Abstract
BACKGROUND:
Information on genetic alterations, notably EGFR mutations, is important for guiding non-small-cell lung cancer (NSCLC) treatment. Circulating tumor DNA (ctDNA) analysis represents a less invasive alternative to tissue biopsy for analyzing mutation status, but its clinical value may vary across disease stages.
OBJECTIVE:
To explore clinical correlates of ctDNA and tissue/plasma-based EGFR mutation (EGFRm) status across all NSCLC stages.
METHODS:
Ninety patients were analyzed, representing three cohorts: newly-diagnosed early-stage, advanced-stage, and recurrent NSCLC. Relationships among clinical/surgical parameters, ctDNA, EGFRm status, and survival outcomes were analyzed.
RESULTS:
Plasma/tissue EGFRm concordance was lower in early-stage (58.6%) than in advanced-stage patients (87.5%). In early-stage patients, ctDNA levels were variable and not significantly associated with clinical/surgical parameters. In advanced-stage patients, time to EGFR-TKI treatment failure (TTF), but not overall survival (OS), was significantly longer in EGFRm-positive vs. EGFRm-negative patients. In patients with recurrent disease, 40% of plasma samples were EGFR T790M-positive at recurrence. In T790M-positive patients, we noted slight trends toward longer OS with vs. without osimertinib treatment and longer OS and TTF with second-line vs. later-line osimertinib.
CONCLUSIONS:
Our results affirm the use of ctDNA testing in advanced-stage and recurrent NSCLC. Further studies on osimertinib as early-line therapy, clinical correlates and the utility of plasma-based testing in early-stage NSCLC are warranted.
Introduction
Activating mutations in the epidermal growth factor receptor (EGFR) gene are among lung cancer’s most common driver mutations [1, 2]. In non-small cell lung cancer (NSCLC), EGFR mutations have been identified in 10–15% of Caucasian patients, and 30–50% of Asian patients [3, 4, 5]. The most frequently identified EGFR mutations are exon 19 deletions and exon 21point mutations [6, 7]. These mutations accounted for
However, even though treatment with first- and second-generation EGFR-TKIs is associated with excellent treatment responses, nearly all patients eventually develop resistance and recurrent disease. Development of EGFR-TKI resistance is most commonly associated with the presence of the EGFR T790M mutation, which has been identified in 40–60% of cases with disease recurrence [12, 13, 14]. Osimertinib, a third-generation EGFR-TKI that targets both EGFR-sensitizing and T790M resistance mutations, has been shown to prolong survival with high efficacy and safety [15, 16]. The US FDA approved osimertinib as a standard second-line treatment for T790M-positive patients and in the first-line setting for patients with EGFR mutations (exon 19 deletion or L858R) detected by any approved test.
Although tissue testing from surgical resection specimens is considered the gold standard for EGFR mutation detection, alternatives are needed in some clinically important scenarios [17]. One such scenario involves NSCLC patients who present with advanced or inoperable disease. In these circumstances, only small tissue specimens can be obtained from the biopsy under bronchoscopy, transthoracic needle biopsy, or cytology. A second common scenario involves EGFRm NSCLC patients who develop recurrent or progressive disease after EGFR-TKI treatment. In such patients, a repeat biopsy is very important to identify genetic alterations that drive resistance, especially the T790M mutation. However, it is often challenging to obtain a repeat biopsy due to the advanced stage of the disease, poorer performance status, or risk of complications from invasive interventions. Tumor heterogeneity across various metastatic sites also complicates the interpretation of test results.
Analysis of circulating tumor DNA (ctDNA) from “liquid biopsies”, such as a plasma or urine samples, is a promising method for EGFR mutation detection in NSCLC patients [18, 19]. However, due to the small size of cell-free DNA fragments, the low fraction of tumor-derived cell-free DNA, and individual variation among patients, EGFR mutation detection in ctDNA requires more sensitive detection methods than tissue testing [20].
Sensitivities for mutation detection in plasma compared with tissue samples ranged from 70–100% for EGFR-TKI-sensitizing mutations and up to 70–80% for T790M; a high concordance between different platforms has been observed [20].
Our study aimed to compare EGFR mutation status between matched tissue and plasma specimens in early-stage and advanced-stage treatment-naïve NSCLC patients and identify EGFR T790M mutations in patients previously treated with EGFR-TKIs, using a ddPCR assay. Several digital and non-digital PCR-based detection methods have been evaluated for plasma ctDNA-based EGFR mutation testing, including the Cobas and therascreen PCR assays, BEAMing dPCR, and digital droplet PCR (ddPCR) [21].
