Abstract
BACKGROUND:
The albumin-globulin ratio (AGR) could be a prognostic biomarker in patients with cancer, although the data is limited in patients treated with immune-checkpoint inhibitors (ICIs).
OBJECTIVES:
We aimed to evaluate the association between AGR and survival in ICI-treated patients.
METHODS:
The data of 212 advanced-stage patients were retrospectively evaluated in this cohort study. The association between AGR with overall (OS) and progression-free survival (PFS) were evaluated with multivariate analyses. Additionally, receptor operating curve (ROC) analysis was conducted to assess the AGR’s predictive power in the very early progression (progression within two months) and long-term benefit (more than twelve months survival).
RESULTS:
The median AGR was calculated as 1.21, and patients were classified into AGR-low and high subgroups according to the median. In the multivariate analyses, patients with lower AGR (
CONCLUSIONS:
In our experience, survival with ICIs was impaired in patients with lower AGR. Additionally, the AGR values could detect the very early progression and long-term benefit ICIs.
Introduction
The immune checkpoint inhibitors (ICIs) became the fifth pillar of cancer treatment [1] and entered treatment algorithms in almost all tumors [2]. The ICIs improved outcomes in several tumors, including non-small cell lung cancer (NSCLC) [3], renal cell carcinoma (RCC) [4], melanoma [5], head and neck cancer (HNC) [6], and Hodgkin lymphoma [7]. However, the outcomes with ICIs are still far from desired, with less than 30% overall response rates in most settings [8], especially in the later treatment lines. Additionally, class-specific immune-related adverse events (IrAEs) [9] and the financial burden of the ICIs [10] make biomarker-based patient selection paramount.
Despite the stunning speed of the therapeutic field developments, the development and implementation of prognostic and predictive biomarkers was a bit slow, possibly due to limited access to complex platforms in most countries and limited tissue availability in the metastatic setting [11, 12]. So, biomarker-based treatment tailoring did not become standard other than tumor PD-L1 expression in the first-line treatment of NSCLC [3] and two tumor agnostic markers, microsatellite instability, and tumor mutational burden, which are present in a limited number of patients [13]. Additionally, these tissue-based biomarkers evaluate the tumor characteristics only and have limited power to reflect the complex host-tumor interactions [14], and have limited benefit in monitoring the dynamic anti-tumor immune response [15].
Recently, the problems and limitations with the tissue-based biomarkers with ICIs paved the way for blood-based biomarkers with a particular interest in the simple biomarkers obtained from the blood count and biochemistry parameters reflecting the immune-inflammatory status and aiding in the prognosis prediction [16]. The studies with C-reactive protein levels, neutrophil-lymphocyte ratio (NLR), and platelet-lymphocyte ratio (PLR) demonstrated poorer outcomes in patients with increased inflammatory pressure [17, 18, 19]. Similarly, the albumin-globulin ratio (AGR) could also reflect the host’s inflammatory status and have the additional advantage of reflecting nutritional status [20], although the data on the ICI-treated patients with AGR is very limited [21]. From this point, we aimed to evaluate the association of AGR values and survival outcomes in a large cohort of ICI-treated patients. Furthermore, we assessed the predictive power of AGR to predict patients garnering a long-term benefit and patients progressing in a short time under ICIs.
Materials and methods
Patients
The data of adult advanced (recurrent or metastatic) cancer patients treated with any ICIs between January 2014 and August 2020 were included in this retrospective study. Additionally, the data of patients treated with ICIs between August 2020 and September 2021 in two institutions (Hacettepe University and Istinye University) used for the validation of the results (validation cohort). Patients with acute or chronic infections and missing data for AGR were excluded. Additionally, patients with microsatellite instable tumors and lung cancer patients with PD-L1 expression of 50 or higher treated with ICIs in the first-line were excluded from the analyses to prevent confounding. All other patients treated within prespecified dates were included in the study due to the limited number of patients and no power analyses were conducted to determine the sample size. The patients were treated with ICIs without a biomarker-based selection (PD-L1, TMB) due to unavailability in most cases.
