate cancer by regulating a variety of signaling pathways including PI3K-AKT signaling pathway [43].
Our study found that salidroside could inhibit the proliferation and migration of prostate cancer PC3 and DU145 cells by inhibiting the activity of PI3K. But, the exact mechanisms by how salidroside modulates PI3K activity are not fully understood yet. One possible mechanism is that salidroside may interact directly with the catalytic or regulatory subunits of PI3K, thereby altering their conformation or stability. This may affect the recruitment of downstream effectors, such as Akt or mTOR, and the subsequent activation of their signaling pathways. Another possible mechanism is that salidroside may modulate PI3K activity indirectly by regulating upstream or parallel signaling pathways. For example, salidroside was found to activate AMPK, a cellular energy sensor that can inhibit or activate PI3K depending on the context. Salidroside was also shown to modulate the expression or activity of other proteins involved in PI3K signaling, such as PTEN, PDK1, and IRS-1 [12, 13]. Compared to other phenolic compounds, such as flavonoids and polyphenols, salidroside have a unique chemical structure that may contribute to their ability to regulate PI3K activity. Specifically, salidroside contain a triterpenoid backbone with one or more sugar moieties attached at various positions. These sugar moieties may be responsible for the interaction between salidroside and PI3K.
In this study, we elucidated the anti-cancer effect of salidroside on PCa for the first time. PI3K/AKT signaling pathway is one of the signaling pathways that plays an important role in basic intracellular functions and involved in regulation of cell proliferation, growth, cell size, metabolism and motility [46]. The function of genes of PI3K/AKT pathway have been extensively studied and were reported to be mostly activated in human cancers. PI3K/AKT pathway plays multiple roles in the carcinogenesis of PCa. Inflammatory cytokines (CCR9, IL-6 and TLR3) regulate PI3K/AKT signaling during PCa cell apoptosis. PI3K/AKT participates in androgen and prostaglandin-associated mechanisms and is regulated by ErbB, EGFR, and the HER family during cell growth. Our results revealed that salidroside manipulates the activation of PI3K/AKT pathway in PCa cells.
Conclusion
Our study demonstrated that salidroside has anti-cancer activity in PCa cells. Salidroside promotes apoptosis and inhibits viability, colony formation and migration of PC3 and DU145 cells. We found salidroside down-regulates the phosphorylation of PI3K and AKT, and 740Y-P, a PI3K agonist, can eliminate the inhibition of PI3K and AKT induced by salidroside. This suggests that PI3K/AKT pathway is the target for the anti-cancer effect of salidroside in PCa cells. Our findings expand the potential use of salidroside in antitumor therapy and provide new drug candidates for the treatment of prostate cancer.
Footnotes
Acknowledgments
None.
Conflict of interest
The authors declare no competing interests.
Author contributions
Conception: Ru-Han Liu, Teng-Fei Ma, Jin-Song Zhang and Chi-Hua Wang.
Interpretation or analysis of data: Qin Yang, Wen-Chang Xiao, Lu Yin and Miao Yin.
Preparation of the manuscript: Ru-Han Liu and Jin-Song Zhang.
Revision for important intellectual content: Ru-Han Liu, Teng-Fei Ma and Chi-Hua Wang.
Supervision: Chi-Hua Wang.
