Abstract
Background
The serum isocitrate dehydrogenase 1(IDH1) level is significantly elevated in patients with non-small cell lung cancer (NSCLC) and has important clinical value as a marker for early diagnosis. This study examined the dynamic changes of serum IDH1 levels of patients with NSCLC undergoing surgery or medical treatment, to evaluate its potential prognostic value.
Methods
The study cohort included 83 NSCLC patients who underwent surgery, 37 NSCLC patients who underwent medical treatment, 50 healthy controls, and 52 disease controls. Serum levels of IDH1 were assayed by enzyme-linked immunoassay. Tumor biomarkers including carcinoembryonic antigen, squamous cell carcinoma, neuron-specific enolase, CYFRA21-1, and pro-gastrin-releasing peptide—which are currently used in clinical practice—were measured by automatic immunoanalyzers.
Results
Serum IDH1 was significantly higher in patients with NSCLC compared with healthy people or patients with benign lung diseases (p < 0.001). The area under the receiver operating characteristic curve for diagnosis and differential diagnosis were 0.897 and 0.879, respectively, which were superior to the five tumor markers. Serum IDH1 levels decreased in most patients after surgery, with the most dramatic changes in patients with stage I tumors compared with stage II and III. Analyses of changes in the serum IDH1 level of patients after receiving chemotherapy or targeted therapy revealed that for patients with progressive disease, serum IDH1 increased significantly after treatment; for patients with partial response or stable disease, it decreased steadily.
Conclusion
IDH1 has potential prognostic value and may be used as a marker for the monitoring of treatment efficacy.
Introduction
Lung cancer currently has the highest mortality rate in the world. 1 Early diagnosis is essential for improving the survival rate of patients. However, most patients are diagnosed as advanced lung cancer at the first diagnosis and thus are ineligible for surgical treatment. Tumor imaging and serological testing have become the most ideal strategy for cancer screening because of their advantages of non-invasiveness, low cost, and speed. 2 Among them, tumor serum and plasma markers have been widely used in clinical practice. Carcinoembryonic antigen (CEA), cytokeratin 19 fragment 21-1 (CYFRA21-1), squamous cell carcinoma antigen (SCC-antigen), neuron specific enolase (NSE), and pro-gastrin-releasing peptide (pro-GRP) are widely used tumor biomarkers for diagnosing non-small cell lung cancer (NSCLC).3–6 However, the sensitivity and specificity of these commonly used tumor markers are not ideal and the identification of novel tumor markers has been a focus of research.
Among the new tumor markers identified in recent years, isocitrate dehydrogenase 1 (IDH1) has shown efficacy in the early diagnosis of NSCLC.7, 8 IDH1 is a key enzyme of the tricarboxylic acid cycle whose main function is to oxidatively decarboxylate isocitrate in the cytoplasm to α-ketoglutarate and generate NADPH. 7 The IDH1 mutation usually occurs at the active center of the enzyme caused by the codon 132 mutation, which consumes NADPH and reduces the production of α-ketoglutarate, which in turn produces a new metabolite, 2-hydroxyglutarate.9, 10 IDH1 has been associated with the occurrence and development of a variety of tumors. For example, IDH1 mutations accompany the development of glioma and are associated with poor prognosis. 11 Multivariate analysis has shown that high expression of IDH1 is related to the tumor grade of colorectal cancer and the patient’s prognosis. 12 Another study showed that the expression level of IDH1 in the peripheral blood of NSCLC patients is significantly higher than that of healthy people and other patients with benign lung diseases. 8 Lung cancer patients with high IDH1 expression in lung cancer have a shorter survival time than patients with low IDH1. 7 IDH1 expression is elevated in the serum of patients with esophageal cancer, and patients with high serum IDH1 levels have shorter overall survival (OS) and progression-free survival (PFS). 13 In the above studies, IDH1 not only shows good diagnostic ability, but also has potential value as a prognostic marker. Studies also showed that IDH1 increased the chemotherapy sensitivity of endometrial cancer cells to metformin through IDH1-Nrf2 signaling. 14 In cholangiocarcinoma, cisplatin exposure in IDH1 mutant cancer cells results in higher levels of reactive oxygen species (ROS), DNA double-strand breaks, and cell death compared with IDH1 wild-type cells. 15 However, research on the role of IDH1 chemoresistance in lung cancer is lacking.
In this study, we analyzed the correlation between IDH1 serum levels and the clinical characteristics of NSCLC patients. We monitored the dynamic changes of serum IDH1 levels before and after surgery and medical treatment, and evaluated the potential of serum IDH1 changes as a marker for monitoring treatment response and efficacy.
Methods
Patients and specimens
Serum samples were collected during April 2019 to December 2019 at Beijing Chest Hospital. The study cohort included 83 NSCLC patients who underwent surgery, 37 NSCLC patients who underwent medical treatment, 50 healthy controls, and 52 disease controls. The NSCLC patients were diagnosed based on x-ray, computed tomography, and biopsy according to the World Health Organization’s Classification of Tumors of the Lung; tumor stage was defined according to the 7th International Association for the Study of Lung Cancer/American Joint Committee on Cancer tumor node metastasis (TNM) staging system. In the NSCLC surgery group, none of the patients received neoadjuvant therapy, and both pre-operation and post-operation serum samples were collected. In the NSCLC medical treatment group, serum samples were collected at three time points: baseline (before treatment), after two cycles of treatment, and after four cycles of treatment. Healthy controls were recruited during annual physical examination and did not show any sign of tumor. Disease controls included patients with non-malignant lung disease, which were mainly pneumonia and tuberculosis. In total, 368 serum samples were collected. Patient information was kept confidential, and the study was approved by the Research Ethics Committee of the Beijing Chest Hospital.
