Abstract
INTRODUCTION:
Nodal peripheral T-cell lymphomas [nPTCL] constitute a heterogeneous group of rare malignancies with aggressive biological behavior and poor prognosis. Epigenetic phenomena involving genes that control DNA-methylation and histone deacetylation play a central role in their pathogenesis. However, the mutational landscape involving epigenetic regulators has never been reported in Latin American patients and their prognostic impact remains controversial.
PATIENTS AND METHODS:
From 2000 to 2019, 59-Brazilian patients with nPTCL were eligible for screening mutations in the IDH-1, IDH-2, RHOA, TET-2 and DNMT3A genes by Sanger sequencing at Formalin-Fixed Paraffin-Embedded samples [FFPE] of diagnosis. We reported the frequency, distribution and potential prognosis of these mutations.
RESULTS:
With a median follow-up of 3.70 years, estimate 2-year OS and PFS were 57.1% and 49.2%, respectively. Mutations in the IDH-1 gene were not found, mutations in the IDH-2 occurred in 3.4% (2/59), RHOA in 23.7% (14/59), TET-2 in 50.8% (30/59) and DNMT3A in 62.7% (37/59). RHOA gene mutations were more frequent in PTCL, NOS and AITL (
CONCLUSION:
Mutations in RHOA, TET-2 and DNMT3A were frequent in Brazilian patients with nPTCL. TET-2 mutations were associated with lower ORR for CHOP-like chemotherapy, decreased PFS and unfavorable clinical-biological characteristics in non-ALCL (PTCL, NOS and AITL). Further studies using a larger cohort may validate our findings.
Introduction
Nodal peripheral T-cell lymphomas [nPTCL] are a heterogeneous group of rare malignancies derived from monoclonal proliferation of T-helper (CD4
Although rare, nPTCL are the most frequent group of mature T-cell lymphomas, accounting for 5–7% of all non-Hodgkin’s lymphomas [NHL] [1, 3]. These neoplasms show characteristic geographic distribution, being more prevalent in Latin America, where it represents up to 15–20% of all NHL [2, 4]. Its diagnosis is challenging, because their four main entities present morphological, immunophenotypical, molecular and genetic overlapping [1, 2, 3].
These tumors usually show aggressive behavior and poorer prognosis in comparison to aggressive B-cell NHL. Frequently, patients present unfavorable biological findings at diagnosis, including inactivation of the tumor suppressor genes TP53 and p15/p16, high expression of P-glycoprotein [Pgp] and multi-drug resistance phenotype [MDR]. They are usually resistant to anthracycline-based chemotherapy with high-rates of refractoriness to CHOP-like regimens and early relapses [5, 6, 7, 8]. However, nPTCL exihits marked heterogenity of prognosis with 5-year overall survival [OS] of 30–40% for PTCL, NOS and AITL, in comparision of 70–80% for ALK-positive ALCL [3, 9]. This heterogeneity is provided by the histopathological subtype, International Prognostic Index [IPI], gene expression profile [GEP] and mutational status of proto-oncogenes, tumor suppressors genes and the landscape of the epigenetic regulators genes [3, 10, 11, 12, 13].
Moreover, abnormalities of epigenetic regulators have been found in different subtypes of nPTCL, especially in Thf-phenotype lymphomas. Therefore, mutations promoting loss of function in genes involving in DNA-methylation and histone deacetylation, like TET-2, DNMT3A, IDH-2 and RHOA have been stated as essential in nPTCL pathogenesis [14, 15, 16, 17, 18, 19, 20]. These data have brought a new strategy of treatment for nPTCL with hypomethylating agents (5-azacytidine and decitabine) and histone-deacetylase inhibitors (romidepsin, vorinostat and belinostat) [21, 22]. However, the frequency and distribution of the epigenetic abnormalities in nPTCL has been widely variable through studies and they are unknown in Latin American population, where those neoplasms are more prevalent, comprising up to 25% of all NHL. Furthermore, the impact of prognosis of these abnormalities in nPTCL is also controversial.
We recently described that pre-treatment peripheral monocytosis
Patients and methods
Study design
This is a single-center and retrospective study performed at Hospital das Clínicas, Faculty of Medicine, University of São Paulo [HC-FMUSP], Brazil. It was approved by the Research Ethics Committee of the University of São Paulo in 2013 (CAPPesq-USP number: 02975012.0.000068) and was allowed to waive the application of the Informed Consent Term [ICT]. Clinical, laboratory and epidemiological data were captured from electronic medical records and Database of the Non-Hodgkin’s Lymphoma Group at the University of São Paulo.
