Abstract
BACKGROUND:
Genetic mutations, peritoneal metastasis and frequent development of chemoresistance worsen the prognosis of ovarian carcinoma.
OBJECTIVE:
The objective of the study is to determine mutations in cancer susceptibility genes in relation with chemotherapy response.
METHODS:
In this follow up descriptive study, 47 consenting female patients diagnosed with surface epithelial ovarian cancer were observed for six months after completion of chemotherapy to see the treatment response. For genetic analysis, the DNA extraction was done and the genomic regions of different exons of BRCA1/2, PALB2, CHEK2, BAP1, CTNNB1, HOXB13, and PIK3CA were amplified using gene specific primers followed by Sanger Sequencing.
RESULTS:
86.7% of the patients were sensitive to chemotherapy whereas 13.3% showed resistance. Genetic variants of BRCA1 in 7%, BRCA2 in 4.7%, PIK3CA in 9.3%, PALB2 in 7%, CHEK2 in 2.3%, BAP1 in 2.3%, and CTNNB1 in 2.3% of the patients were found. There was also a significant association between TNM stage and the treatment response (
CONCLUSION:
Our study exhibits the pivotal role of genetic analysis in predicting the treatment response and paving pathway for patient tailored targeted therapy in Pakistani population.
Introduction
Surface epithelial ovarian cancer (SEOC), is the most fatal gynecological malignancy, having rapidly increasing incidence and mortality. GLOBOCAN 2020 by the International Agency for Research on Cancer reported 313,959 (1.6%) new cases of ovarian carcinoma worldwide with 207,252 (2.1%) deaths per year [1]. In Pakistan, 4326 (2.4%) cases have been reported with 2946 (2.5%) deaths in 2020 with cumulative risk of having ovarian carcinoma is 0.55% [2]. The treatment of ovarian cancer is complex because of heterogeneity of tumor. The reason for heterogeneity is diverse histological subtypes of SEOC because of different cellular origin, diverse mutational spectrum, which leads to different prognosis for every patient [3]. Different molecular subtypes can be present within one histological type with different prognoses [4]. Late stage diagnosis with peritoneal metastasis and chemoresistance has worsened the prognosis and survival rate [5].
SEOC is thought to be related to an accumulation of genetic alterations that can lead to alterations in expression of various genes. Along with deleterious mutations in homologous recombination genes, other cancer susceptibility genes also play an important role in the magnitude of cancer risk which could be helpful in making decisions regarding treatment protocols according to the local clinical oncology practice guidelines [6]. There is an increasing demand, by both health care providers and patients, for genetic testing due to the perceived clinical and economic advantages of this approach. The panels of mutational analysis include various homologous recombination genes (BRCA1, BRCA2, BARD1, ATM, BRIP1, CHEK2, NBN, PALB2, MRE11A, RAD50, RAD51D, RD51C, and XRCC2) along with other cancer susceptibility genes. Genetic testing provides a detailed insight of SEOC risk and progression thus playing an important role in ovarian carcinogenesis [7, 8].
BRCA mutated breast and ovarian cancers are mostly sporadic. The large prospective study in the United Kingdom observed that germline BRCA mutations were seen in 12.4% women out of which 5.4% represents BRCA2 mutations while 7.4% were of BRCA1 type [9]. Various studies reported that 10% incidence of BRCA mutations were reported in absence of family history of ovarian and/or breast cancers. BRCA mutation had been reported in 60–70% cases with family history of breast and/or ovarian cancers [10, 11]. The recent study conducted in Pakistan had detected 18.2% pathogenic BRCA mutations in cases of family history [12]. BRCA1/2 has an important role as a biomarker for specific diagnostics as well as a predictor for therapy. Previous studies have validated that patients with BRCA-mutations are more sensitive to first line chemotherapy as well as these cases show promising response after treatment with PARPi (inhibitors of Poly ADP Ribose Polymerase) [13, 14]. Presently in Pakistan, treatment options to these patients are not based on surveillance data or recommended international guidelines. To date, there is no study conducted for cancer susceptibility and prognostic genes in patients of SEOC in Pakistan and molecular targets relating the treatment response in Pakistan are yet to be defined from scratch. This follow up study was conducted to address the mutational status of a set of genes related to SEOC that aims to better define treatment response and outcome thus minimizing the burden of morbidity and mortality. The objective of the study is to give insight about the mutations in a set of cancer susceptibility genes in platinum resistant and sensitive patients of primary SEOC which may help in future studies with larger number of patients in modifying treatment modalities.
