Abstract
BACKGROUND:
Despite extensive research on endometrial cancer and tumor hypoxic microenvironment, there are no reports exploring the role of DDIT4 in endometrial cancer.
OBJECTIVE:
This study aimed to elucidate the significance of DDIT4, as a prognostic biomarker for endometrial cancer by immunohistochemical staining and statistical analysis.
METHODS:
Four endometrial cancer cells were cultured under normoxia and hypoxia, and the differentially expressed genes were examined using RNA-seq. Immunohistochemical staining for DDIT4 and HIF1A was performed in 86 patients with type II endometrial cancer treated at our hospital, and their correlation with other clinicopathological factors and the prognostic role was analyzed using statistical methods.
RESULTS:
The expression analysis of hypoxia-inducible genes using four types of endometrial cancer cells revealed that DDIT4 was among the 28 genes that were upregulated in all cells. Based on our results of immunohistochemistry of DDIT4 expression in endometrial cancer tissues, univariate and multivariate analyses based on COX regression analysis showed that high DDIT4 expression significantly correlated to favorable prognosis in both progression-free survival and overall survival. Limited to recurrent cases, metastasis to only lymph nodes was significantly related to high DDIT4 expression, whereas metastasis to other parenchymal organs was significantly dominant in patients with low DDIT4 expression.
CONCLUSIONS:
The expression of DDIT4 enables to predict survival and recurrence in type II endometrial cancer.
Introduction
Endometrial cancer is the most frequent gynecological malignancy in Western countries [1]. In Japan, the number of patients with endometrial cancer is on the rise, as is the number of patients with cervical cancer [2]. Treatment of endometrial cancer consists of a combination of surgery and postoperative chemotherapy or postoperative radiation therapy based on the pathological findings [3]. The adverse risk classification determines the need for postoperative treatment. As a result, more than 90% of patients with early-stage endometrial cancer survive more than 5 years without recurrence [2]. However, some patients with early-stage and considerable patients with advanced endometrial cancer experience recurrence, despite the implementation of postoperative therapy. Once the disease relapses, it is difficult to treat due to limited treatment options with few effective chemotherapy regimens. Grade 3 endometrial carcinoma, serous carcinoma, and clear cell carcinoma, considered particularly high-risk in histology, are defined as type II endometrial cancer [4]. Type II endometrial cancer is clinically distinct from type I Grade 1-2 endometrial carcinoma caused by unopposed estrogen and accounts for approximately 20% of all endometrial cancer [5]. To our knowledge, there are currently no useful prognostic biomarkers for type II endometrial cancer other than clinical stage established by the International Federation of Gynecology and Obstetrics (FIGO). Accurate prognostic biomarkers will increase the possibility of improving prognosis through individualized treatment.
Hypoxia contributes to a more aggressive cancer phenotype, including increased cell proliferation, resistance to chemotherapy, promotion of angiogenesis, mimicking angiogenesis, and maintenance of cancer cell stemness, as well as increasing the invasiveness and metastatic potential of cancer cells [6]. A representative hypoxia-inducible factor, the hypoxia-inducible factor 1
To the best of our knowledge, there are no reports exploring the significance and relationship of DDIT4 in endometrial cancer. In this study, we aimed to determine the role of DDIT4 as a prognostic biomarker for endometrial cancer by immunohistochemical staining and statistical analysis.