Patients
We identified 124 patients with a biopsy-proven diagnosis of nPTCL treated at our institution from January 2000 to December 2019. Of these, 59 (59/124–47.6%) were included in this study. Patients with symptomatic heart failure or cardiac ejection fraction
Clinical and laboratory data, including age, gender, histopathological variant, IPI, Prognostic Index for T-cell lymphoma [PIT], number of extranodal sites involved by tumor, lactic dehydrogenase [LDH] serum, performance status by Eastern Cooperative Oncology Group [ECOG] scale, bulky disease
Date of diagnosis, the obtainment of remission, relapse and the first and last cycle of therapy or radiotherapy, date and cause of death, and the last follow-up were also taken to access the overall response rate [ORR], overall survival [OS] and progression-free survival [PFS]. Prior to commencing therapy, we collected complete blood count, biochemical tests, including renal and liver function, LDH and
Therapy, response assessment and follow-up
Patients were treated with 6 to 8 cycles of antracycline-based therapy, including CHOP-21 [cyclophosphamide 750 mg/sqm I.V. on D1, doxorubicin 50 mg/sqm I.V. on D1, vincristine 1.4 mg/sqm (maximum 2.0 mg) I.V. on D1 and prednisone 100 mg/day P.O. on D1 to D5] or CHOEP-21 regimens [CHOP plus etoposide 100 mg/sqm I.V. D1 to D3]. CHOEP-21 was preferentialy indicated for patients under 60 years, ECOG
Patients presenting paranasal sinuses, paravertebral mass, kidney, adrenal, testes, ovaries, breast involvement, high-intermediate or high-risk IPI/PIT received central nervous prophylaxis with intrathecal injection of 12 mg of methotrexate and 2 mg of dexamethasone during the first four cycles of chemotherapy plus with two cycles of high-dose methotrexate (3,000 mg sqm I.V.) after the last cycle of chemotherapy.
Out of ALK-positive ALCL, patients under 65 years, ECOG 0-1 and no significant comorbidities received ASCT in first complete response [CR] or partial response [PR]. Interim response assessment after the 4th cycle and at the end of treatment was performed using CT or 18-FDG-PETCT [26]. Clinical follow-up was done every 3 months in the first two years, every 6 months in the third and fourth years and annually after five years for patients with CR.
Histology and immunohistochemistry
Tissue sections of 5-
Biopsies showing atypical lymphoid infiltrate of small and medium size cells along with polymorphic and inflammatory background, intense vascular proliferation and with follicular dendritic cells [FDC] irregularly distributed were complemented with follicular T-helper markers as CD10 (Novocastra, S6C6, 1/2000), BCL-6 (Abcam, EPR11410-43, 1/500), ICOS (Abcam, SP98, 1/100), CXCL-13 (Abcam, Ab112521, 1/300) and PD-1 (Abcam, NAT105, 1/1000). Likewise, it was perfomed IHC for vascular markers [CD31 (Dako, JC/70A, 1/100) and CD34 (Dako, QBEand-10, 1/2000)], FDC antigens [CD21 (Novocastra, 2G9, 1/800) and CD23 (Biocare, 1B12, 1/1000)] and in situ hybridization (ISH) for Epstein-Barr virus using ZytoFast Plus CISH
However, presence of atypical lymphoid infiltrates of large and pleomorphic cells (“hallmark cells”) expressing Ki67
Molecular biology
Total DNA was isolated from five cuts of 10-
Amplification of target DNA fragments of TET-2, DNMT3A, IDH-1, IDH-2 and RHOA genes were run in the Veriti
The sequencing by Sanger using BigDye™Terminator v 3.1 Cycle Sequencing Kit (Thermo Fisher Scientific, Wilmington, DE, USA) and M13 primers was performed in the Veriti
Statistical analysis
The sample characterization was described in absolute and relative frequencies for categorical variables and by measures of central tendency, dispersion and position for numerical variables. All variables were accessed for association with mutations in TET-2, DNMT3A, RHOA, IDH-1 and IDH-2 genes. Mutations were described in absolute and relative frequencies for the global cohort and according to distribution by histopathological subtypes of nPTCL.
Log-rank test was applied to determine association between gene mutation and prognosis. Chi-square test with Bonferroni correction was used to test the association between mutational status and clinical-laboratorial features and prognostic factors. Univariate analysis to determine variable of prognosis was performed using the Cox-regression method. Due to the relative small sample size, multivariate analysis was not performed.