Detection of serum IDH1 levels
Serum levels of IDH1 were measured by enzyme-linked immunoassay (ELISA) using a commercial diagnostic kit (Beijing Modern Gaoda Biotechnology, Beijing, China) according to the manufacturer's recommendations. The concentrations of IDH1 were calculated according to a standard curve, which was fitted using the standard values. Measurements were undertaken twice in duplicate. The threshold of IDH1 was set at 5 ng/mL.
Detection of serum tumor biomarkers levels
Serum levels of the tumor biomarkers CEA, CYFRA21-1, SCC-antigen, NSE, and pro-GRP were measured by an automatic flow fluorescence immunoanalyzer (Roche Applied Science, Penzberg, Germany). The detection kit operators were blinded to diagnostic information. The cutoff values of each tumor biomarker were defined as: 6 ng/mL for CEA, 6 ng/mL for CYFRA21-1, 4 ng/mL for SCC-antigen, 40 ng/mL for NSE, and 80 pg/mL for pro-GRP.
Statistical analysis
Data analyses and graph plotting were performed with GraphPad Prism software (version 7) and R (version 3.6.5). Differences between independent groups were tested with the Mann–Whitney U test (continuous variables and non-parametric analyses). Differences between pre-operation and post-operation samples were tested with Wilcoxon matched-pairs test. Differences of serum IDH1 level change between subgroups were tested with covariance analysis and general linear regression.
Bioinformatic analysis
IDH1 mutation data were downloaded from the International Cancer Genome Consortium data portal (ICGC, https://dcc.icgc.org). 16 Mutation rate comparation and cohort survival comparation was performed using online tools of the ICGC. IDH1 expression data and patient clinical information was from The Cancer Genome Atlas (TCGA) transcriptome sequencing data. Survival analysis was performed by the web tool Gene Expression Profiling Interactive Analysis (GEPIA). 17
Results
Serum IDH1 levels in NSCLC patients and controls
Patient characteristics of the 222 NSCLC patients and controls are shown in Table 1. The distributions of gender and age between the NSCLC and control groups were not significantly different.
Characteristics of the study cohort.
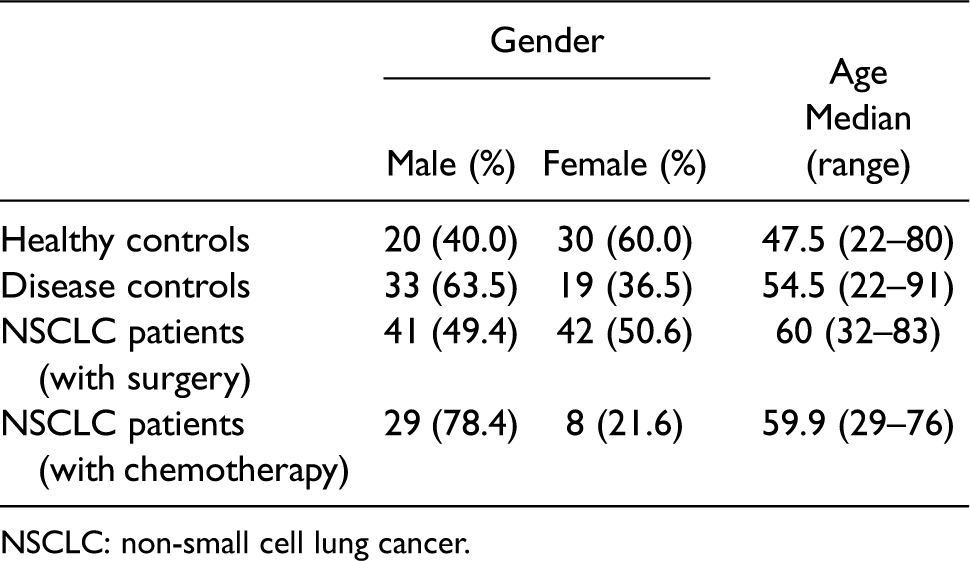
NSCLC: non-small cell lung cancer.
To evaluate the diagnostic value of IDH1, we examined the pre-operation serum levels of IDH1 in NSCLC patients compared with healthy individuals and benign lung disease patients using ELISA. Serum IDH1 levels were slightly (but significantly) higher in the disease controls (2.69 ± 1.41 ng/mL; p < 0.001) and significantly (and markedly) higher in NSCLC patients (6.42 ± 3.48 ng/mL; p < 0.001) compared with healthy individuals (1.99 ± 1.63 ng/mL) (Table 2). Serum IDH1 levels showed no significant difference according to gender, age, tumor pathological type, and tumor-driven gene (TDG) mutation burden (p > 0.05). Higher levels of serum IDH1 were detected in patients with early-stage NSCLC (stage I and II) compared with advanced NSCLC patients (stage III and IV), although the difference was not significant (p = 0.164).
Clinical features of serum IDH1 levels in patients and controls.