Material and method
Study population and sample collection
This was a follow up descriptive study conducted in INMOL (Institute of Nuclear Medicine and Oncology), Jinnah Hospital, Services Institute of Medical Sciences and Mayo Hospital, Lahore, Pakistan. Clinico-pathological/laboratory work was carried out in the Department of Morbid Anatomy and Histopathology, while, molecular studies were carried out at the Department of Physiology and Cell Biology, University of Health Sciences, Lahore. After approval from the institutional Ethical Review Board, a written informed consent was taken from each patient recruited in this study (Ethical review letter no.: UHS/REG-19/ERC/2071, dated: 12 June, 2019). The study comprised of forty seven consenting female patients diagnosed for the first time with resectable surface epithelial ovarian cancer presenting at the above mentioned tertiary care hospitals of Lahore. Relevant demographic information, selective present and past medical history, family history was taken and recorded on predesigned proformas after taking informed written consent following code of ethics of the World Medical Association (Declaration of Helsinki). After making a complete clinicopathological and radiological evaluation, surgery followed by chemotherapy was planned by the oncologists. Immediately after surgical resection, a representative portion of the excised malignant ovarian tissue and adjacent healthy tissue for comparison (0.5–1 gm) was snapped frozen in liquid nitrogen separately and stored at
Flow diagram for registration of study participants, use of screening methodology and frequency of genetic mutations in the studied population (During treatment 2 patients died whereas insufficient DNA amount was extracted from two samples. Finally, 43 patients were followed and observed for 6 months after treatment).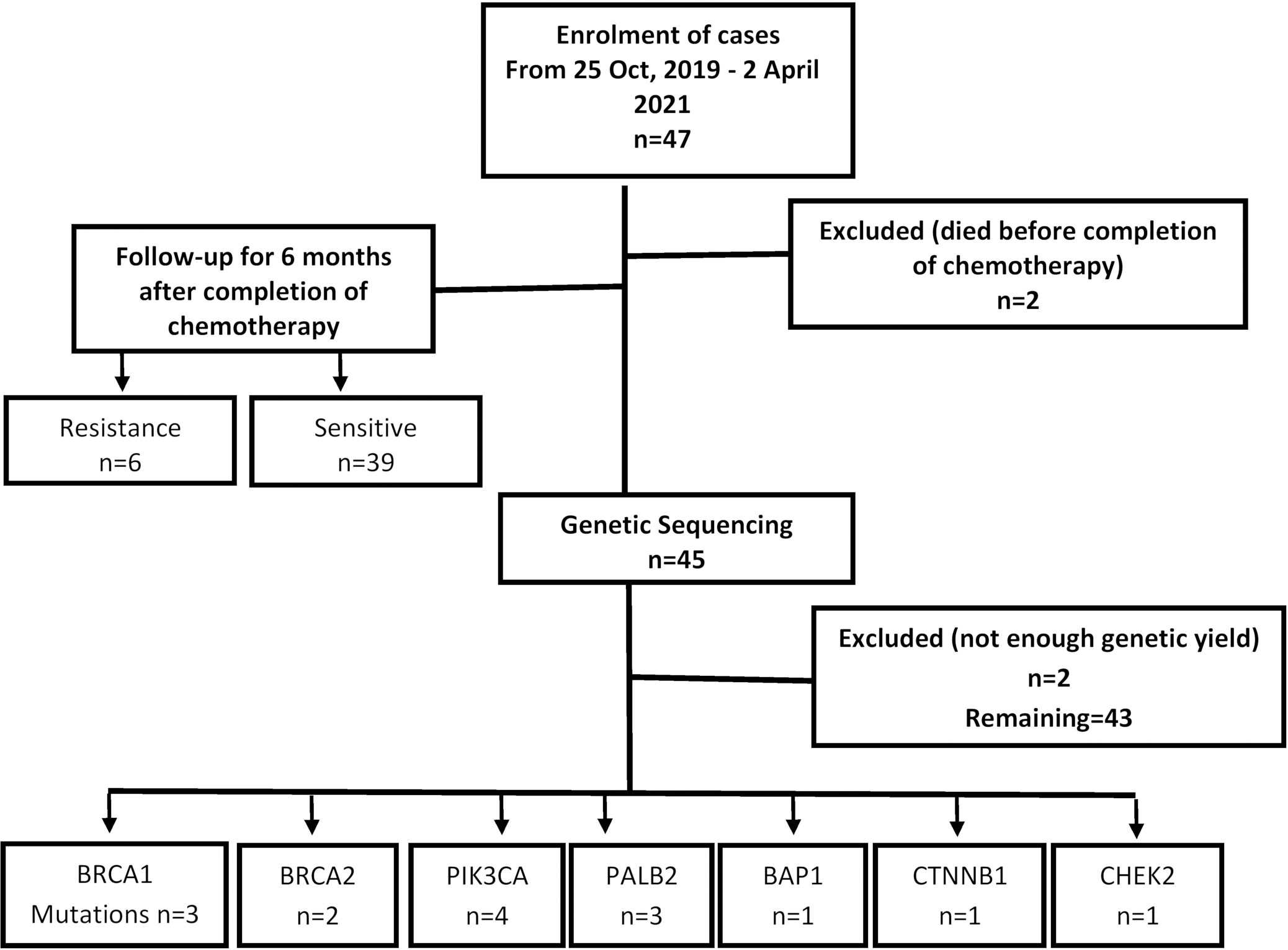
Updated International Federation of Gynaecology and Obstetrics (FIGO) (Jan. 2014)/TNM staging was done for each enrolled patient to ascertain the extent of tumor and the treatment options. Pre-surgery serum CA-125 levels were determined through ELISA.