OS and PFS curves were estimated by the Kaplan-Meier method. For survival analysis, silent mutations were removed, only missense and nonsense mutations of RHOA and TET-2 genes were considered. OS was considered from the date of diagnosis to death and PFS from the date of diagnosis to disease progression, death or last follow-up. Data were censored at the last follow-up. Statistical tests were performed using STATA 12.0 softwares and a
Results
Clinical features
Table 1 displays the clinical features and therapy modalities of the 59 patients included in the study. The median age was 50 years (interquartile range [IQR]: 38–61 years) and 57.6% (34/59) were male. Of them, 37.3% (22/59) were ALK-negative ALCL, 27.1% (16/59) PTCL, NOS, 20.3% (12/59) ALK-positive ALCL and 15.2% (9/59) AITL. The LDH median was 488 U/L (297–737). Bulky was observed in 39% (23/59) of patients, B-symptoms in 83% (49/59), and 11.9% (7/59) presented bone marrow infiltration. ECOG
Clinical and laboratory characteristics and therapy in 59-Brazilian patients with nPTCL
Clinical and laboratory characteristics and therapy in 59-Brazilian patients with nPTCL
nPTCL: nodal peripheral T-cell lymphoma; IQR: interquartile range; ALCL: anaplastic large-cell lymphoma; PTCL, NOS: peripheral T-cell lymphoma, not otherwise specified; AITL: angioimmunoblastic T-cell lymphoma; IPI: International Prognostic Index; PIT: Prognostic Index for T-cell lymphomas; LDH: lactate dehydrogenase; ECOG: Eastern Cooperative Oncology Group; BM: bone marrow; CS: clinical stage; ASCT: autologous stem-cell transplantation.
CHOEP-21 was given as first-line for 47.4% (28/59) of patients, CHOP-21 for 28.8% (17/59), 15.2% (9/59) received other regimens and 8.5% (5/59) did not undergo any therapy due to early death. Patients received a median of 6 cycles (IqR 4–8) of chemotherapy and 33.9% (20/59) received ASCT in first CR/PR and 23.7% (14/59) were consolidated with involved-field radiotherapy.
The ORR rate was 59.3% (35/59–95% CI: 45.7–71.9%), 54.2% (32/59) reached CR and 3/59 (5.1%) PR. Lymphoma-relapse between 6 and 12 months after the end of primary therapy occurred in 8.4% (5/59) of patients and early relapse rate [ERR] of 14.3% (95% CI: 4.8–30.3%). The death rate [DR] was 52.5% (31/59–95% CI: 39.1–65.7%), the majority due to progression disease [PD] (15/31–48.3%), followed by infectious complications (10/31–32.2%). Cardio-respiratory complications caused 5/31 (16.1%) deaths and 1/31 (3.2%) patients died in consequency of a second neoplasm.
The median follow-up for the all cohort of nPTCL was 3.70 years (0.9–12.4), with a median OS of 4.2 years (95% CI: 1.1–not reached) and 2-year OS of 59.1% (95% CI: 45.5–70.4%). Median PFS was 1.4 years (95% CI: 0.6–3.1%), with 2-year PFS of 47.2% (95% CI: 34.0–59.2%) [Fig. 1]. Estimate 2-year OS was 72.7% (95% CI: 49.1–86.7%) for ALK-positive ALCL, 58.3% (95% CI: 27.0–80.1%) for ALK-negative ALCL, 49.2% (95% CI: 23.5–70.6%) for PTCL, NOS, and 44.4% (95% CI: 13.5–71.9%) for AITL. Estimate 2-year PFS for ALK-positive ALCL was 68.2% (95% CI: 44.6–83.4%), 58.3% (95% CI: 27.0–80.1%) for ALK-negative ALCL, 12.5% (95% CI: 2.0–32.8%) for PTCL, NOS and 22.2% (95% CI: 3.3–51.3%) for AITL.