Materials and methods
Refinement of hypoxia-inducible molecules in endometrial cancer cells
Total RNA was extracted from samples of four endometrial cancer cell lines (AMEC, HEC1A, HEC50, and RL50) cultured in normal oxygen concentration (O2 21%) and hypoxia (O2 1%) for 72 hours, using RNeasy Mini Kit (Qiagen, Tokyo, Japan). RNA sequencing was conducted by Rarevariant, Inc. (Tokyo, Japan). The expression level of each gene was quantified using Kallisto, and the data were summarized using the tximport package (ver. 1.18.0) of R software (ver. 4.0.3) and RStudio (RStudio, Boston, MA). Differentially expressed genes (DEGs) between normoxia and hypoxia were determined by the absolute value of log2 fold change
Target patients and clinical information
Representative images of immunohistochemical staining. Representative images of immunohistochemical staining of DDIT4 in the upper panel and HIF1A in the lower panel are presented. The images were scored 0–3 based on staining intensity.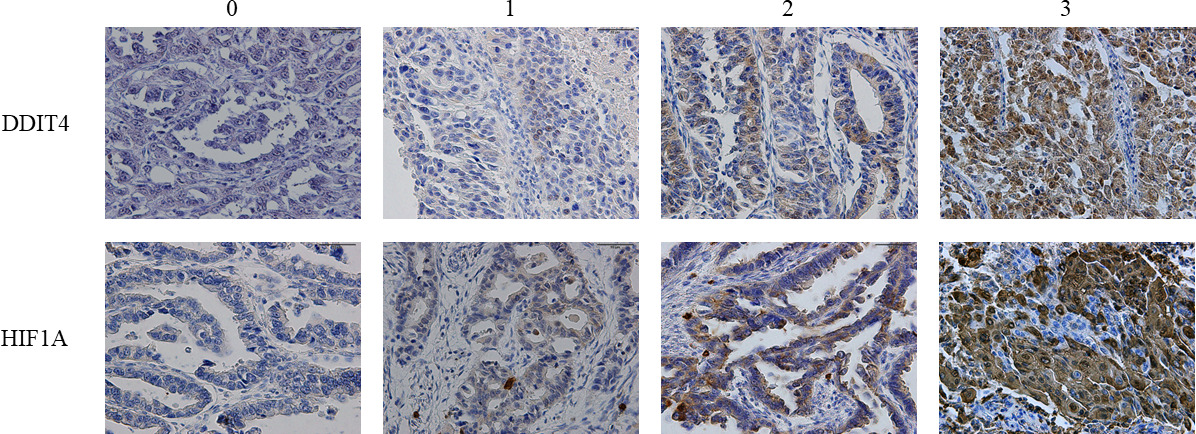
Clinical information of 546 consecutive patients with endometrial cancer treated at our hospital from January 2002 to January 2017 was analyzed retrospectively. Of these, 95 (17.4%) were diagnosed with type II endometrial cancer, including patients with grade 3 endometrial cancer, serous carcinoma, clear cell carcinoma, or a mixed type of these tumors. All patients underwent computed tomography (CT) before primary surgery to assess the local progression and systemic metastasis. In principle, all patients with type II endometrial cancer are operated on by laparotomy. Nine patients were excluded due to a lack of preoperative clinical information. The remaining 86 cases were analyzed in this study. Age at diagnosis, pathologic histology, clinical stage according to the 2008 International Federation of Gynecology and Obstetrics (FIGO) staging system, ascites cytology at the primary surgery, presence of lymphovascular invasion, depth of myometrial invasion, body mass index (BMI) at diagnosis, history of diabetes mellitus, surgical radicality (with or without lymphadenectomy) and CA125 values at diagnosis were extracted from the medical records. Pathological findings were evaluated by at least one pathologist and one gynecologic oncologist. Due to the high risk of recurrence of type II endometrial cancer, paclitaxel and carboplatin (TC) combination chemotherapy was principally proposed as postoperative adjuvant chemotherapy for all patients. This study was approved by the Nagoya University Ethics Committee (approval number 2019-0106). The Committee issued a waiver of written informed consent because this study was a retrospective analysis and no additional samples or images were collected.
After the final treatment, patients were followed up every 1 to 3 months during the first year and every 3 to 6 months after the second year until the fifth or tenth year. Routine follow-up included physical examination, transvaginal ultrasonography, and blood tests (including CA125). Follow-up CT was performed at least once every 6 months for the first 2 years. The diagnosis of recurrence was determined based on CT imaging. For survival, the outcomes were progression-free survival (PFS) and overall survival (OS), where PFS was defined as the beginning of the primary treatment to relapse or progression and OS was defined as the time from the start of first treatment to death for any reason. The follow-up period was defined as the time from the start of the first treatment to the last follow-up date or death.