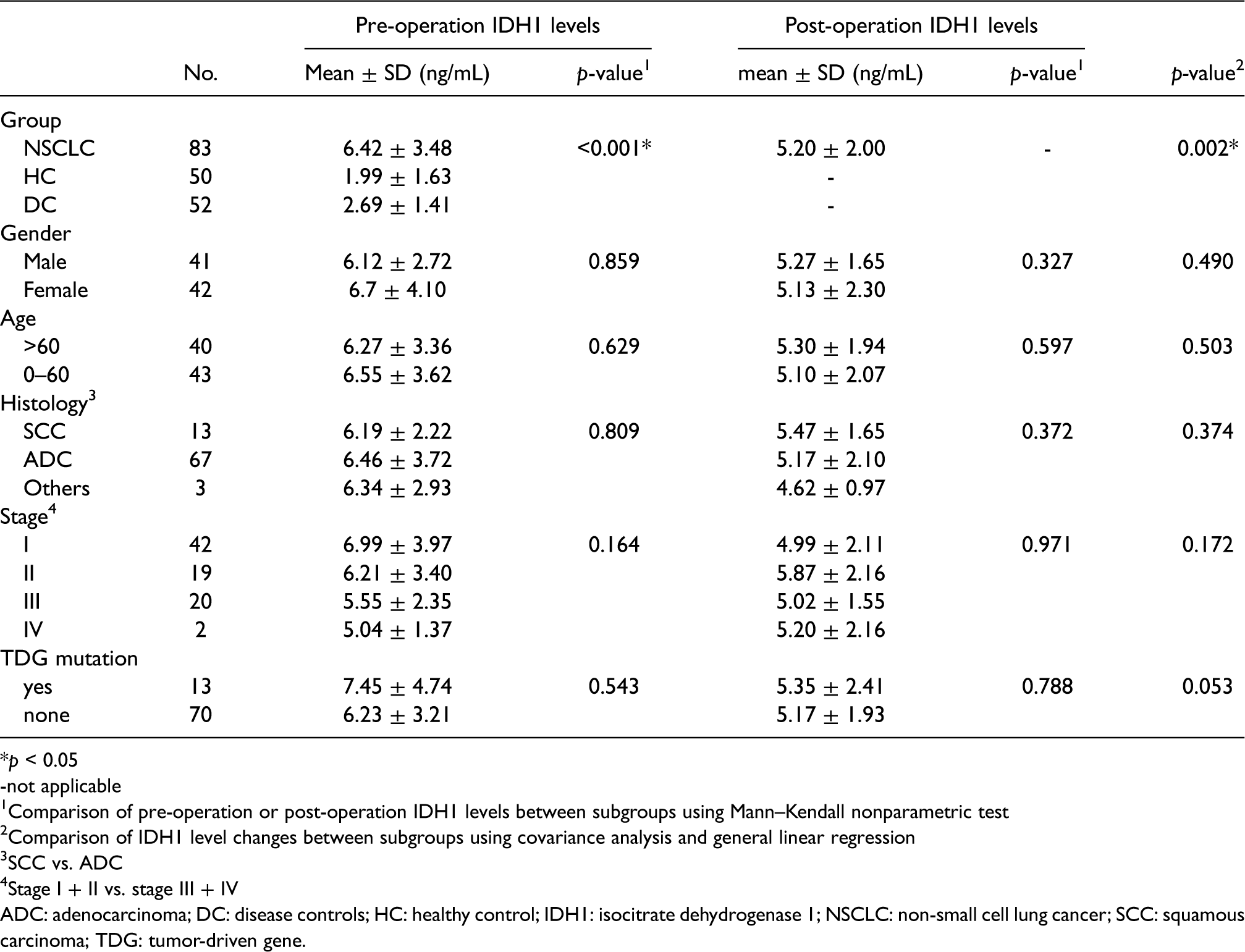
*p < 0.05
-not applicable
Comparison of pre-operation or post-operation IDH1 levels between subgroups using Mann–Kendall nonparametric test
Comparison of IDH1 level changes between subgroups using covariance analysis and general linear regression
SCC vs. ADC
Stage I + II vs. stage III + IV
ADC: adenocarcinoma; DC: disease controls; HC: healthy control; IDH1: isocitrate dehydrogenase 1; NSCLC: non-small cell lung cancer; SCC: squamous carcinoma; TDG: tumor-driven gene.
It was reported that plasma IDH1 levels could be used as an early diagnostic marker. 8 We next analyzed the diagnostic potential of serum IDH1 levels in NSCLC patients, benign lung disease patients, and healthy individuals. The area under the receiver operating characteristic (ROC) curve (AUC) was 0.897, which indicated a good diagnostic capability (Figure 1(a)). Since the commercial IDH1 serum test kit is currently used in clinical practice, we analyzed the detection efficiency of the recommended detection threshold of the test kit in our cohort. At the cutoff of 5 ng/mL, serum IDH1 has a sensitivity of 60.2% and a specificity of 91.2% for discriminating NSCLC patients from benign lung disease patients and healthy individuals. In addition to IDH1, five conventional NSCLC tumor markers were also measured in the NSCLC patients and benign lung disease patients: CEA, CYFRA21-1, SCC-antigen, NSE, and pro-GRP. The ROC curve of IDH1 and the five NSCLC tumor markers in discriminating NSCLC from benign lung diseases are shown in Figure 1(b). The AUC of IDH1 was 0.879, which was superior to the AUC for the five tumor markers. The AUC of these tumor markers were 0.699 (CEA), 0.598 (SCC), 0.568 (NSE), 0.669 (CYFR21-1), and 0.590 (pro-GRP).