Overall survival curve (A) and progression-free survival curve (B) for 59-Brazilian patients with nPTCL.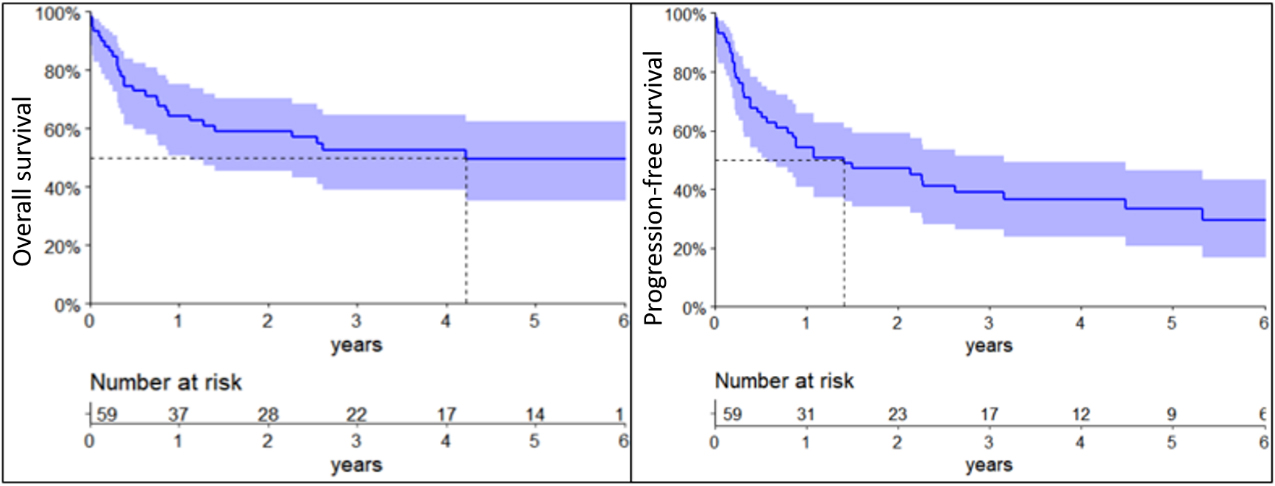
Diagram showing the frequency and distribution of IDH-1, IDH-2, RHOA, TET-2 and DNMT3A genes mutations according in the main subtypes of nPTCL (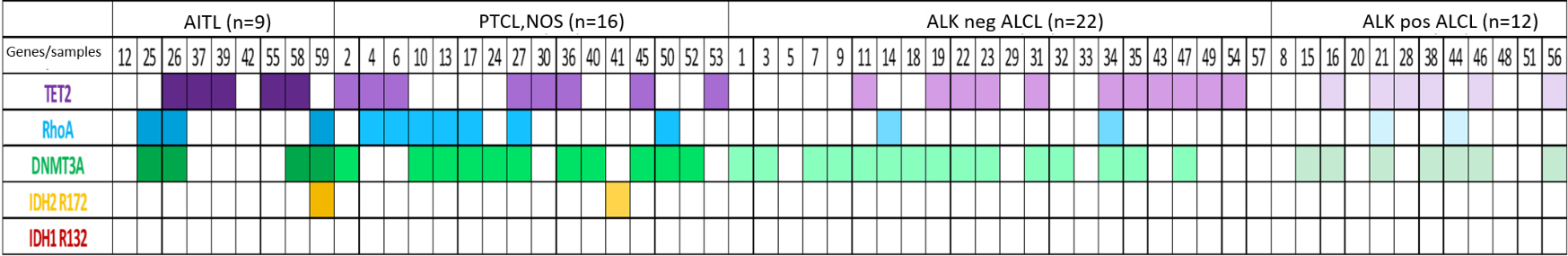
For the all cohort of nPTCL (
The IDH-2 R172 mutation occurred in 11.1% (1/9) of AITL, 6.2% (1/16) of PTCL, NOS and did not occur in any case of ALCL. RHOA gene mutations were seen in 33.3% (3/9) of AITL, 43.7% (7/16) PTCL, NOS and only 11.7% (4/34) of ALCL. TET-2 mutations were present in 55.5% (5/9) AITL, 50% (8/16) PTCL, NOS, 50% (11/22) ALK-negative ALCL and 50% (6/12) ALK-positive ALCL. The DNMT3A mutations were observed in 44.4% (4/9) of AITL, 68.7% (11/16) of PTCL, NOS, 68.1% (15/22) of ALK-negative ALCL and 58.3% (7/12) of ALK-positive ALCL [Fig. 2].
