Abstract
Melanoma, a skin cancer derived from malignant melanocytes, is characterized by high aggressiveness and mortality. However, its exact etiology is unknown. Recently, the roles of exosomes and exosomal microRNAs (miRNAs) in the progression and therapy of various disorders, including melanoma, have gained attention. We investigated the impact of miR-138-5p from exosomes released by human mesenchymal stem cells (HMSCs) on the pathogenesis of melanoma. We isolated exosomes from HMSCs (HMSC-exos) by ultracentrifugation and verified them by specific biomarkers and transmission electron microscopy. We used CCK8, flow cytometry, quantitative real-time PCR (qRT-PCR), and Western blots to investigate cell proliferation, apoptosis, and mRNA and protein levels, respectively. Additionally, we used luciferase assays to examine the relationship between miR-138-5p and SOX4. Administration of HMSC-exos dramatically repressed the growth of melanoma cells. Elevated miR-138-5p levels in HMSC-exos were linked to increased cell apoptosis, and miR-138-5p downregulation had the opposite effects on cells. SOX4 was targeted by miR-138-5p through direct binding to the SOX4 3’UTR. In melanoma tissues, miR-138-5p was downregulated, and SOX4 was upregulated and was negatively correlated.
MiR-138-5p plays a crucial role in melanoma progression. The negative regulation of SOX4 transcription mediates the function of miR-138-5p. These findings provide a novel concept of melanoma pathogenesis and identify a valuable target (miR-138-5p/SOX4 axis) in treating this disease.
Keywords
Introduction
Melanoma, which evolves from the malignant proliferation of melanocytes, is a severe skin cancer with high aggressiveness, metastasis, mortality, and recurrence [1, 2]. Furthermore, a rapid increase in melanoma incidence has occurred over the past few decades, reaching 287,723 cases worldwide in 2018 [1, 3, 4]. Early-stage melanoma can be removed surgically, while metastasized melanoma requires multiple treatment strategies, including chemotherapy, radiotherapy, targeted therapy, and immunotherapy [1]. However, the outcomes and prognoses of most current therapies are under satisfactory. Therefore, exploring the pathogenic mechanisms of melanoma and identifying novel drug targets for the treatment of melanoma are urgently needed.
Although melanoma is related to genetic variance and ultraviolet light exposure, its exact etiology and mechanisms are still unclear. Recent publications highlighted the importance of microRNAs (miRNAs) in the development and prognosis of melanoma [5, 6, 7]. Moreover, aberrant miRNA expression has been linked to melanoma progression [8, 9, 10]. For example, upregulated expression of the miR-17-92 cluster was observed in many tumors and is linked to the unconstrained proliferation of cancer cells [11]. Conversely, overexpression of miR-138 suppresses proliferation and invasion and promotes apoptosis of tumor cells [12, 13], suggesting its potential as a molecular target. However, the functional mechanism of miR-138 in melanoma is unclear.
Exosomes are bioactive lipid bilayer vesicles secreted by most cells. Exosomes mediate intercellular communication and communication of cells with the surrounding environment by transmitting various molecules like miRNAs [14, 15]. Previous report has illustrated that exosomes show great promise in cancer immunotherapy because of their immunogenicity and molecular transfer function [16]. Moreover, miR-200b in exosome from colorectal cancer cells promoted colorectal cancer proliferation upon TGF-
MiRNAs elicit responses in cells by targeting specific target genes/proteins. For instance, miR-14-5 and miR-30a-5p elicit changes in melanoma through targeting SOX4 [18, 20]. As an essential developmental transcription factor, SOX4 has versatile roles in many physiological processes and modulate multiple signaling pathways [18, 20]. Whether SOX4 mediates the action of miR-138-5p in the context of melanoma remains undetermined. This study aimed to determine the role of miR-138-5p from MSC-derived exosomes, in the melanocyte survival and investigate the molecular mechanisms involved.
