Abstract
BACKGROUND:
Biomarkers that predict which patients with early stage NSCLC will develop recurrent disease would be of clinical value. We previously discovered that an autoantibody to a complement regulatory protein, complement factor H (CFH), is associated with early stage, non-recurrent NSCLC, and hypothesized that the anti-CFH antibody inhibits metastasis.
OBJECTIVES:
The primary objective of this study was to evaluate the anti-CFH antibody as a prognostic marker for recurrence in stage I NSCLC. A secondary objective was to determine if changes in antibody serum level one year after resection were associated with recurrence.
METHODS:
Anti-CFH antibody was measured in the sera of 157 stage I NSCLC patients designated as a prognostic cohort: 61% whose cancers did not recur, and 39% whose cancers recurred following resection. Impact of anti-CFH antibody positivity on time to recurrence was assessed using a competing risk analysis. Anti-CFH antibody levels were measured before resection and one year after resection in an independent temporal cohort of 47 antibody-positive stage I NSCLC patients: 60% whose cancers did not recur and 40% whose cancers recurred following resection. The non-recurrent and recurrent groups were compared with respect to the one-year percent change in antibody level.
RESULTS:
In the prognostic cohort, the 60-month cumulative incidence of recurrence was 40% and 22% among antibody negative and positive patients, respectively; this difference was significant (Gray’s test,
CONCLUSIONS:
The anti-CFH autoantibody may be a useful prognostic marker signifying non-recurrence in early stage NSCLC patients. However, change in the level of this antibody in antibody-positive patients one year after resection had no association with recurrence.
Introduction
Early stage non-small cell lung cancer (NSCLC) patients are typically treated with surgical resection [1]. However, overall 5-year survival for stage I NSCLC is
A variety of biomarker-based approaches, whether tumor- or blood-based, have been extensively explored for prognosis in early stage NSCLC (reviewed in [6, 7, 8, 9]). Within the tumor microenvironment, the presence of CD4
We previously reported autoantibodies against complement factor H (CFH) in the sera of patients with early stage, non-metastatic NSCLC [30]. Patients with stage I NSCLC had a significantly higher incidence of anti-CFH autoantibody than those with late-stage NSCLC (
Materials and methods
Study population and patient samples
The Duke University Institutional Review Board (IRB) approved this study. The current protocol number for this study is Pro00012914, with only minimal revisions from previous versions. The protocol is reviewed and renewed annually by the Duke IRB. Experiments were undertaken with the understanding and written consent of each subject, and the study conforms with The Code of Ethics of the World Medical Association (Declaration of Helsinki), printed in the British Medical Journal (18 July 1964). Study design, reporting, and analysis adhered to the principles summarized in ref [33] with respect to the STROBE [34] and QUIPS [35] guidelines for prognostic studies. In this report, a non-matched case-control study of autoantibody positive vs. negative patients with respect to outcome is followed by a temporal study of outcome of autoantibody positive patients as a function of change in autoantibody levels.
From our prospective database, we selected two independent groups of sequential stage I NSCLC patients: 157 patients for the prognostic cohort and 47 patients for the temporal cohort who qualified on the basis of anti-CFH autoantibody positivity, from an initial group of 203 patients tested. All patients had a new diagnosis of pathologic stage I (T1 N0 M0 or T2 N0 M0) NSCLC as defined by the International System of Staging for Lung Cancer [36]. All patients underwent surgical resection and lymph node dissection at our institution, and all but two were followed for at least 2 years or until recurrence was documented; the exceptions were two non-recurrent patients in the temporal cohort who were followed for 17 and 20 months.
A serum sample was collected from all sequential patients who consented to provide a serum sample at the time of diagnosis (March 1997 to September 2012 for the prognostic cohort and April 1998 to September 2004 for the temporal cohort) before any treatment, and again in the temporal cohort at approximately one year post-resection. Recurrence was defined as either local or distant disease, histopathologically confirmed not to be a second primary. Sera were stored in our IRB-approved
Detection of anti-CFH autoantibody by ELISA
Two different ELISAs that give qualitatively similar results were used to assign anti-CFH autoantibody status: in one ELISA the immobilized target was a peptide containing the antibody’s epitope; in the other ELISA, the full length protein was used. Independent evaluation demonstrated that samples defined as positive or negative in one ELISA were defined the same way in the other (data not shown).