IDH-2, DNMT3A and TET-2 mutations were homogeneously distributed among the different subtypes of nPTCL, with
Mutations in TET-2, DNMT3A and RHOA genes. Distribution according to histopathological variants of nPTCL and region of the genes of interest. Classification of mutations described by subtype (synonymic, missense or nonsense).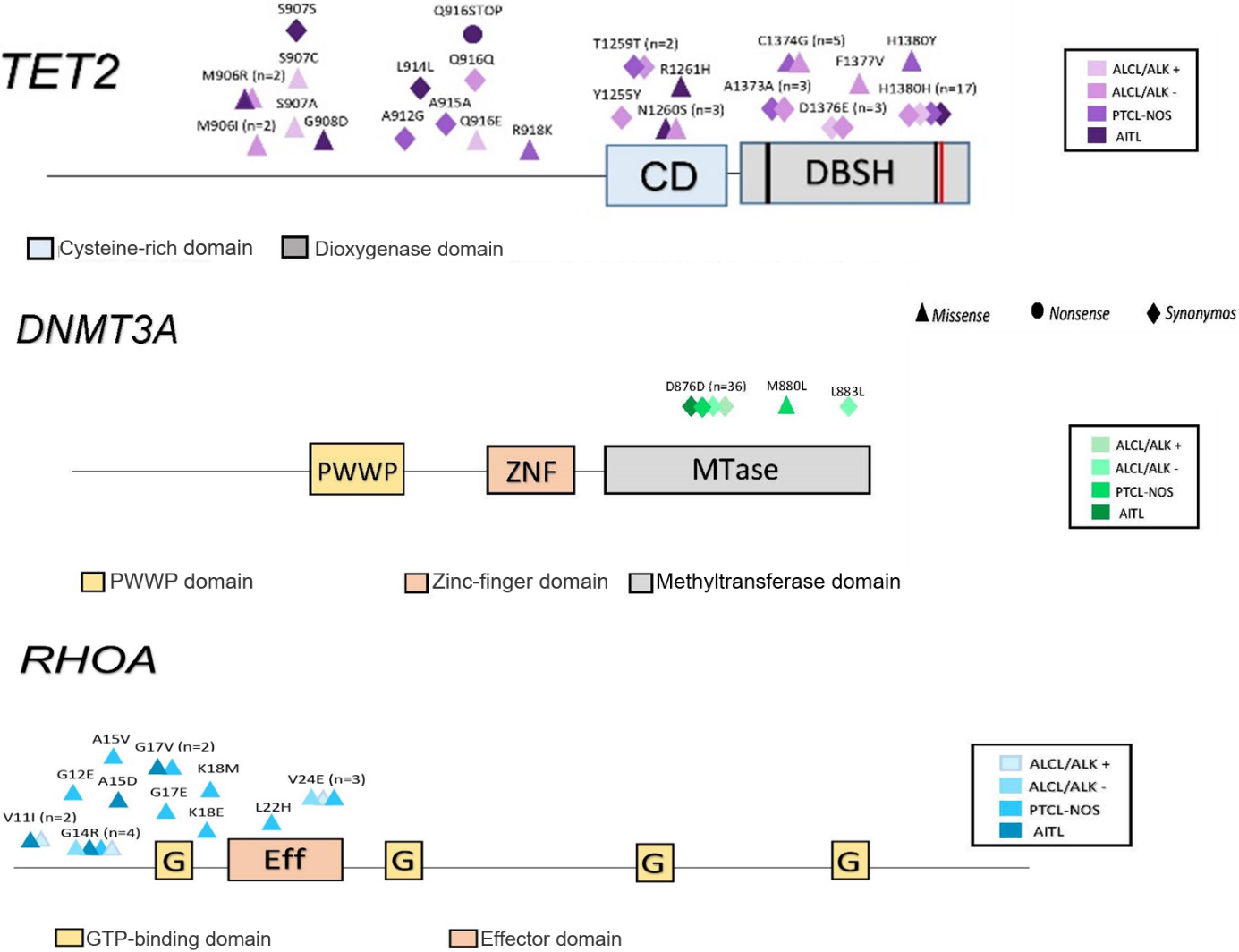
PFS according to mutational status of the RHOA gene in 59-Brazilian patients with nPTCL. The yellow line represents the RHOA-mutated patients and the blue line represents the RHOA-wild type patients (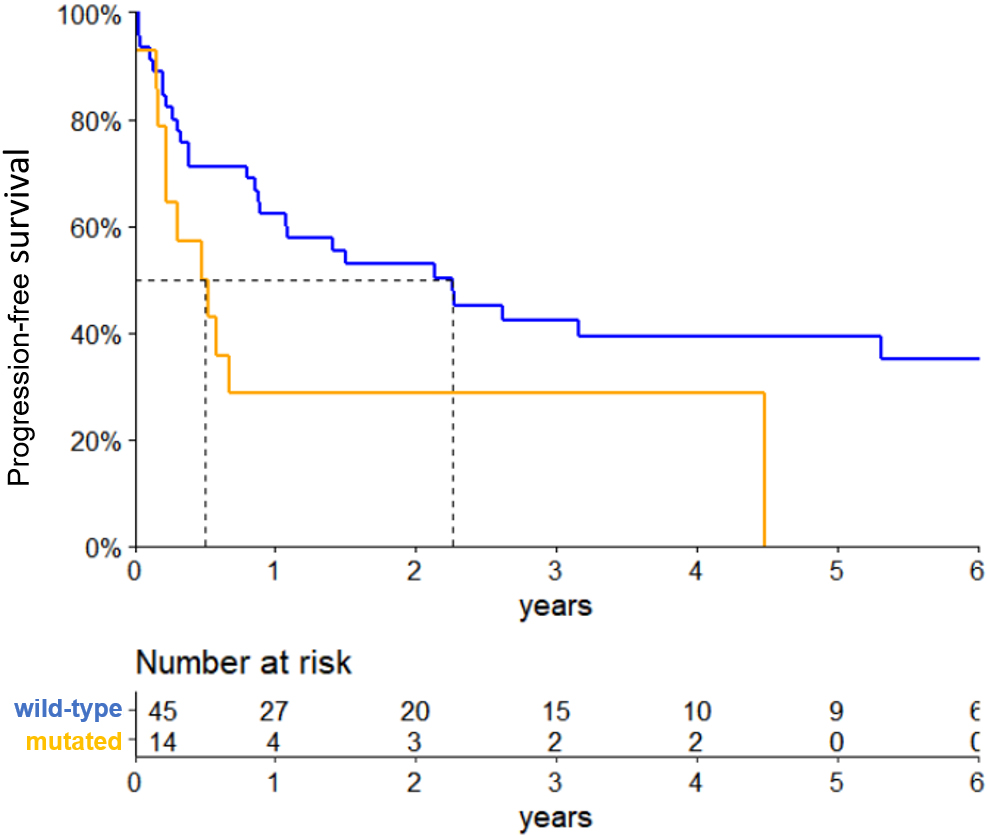
PFS according to mutational status of the TET-2 gene in the 25-Brazilian patients with PTCL, NOS and AITL. The yellow line represents the TET-2-mutated patients and the blue line represents the TET-2-wild type cases (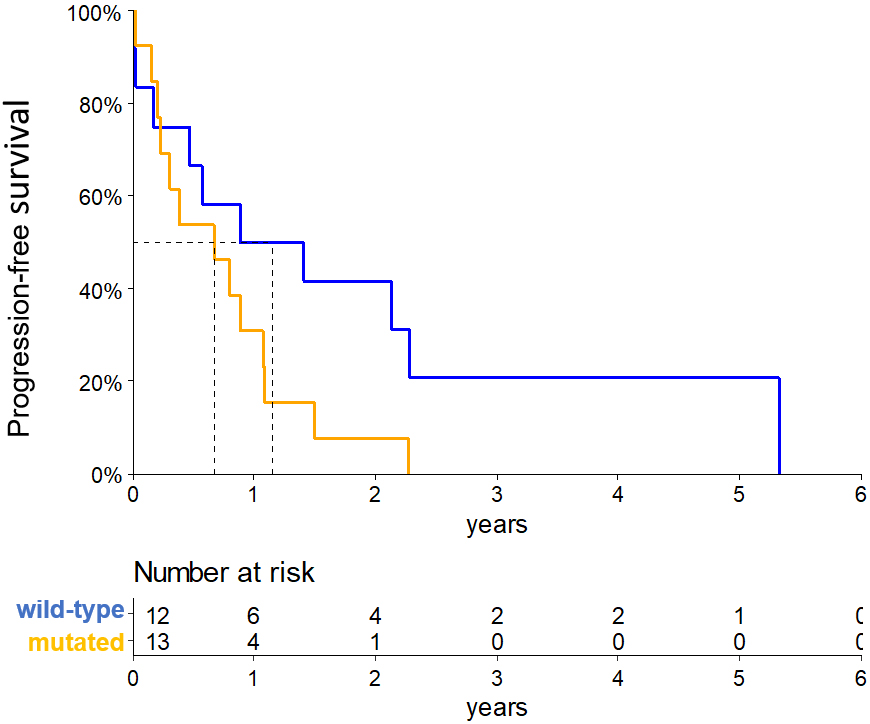
For the entire cohort of nPTCL (
For ALCL (
For non-ALCL (PTCL, NOS and AITL) (
Clinical and biological features and mutational status
In the total cohort (
In the subgroup of PTCL, NOS and AITL (
Discussion
This is the first study showing frequency, distribution and prognostic impact of the mutational landscape of epigenetic regulators genes in patients with nPTCL from Latin America. Here, we demonstrated that IDH-2, RHOA, TET-2 and DNMT3A mutations were recurrent in Brazilian patients with nPTCL. Their frequency were similar to previously reported in European and American patients [15, 17]. These findings support the relevance of epigenomic phenomena in the pathogenesis of nPTCL. Moreover, in our casuistic, RHOA gene mutations were more prevalent in PTCL, NOS and AITL and more prevalent in patients with high-tumor burden in the entire cohort.