Materials and methods
Human samples
The independent ethics committee of Fudan University Shanghai Cancer Center authorized this study, which was complied with the Declaration of Helsinki. All patients signed a consent agreement before the experiment. We collected 50 human primary melanoma tissues (I–III,
Cell culture
We acquired the cells used in the present study from the cell bank of Shanghai Biology Institute (Shanghai, China), including human mesenchymal stem cells (HMSCs), melanoma (A2058, A375, SKMEL1 and WM115) and a corresponding normal human epidermal melanocyte HEMA-LP. Cells were grown in DMEM (Trueline, Kaukauna, WI, USA) supplied with a 10% FBS (Thermo Fisher Scientific), 2 mM L-glutamine, and 1% penicillin/streptomycin (Solarbio, Beijing, China) and were maintained in an incubator with 5% CO
Isolation and characterization of human mesenchymal stem cell-derived exosomes (HMSC-exos)
We prepared HMSC-exos as described previously, with some modifications [22]. We collected conditioned media from cultured HMSCs and centrifuged them at 1,000 g for 20 min to clear away cell debris, followed by sequential centrifugation at 2,000 g and 10,000 g for 20 min each. The supernatants were then gathered and condensed with a molecular weight cut-off (MWCO) of 100 kDa (Millipore) at 1,000 g for 30 min. The condensed supernatants were layered over 5 mL of 30% sucrose/D2O and subjected to ultracentrifugation at 100,000 g for 1 hour (optimal-90K; Beckman Coulter). The fraction with enriched exosomes was diluted with PBS and subjected to further centrifugation 3x at 1,000 g for 30 min using a 100 kDa MWCO. The purified exosomes were filtered through a 0.22
Validation of exosome uptake by fluorescent labeling
Green fluorescent linker PKH67 (UR52303, Umibio, Shanghai, China) was applied to stain exosomes according to manufacturer’s protocols. Briefly, we seeded SKMEL1 and WM115 cells into 24-well plates at a density of 50,000 cells/well and prepared 5 wells for each group. Normal culture media or culture media with PKH67 labeled exosomes (20
Real-time quantitative PCR (RT-qPCR)
Total RNA was extracted from samples using TRIzol Reagent (Invitrogen, Waltham, MA, USA). Corresponding cDNAs were synthesized from isolated RNAs using a kit (Thermo Fisher Scientific, Waltham, MA, USA) following the manufacturer’s instructions. The conditions used for RT-qPCR were: 95
Western blot
We prepared total protein from various samples using RIPA lysis buffer (JRDUN, Shanghai, China) plus EDTA-free Protease Inhibitor Cocktail (Roche, Heidelberg, Germany). Protein concentrations were determined using the BCA method (Thermo Fisher Scientific). Total protein (25
Knockdown and overexpression of SOX4
Short interfering RNAs targeting human SOX4 (siSOX4-1: 5’-GGAAGCUGCUCAAAGACAGTT-3’; siSOX4-2, 5’-GACAGCGACAAGAUCCCUUTT-3’; and siSOX4-3 5’-GCGACAAGAUCCCUUUCAUTT-3’) and negative control siRNA (siNC, 5’-UUCUCCGAACGUGUCACGUTT-3’) were synthesized (Major Industrial Co., Ltd, Shanghai, China) and cloned into lentiviral plasmids (pLKO.1). For SOX4 overexpression, we prepared a pLVX-puro lentiviral plasmid containing SOX4 (NM_ 003107.3) cDNA (oeSOX4), along with a mock plasmid as a negative control (oeNC). Lipofectamine 2000 was used to transfect cells following the manufacturer’s instructions and cells were cultured for 48 hours before the subsequent analysis.
Cell proliferation
We measured cell proliferation was measured using a cell Counting Kit-8 (CCK-8) assay (Signalway Antibody, Maryland, USA). At 0, 24, 48, and 72 h, we incubated cells with CCK-8 solution (1: 10) for 1 h. Cell proliferation was quantified using a microplate reader (Pulangxin, Beijing, China) with optical densities at a wavelength of 450 nm. We performed each experiment in triplicates.
Flow cytometry
Cells were harvested 48 h post-transfection and stained with Annexin V-fluorescein isothiocyanate (Beyotime, Beijing, China). Apoptosis was measured by flow cytometry (BD, San Diego, CA, USA). Annexin V
Dual-luciferase reporter gene assay
Potential binding sites for miR-138-5p in the SOX4 3’-UTR were predicted using TargetScan and Starbase. According to the predictions, DNA fragments containing wild-type or mutant sequences for miR-138-5p binding were synthesized and cloned into luciferase reporter vectors (pGL3-Basic). We introduced these constructs (namely, wild-type (WT) and Mut 3’UTR), with an internal reporter plasmid and miR-138-5p mimic or inhibitor, into human 293T cells and measured luciferase activities 48 h after transfection using a dual-luciferase reporter gene kit (Beijing Yuanpinghao Biotechnology Co., Ltd.).