Histopathological analysis
Processing/grossing of the surgical specimens’ (unilateral or bilateral oophorectomy, salpingo-oophorectomy either by laparotomy or laparoscopically, subtotal resection or removal of tumor fragments and hysterectomy with salpingo-oophorectomy) was done according to College of American Pathologists (CAP) protocol. Representative sections were taken for routine tissue processing and Hematoxylin and Eosin (H & E) staining. Histopathological reporting of the tumor was done according to CAP guidelines.
DNA extraction
For genetic analysis, the frozen tissue samples (0.5–1 gm) were grounded into small pieces in liquid nitrogen by using tissue homogenizer. Tissue sample was suspended in chilled lysis buffer. Protease inhibitor cocktail was added to avoid proteolysis during tissue lysis. Vigorous vortexing and centrifugation at 14,000 rpm for one hour at 4
Chemotherapy response
Descriptive characteristics of socio-demographic and clinical variables (
45)
Descriptive characteristics of socio-demographic and clinical variables (
After initial resection, the patients were enrolled for standard first-line chemotherapy comprising of a Taxane with the addition of Carboplatin (Inj. Paclitaxel intravenous with carboplatin in combination at one time, 3 weekly for 08 cycles with dose of paclitaxel ranging from 135 to 175 mg/m
The data was entered and analyzed using IBM SPSS (Statistical Package for Social Sciences) version 24. Mean
Results
Table 1a shows socio-demographic and clinical variables of 45 patients in studies population. The mean age with standard deviation of the ovarian cancer patients in the study was 46.04 (
Serum levels of CA125 in response to chemotherapy. (a) CA125 levels pre-chemotherapy with respect tor treatment response. (b) CA125 levels post-chemotherapy with respect tor treatment response. (c) Median differences between CA125 levels at the time of diagnosis and after administration of chemotherapy.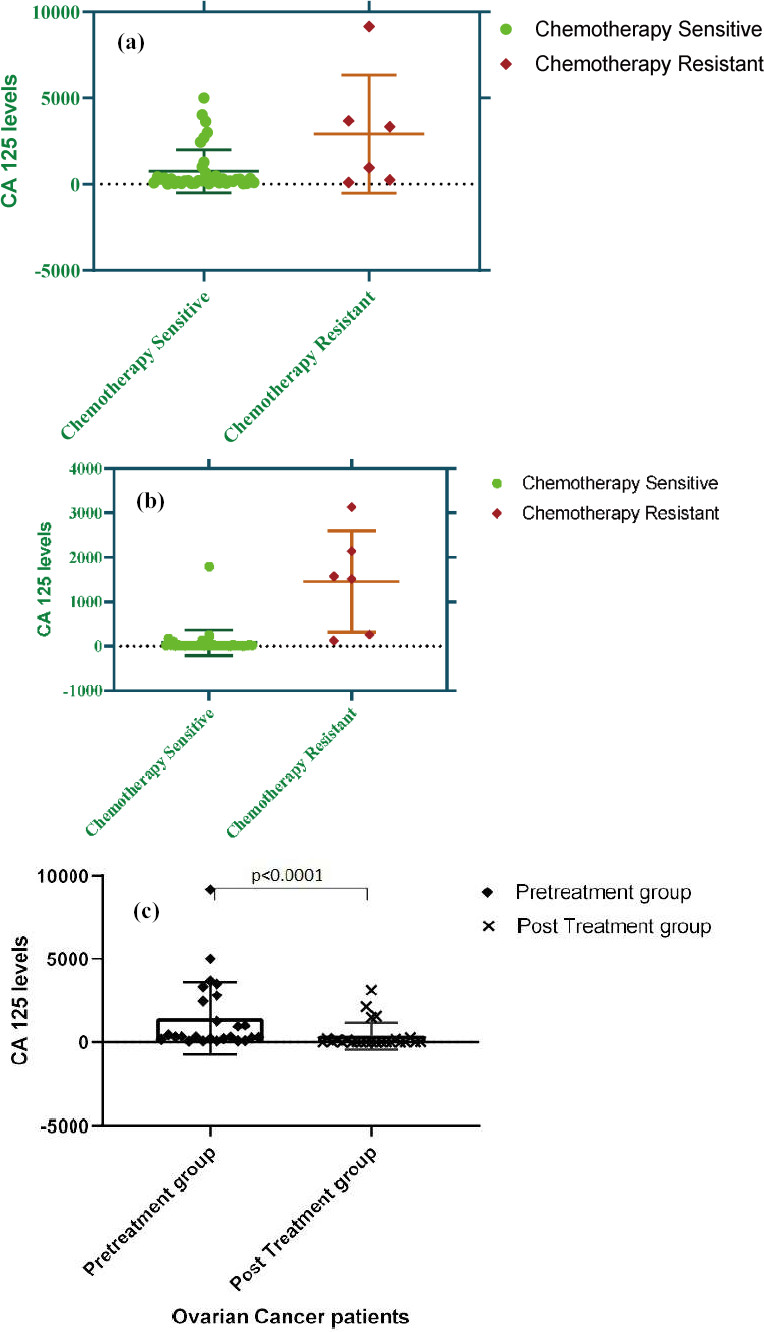
Frequency of genetic mutations in the studied population (
Cross tabulations of socio-demographic and clinical variables in the absence/presence of BRCA 1/2 mutations (