Methods
Patients and samples
This study was conducted between April 2015 and March 2016 at the Ramathibodi Hospital (Bangkok, Thailand), a major tertiary referral center. Patients with histologically confirmed pulmonary adenocarcinoma and available tumor tissue and blood samples were eligible for the study. Patients without adequate documentation of clinicopathologic and treatment characteristics or survival follow-up data for the planned analyses were excluded from the study.
Three cohorts of patients were analyzed, representing the newly diagnosed (I) early-stage and (II) advanced-stage disease, and (III) recurrent disease settings. The newly-diagnosed early-stage group included treatment-naïve patients with resectable stage I–IIIA tumors. For this group, the tissue samples were obtained from surgical resection specimens, and matched peripheral blood samples were collected pre-operatively (one day before surgery). The newly-diagnosed advanced-stage group included treatment-naïve patients with unresectable locally advanced or metastatic disease (stage IIIB–IV). For this group, tissue samples were obtained from small biopsies or cytology specimens from primary tumors or metastatic sites, and matched peripheral blood samples were collected before EGFR-TKI therapy. The recurrent disease group included EGFRm patients with advanced-stage/metastatic disease previously treated with EGFR-TKI. Tissue samples from these patients were obtained from small biopsies or cytology specimens from primary tumors or metastatic sites. The matched plasma samples were those collected after disease progression on EGFR-TKI therapy.
Patients were enrolled after providing written informed consent, and the study conforms with The Code of Ethics of the World Medical Association (Declaration of Helsinki). The study was approved by the Ethics Committee of Ramathibodi Hospital.
Tissue-based EGFR mutation testing
DNA was extracted from formalin-fixed paraffin-embedded (FFPE) tissue samples using the QIAamp DNA FFPE Tissue Kit (QIAGEN), according to the manufacturer’s instructions. EGFR mutation testing was performed using the therascreen EGFR RGQ PCR Kit version 2 (QIAGEN) or the Cobas EGFR Mutation Test v2 (Roche Molecular Systems), as per routine practice in our institution.
Plasma-based EGFR mutation testing
Sample preparation and DNA extraction
Peripheral blood samples were collected in EDTA tubes and centrifuged once at 2,500
Digital droplet PCR detection of EGFR mutations
Extracted ctDNA was tested for EGFR mutations (exon 19 deletion, L858R, T790M, S768I, L861Q, and G719X) using the Droplet Digital PCR system (Bio-Rad Laboratories). Reactions were performed in 20
Outcomes
Follow-up and treatment response evaluations based on clinical or imaging criteria were performed as per routine practice in our institution.
In the early-stage and advanced-stage cohorts, overall survival (OS) was defined as the time from diagnosis (EGFR mutation testing) to death from any causes or last follow-up. In early-stage patients, recurrence-free survival (RFS) was defined as the time from diagnosis to disease recurrence, death from any causes, or last follow-up. In advanced-stage patients, time to treatment failure (TTF) was defined as the time from initiation to end of first-line treatment.
In the recurrent-disease cohort, OS was defined as the time from disease recurrence (established by T790M mutation testing) to death from any causes or last follow-up. TTF was defined as the time from initiation to end of osimertinib treatment.
Characteristics of patients
Characteristics of patients
For analysis of matched tissue and plasma samples, concordance rate, sensitivity and specificity, and positive and negative predictive value (PPV, NPV) were calculated with reference to tissue EGFR mutation status. Two-sample Wilcoxon rank-sum (Mann-Whitney), Kruskal-Wallis, or Spearman’s rank correlation tests were used to analyze relationships between clinical/surgical parameters and ctDNA concentration, and between these parameters and EGFR T790M status. The Kaplan-Meier method was used to estimate survival times. Log-rank tests were performed to evaluate the significance of between-group differences in survival outcomes. Univariate and multivariate Cox regression models were used to analyze relationships between clinical/surgical parameters and survival outcomes.
All statistical analyses were performed using Stata software (version 16). Significance tests were performed at the 5% significance level (
Results
Concordance rate for EGFR mutation status in tissue and matched plasma specimens from newly-diagnosed early- or advanced-stage patients
Concordance rate for EGFR mutation status in tissue and matched plasma specimens from newly-diagnosed early- or advanced-stage patients
PPV, positive predictive value; NPV, negative predictive value.