Statistical analysis
We conducted statistical analyses with GraphPad Prism Software Version 7.0 (La Jolla CA, USA). Data are presented as mean
Results
MiR-138-5p expression is reduced in human melanoma tumors
The relative levels of miR-138-5p in 50 human melanoma tissues were divided into two groups (I–III,
miR-138-5p was downregulated in human melanoma tumors. 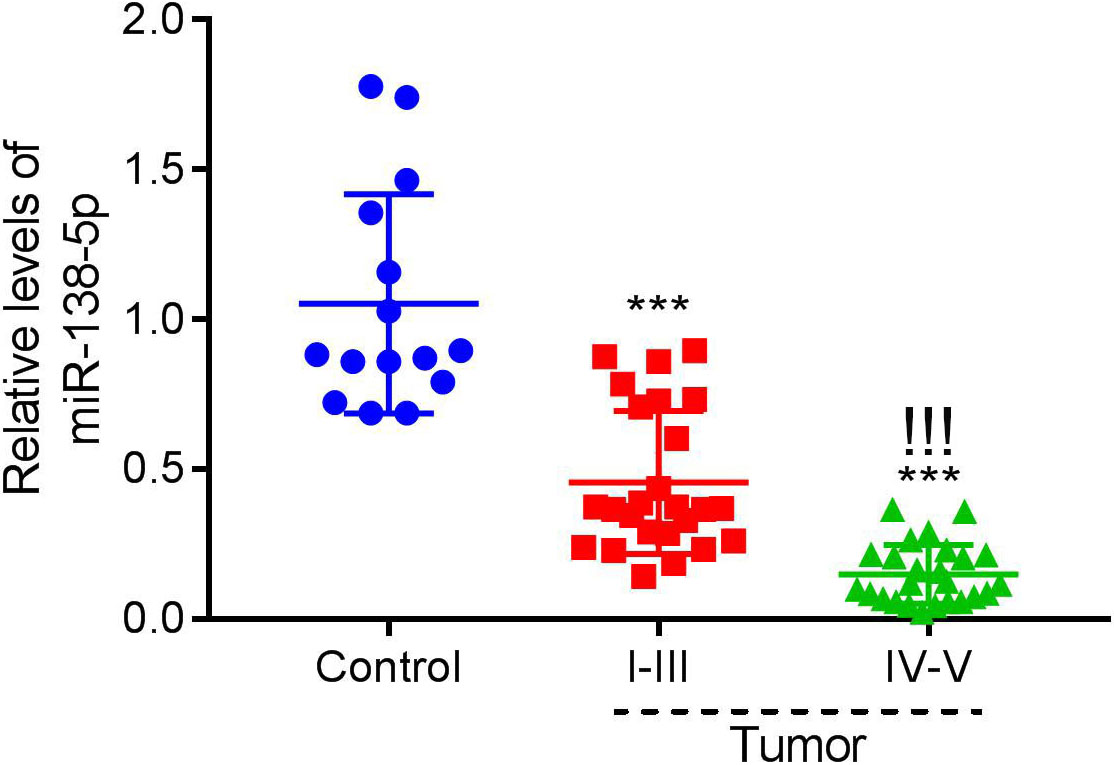
Human mesenchymal stem cell (HMSC)-derived exosomes (HMSC-exo, HE) inhibited the proliferation of human melanoma cells in a dose-dependent manner. A and B. The uptake of control-exos or HMSC-exos labeled with PKH-67 by human SKMEL1 and WM115 cells was determined using immunofluorescence. C and D. CCK8 assays were used to examine the proliferation of SKMEL1 and WM115 cells after co-culturing with 0, 100, 200, and 500 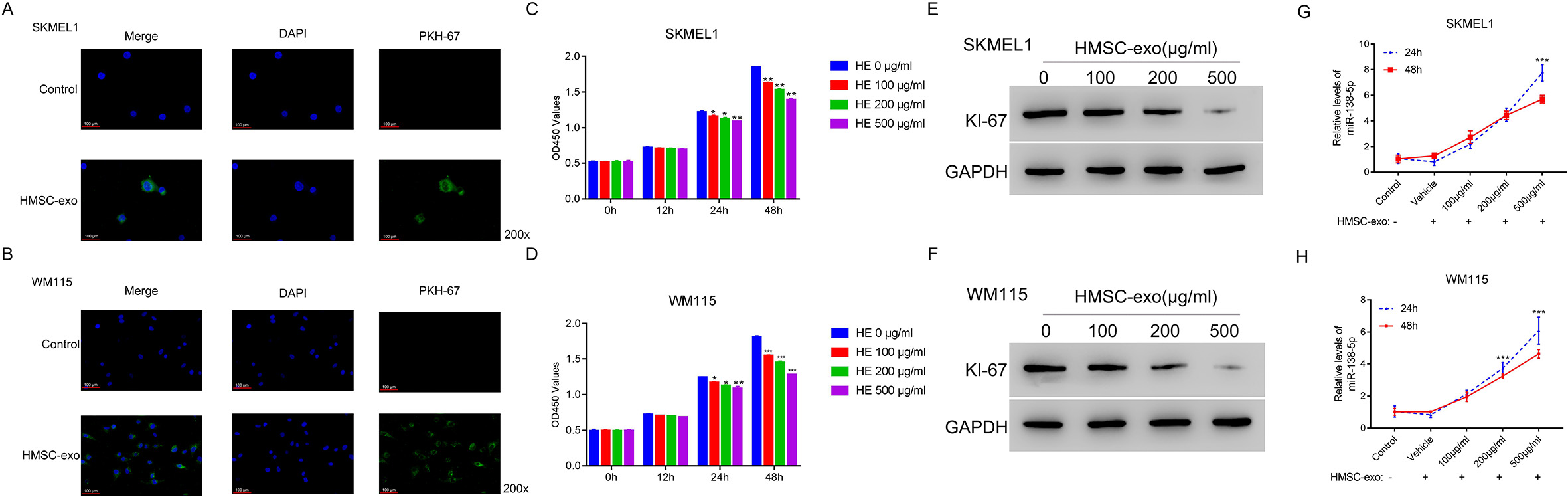
Dysregulated miR-138-5p affects the impact of HMSC-exo on human melanoma cell apoptosis. A and B. Flow cytometry was used to examine the apoptosis of SKMEL1 and WM115 cells after co-culturing with miNC-exo, mimic-exo, or inhibitor-exo. 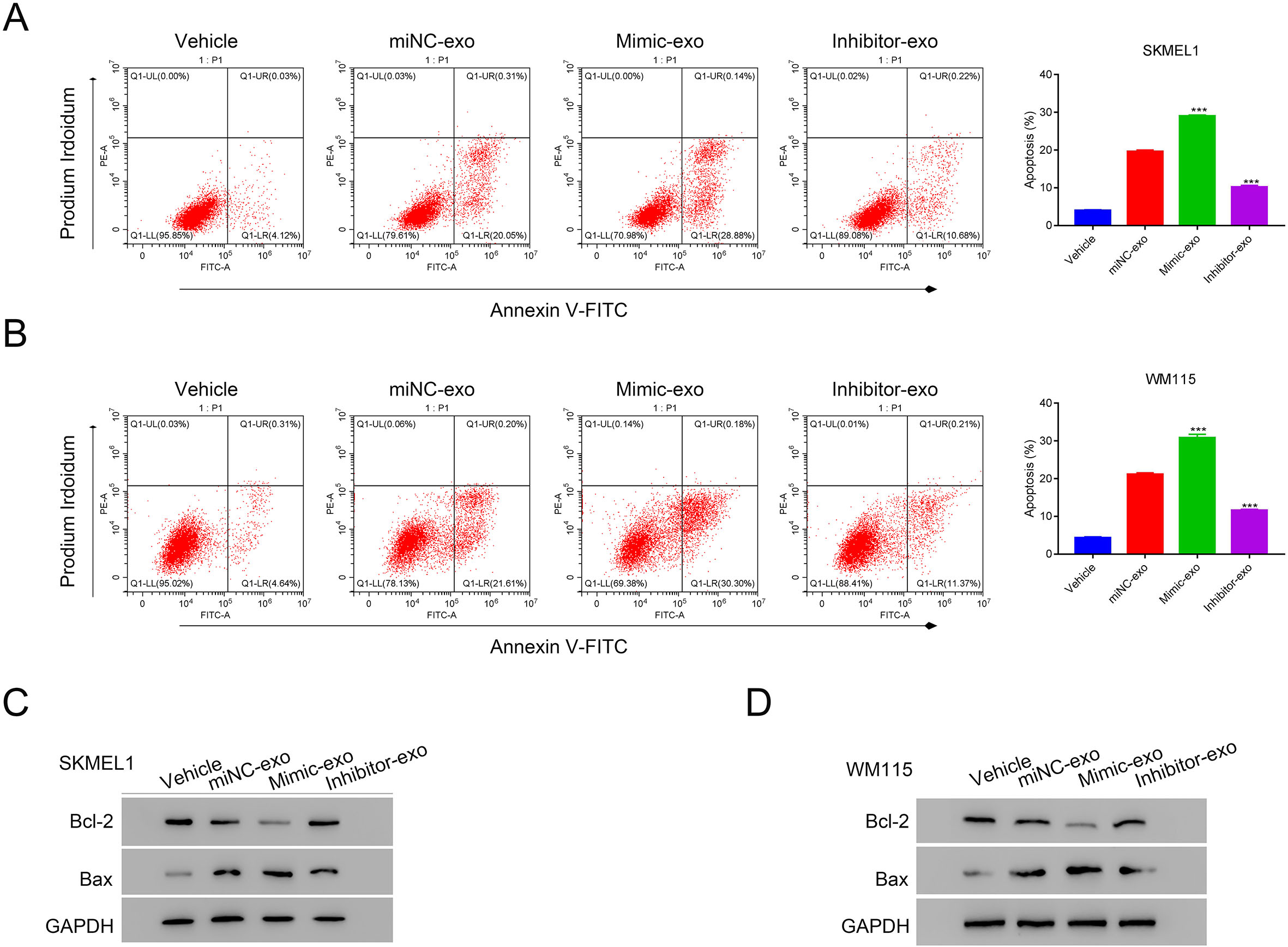