Immunostaining
The primary tumor was a paraffin-embedded block taken at the time of surgery. Tumors were serially sectioned at 4
Statistical analysis
Comparisons between the two groups were performed with the Student’s t-test or Mann-Whitney U test for continuous variables. Categorical variables were compared with the chi-squared or Fisher’s exact test. Kaplan-Meier curves were estimated for OS and PFS. Univariate and multivariate Cox regression analyses were used to assess the hazard ratio for OS and PFS. Significance was set as two-sided with a
Results
Identification of hypoxia-inducing molecules in endometrial cancer cells
The RNA profiles of endometrial cancer cells under normoxia and hypoxia. (A) The Venn diagram for upregulated genes under hypoxia in endometrial cancer cells. (B) The hierarchical clustering and heatmap analysis of the RNA profiles of endometrial cancer cells under normoxia and hypoxia. (C) Principal component analysis of the RNA profiles of endometrial cancer cells under normoxia and hypoxia.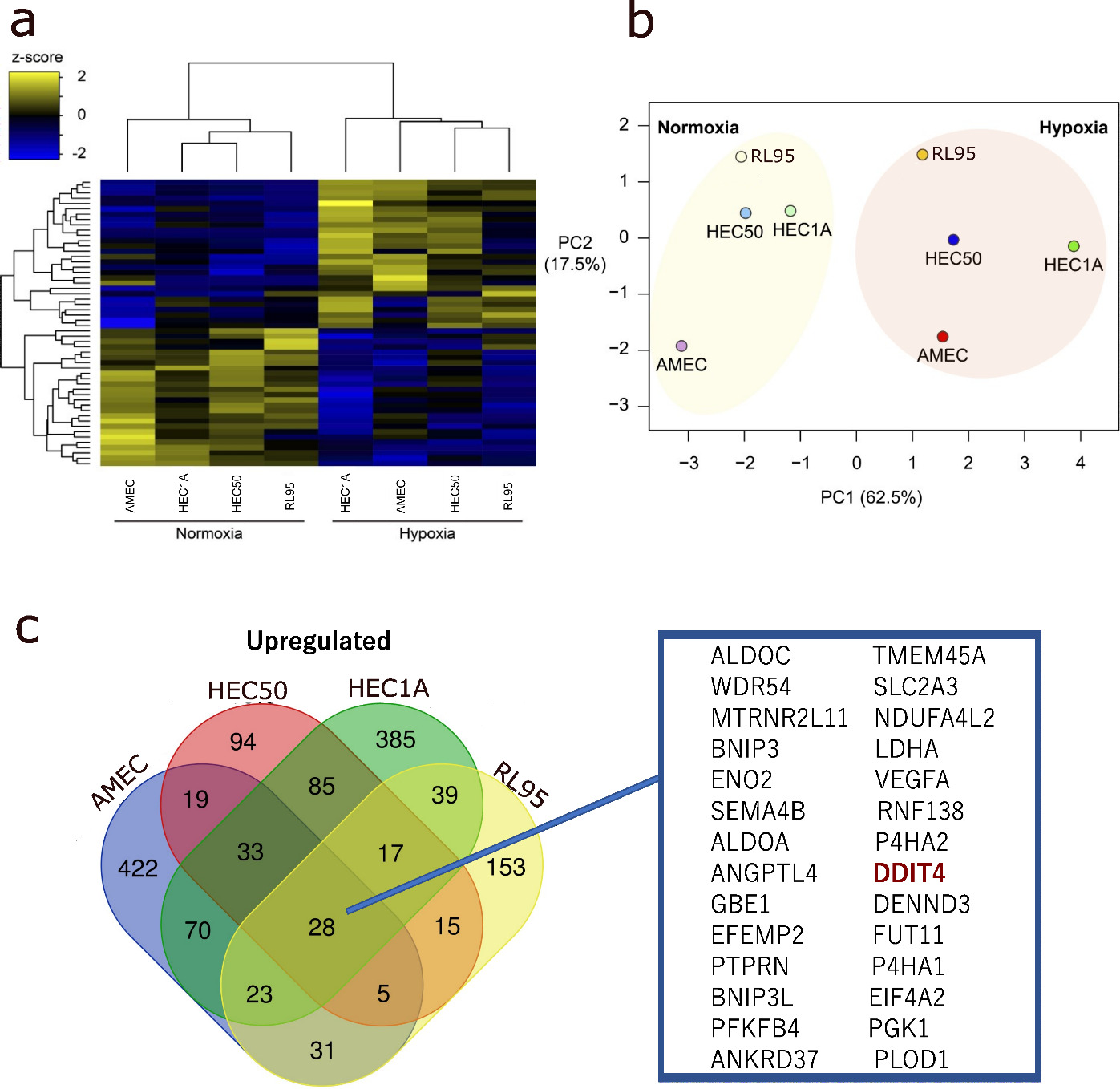
First, to identify hypoxia-inducing molecules in endometrial cancer cells, transcriptome analysis was performed using four endometrial cancer cell lines under normoxia and hypoxia. Of the molecules commonly upregulated by hypoxia, 28 genes were elevated in all four cell lines, including DDIT4 but not HIF1A (Fig. 2a). The heatmap showed that the gene expression profile of endometrial cancer cells under hypoxia was quite different from those under normoxia (Fig. 2b). The principal component analysis also showed that hypoxic stimulation-induced changes in a common direction in all endometrial cancer cells (Fig. 2c). These results indicate that hypoxia induces a common set of genes in endometrial cancer cells, including DDIT4.