Table 2 shows the association of socio-demographic and clinical variables with BRCA 1/2 was analyzed using Fisher’s Exact analysis in 43 patients. The data shows that in patients whom CA125 levels decreased after chemotherapy, 9.8% were positive for BRCA 1/2 mutations, whereas a large number of patients (90.2%) were negative for BRCA 1/2 mutations. With respect to mutations present in patients, BRCA 1/2 was found positive in 15.2% of the patients and negative in 84.8% of the patients (Table 2).
Socio-demographic and clinical variables with outcome of chemotherapy (
Association of the genetic mutations with outcome of chemotherapy (
Table 3a presents the cross tabulations of socio-demographic and clinical variables with chemotherapy response in 45 patients. The results further showed that there was a significant association between mean CA125 levels pre- and post-therapy (
Chemotherapy response with presence/absence of BRCA1/2 mutations in the studied population.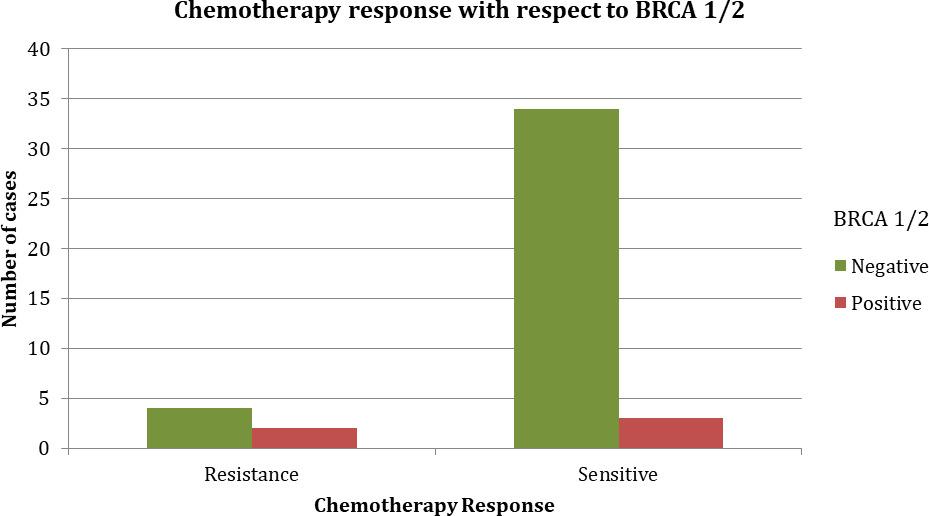
The graphically represented results further showed in Fig. 2c that the median differences between CA125 levels at the time of diagnosis and after administration of chemotherapy, as analyzed through related samples using the Wilcoxon Signed Rank Test was statistically significant (
Figure 3 depicts the chemotherapy response with respect to BRCA1/2 mutations. The studied population showed 6 cases of chemoresistance while 39 cases of chemosensitive response. Out of 3 cases of BRCA1 mutation positive, one case (33.3%) was resistant to chemotherapy. While out of 2 cases of BRCA2 mutation positive, one case (50%) was resistant. So out of 5 BRCA 1/2 positive cases, only 2 were resistant while 3 were sensitive. Moreover, both the resistant cases were of stage IV.
Table 4 depicts the mutational analysis in individual cases alongwith response to chemotherapy in studied population. There was no single case that had more than one mutation.
Mutational analysis of studied population with chemotherapy response
Footnotes
Conflict of interest
None.