Has-miR-138-5p inhibited the transcription of SOX4 through binding the SOX4 3’UTR in SKML1 and WM115. A–D. The relative levels of miR-138-5p and SOX4 were examined in SKML1 and WM115 cells in the presence of miNC, inhibitor, or mimic. 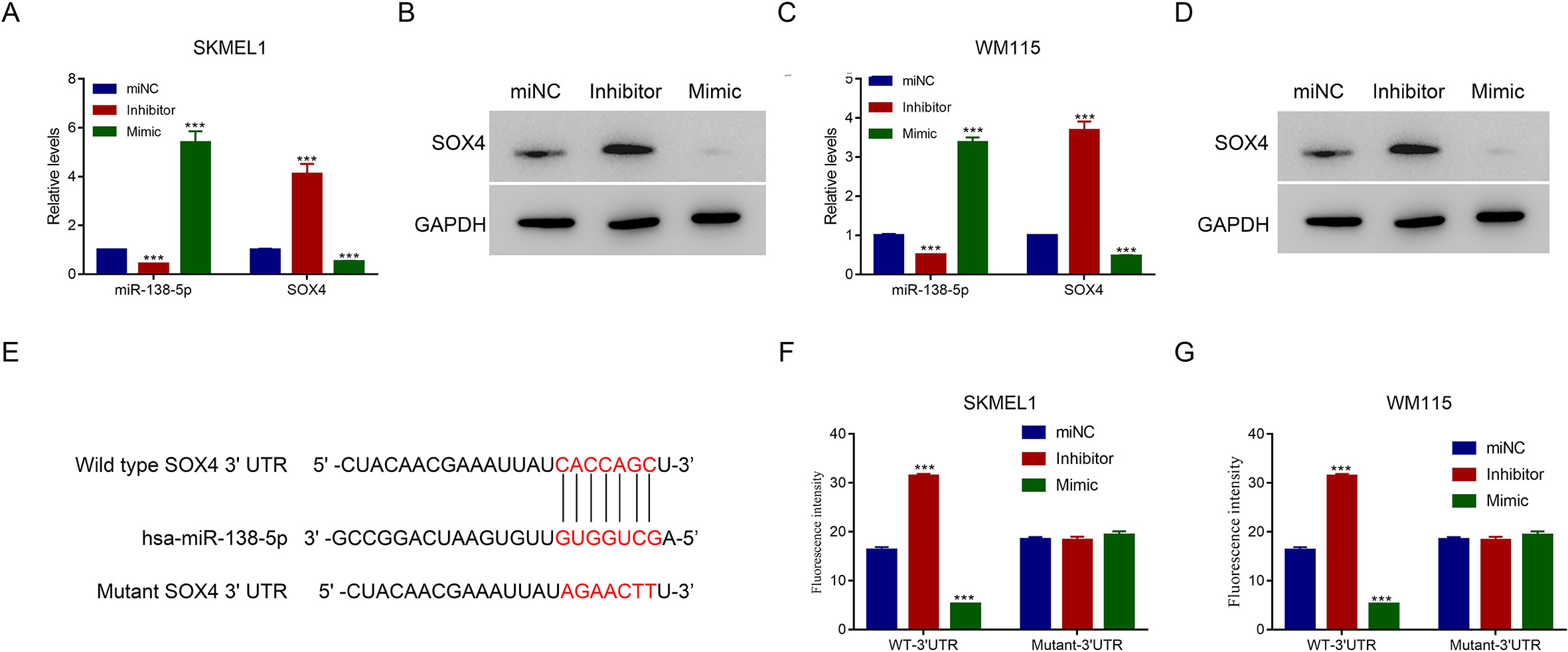
Negative correlation between miR-138-5p and SOX4 in human melanoma tissues. A. The relative mRNA level of SOX4 was upregulated in human melanoma tissues. 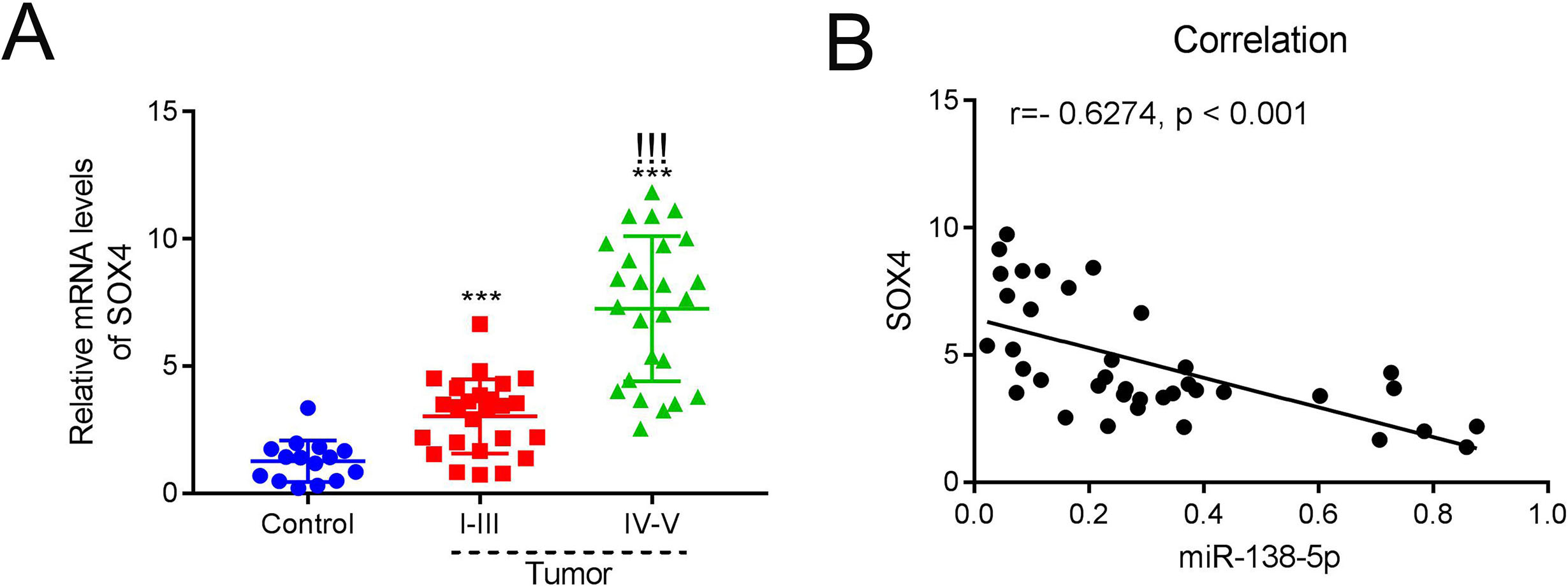
Overexpression SOX4 disrupts the effects of HMSC-exo and miR-138-5p mimic on apoptosis in WM115 cells. A. Flow cytometry was used to examine the apoptosis of WM115 cells after transfecting with oeNC and oeSOX4 in the presence of HMSC-exo, 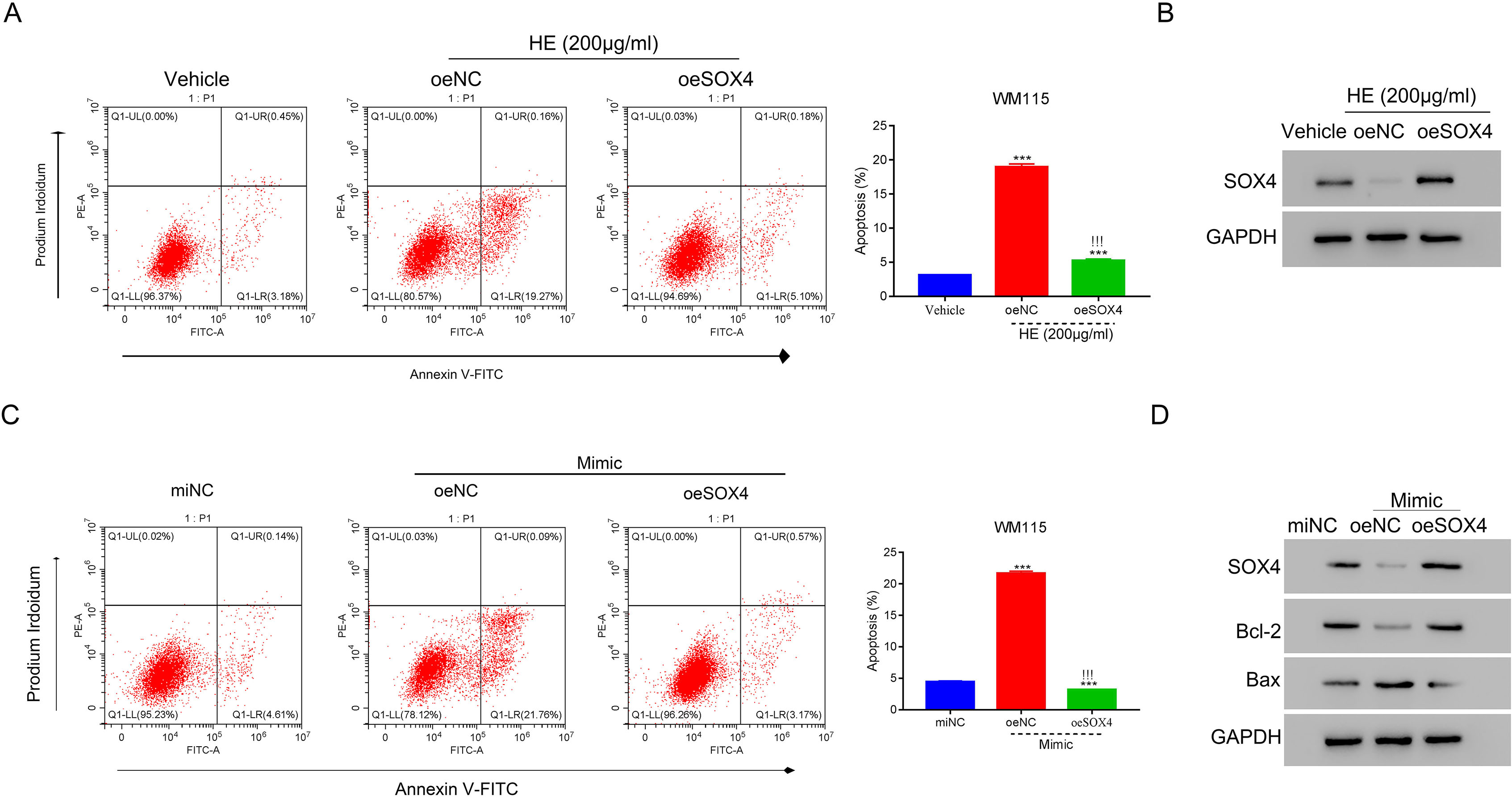
We characterized HMSCs and HMSC-derived exosomes (HMSC-exos). Flow cytometry revealed that over 95% of prepared HMSCs were CD90 and CD105 positive and CD34 and CD45 negative (Fig. S1A). WE observed the expected morphology of isolated HMSC-exos by TEM (Fig. S1B). The CD9 and CD63 biomarkers were present on the exosomes (Fig. S1C).
Then, we traced the endocytosis of control or HMSC-exos by human SKMEL1 and WM115 cells by PKH-7 labeling. We observed immunofluorescent signals in SKMEL1 and WM115 cells incubated with HMSC-exos (200