Surprisingly, TET-2 mutations were statistically associated with decreased PFS, lower ORR and were associated with unfavorable clinical features in PTCL, NOS and AITL. Therefore, it is reasonable to suggest TET-2 mutations as a potential biomarker of poor prognosis in this setting.
TET-2 [ten-eleven translocation-2] gene encodes a Fe
More recently, TET-2 gene inactivating mutations were described in nPTCL, especially in PTCL, NOS and AITL (40%–50%) [15, 16]. Experimental studies showed that TET-2 mutations are essential for nPTCL development driving it lymphomagenesis [33]. However, the prognosis of TET-2 mutations remains controversial in nPTCL.
We also found that PTCL, NOS and AITL patients carrying TET-2 mutations had decreased 2-year PFS than those with no mutation (2 year-PFS: 7.6% vs 41.7%,
Our data is in agreement with other authors. Lemonier et al. examined TET-2 mutations in 190 nPTCL patients. They showed TET-2 mutations in 47% AITL and 38% PTCL, NOS cases. In this French cohort, TET-2 mutations were predictive of lower PFS (
Recently, Yingying et al. evaluated FFPE biopsies of 53 nPTCL cases by next-generation sequencing with a panel of 659 genes. They showed that TET-2 mutations were also frequent (64%) in nPTCL, particularly in AITL. Furthermore, TET-2 and TP53 mutations were associated with dismal survival (HR: 3.574, 95% CI: 1.069–11.941,
We also found that TET-2 mutations predominated, associated with unfavorable clinical features in PTCL, NOS and AITL patients. TET-2 mutations were more common in patients with intermediate-high and high-risk IPI (
Wang et al. investigated 92 non-ALK-positive nPTCL (PTCL, NOS, AITL and ALK-negative ALCL) searching for mutations involving IDH-2, TET-2, RHOA and DNMT3A genes by Sanger sequencing and pyrosequencing. They showed TET-2 mutations in 82.1% (32/39) AITL, 46.3% (19/41) PTCL, NOS and 33.3% (4/12) ALK-negative ALCL. In PTCL, NOS, TET-2 mutations were prevalent in both TBX-21 and GATA-3 subtypes. DNMT3A mutations also were recurrent (30–40%) in PTCL, NOS and AITL. As observed in our study, DNMT3A mutations were more frequent in the MTase-binding domain and TET-2 in the Cys-rich and double-stranded
Experimental studies have shown that TET-2 mutations may be concurrent with hypermethylation in it promotor region, causing it silencing and consequently loosing it function of tumor suppression [35]. Moreover, TET-2 gene is a critical element for genome stability and its deregulation disrupts DNA repair response assemblying a favorable ambient for clonal hematopoiesis development [36]. AML carrying TET-2 mutation is associated with hyperleucocytosis and high percentage of blasts [37]. Similarly, in our casuistic TET-2 mutations in PTCL, NOS and AITL were associated with high tumor burden, represented by high levels of LDH, bulky disease and high-risk IPI/PIT.
Notably, TET-2 mutations are frequent in hematopoietic precursor cells of healthy individuals, especially in elderly. These mutations drive clonal hematopoiesis of undetermined potential [CHUP] associated with higher risk of hematological malignancies [38, 39]. Even though TET-2 constitutive mutations are not enough to promote neoplastic transformation, the existence of pre-malignant clones carrying TET-2 mutations are highly susceptible to acquire and accumulate additional genetic abnormalities [40].
In this study, we did not search for TET-2 mutations in DNA of fibroblasts to distinguish if it was germline or somatic. However, the pathological meaning of TET-2 mutations identified in our cases was confirmed in the PolyPhen-2, Mutation Tasting platform, COSMIC and GDC databases. Another limitation of our study refers to the use of the Sanger methodology for sequencing the genes of interest. The use of a next-generation sequencing (NGS) platform would allow for complete gene mapping and the detection of a greater number of mutations. However, this study was conducted to search for specific mutations in target-genes based on the results of previous studies involving PTCL patients. To overcome this limitation, we are currently expanding our cohort in a multi-institutional task force that aims to identify new diagnostic and prognostic biomarkers in PTCL using whole exome sequencing (WES) by NGS.