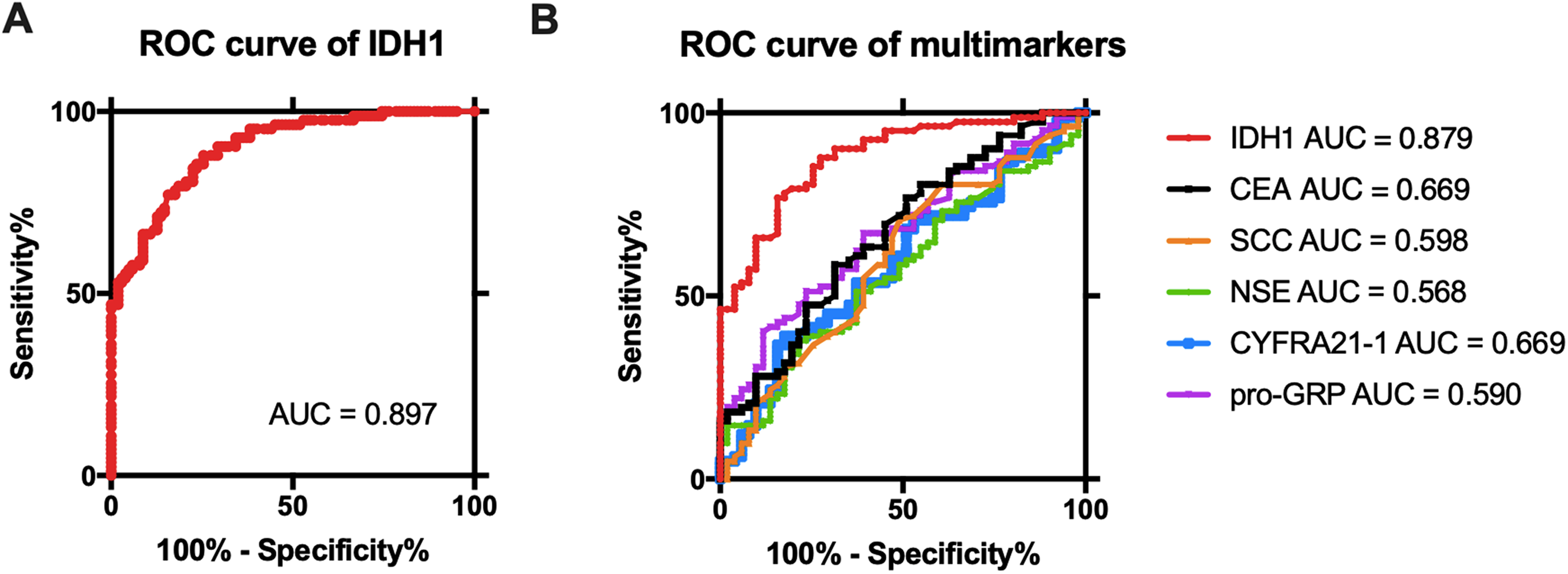
ROC curve of serum IDH1 and five other tumor biomarkers in NSCLC patients, benign lung disease patients, and healthy individuals. (a) ROC curve of serum IDH1 in discriminating NSCLC patients from benign lung disease patients and healthy individuals. (b) ROC curve of IDH1, CEA, SCC, NSE, CYFRA21-1, and pro-GRP in discriminating NSCLC patients from benign lung disease patients. The AUC of each biomarker is listed.
Serum IDH1 level changes before and after surgery
For the 83 NSCLC patients who underwent surgery, we collected serum during the postoperative review. We found that the serum IDH1 level of most of the NSCLC patients who received surgery (55/83, 66.3%) was significantly decreased compared with pre-operative serum IDH1 level (p = 0.002). However, the serum IDH1 levels of some patients (18/83, 33.7%) increased during postoperative follow-up visits (Figure 2(a)). We then investigated the characteristic that might be related to the changes in IDH1. However, no significance was observed regarding gender, age, tumor histological type, tumor stage, or TDG mutation burden (Supplementary Table 1), and the proportion of patients in each subgroup with IDH1 was similar.
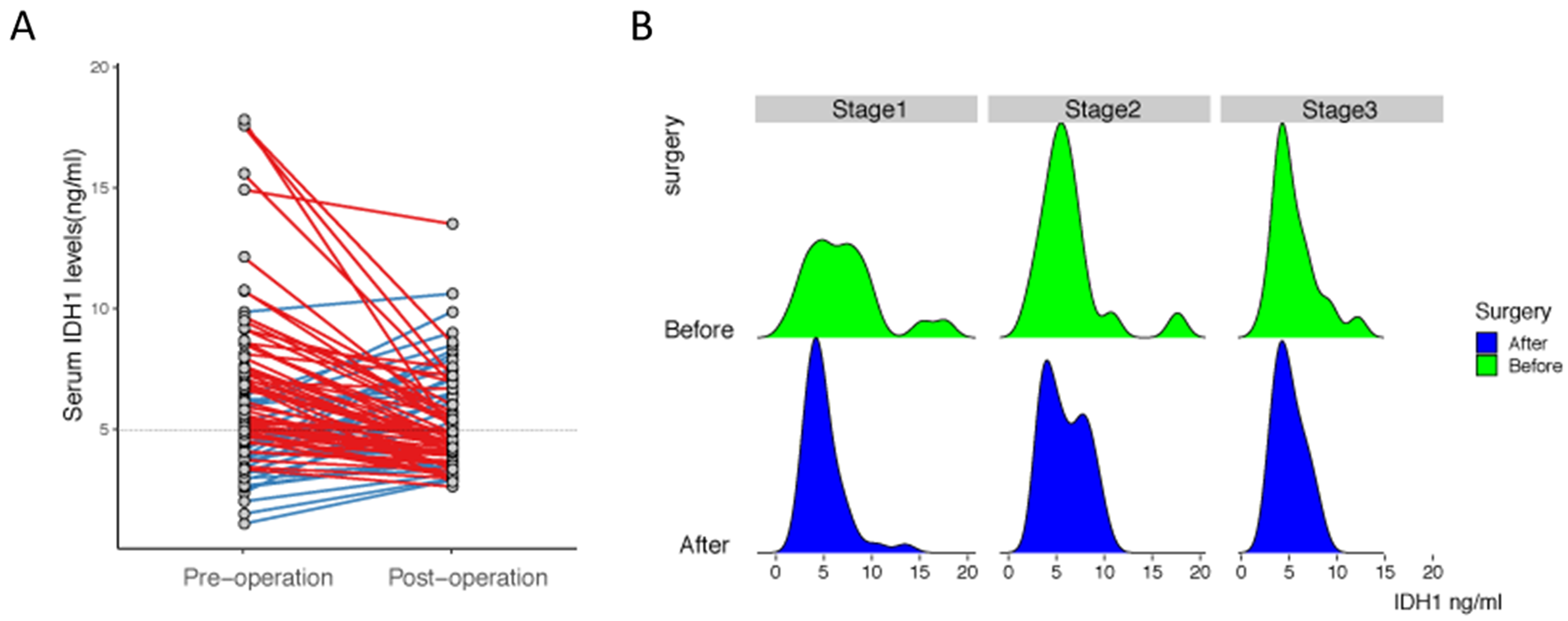
Dynamic change of serum IDH1 in patients after surgery. (a) Serum levels of IDH1 in the NSCLC patients, pre-operation and post-operation. Red lines indicate a decrease of serum IDH1 levels, and blue lines indicate an increase of serum IDH1 levels. (b) Distribution of serum IDH levels in NSCLC patients with different tumor stages.
