Abstract
BACKGROUND:
The most commonly used prognostic factors in acute myeloid leukemia (AML) are cytogenetic, molecular, and morphological markers. However, AML prognosis is still unfavorable particularly in adults. So, further reliable markers are urgently needed to improve the risk stratification and treatment decisions. CUB domain-containing protein 1 (CDCP1; CD318) and endoglin (CD105) are new markers correlated with poor prognosis in different solid tumors, but their role in AML prognosis is not fully evaluated.
OBJECTIVES:
This work aimed to evaluate the prognostic role of CD318 and CD105 in AML and their impact on the outcomes.
METHODS:
Sixty-five newly diagnosed AML patients were included in this study. CD318 and CD105 expression was assessed by quantitative real-time polymerase chain reaction. Patients were followed up for
RESULTS:
Patients with high CD318 and CD105 showed higher white blood cell (WBC) count, M2 subtype, poor cytogenetic risk, reduced complete remission, and a greater number of deaths compared to low CD318 and CD105. CD318 was correlated with CD105, and both were correlated with WBC count, bone marrow blasts, and peripheral blood blasts. After a follow-up period of up to 24 months, relapse-free survival for high CD318 and CD105 was significantly different (42.1% and 52.6% vs. 64.5% and 58.1% for low CD318 and CD105, respectively). Survival was worse in patients with high CD318 and CD105, as the mean survival time was 13.9 and 13.3 months compared to 24 and 22.7 months in low CD318 and CD105, respectively.
CONCLUSIONS:
CD318 and CD105 are upregulated in AML patients. Their overexpression was associated with poor response to treatment and poor outcomes. Therefore, CD318 and CD105 can be useful prognostic markers in AML.
Keywords
Abbreviations
Introduction
Acute myeloid leukemia (AML) is the most common form of acute leukemia in adults [1]. Despite advancement in the treatment of AML have led to significant improvements in outcomes for younger patients, prognosis in the elderly who account for most new cases remains poor [2]. The elderly AML patients aged from 65 to 74 years die within one to two years following AML diagnosis because they are often unable to tolerate current treatment regimens [3].
In the United States, the age-adjusted annual incidence of AML is 4.3 per 100,000. Incidence increases with age, with a median age at diagnosis of 68 years. AML can arise in patients with an underlying hematological disorder, or as a result of previous therapy (topoisomerases II, alkylating agents or radiation) [4]. However, in majority of cases, it appears as a de novo malignancy in previously healthy individuals. The pathogenesis of AML involves the abnormal proliferation and differentiation of a clonal population of myeloid stem cells. In most AML cases, the etiology is unclear. However, AML is sometimes caused by previous exposure to therapeutic, occupational, or environmental DNA-damaging agents [1].
AML is a highly heterogeneous disease; although cases can be stratified into favorable, intermediate, and adverse-risk groups based on their cytogenetic profile, prognosis within these categories varies widely. Its prognosis usually depends on cytogenetic and morphological markers [5]. The commonly used cytogenetic and molecular features include inversion (16), translocation (8; 21), translocation (15; 17). Translocations (8; 21) in core-binding factor AML (CBF-AML) or (15; 17) in acute promyelocytic leukemia (APL) result in the formation of chimeric proteins (RUNX1-RUNX1T1 and PML-RARA, respectively), which alter the normal maturation process of myeloid precursor cells [6].
Besides, genetic mutations are identified in more than 97% of cases [6] often in the absence of any large chromosomal abnormality [7]. Animal models proposed two-hit model of leukemogenesis provided two classes of mutation [8]. Class I mutations activates the pro-proliferative pathways, such as FLT3-ITD, TKD, K/NRAS, TP53 and c-KIT [7]. Class II mutations which impair normal hematopoietic differentiation [9] such as NPM1 and CEBPA and have a better prognosis [7]. Epigenetic regulation has recently considered the third class of mutations, with downstream effects on both cellular differentiation and proliferation including mutations in the DNA-methylation related genes DNMT3A, TET2, and IDH-1 and IDH-2 [6]. Proteomic biomarkers include PTP, PTK and PIP [10].
The identification of current genetic mutations, such as FLT3-ITD, NMP1, MLL and CEBPA are used to refine individual prognosis and guide management [11]. However, new markers are required for the improvement of the risk stratification and treatment decision guiding in AML.
CUB domain-containing protein 1 (CDCP1), also known as CD318, is a transmembrane protein expressed on the surface of hematopoietic mesenchymal and neural stem cells and in fibroblasts [12]. CD318 promotes solid tumor metastasis via interaction with integrins and antiapoptotic signaling via Akt [13, 14]. CD318 expression has been observed on CD34- and CD133-positive AML blasts. CD318 overexpression has been correlated with poor overall survival (OS) in the colon, breast, renal, hepatocellular (HCC), and pancreatic carcinoma [13, 14, 15, 16, 17].