For the prognostic cohort, serum anti-CFH autoantibody status was assessed using the peptide ELISA. Each sample was assayed in triplicate in 96-well plates. Wells were coated with 100
We defined a positive autoantibody response as a mean fluorescence intensity of the synthetic peptide-coated wells that was statistically greater (
For the temporal cohort, an indirect ELISA with full-length human CFH (Complement Technology, Inc., Tyler, TX) immobilized in the wells was employed. Prior to immobilization, the CFH autoantibody epitope was exposed by incubating the CFH for 30 minutes in 15 mM Tris (2-carboxyethyl) phosphine (TCEP)/0.1 M Tris-HCl, pH 8. Following reduction, the CFH was diluted to 2
Statistical analysis
To assess the effect of antibody status on time to recurrence, a competing risk framework was using since death and recurrence are competing risks [38]. Using SAS PROC TEST and PROC PHREG, cumulative incidence curves were generated for each stratum, and Gray’s test was used to compare curves [39]. We used the SeSpPPVNPV function within the R software package timeROC to obtain performance measures of the antibody as a biomarker for time to recurrence [40].
A Wilcoxon two-sample test was used to compare the percent change in CFH at 1 year among patients with and without recurrence within the second temporal cohort.
Results
Study population, antibody and recurrence status
Baseline demographics and clinical characteristics, including gender, race, pathologic stage, and histology, for the 157 early-stage lung cancer patients analyzed in the prognostic cohort are reported in Table 1. Included in this study population were 95 patients with stage I NSCLC who had not developed recurrence at least 2 years after resection, and 62 patients with stage I NSCLC who did develop recurrent disease after resection. All diagnoses were confirmed by a board certified pathologist. Recurrences were diagnosed by CT-PET imaging and/or biopsy. The mean age was 67 years (SD, 8.6 years) and mean smoking history was 46.4 pack years. By ELISA, 42 (27%) of stage I NSCLC patients were antibody-positive and 115 (73%) were antibody-negative. The distribution of the 157 patients according to their antibody status, recurrence, and survival status is shown in Table 2.
Prognostic study: Patient demographics and clinical characteristics
Prognostic study: Patient demographics and clinical characteristics
Prognostic study: Grouping by anti-CFH antibody, recurrence, and survival status
For the temporal cohort, sera from 203 stage I NSCLC patients were first screened for anti-CFH antibody in serum collected at time of resection and again approximately one year later. Of the 203 patients, 47 (23%) were antibody positive, including 28 patients with stage I NSCLC who had not developed recurrence at least 2 years after resection (with the exception of two patients not followed beyond 17 and 20 months), and 19 patients with stage I NSCLC who did develop recurrence. Among patients with recurrence, time to recurrence averaged 3 years (range 0.7–8.9 years). Baseline demographics and clinical characteristics for the 47 early-stage lung cancer patients analyzed in the temporal study are reported in Table 3.