Patients’ characteristics stratified by DDIT4-low and DDIT4-high groups
Patients’ characteristics stratified by DDIT4-low and DDIT4-high groups
SD, standard deviation; BMI, body mass index.
Based on RNA-seq results, we focused on the predictive value of DDIT4 expression in endometrial cancer tissues. A total of 86 patients were enrolled in the clinical analysis. The Median follow-up was 51.4 months (range: 1–155 months). The median age was 62 years. Thirty-four patients (39.5%) had stage I endometrial cancer, and 38 (44.2%) had positive cytology of the ascites at the primary surgery. Postoperative adjuvant therapy consisted of chemotherapy in 72 patients (83.7%), radiation therapy in 1 patient (1.1%), and no postoperative treatment in 13 patients (15.1%). Patient background information is summarized in Table 1. Kaplan-Meier analysis revealed 5-year PFS and OS rates of 58.6% and 73.8%, respectively, for all patients.
First, immunohistochemical staining for HIF1A and DDIT4 was performed on the primary lesions of surgical specimens from endometrial cancer patients; DDIT4 staining was quantified based on the H-score because of staining heterogeneity in the tumors, and HIF1A staining intensity in the tumors was categorized into 4 levels (0–3). To calculate the optimal value for detecting the presence or absence of recurrence using the H-score of DDIT4, the cutoff value was set at H-score
Univariate analysis of factors associated with prognosis
Results of univariate analysis for progression-free survival and overall survival
Results of univariate analysis for progression-free survival and overall survival
PFS, progression-free survival; OS, overall survival; BMI, body mass index; HR, hazard ratio; CI, confidence interval.
Kaplan-Meier curves show overall survival in all stage and stage III–IV. (A) The 5- and 10-year OS rates in the DDIT4-high group were both 79.2% and those in the DDIT4-low group were 62.8% and 54.9%, respectively. (B) The Kaplan-Meier curve shows OS stratified by DDIT4 expression in stage III–IV patients.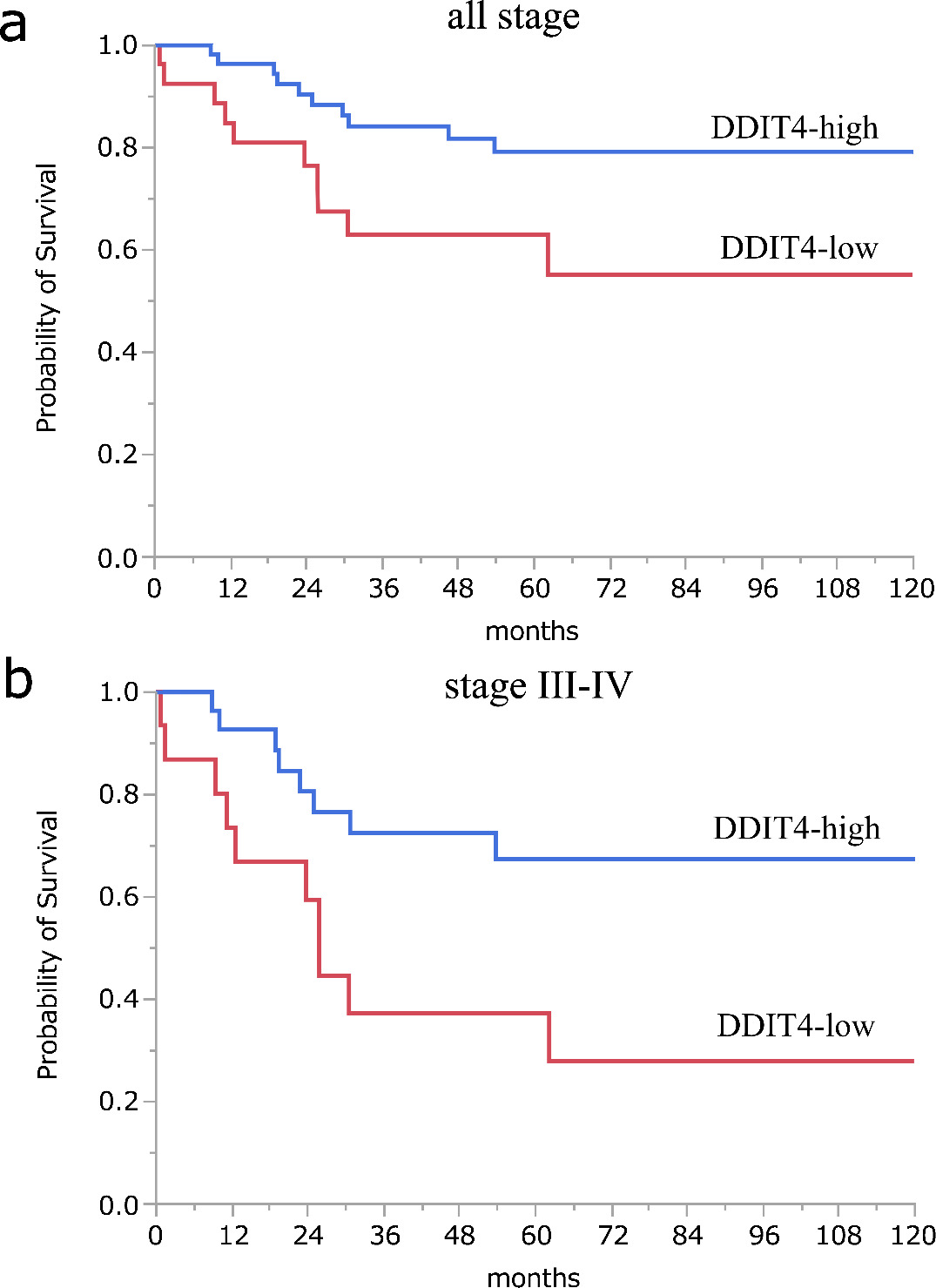