We further examined which factor might be related to the change in IDH1. Covariance analysis suggested that serum IDH1 change was not significantly influenced by gender, age, tumor histological type, tumor stage, or TDG mutation burden (Table 2). However, we found that stage I lung cancer patients showed the greatest decrease in serum IDH1 level compared with stage II and III patients (Figure 2(b)). Before surgery, NSCLC patients with stage I showed the greatest variation in serum IDH1 levels and the histogram did not show sharp identifiable peaks, compared with serum IDH1 levels in patients with stage II or III. After the operation, serum IDH1 levels of stage I patients showed a single sharp peak. The Wilcoxon matched-pairs test revealed that the serum IDH1 level of NSCLC patients with stage I decreased significantly after surgery (p < 0.001), while the decrease of serum IDH1 in patients with stage II and III after surgery was not significant (p = 0.968, p = 0.263, respectively).
Serum IDH1 level increase after medical treatment predicts disease progression
Recent reports have shown that low-grade glioma has a high frequency of IDH1 mutation, which had a high impact on disease progress. 11 To discover IDH1's prognostic value in NSCLC, we first evaluated the IDH1 mutation rate in NSCLC. According to the ICGC data portal, 16 the IDH1 mutation rate in a Korea lung cancer cohort is lower than 10%, much less than in glioma (Figure 3(a)). The IDH1 mutation rate in the USA lung cancer cohort from the TCGA database is about 1%, even less. Unlike in low-grade glioma, IDH1 mutation could not predict patient outcome in OS or disease-free survival (DFS) (Figure 3(b) and (c), supplementary Figure 1(a) and (b)).