Relationship between ctDNA concentration and surgical parameters in early-stage patients
A total of 90 patients were included in the study. Of these, 58 patients were diagnosed with early-stage (TNM stage I to IIIa;
EGFR mutation status in matched plasma and tissue specimens from newly-diagnosed NSCLC patients
In early-stage patients, EGFR mutations were identified in 14 out of 29 tissue specimens (48%) (Table 1). The matched plasma samples were also positive in two of these 14 tissue EGFRm-positive cases. In advanced-stage patients, EGFR mutations were identified in 18 of 29 tissue samples (62%), and 15 of these 18 matched plasma specimens were also positive. The plasma/tissue EGFR mutation concordance rate, sensitivity and specificity, and positive and negative predictive value of plasma tests are shown in Table 2.
Early-stage patients: Relationship between ctDNA concentration and clinical/surgical parameters
Plasma ctDNA concentration showed wide variation (range: 754–18440 ng/ml of plasma, median 3473 ng/ml). There was no significant association between ctDNA concentration and disease stage or any other clinical, or surgical resection parameters analyzed, including age, gender, smoking status, tumor size, lymphovascular invasion LVI, pleural invasion (PI), or nodal (N) metastasis (Table 3).
Early-stage patients: Relationship between clinical/surgical parameters and survival outcomes
Relationship between survival outcomes and clinical/surgical parameters in early-stage cases
Relationship between survival outcomes and clinical/surgical parameters in early-stage cases
The median overall survival (OS) in this group of patients was 54.4 months (95% CI: 45.7 months–not estimable). Univariate and multivariate analyses of relationships between RFS or OS and various clinical/surgical parameters are shown in Table 4. In multivariate models, shorter RFS was significantly associated with current-smoker status (
The median OS in this group was 15.5 months. Median OS in EGFRm tissue-positive patients (
Analysis of factors associated with the presence of EGFR
T790M in patients with recurrent disease