Next, we compared the prognosis between DDIT4-low and DDIT4-high groups. Kaplan-Meier analysis showed that the 5- and 10-year OS rates in the DDIT4-high group were both 79.2% and those in the DDIT4-low group were 62.8% and 54.9%, respectively, showing significant differences between the two groups (
Results of multivariate analysis for progression-free survival and overall survival
Results of multivariate analysis for progression-free survival and overall survival
PFS, progression-free survival; OS, overall survival; HR, hazard ratio; CI, confidence interval.
Site of recurrence stratified by DDIT4-low and DDIT4-high groups
From the results of the univariate analysis, we proceeded to subsequent multivariate analyses (Table 3). In the PFS analysis, we selected myometrial invasion, lymphovascular invasion, FIGO stage, CA125 value, ascites fluid cytology, and DDIT4 category as variables. For the OS analysis, the chosen variables included FIGO stage, myometrial invasion, ascites fluid cytology, and DDIT4 category. Patients with stage III–IV endometrial cancer had a significantly higher risk of disease progression compared to patients with stage I–II disease (HR 5.136 (95% CI
The site of recurrence was examined as a factor associated with DDIT4 expression and prognosis. The recurrence site was divided into two groups, a locoregional recurrence group and a distant recurrence group, and the association with DDIT4 category was tested by chi-square test. The locoregional recurrence group was significantly associated with high expression of DDIT4 (Table 4).
Discussion
In type II endometrial cancer, DDIT4 expression can be a useful prognostic biomarker other than FIGO stage. To the best of our knowledge, this is the first report to clarify the prognostic significance of DDIT4 in endometrial cancer. High expression of DDIT4 is a common feature in hypoxic tumor microenvironments regardless of cancer type and is more representative of changes in endometrial cancer cells than HIF1A according to our RNA-seq analysis. Contrary to expectations, high DDIT4 expression in type II endometrial cancer correlated with a favorable prognosis. This may be partly because the recurrence pattern in patients with endometrial cancer expressing high DDIT4 in IHC is more likely to occur as a locoregional recurrence amenable to local therapy such as surgery or radiotherapy.