Demographic features, ICI types, baseline height and weight, Eastern Cooperative Oncology Group (ECOG) performance status, previous treatment lines, comorbidities, regularly used drugs were recorded together with the best response to immunotherapy and survival data. The baseline lactate dehydrogenase, neutrophil, platelet, lymphocyte, albumin, and globulin levels were collected and recorded from the routine laboratory tests ordered in the preceding one week before the first immunotherapy dose. The comorbidities were classified according to Charlson-Comorbidity Index (CCI) [22]. The AGR was calculated by dividing the albumin (gr/dL) to total protein-albumin values.
Statistical analyses
Descriptive statistics were expressed by the median, interquartile range (IQR; 25th-75th percentile), standard errors for continuous variables, and frequency and percentages for categorical variables. Independent group comparisons were made using Mann-Whitney U and Chi-square tests for continuous and categorical variables, respectively. The median values were used as the cut-off for the definition of AGR-high and low groups. The Spearman test was used to evaluate the correlation between the AGR values and NLR/PLR. The overall response rate (ORR) was calculated by dividing the patients with a complete or partial response to all response-evaluated patients. The overall survival (OS) time was defined as the period from treatment initiation to the last follow-up and/or death, and progression-free survival (PFS) time was defined as the period between treatment initiation to disease progression and/or death. Univariate survival analyses were conducted with Kaplan-Meier analyses and comparisons between prognostic groups were made by log-rank tests. Multivariate survival analyses were conducted by a backward Cox-regression model, including the significant parameters in the univariate analyses, and hazard ratios were calculated together with 95% confidence intervals (CI). The discriminative ability of AGR in very early progression (within two months) and long-term benefit (more than oneyear survival) was evaluated with receiver operating characteristics (ROC) curve analysis [23]. All statistical analyses were performed in SPSS, version 25.0 (IBM Inc., Armonk, NY, USA), and a type-I error level of 5% (
Results
Baseline characteristics
A total of 253 patients were treated within the prespecified dates. After the exclusion of the patients treated within early access programs (