Analysis of factors associated with the presence of EGFR T790M in patients with recurrent disease
Overall survival (OS) and time to treatment failure (TTF) in advanced-stage NSCLC patients as assessed by clinical or imaging criteria as per routine practice of the institution. (A) OS and (B) TTF curves plotted for all advanced-stage patients (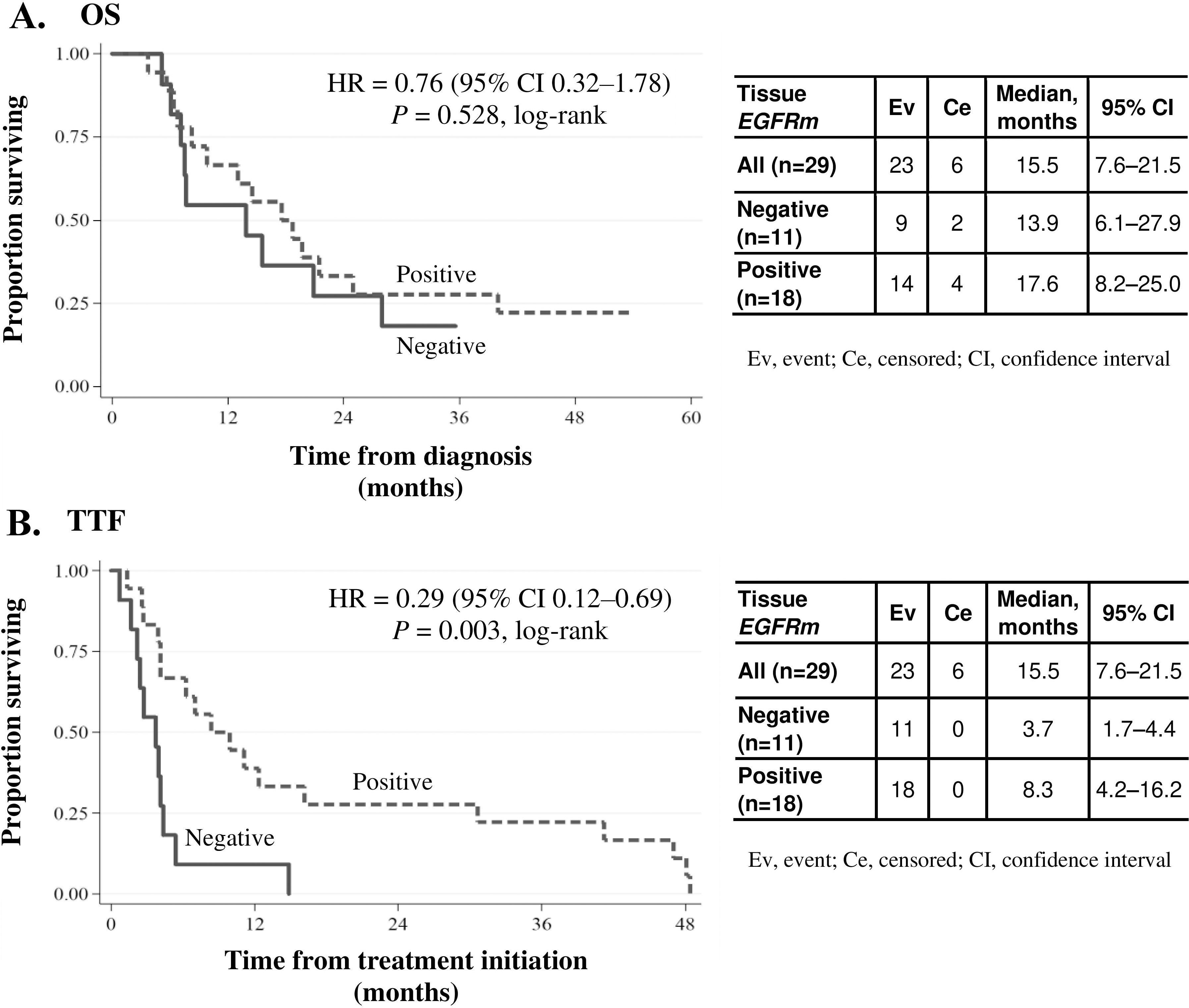
The median TTF was 44 (95% CI: 3.7–9.9) months. TTF showed a statistically significant relationship with tissue EGFRm status (Fig. 1B). Median TTF in EGFRm tissue-positive patients (
Overall survival (OS) and time to treatment failure (TTF) in patients with recurrent disease. (A) OS curve among EGFR
T790M-positive patients (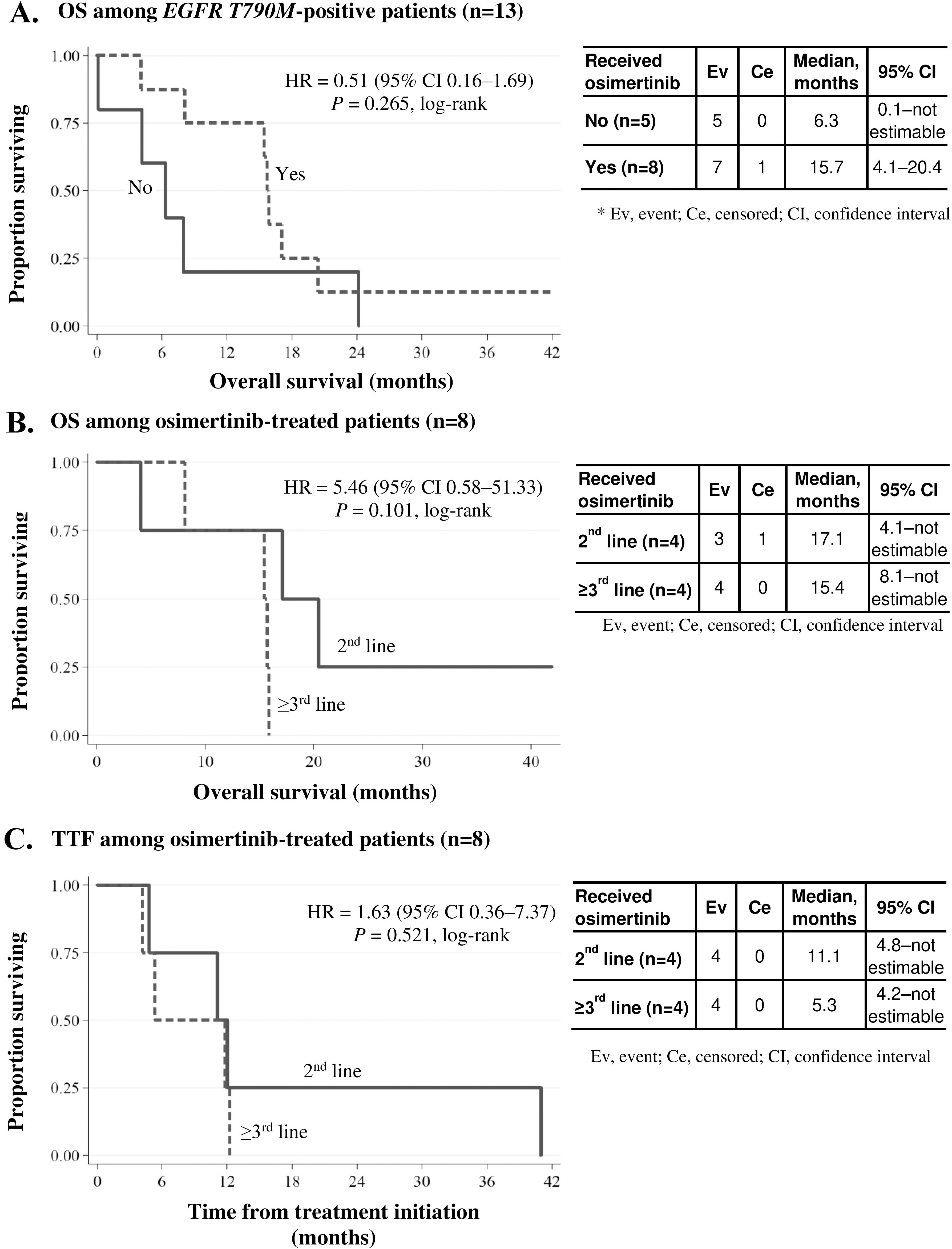
The median OS after disease recurrence was 12.2 months in this group (
Eight of the 13 T790M-positive patients were treated with osimertinib. As shown in Fig. 2A, OS appeared longer among osimertinib-treated patients, with a median OS (95% CI) of 15.7 (4.1–20.4) months versus 6.3 (0.1–not estimable) months among those who did not receive osimertinib. However, the difference was not statistically significant (
Among osimertinibtreated patients (
To identify potential predictors of T790M acquired resistance and disease recurrence, we also analyzed relationships between plasma T790M status and clinical variables (Table 5). In the T790M-positive group, plasma ctDNA concentrations and mutant EGFR DNA concentrations were significantly higher than in the T790M-negative group (
Plasma-based ctDNA testing shows promise for quantitative assessment of driver and resistance mutations in lung cancer and is becoming more widely used as an alternative to tissue-based testing. A 2018 IASLC position paper outlined two scenarios in clinical practice in which plasma-based testing may offer advantages: initial molecular diagnosis in treatment-naïve advanced EGFRm NSCLC patients, and detection of acquired resistance mutations in patients with disease progression on EGFR-TKIs and other targeted therapy [17].