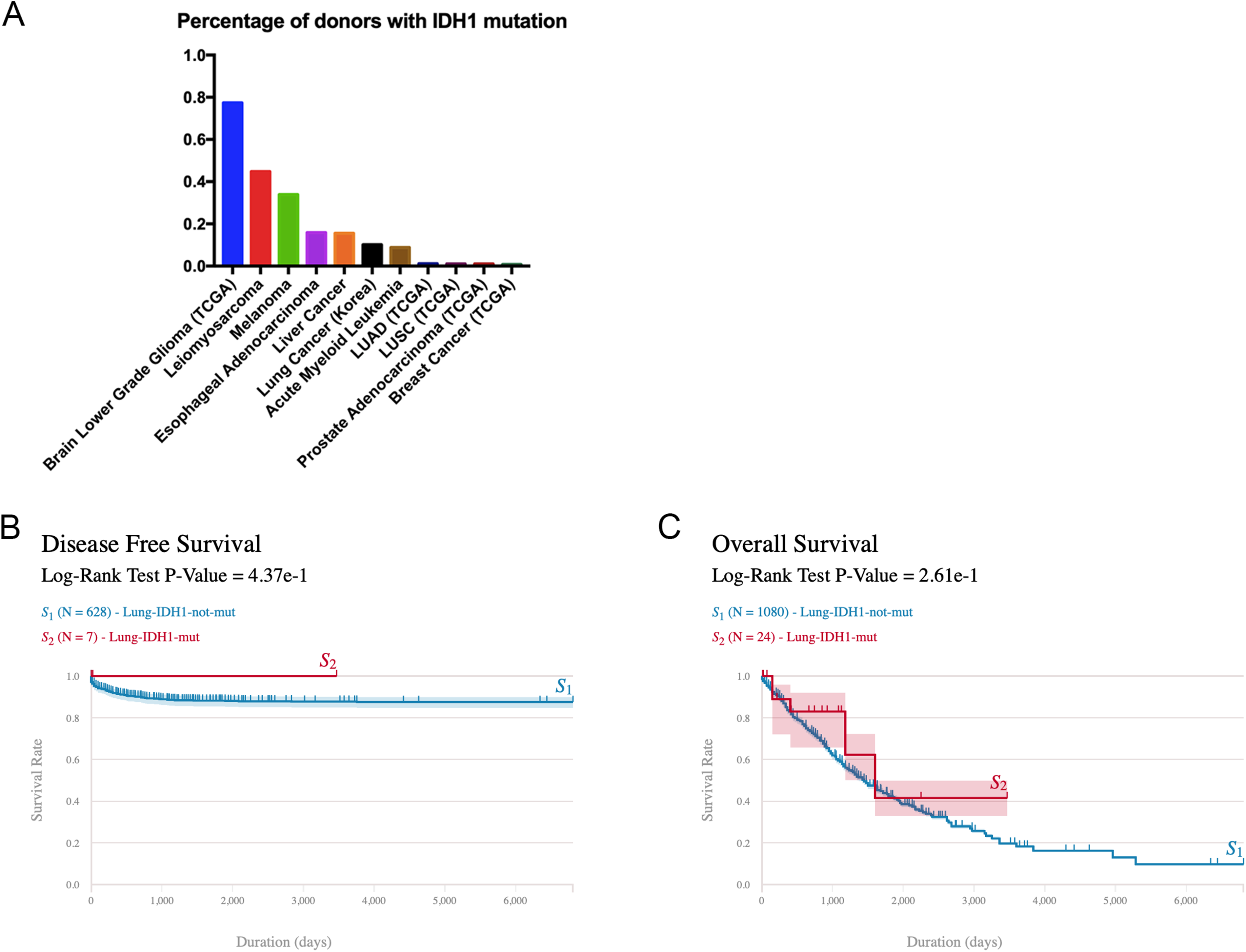
IDH1 mutation on lung cancer. (a) Comparison of IDH1 mutation rate among different types of cancer. (b) Disease-free survival. (c) Overall survival of lung cancer cohort based on IDH1 mutation condition. Data were downloaded from the data portal ICGC. Survival analysis was performed by ICGC online tools.
Then, we investigated whether patient serum IDH1 level was associated with disease progress. However, serum IDH1 levels either before therapy treatment or after therapy treatment could not predict patient DFS (Figure 4(a) and (b)). The IDH1 expression data in NSCLC tumor tissue were downloaded from the TCGA data portal. DFS analysis failed to separate patients with different IDH1 expression levels as well (Figure 4(c)). We extracted lung adenocarcinoma data with treatment outcome information, yet IDH1 expression had no significant differences between different outcome groups (Figure 4(d)).
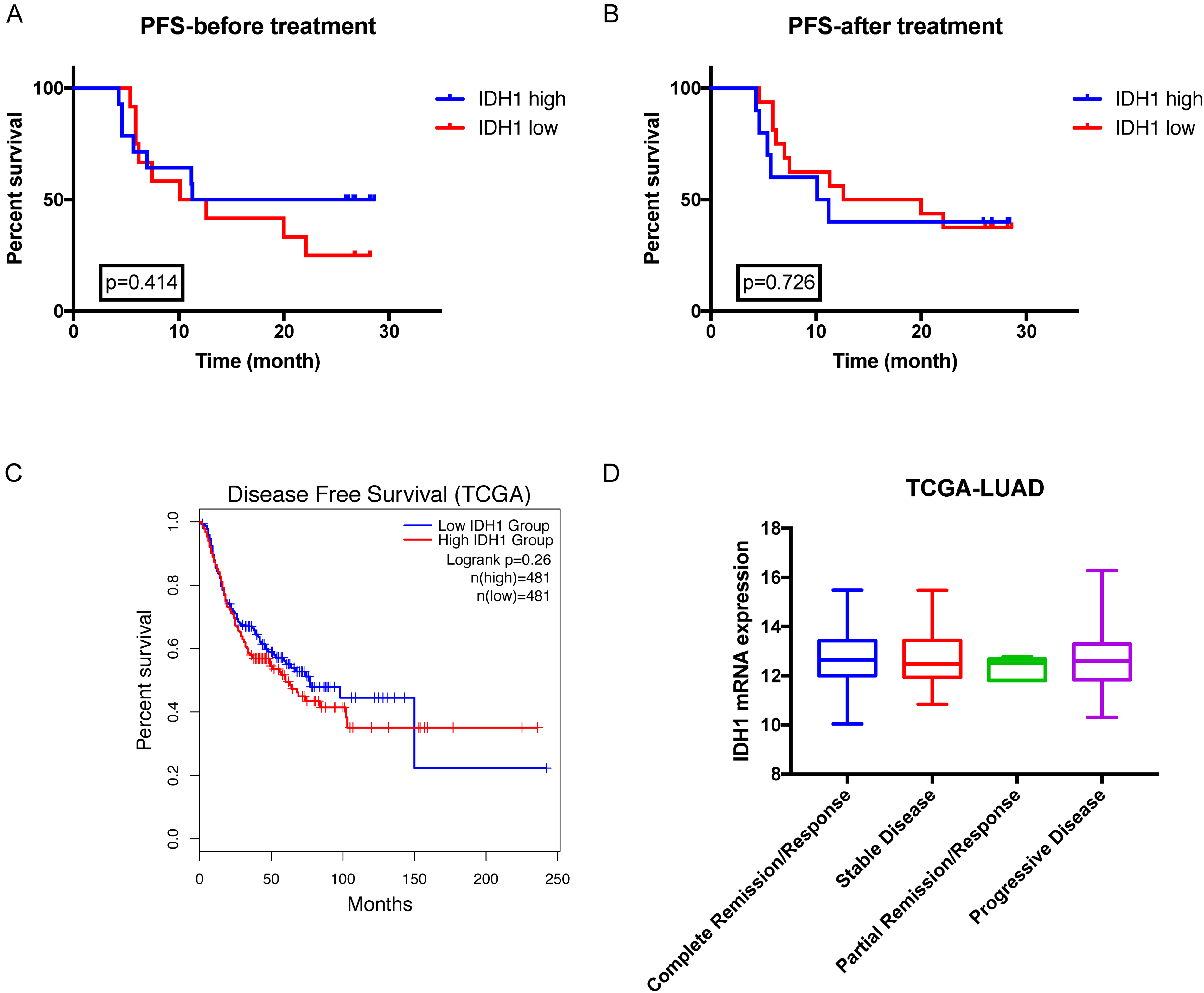
Relationship of IDH1 expression level and patient prognosis. Progression-free survival analysis of patients serum IDH1 level. (a) before chemotherapy and (b) after four cycles of chemotherapy. (c) Disease-free survival analysis of tumor IDH1 mRNA expression in lung cancer. Data were downloaded from the TCGA database. Survival analysis was performed by GEPIA online tools. (d) Tumor IDH1 mRNA expression level between patients with different clinical outcome. Data were downloaded from the TCGA database.