Baseline patient characteristics of study population
Baseline patient characteristics of study population
Comparison of baseline characteristics in the AGR low and AGR high groups
The median BMI of the cohort was 26.12 (IQR 23.40–29.89) kg/m
The association between NLR (left) and PLR (right) with AGR values.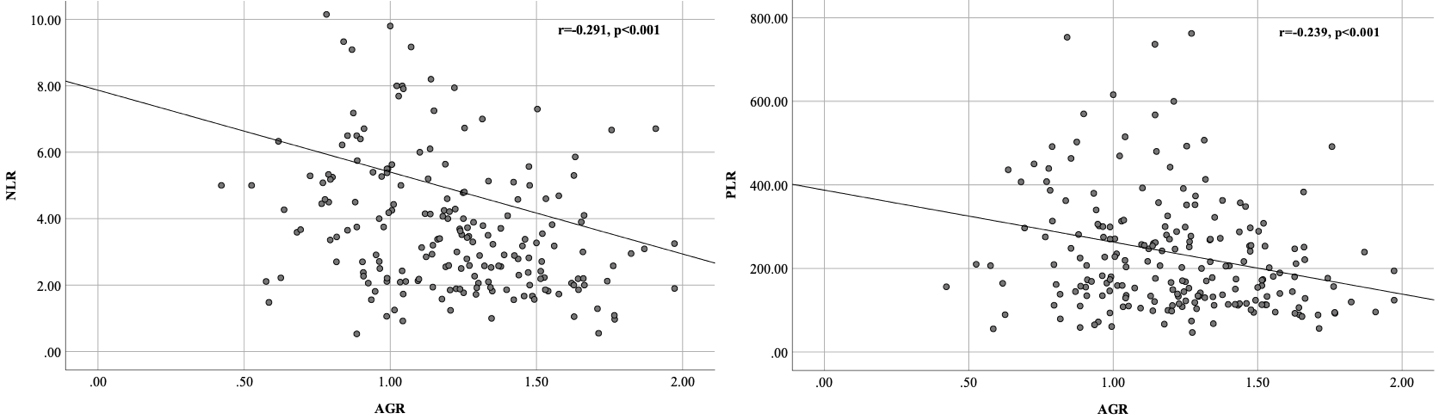
The association between response (complete or partial) with OS (left) and PFS (right).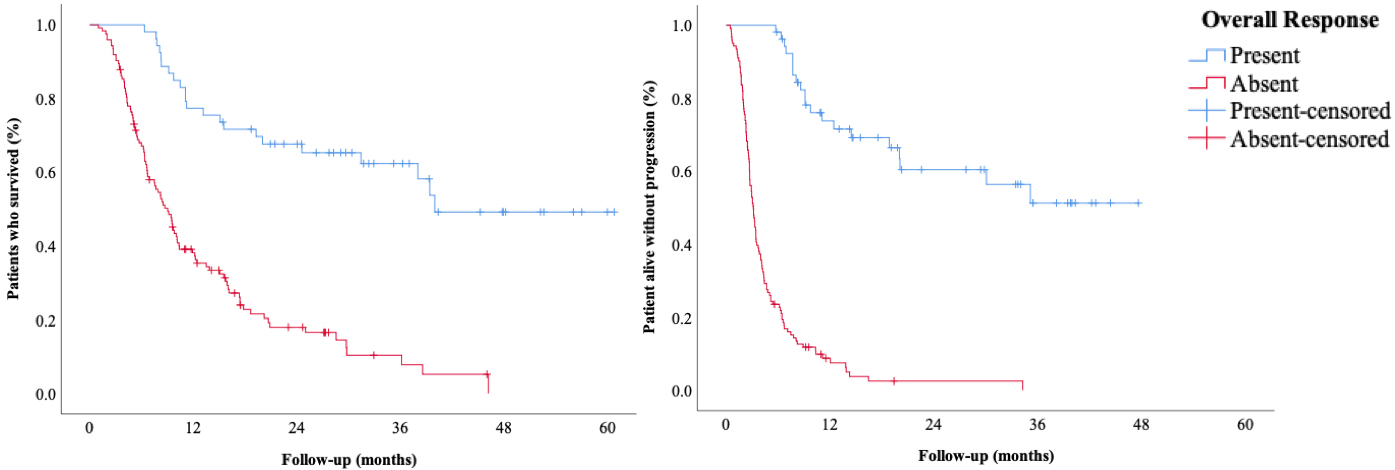
During a median follow-up of 9.56 (IQR 4.97–20.91) months, 154 patients died (72.6%), and 166 patients (78.3%) had any PFS event. The median OS was 9.76
In univariate analyses, patients with higher AGR (
The association between AGR with OS (left) and PFS (right).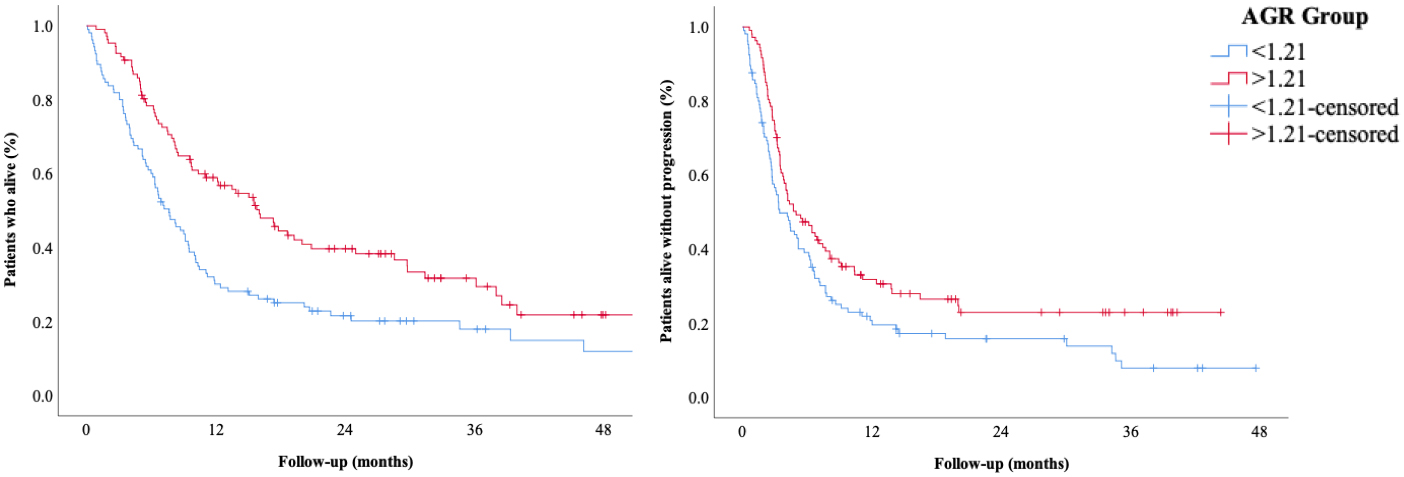
The association between AGR quartiles and OS.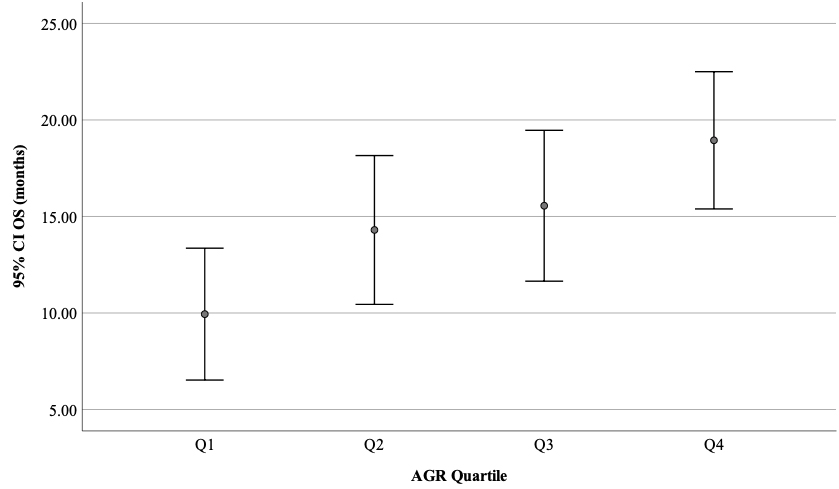
The association between clinical factors with overall and progression-free survival in multivariate analyses
The ROC analyses for AGR values in progression in two months (left) and long-term benefit (right).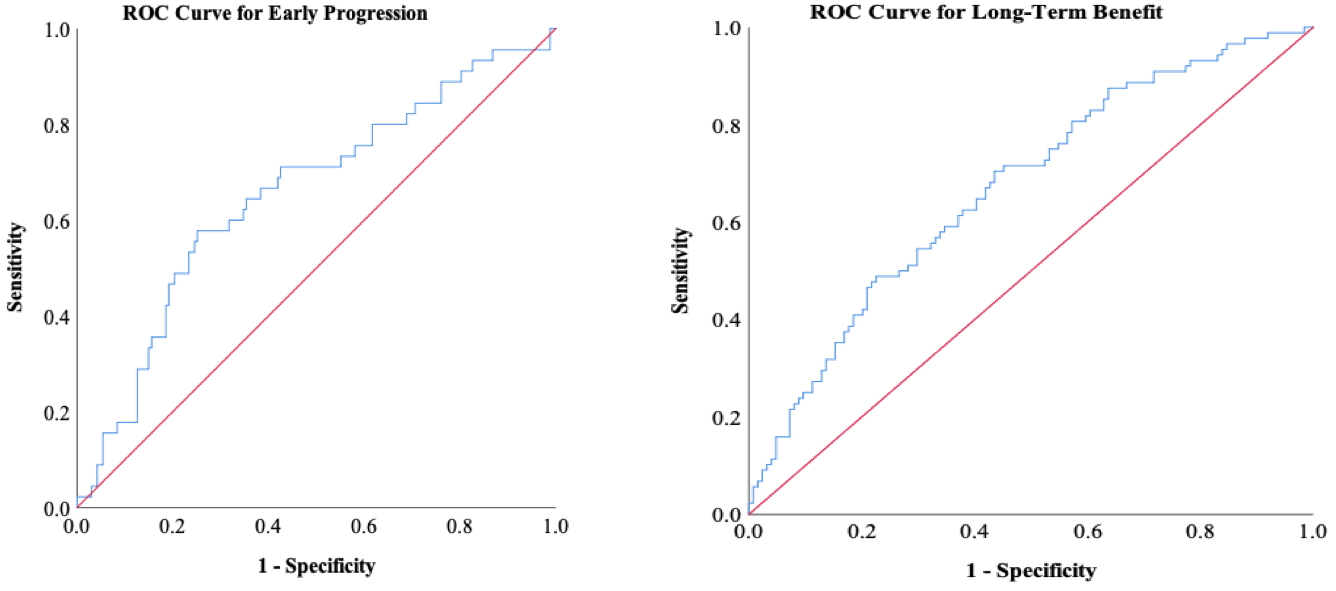
A multivariate analysis model was constructed and included the following parameters: ECOG performance status (0 vs.