On the other hand, the clinical value of plasma-based testing is less clear in early-stage EGFRm NSCLC, when ctDNA levels tend to be low and highly variable. In this study, we used quantitative digital droplet PCR, a highly sensitive detection method, to examine EGFRm status in plasma ctDNA samples from patients treated at our center in three different clinical settings: newly-diagnosed early-stage and advanced-stage NSCLC and recurrent NSCLC after first-line EGFR-TKI therapy.
Our analysis detected EGFR mutations in 71% of NSCLC patients based on tissue samples. This is generally consistent with reports from other institutions in Thailand (49.1–57.4%) [22, 23, 24], and similar to results from next-generation sequencing (NGS) mutation testing in our institution (68.1%) [24]. The most frequently detected EGFR mutation was the exon 19 deletion (32.2%), within the range documented in other studies from Thailand and other Asian countries (31.8–52.7%) [23, 25]. L858R mutations were also common (31.1% overall) in our study; this is consistent with the range of 9.3–37.6% (either alone or in combination with another mutation) reported by other studies [8, 23, 25]. The findings are also consistent with an earlier report from the EGFR Mutation Database, which estimated that 88% of all EGFR mutations occurred in exons 19 and 21 [7].
In newly-diagnosed patients with early-stage NSCLC, concordance between plasma and tissue EGFRm status was only 58.6%, with low detection sensitivity (14.3%). In these early-stage patients, we found that ctDNA levels were low and highly variable and showed no significant relationship with tumor size, lymphovascular or pleural invasion, or nodal metastasis. These findings are in broad agreement with previous analyses of ctDNA-based EGFRm detection sensitivity at different stages of the disease with a range of 10.0–22.2% for early-stage NSCLC, and 56.0–72.7% for advanced-stage NSCLC [18, 26, 27]. As the extent of disease is limited in early-stage NSCLC, even with lymphovascular invasion, the EGFR mutation levels in these patients’ samples may have been too low to detect, even with the highly-sensitive ddPCR method. Further research is needed to evaluate the value of plasma testing in early-stage NSCLC.