According to these results, the cross-section expression level of IDH1 might not be associated with the prognosis. Since we continuously test patients’ serum IDH1 levels during follow-up, we examined whether dynamic changes in those levels were related to patient treatment response. We examined 26 patients with advanced NSCLC (stage III and IV) who underwent chemotherapy or targeted therapy after surgery, and collected serum before chemotherapy treatment after two cycles of treatment, and after four cycles of treatment. These patients included 19 cases of lung adenocarcinoma (ADC) and 7 of small cell lung cancer (SCLC). Based on the TDG mutation status, 7 patients received epidermal growth factor receptor tyrosine kinase inhibitor treatment, and 19 patients received platinum-based chemotherapy. Patients received a therapy efficacy evaluation according to the response evaluation criteria in solid tumor (RECIST 1.1) after four cycles of treatment. Patients’ response to therapy was defined as stable disease (SD), partial response (PR), and progressive disease (PD). As the treatment progressed, serum IDH1 levels dramatically increased in PD patients, while serum IDH1 levels gradually decreased in SD/PR patients (Figure 5(a)). Repeated measures analysis of variance showed that the changes in the two groups were statistically significant (p < 0.001). We observed that the serum IDH1 level of PD patients alredy increased at the middle of four cycles of treatment. Therefore, we expanded the analysis to include patients who received at least two cycles of treatment, leading to a total of 37 patients, including 23 cases of ADC, 4 SCC, and 10 SCLC. As shown in Figure 5(b), serum IDH1 of PD patients increased after treatment, while serum IDH1 decreased in PR/SD patients. Although it did not meet the statistical requirements, DFS analysis results showed that patients with serum IDH1 increase had a shorter survival time (Figure 5(c)).
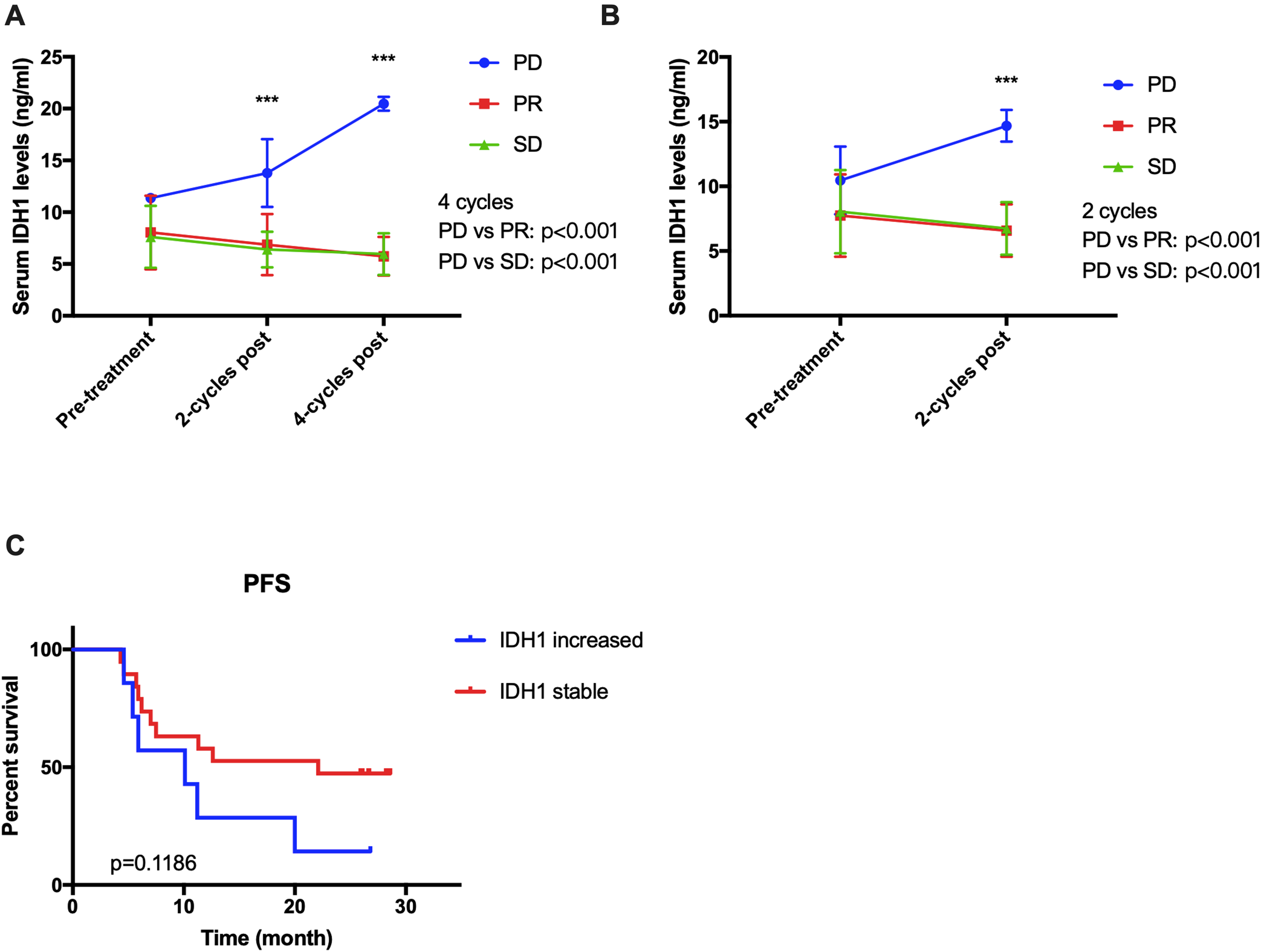
Dynamic changes of serum IDH1 before and after medical treatment. Patients who underwent four cycles of treatment (n = 27) (a), or at least two cycles of treatment (n = 37) (b), were divided according to response: PD, PR, and SD. Statistical analysis between PD and PR/SD groups using repeated measures analysis of variance. (c) Patients were separated into two groups according to their serum IDH1 level changes after four cycles of chemotherapy. Progress-free survival analysis was performed on patients.
Discussion
Although there have been many clinical applications of tumor markers, the sensitivity and specificity of these markers are not high enough for successful early tumor diagnosis and prognosis. 18 Therefore, the identification of novel tumor markers remains one of the current research hotspots. Analyses of serum samples show advantages in being non-invasive and easy to obtain. IDH1 is a serum marker that was recently discovered for NSCLC. Although the original discovery of IDH1 overexpression in NSCLC was in patiets plasma. Yet serum is more convenient to use than plasma. It is good to know that the diagnostic power of IDH1 in serum is no less than in plasma. 