Concerning RHOA gene, it encodes a small GTP-ase working in the conformation of the cytoskeleton, T-cell receptor (TCR) signaling, and driving T-lymphoid ontogeny [41]. The relationship between RHOA and AITL lymphomagenesis has been coming out since mutations in this gene occur in up to 60% of this lymphoma. Indeed, RHOA mutations are highly specific for AITL and nPTCL of Thf-phenotype. Therefore, RHOA mutations are considered specific biomarkers of these lymphoproliferative disorders [18, 42]. However, the prognosis of RHOA mutations remain controversial. RHOA G17V mutation was associated with unfavorable clinical characteristics by Nagao et al. [19]. In opposition, recent meta-analysis did not find any evidence of RHOA mutations as predictor of survival [42].
Similarly to other collaborative groups [15, 17], we were also able to demonstrate association of RHOA mutations and PTCL, NOS and AITL (
As previously reported, concurrent mutations in distinct epigenomic gene regulators were found in almost half of our casuistic. The cooperation between RHOA and TET-2 mutations in nPTCL-Thf pathogenesis has been previously questioned [20]. In this study, 42.8% (6/14) of cases that presented RHOA mutations had TET-2 mutations concomitantly, corroborating this theory Surprisingly, we also found that in the entire cohort of nPTCL RHOA and TET-2 mutations were correlated with high-tumor burden, with
The high frequency of epigenetic abnormalities occurring in the nPTCL setting, particularly in DNA methylation and histone regulation, contributed to come out a new therapeutic rationale targeting epigenetic regulation. Nowadays, a series of trials have been focused on testing efficacy and safety of hypomethylating agents such as 5-azacytidine plus histone deacetylase inhibitors (romidepsin, vorinostat and belinostat) for relapsed/refractory nPTCL patients. The results have been promising. As first demonstrated for myeloid neoplasms, recent studies have shown nPTCL carrying TET-2 and IDH-2 mutations may be better treated with epigenetic agents instead of conventional chemotherapy [44, 45].
Conclusion
In conclusion, we showed mutations in epigenetic regulators IDH-2, TET-2, DNMT3A and RHOA genes as recurrent genetic abnormalities in Latin American nPTCL patients. Their incidence and distribution were similar to described in the literature. RHOA mutations were more prevalent in THf-phenotype nPTCL and concurrent RHOA and TET-2 mutations were frequent and predominant in patients with high-tumor burden. As previously hypothesized, we also speculate if these concurrent mutations cooperate in nPTCL lymphomagenesis. TET-2 mutations were predictive of poor-PFS, lower ORR and were associated with unfavorable features in PTCL, NOS and AITL. These exploratory results should be validated further.
Author contributions
Conception: LAPCL and JP.
Interpretation or analysis of data: LAPCL, GCB, HFC, JBC, LBOA, LN, IB, MCNZ and JP.
Preparation of the manuscript: LAPCL.
Revision for important intellectual content: JP and VR.
Supervision: JP.
Availability of data and materials
All data generated and analyzed during this study is included in this published article. The raw data for this study are with the corresponding author and may be made fully available upon request via e-mail to the corresponding author.
Competing interests
The authors declare that they have no competing interests.
Ethical considerations
This study was approved by the local Ethic Committee (Sao Paulo University Ethic Committee) in 2013, under the number 02975012.0.0000.0068. All procedures followed were in accordance with ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2008. The application of the Free and Informed Consent Form (FICF) was waived by the Ethics Committee for Local Research (CAPPESQ – Ethics Committee for the Analysis of Research Projects at Hospital das Clínicas, Faculty of Medicine, University of São Paulo/HC-FMUSP).
Funding
The research project was funded by FAPESP – Fundação de Amparo à Pesquisa do Estado de São Paulo [process number 2012/50495-3].
Supplementary data
The supplementary files are available to download from http://dx.doi.org/10.3233/CBM-220013.
sj-docx-1-cbm-10.3233_CBM-220013.docx - Supplemental material
Supplemental material, sj-docx-1-cbm-10.3233_CBM-220013.docx
Footnotes
Acknowledgments
The authors would like to thank Ciro Chrispim for providing English review and editing. Ciro Chrispim for english review and editing.