DDIT4 is one of 28 genes commonly up-regulated in all four of our endometrial cancer cell lines and has been reported to be induced in cells exposed to severe microenvironments such as hypoxia, irradiation, and low nutrition [14]. Numerous studies indicate that hypoxia stimulates invasiveness, cell proliferation, resistance to chemotherapy, and maintenance of cancer cell stemness in cancer cells, contributing to more aggressive cancer phenotypes. A recent review on endometrial cancer showed that HIF-1
There have been no reports on the correlation between DDIT4 expression and prognosis in endometrial cancer. In silico analysis using the KM plotter and SurvExpress showed that high DDIT4 expression correlated with poor prognosis in acute myeloid leukemia, rectal cancer, and lung cancer, whereas high DDIT4 expression correlated with a good prognosis in gastric cancer [19, 24]. These results imply that the prognostic significance of DDIT4 expression differs among cancer types and that the molecular mechanisms induced by hypoxia differ among tissues or organs. In addition, a recent meta-analysis has shown a negative correlation between HIF1A expression and survival in endometrial cancer [21]. On the other hand, the expression of hypoxia-inducible molecules including HIF1A does not necessarily correlate with poor prognosis, and its biological significance is not necessarily tumor-promoting [9, 10]. Although there are reports that hypoxia promotes tumor growth and invasion, the role of hypoxia in the tumor microenvironment is complex, and it is premature to assume that simply tumor cell phenotype correlates with prognosis [25, 26]. Immune-related cells other than tumor cells such as macrophages, lymphocytes, and fibroblasts constitute the tumor microenvironment, and not only the function of tumor cells but also their interaction with the microenvironment may have a significant impact on metastasis and recurrence. Therefore, our results are not necessarily inconsistent with previous findings.
In our study, high DDIT4 expression was associated with improved PFS and OS in endometrial cancer patients. This result suggests that DDIT4 expression may serve as a prognostic biomarker for identifying patients at a higher risk of disease progression and poor outcomes. By incorporating DDIT4 expression levels into existing risk stratification models, clinicians may be able to better tailor treatment strategies and follow-up schedules for individual patients. Furthermore, limited to cases of recurrence, high DDIT4 expression was higher in cases of locoregional recurrence with retroperitoneal lymph node metastasis, while DDIT4 expression was lower in cases of distant recurrence including peritoneal dissemination and/or other parenchymal organs. Lymphatic, hematogenous, and peritoneal metastasis are representative modes of recurrence of endometrial cancer, each of which may depend not only on differences in their anatomic pathways but also on differences in the molecular mechanisms of the primary tumor. Previous reports have shown that hypoxia and DDIT4 expression correlate with lymph node metastasis in lung adenocarcinoma, which supports the results of this study [24]. In terms of the relationship between recurrence mode and prognosis in endometrial cancer, some reports suggest that radiotherapy for patients with lymph node recurrence may be associated with long-term survival [27]. High expression of DDIT4 in endometrial cancer was found to be characterized by a pattern of locoregional recurrence. DDIT4 exhibits high expression in the majority of cases with low aggression, while low expression is observed in cases with high aggression that involve distant recurrence. This could be interpreted as a result of molecular pathological changes that DDIT4 is upregulated and maintained in the early stages of carcinogenesis, but there is a possibility that its expression decreases during the acquisition of aggressive traits such as metastasis and recurrence.
Given the relatively small number of patients in this study (
In conclusion, in the present study, high expression of the hypoxia-inducible molecule DDIT4 in type II endometrial cancer was associated with a favorable prognosis, especially characterized by locoregional recurrence.
Footnotes
Acknowledgments
This section should contain all acknowledgements, including any funding source for the research.
Author contributions
Conception: Yoshikawa N.
Interpretation or analysis of data: Kosuke Y, Wenting L, Tetsuya M, Satomi H, Satoshi T, Yoshiki I, Akira Y, Yusuke S.
Preparation of the manuscript: Yoshikawa N.
Revision for important intellectual content: Kaoru N.
Supervision: Hiroaki K.