A statistically significant association between AGR with OS (HR: 1.517, 95% CI: 1.092–2.108,
Receiver operating characteristics (ROC) analyses
The receiver operating characteristics (ROC) curve analyses demonstrated a moderate ability of AGR to predict OS (area under curve (AUC): 0.637, 95% CI: 0.555–0.719,
Validation cohort
We evaluated the performance of AGR as a prognostic and predictive biomarker in a validation cohort of 128 patients. The median age of the validation cohort was 60 (IQR 52–68) and 53.9% of the patients were male. The RCC (15.6%), melanoma (13.3%), and NSCLC (11.7%) were the most frequent diagnoses. Similar to the development cohort most patients (80.2%) had a good ECOG performance status (0 or 1). The median follow-up was 6.16 (IQR 3.56–9.87) months in the validation cohort. During the follow-up, 51 (39.8%) patients died and 86 patients (67.2%) had any PFS event. The median PFS was 4.01
Similar to the development cohort, AGR values had moderate ability to predict OS (AUC: 0.676, 95% CI: 0.543–0.810,
Discussion
In this study, we observed significantly lower OS and PFS in patients with lower AGR levels and higher ECOG performance status scores. Additionally, the patients with higher CCI had decreased OS. The decreased overall survival in the AGR-low patients was independent of the response to immunotherapy, the NLR levels and the use of ICI alone or ICI plus CT or targeted therapy.
Malnutrition is a widespread problem in patients with cancer and is related to decreased performance status, poor treatment tolerance, and impaired overall survival [24, 25, 26, 27]. There are several tools to evaluate malnutrition, including anthropometric measurements and questionnaires [28, 29]. The albumin levels could also serve as a denominator of malnutrition [30]. Besides, it has close relations with the host’s inflammatory status as its levels decrease with inflammation [31, 32, 33]. These features make albumin a candidate biomarker to reflect both the nutritional and the inflammatory status. Not surprisingly, the patients treated with chemotherapy or surgery with lower albumin levels had decreased overall survival [34]. However, the data on the value of albumin in ICI-treated patients is relatively limited. In a recent study on 106 NSCLC patients treated with ICIs, patients with low albumin levels (
The globulin levels reflect the combined antibody and cytokine burden in the body and increase in almost all chronic inflammatory states, including cancer [38, 39]. The cytokines produced during the inflammatory stress are reflected in the globulin constituent of the blood and have instrumental roles in tumor progression and immune exhaustion [40]. Based on this, globulin levels could be an indirect reflector of uncontrolled inflammation and tumoral progression. So, AGR has a solid biologic rationale to predict prognosis in cancer. An association between the lower AGR values and decreased OS was demonstrated in various tumors, including but not limited to urothelial cancer [41], RCC [42], hepatocellular cancer [43, 44], colorectal cancer [39], testicular cancer [45], breast cancer [46] and pancreatic cancer [47]. However, the cut-offs for AGR and sample sizes were very variable. A recent combined analysis of 14 studies encompassing 4136 patients with cancer demonstrated lower OS in patients with lower AGR values (HR: 1.87, 95% CI 1.50–2.34,
We observed an association between poorer ECOG status and decreased OS and PFS. The ECOG status is a general nominator of overall status and disease and symptom burden [50]. As in our study, a strong correlation between the ECOG status and survival was consistently demonstrated in patients treated with chemotherapy [51, 52] or immunotherapy [53]. The CCI could also serve similarly and successfully quantify the comorbidity burden [22] and aid in treatment individualization. Although relatively understudied in the ICI-treated patients, in a recent study on 66 NSCLC patients, patients with higher CCI had decreased OS. Like our study, the association between the PFS and CCI did not reach statistical significance [54], possibly due to the limited sample size.