Temporal study: Patient demographics and clinical characteristics
Cumulative incidence curves for the 157 anti-CFH early stage NSCLC patients are shown in Fig. 1. The difference between the two cumulative incidence curves is statistically significant (
Performance measures of anti-CFH antibody as a biomarker for time to recurrence
Performance measures of anti-CFH antibody as a biomarker for time to recurrence
Abbreviations: Se, sensitivity; Sp, specificity; PPV, positive predictive value; NPV, negative predictive value. Predictive accuracy measures were estimated at cutpoint
Cumulative incidence functions according to anti-CFH antibody status.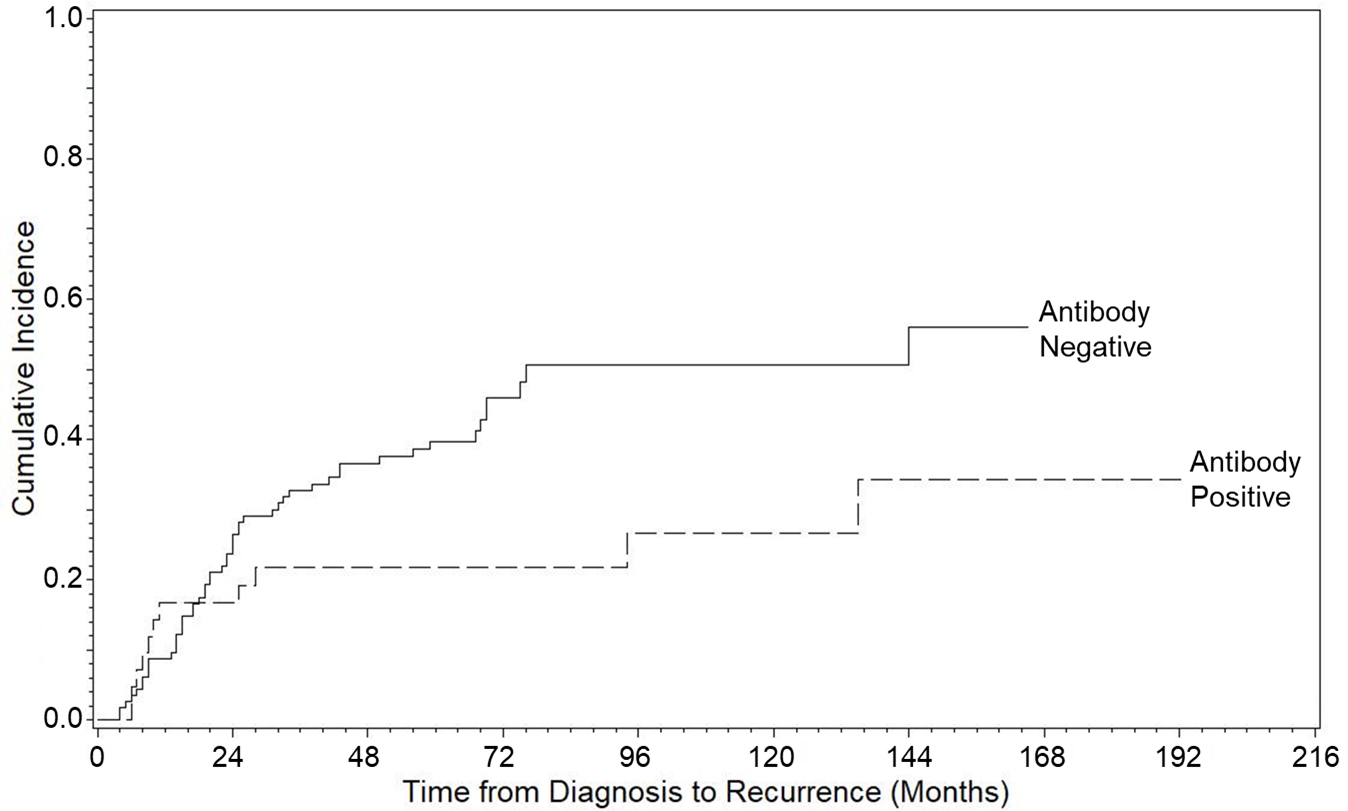
The change in serum anti-CFH antibody level between diagnosis and one year after diagnosis was computed. The distribution of change within each recurrence group is shown in Fig. 2. The comparison of the two groups using an exact Wilcoxon two-sample test is not statistically significant (
Box-and-whiskers plot displaying distribution of percent change for the recurrent and non-recurrent groups. An analysis of variance was used to compare percent change from baseline among patients in the recurrent and non-recurrent groups. A percent change greater than 0 is an indication that the follow-up assessment at 1 year is greater than the baseline assessment at time of resection.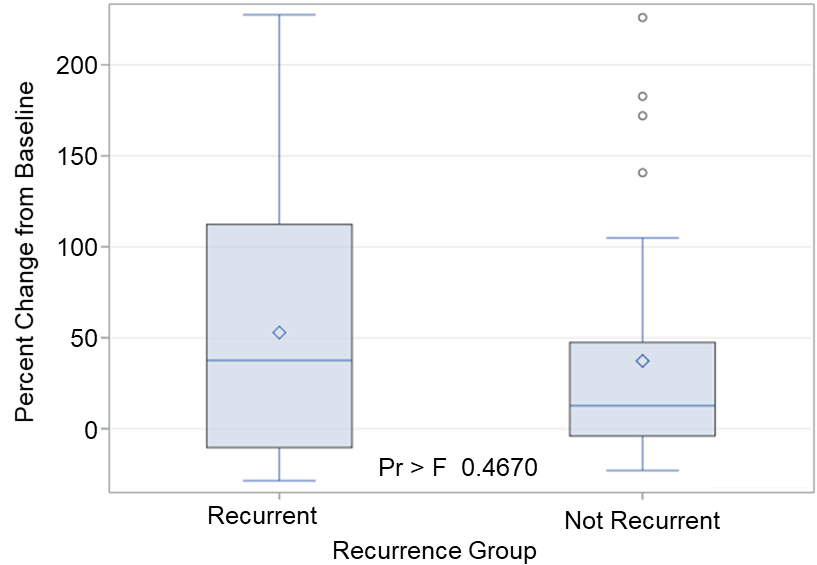
While a variety of strategies have been used to develop prognostic markers for lung cancer, there remains no consistent method to determine which patients with early stage disease will recur following resection. A biomarker or panel of markers that could make this distinction at the time of diagnosis would potentially improve outcomes, as these select patients may benefit from adjuvant therapy.
In studying the host immune response to NSCLC [13, 20, 30, 41], and in light of reports that suggest tumor rejection can be initiated by antitumor antibodies [31, 32], we hypothesized that some patients have developed specific autoantibodies against tumor cells that could potentially be used for both diagnostic and therapeutic purposes. We previously reported an antibody against CFH that is associated with early stage, non-metastatic disease [30]. We cloned the antibody from a single human B cell, and have shown it causes complement dependent cytotoxicity of tumor cells, inhibits tumor growth, and modulates the adaptive immune response, lending mechanistic relevance to the association of the anti-CFH antibody with non-recurrence [42].