19 Therefore, the current commercial kits mainly use serum samples to determine IDH1 levels. In our study, serum IDH1 levels showed high sensitivity in the diagnosis of NSCLC. The average serum IDH1 level in patients with stage I lung cancer was the highest compared with levels in patients with stage II and III tumors. In addition, serum IDH1 was very effective in distinguishing tumors and benign lung diseases (Figure 1(a)). Serum IDH1 testing was also specific to lung cancer. The ROC curve performed poorly in diagnosing other cancers, including esophageal cancer, gastric cancer, and liver cancers. 8
We compared the dynamic changes of serum IDH1 before and after surgery; similar to other tumor biomarkers,20, 21 serum IDH1 levels decreased after surgery (Figure 2(a)). The changes in patients with stage I tumors were the most dramatic. However, among the 83 patients who underwent an operation, 28 showed elevated serum IDH1 levels after surgery. This ratio is higher than the results in other studies.7, 8 We attribute the reason to the timing of our postoperative serum collection in our study; we collected the serum samples during patients’ follow-up visits, while other studies collected serum samples within 3 days after surgery. 7 Together our results suggest that IDH1 may be used as a marker for tumor prognosis in NSCLC.
We also compared the dynamic changes of serum IDH1 before and after treatment. We found that after two or four cycles of treatment, the serum IDH1 levels increased in PD patients, while in PR/SD patients, serum IDH1 displayed a steady or slow decline. Although the tumor had not progressed in these four-cycle PD patients at the end of the second cycle, their serum IDH1 expression had begun to increase (Figure 3(b)). These results suggested that IDH1 has potential value for prognosis in NSCLC patients undergoing medical treatment.
Our cohort included patients with stage I–IV tumors. In addition, the treatments also varied among patients, and patients received different strategies of chemotherapy and targeted therapies according to mutated TDGs. Previous studies reported that IDH1 mutations are associated with chemotherapy response in patients with gliomas and leukemias.22, 23 In our research cohort, serum IDH1 levels also predicted the effects of targeted therapies. However, the number of patients in our study was limited; therefore, future studies are required with a greater number of patients to obtain more sufficient evidence. These findings may help elucidate the relationship between serum IDH1 dynamic changes and the clinical characteristics of NSCLC patients.
Supplemental Material
sj-docx-1-jbm-10.1177_17246008211052571 - Supplemental material for The isocitrate dehydrogenase 1 is a potential prognostic indicator for non-small cell lung cancer patients
Supplemental material, sj-docx-1-jbm-10.1177_17246008211052571 for The isocitrate dehydrogenase 1 is a potential prognostic indicator for non-small cell lung cancer patients by Xintong Zhang, Shang Ma, Yan Chen, Yanjun Yin, Wanqiu Bai, Jinjing Tan and Guangli Shi in The International Journal of Biological Markers
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