We additionally evaluated the prognostic role of NLR and PLR in survival outcomes with ICIs. We used the median NLR and PLR values as the cut-offs for NLR and PLR. While the univariate analyses were statistically significant, the association between the NLR or PLR with OS was not statistically significant in the multivariate analyses. However, when we restructure the model with a different NLR cut-off (five), the association of NLR with OS and PFS was significant. Our results support the recent observations pointing out a more robust negative association with survival with higher NLR cut-offs [55, 56]. However, the possibility of lower sensitivity should be kept in mind with this strategy.
Our study has several limitations. First, the study’s retrospective nature and a heterogeneous patient group with a modest patient number prevented us from conducting additional subgroups analyses. Additionally, the limited number of cases caused relatively wide confidence intervals in the multivariate analyses and prevented us from conducting linear logistic regression analyses instead of binary analyses with a patient dichotomization according to an AGR cut-off. Most of our patients were treated in the second or later lines of the treatment that limited our results’ generability to patients treated in the clinical trials and countries with access to immunotherapy in the earlier lines. The additional adjustments according to pathologic characteristics like PD-L1 expression levels and tumor mutational burden were not possible due to a lack of data in most cases. However, PD-L1 levels are not routinely used in the patient selection for the tumor types and treatment lines of our cohort. Despite these limitations, we demonstrated the promise of AGR as a possible biomarker for prognosis prediction.
In conclusion, in our experience, survival with ICIs was impaired in patients with lower AGR values. Additionally, the AGR values could detect the patients with very early progression and patients garnering long-term benefit ICIs. If prospective studies validate our observations, AGR could be a readily available biomarker for treatment tailoring in ICI-treated patients.
Funding
The authors received no financial support for this article.
Compliance with ethical standards
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. The study was approved by the ethics committee of Hacettepe University.
Consent to participate
N/A
Consent for publication
N/A
Availability of data and material
The data was not reposited in the data repositories.
Code availability
N/A.
Author contributions
Conception: Deniz Can Guven, Oktay Halit Aktepe, Mustafa Erman, Suayib Yalcin, Sercan Aksoy, Saadettin Kilickap.
Interpretation or analysis of data: Deniz Can Guven, Oktay Halit Aktepe, Melek Seren Aksun, Taha Koray Sahin, Gozde Kavgaci, Enes Ucgul, Ibrahim Yahya Cakir, Hasan Cagri Yildirim, Gurkan Guner, Serkan Akin, Neyran Kertmen, Saadettin Kilickap.
Preparation of the manuscript: All Authors.
Revision for important intellectual content: All Authors.
Supervision: Mustafa Erman, Suayib Yalcin, Sercan Aksoy, Saadettin Kilickap.
Supplementary data
The supplementary files are available to download from http://dx.doi.org/10.3233/CBM-210349.
sj-pdf-1-cbm-10.3233_CBM-210349.pdf - Supplemental material
Supplemental material, sj-pdf-1-cbm-10.3233_CBM-210349.pdf
sj-xlsx-1-cbm-10.3233_CBM-210349.xlsx - Supplemental material
Supplemental material, sj-xlsx-1-cbm-10.3233_CBM-210349.xlsx
sj-xlsx-2-cbm-10.3233_CBM-210349.xlsx - Supplemental material
Supplemental material, sj-xlsx-2-cbm-10.3233_CBM-210349.xlsx
Footnotes
Conflict of interest
The authors declare that they have no conflict of interest.