Overexpression of miR-138-5p in HMSC-exos promoted the apoptosis of melanoma cells
Gain and lose function assay were used to induce miR-138-5p overexpression or silencing in HMSCs by using corresponding mimic and inhibitor. When miR-138-5p mimic or inhibitor was introduced into HMSCs, we confirmed increased or decreased miR-138-5p levels relative to the control (miNC) (
Apoptosis in SKMEL1 and WM115 cells treated with exosomes, including miNC-exos, mimic-exos, and inhibitor-exos, was visualized by flow cytometry. Compared to miNC-exo-treated cells (either SKMEL1 or WM115), apoptosis increased remarkably in cells treated with mimic-exos, whereas apoptosis significantly decreased in cells treated with inhibitor-exos (
miR-138-5p inhibits SOX4 transcription by binding to its 3’UTR in melanoma cells
MiRNAs elicit cellular responses by targeting genes coding for effector protein(s). We exploited TargetScan and Starbase to predict the target(s) of miR-138-5p. The prediction indicated that SOX4 was a potential target of miR-138-5p. To validate this prediction, we introduced miR-138-5p inhibitor, mimic, or control (miNC) into SKMEL1 and WM115 cells and measured the levels of miR-138-5p and SOX4 by qRT-PCR and/or Western blot. Interestingly, increased miR-138-5p content was linked to decreased SOX4 levels at both the mRNA and protein levels compared with levels in cells treated with the control (