Concordance of EGFRm status in matched plasma and tissue was higher in advanced-stage (87.5%) than in early-stage patients. This is within the range reported by other studies in advanced NSCLC, where plasma-tissue concordance rates were 80% or above for matched samples (79.0–93.0%) [19, 20, 28, 29]. The observed sensitivity and specificity of plasma ctDNA-based EGFRm detection were similar to pooled estimates from meta-analyses (pooled sensitivity 62–75%; pooled specificity 79–96%) [18, 30, 31]. A more recent study reported 51% sensitivity and 77% specificity for plasma ctDNA-based EGFR T790M detection in advanced-stage NSCLC [32].
In 40% of the recurrent-disease patients (13/32), the T790M mutation was detected in plasma samples taken at recurrence, similar to what has been reported in Korean, Japanese, and Chinese patients (34.0–43.3%) [33, 34, 35]. Our study is the first report of the incidence of acquired T790M resistance (40.6%) in Thailand. Within the recurrent-disease group, we found significantly higher plasma ctDNA and EGFR mutant DNA levels in T790M-positive than in T790M-negative patients.
Although TTF on EGFR-TKI therapy was significantly longer in EGFRm-positive patients than in EGFRm-negative patients (whether based on tissue or plasma status), this did not appear to translate to significantly longer survival. However, among T790M-positive patients who received osimertinib for recurrent disease (
In this analysis of plasma ctDNA-based EGFR mutation testing and clinical outcomes in Thai patients with newly-diagnosed and recurrent NSCLC, the observed EGFR mutation patterns and clinical outcomes were broadly similar to other studies in Asia. Due to the small sample size in this study, it was difficult to establish firm conclusions regarding relationships between EGFR mutation status and survival outcomes, especially in subgroups. Our results affirm the use of plasma-based mutation testing in managing advanced and recurrent NSCLC, for example, in detecting acquired resistance mutations at recurrence. However, it will be essential to continue evaluating plasma-based testing in early-stage cases, especially as more sensitive and robust detection methods become available for research or clinical use.
Disclosures
Thanyanan Reungwetwattana: Research grant: AstraZeneca, Roche, Novartis, MSD; Advisory board: AstraZeneca, Roche, Novartis, MSD, Pfizer, Yuhan, Amgen, Takeda, BMS, BI; Speaker honoraria: AstraZeneca, Roche, Novartis, MSD, Pfizer, Takeda, BMS, BI.
Pimpin Incharoen: Advisory board: Novartis.
Author contributions
Conception and study design: Pimpin Incharoen, Ekaphop Sirachainan, Thanyanan Reungwetwattana.
Collection of the specimen and laboratory work: Pimpin Incharoen, Artit Jinawath, Lalida Arsa, Kaettipong Kamprerasart, Narumol Trachu, Nanamon Monnamo, Phichai Chansriwong, Ekaphop Sirachainan, Thanyanan Reungwetwattana.
Interpretation, analysis of data, and result conclusion and suggestion: Pimpin Incharoen, Artit Jinawath, Narumol Trachu, Dittapol Muntham, Ekaphop Sirachainan, Thanyanan Reungwetwattana.
Preparation of the manuscript: Pimpin Incharoen, Lalida Arsa, Kaettipong Kamprerasart, Khantong Khiewn-gam, Narumol Trachu, Dittapol Muntham, Ekaphop Sirachainan, Thanyanan Reungwetwattana.
Revision for important intellectual content: Pimpin Incharoen, Thanyanan Reungwetwattana.
Supervision: Thanyanan Reungwetwattana.
Footnotes
Acknowledgments
The authors would like to thank the following: Ramathibodi Cancer Center for the support of reagents and chemicals; the Division of Medical Oncology, Department of Medicine, Department of Pathology and Research Center, Faculty of Medicine Ramathibodi Hospital, Mahidol University for supporting the clinical data and clinical specimens.
The study was funded by Ramathibodi Cancer Center and Division of Medical Oncology, Department of Medicine, Faculty of Medicine Ramathibodi Hospital, Mahidol University, Bangkok, Thailand.