Here we explored the potential of this anti-CFH antibody as a biomarker to predict time to recurrence in patients with early stage NSCLC. We were able to stratify our group of 157 Stage I patients with either non-recurrent or recurrent disease on the basis of anti-CFH antibody positivity at the time of diagnosis. Possible confounding factors not addressed in this study that might affect recurrence are post-surgical treatment, if any, concurrent other disease, or immune status. Possible sources of bias include attrition bias, in which patients who did not return to the clinic for follow-up visits were eliminated from the study, and outcome bias, in which variations in measurement intervals among patients may have differentially altered time to recurrence measurements.
While antibody titers over time have been studied in infectious diseases, temporal anti-tumor antibody levels are not well understood. It is noteworthy that in this study the antibody does persist in the blood at least one year after resection of the tumor. However, minor changes in antibody level at one year are not associated with recurrence. As more studies characterize anti-tumor antibodies, their functional consequences will be better understood and their diagnostic potentials will be realized.
Although discrimination by the anti-CFH antibody alone showed statistical significance, it is expected that other autoantibodies with prognostic value could be found that augment it in order to identify which patients will recur and potentially benefit from additional therapy. In NSCLC, as well as other cancer types, intratumoral TLS are associated with a favorable prognosis [19]. TLS, like follicles of secondary lymphoid organs, are organized assemblies of tumor antigen-presenting dendritic cells, activated and proliferating T cells, and antibody-producing plasma B cells. Within the germinal centers of intratumoral TLS, B cells are activated, proliferate, hypermutate and express IgG and IgA antibodies, and many of these antibodies are tumor-antigen specific [17, 43]. Intratumoral germinal centers are more prevalent in stage I NSCLC than in later stages [20], and may be a rich source of additional tumor targeting antibodies. This approach of mining the humoral response to tumors is just beginning to be explored and has the potential to transform diagnostic and therapeutic options.
Footnotes
Author contributions
Conception: EFP, JEH, EBG, and MJC
Interpretation or analysis of data: JEH
Preparation of the manuscript: EFP, EBG, and JEH
Performance of experiments: RTB, RG, and MJC
Supervision: EFP and MJC