Negative correlation between miRNA-138-5p and SOX4 in melanoma tissues
We measured the expressions of miR-138-5p and SOX4 in melanoma and para-cancerous tissues. As indicated in Fig. 5A, SOX4 expression in tumor tissues was significantly increased at early stages (I–III) and further enhanced at late stages (IV–V) compared to SOX expression in para-cancerous tissues (
SOX4 overexpression partially abolished the action of HMSC-exos and the miRNA-138-5p mimic on apoptosis
We explored SOX4’s direct influence on the apoptosis of melanoma cells. For this purpose, we measured SOX4 expression in several melanoma cell lines. Intriguingly, WM115 cells exhibited the highest mRNA and protein levels of SOX4 among the cell lines, including normal human epidermal melanocyte HEMA-LP (
Then, we performed apoptotic analyses SOX4 lentiviruses-transduced and HMSC-exos-treated WM115 cells. Relative to the control, HMSC-exos treatment promoted apoptosis in WM115 cells, and SOX4 overexpression almost completely abrogated this effect (
The A2058 cell line was chosen for further investigation since these cells exhibited the lowest level of SOX4 among all examined cell lines (
Discussion
The severity of melanoma, including high morbidity and mortality [1, 3, 4], together with limited outcomes and prognosis after most current therapies [1] indicate the urgent need for extensive investigations into the pathogenesis of melanoma and the identification of novel targets and therapies. In this regard, we focused on miR-138-5p, which was downregulated dramatically in melanoma tissues. This important miRNA in exosomes derived from HMSCs. Our results demonstrate that: a) altered expression of miR-138-5p HMSC-exos severely affects the apoptosis in melanoma cells; b) downregulation of miR-138-5p and upregulation of SOX4 were observed in melanoma tissues, indicating a negative correlation between the two signaling molecules; c) miR-138-5p modulates SOX4 through binding to the SOX4 3’ UTR; and d) altered levels of SOX4 influence the impact of HMSC-exos and miR-138-5p on melanocyte apoptosis. Therefore, this study provides novel insights into the actions of HMSC-exos and confirmed important functions of miR-138-5p and its target SOX4 in the pathogenesis of melanoma.
The occurrence of melanoma is often associated with abnormal proliferation and apoptosis of melanocytes [24, 25]. Thus, inhibiting proliferation or promoting apoptosis of melanocytes can be an effective way to treat melanoma. The beneficial effects of HMSC-exos were previously demonstrated [26, 27]. For instance, HMSC-exos ameliorated hypoxia-induced cardiomyocyte apoptosis [26]. Similarly, exosomes from MSCs protected rats from glucocorticoid-triggered osteonecrosis [27]. In this study, we demonstrate an important action of HMSC-exos on the survival of melanoma cells, thereby expanding the functional scope of the exosomes from MSCs.
The most abundant cargo molecules within exosomes are miRNAs [28]. Therefore, the content and composition of miRNAs have fundamental effects on the functions of exosomes. These miRNAs affect gene expression in a post-transcriptional fashion by targeting corresponding mRNAs. Furthermore, the crucial impact of miRNAs on the progression of melanoma has been established [6, 29, 30]. For example, miR-155, mi-193b, and miRNA-205 modulate melanoma cell proliferation, while miR-145 and miR-182 participate in the survival of melanoma cells [31]. Similarly, the impact of miR-138 on the proliferation of melanoma cells has been verified [13]. Previous reports have indicated that exosomes from highly metastatic melanomas increased the metastatic behavior of primary tumors by permanently ‘educating’ bone marrow progenitors and regulates the polarization of macrophage activation [32, 33]. Moreover, breast cancer-derived exosomes promoted the progression of cell invasion and metastasis [34].
Therefore, tumor-derived exosome not only have roles of control of malignant phenotype in vitro model of melanoma cells but also give contrary effects as to promote cell invasion and metastasis. They have dual possible effects, which is different to the function of HMSC-derived exosome. Here, we validated the impact of miR-138 from HMSC-exos, the current study showed that upregulating miR-138-5p promoted melanoma cell apoptosis, thereby highlighting the important influence of this miRNA on the pathogenesis of melanoma.
The multiple functions of miR-138-5p in physiological and pathological conditions are elicited by targeting effector molecules [35, 36, 37]. For instance, MSC-derived exosomal miR-138-5p from bone marrow protects astrocytes from ischemic stroke via suppression of lipocalin 2 [36]. MiR-138 inhibits the proliferation of melanoma cells via direct targeting of hypoxia-inducible factor-1
In conclusion
Together, our findings indicated that HMSC-derived exosomes inhibited the apoptosis of melanoma cells through delivering miR-138-5p, then targets SOX4. Our results not only indicated the critical role of miR-138-5p/SOX4 in the progression of melanoma cells but also provided evidence to point out the potential value of stem cell-based therapy as a promising management option for melanoma.
Data availability statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Funding
This research was Funded by Natural Science Foundation of the Tibet Autonomous Region (NO. XZ2017 ZRG-87 (Z)).
Author contributions
Conception: ZL.
Interpretation or analysis of data: XW and ZC.
Preparation of the manuscript: XW, ZC and BS.
Revision for important intellectual content: XW and ZC.
Supervision: ZL.
Supplementary data
The supplementary files are available to download from http://dx.doi.org/10.3233/CBM-210409.
sj-pdf-1-cbm-10.3233_CBM-210409.pdf - Supplemental material
Supplemental material, sj-pdf-1-cbm-10.3233_CBM-210409.pdf
Footnotes
Acknowledgments
We sincerely acknowledged the support given by Shigatse People’s Hospital and Fudan University Shanghai Cancer Center for present research.
Conflict of interest
The authors report no conflict of interest