Endoglin (CD105) is a type I transmembrane glycoprotein located on the endothelial cell surface, which serves as a transforming growth factor-
The prognostic significance of CD318 and CD105 in AML has not been fully assessed. Heitmann et al. evaluated the role of CD318 as a new prognostic marker in AML by immunophenotyping of leukemic blasts. Flow cytometric analysis revealed a substantial expression in 57% of all cases. CD318 surface levels were significantly correlated with OS [28]. Kauer et al. generated a new CD105 antibody to analyze the expression and prognostic significance of CD105 protein expression in AML by flow cytometry. Flow cytometric analysis revealed that high CD105 expression correlated significantly with poor OS and progression-free survival [29]. Allogeneic stem cell transplantation for eligible candidates is a treatment modality in AML. Märklin et al. detected high CD105 expression in 65.8% of AML patients following allogeneic hematopoietic stem cell transplantation [30]. No previous studies studied both markers together or evaluated their gene expression to the authors’ knowledge.
Despite advances in the supportive care of AML, the backbone of therapy remains a combination of cytarabine- and anthracycline-based regimens with allogeneic stem cell transplantation for eligible candidates. New molecular targeted therapies provide effective anti-leukemic activity with reduced toxicity. Due to the molecular diversity of AML, targeted therapies are better to be combined. Discovery of new treatments depends on improved genetic profile and risk stratification which may benefit in remission and survival. The recognition of gene expression profile of specific cell surface markers can provide a therapeutic target for recombinant monoclonal antibodies. The key role is selectivity in targeting leukemic myeloid cells while sparing non-malignant myeloid precursors. Besides, the development of well-tolerated oral therapies, will help elderly patients at a higher risk of mortality due to intolerance to current treatment regimens.
So, this study investigated the prognostic role of both CD318 and CD105 overexpression in AML by quantitative real-time polymerase chain reaction (qRT-PCR) and tried to explore their impact on patient outcomes.
Materials and methods
Study design
This prospective study was carried out on 65 patients with newly diagnosed AML. This study was carried out in the Departments of Clinical Pathology, Medical Microbiology and Immunology and Medical Biochemistry and Molecular Biology, Faculty of Medicine, Zagazig University, from March 2018 to June 2021. The diagnosis and classification of AML samples relied on the morphology and cytochemistry of bone marrow (BM) and classified according to the French-American-British (FAB) classification [31]. Research ethical committee approval (ZU-IRB# 4906/810-2018) and informed written consent from all participants in this research were fulfilled.
Bone marrow (BM) cells were collected from all AML patients at the time of diagnosis. BM specimens were collected on potassium ethylene diamine tetra-acetic acid (K-EDTA) (1.5 mg/mL) for morphologic, immunophenotypic, and molecular analysis.
Flow cytometric analysis
Immunophenotyping for acute leukemia using the standard panel of FITC/PE/PerCP-labeled monoclonal antibodies (CD45, CD34, CD38, HLA-DR, MPO, terminal deoxynucleotidyl transferase, CD64, CD14, CD2, CD3, CD5, CD7, CD19, CD79a, CD20, and CD22) was performed (Becton Dickinson, San Diego, CA, USA). Antigens were scored positive using a cutoff value of
Primer sequence of the studied genes
Primer sequence of the studied genes
Total RNA was extracted using easy-Red
qRT-PCR was carried out on an Mx3005P RT-PCR System (Stratagene, La Jolla, CA, USA). Aliquots of 1
Treatment plan
All 50 fit patients were given an anthracycline and cytarabine-based induction chemotherapy regimen; 3
Fifteen patients who were not fit for aggressive chemotherapy were treated by alternative low-intensity therapy, including low-dose cytarabine (10 mg/m
Patients with acute promyelocytic leukemia are treated with all-trans retinoic acid or Vesanoid as a differentiating factor plus chemotherapy: anthracycline alone or anthracycline plus cytarabine.
Hematologic CR is defined as meeting all the following response criteria:
Statistical analysis
Data were analyzed using IBM SPSS 23.0 for Windows (SPSS, Inc., Chicago, IL, USA) and NCSS 11 for Windows (NCSS LCC, Kaysville, UT, USA). Quantitative data were expressed as the mean
Results
Basic clinicodemographic characteristics and outcomes among the participants
According to the FAB classification of AML patients, M4 was the most frequent (29.2%), followed by M2 (26.2%), and the least was M3 (6.2%). Based on cytogenetics, patients were categorized according to the National Comprehensive Cancer Network (NCCN) risk score. The intermediate cytogenetic risk was detected in 38.5% of the patients, followed by favorable risk (30.8%), and poor risk was detected in 24.6% of the patients. Response to chemotherapy was defined according to the European Leukemia Network definition. CR was defined as a presentation with normocellular BM containing
Demographic and basic characteristics among the participants
Demographic and basic characteristics among the participants
FAB: French-American-British; CR: Complete response to treatment, PR: partial response, RR: refractory response, RFS: relapse-free survival.
At a cutoff value of
Relation between CD318 level and basic clinical characteristics of the studied cases
Relation between CD318 level and basic clinical characteristics of the studied cases
FAB: French-American-British; CR: Complete response to treatment, PR: partial response, RR: refractory response, RFS: relapse-free survival.
Patients were divided according to the CD105 expression level at a cutoff value of
Relation between CD105 level and basic clinical characteristics of studied cases
Relation between CD105 level and basic clinical characteristics of studied cases
FAB: French-American-British; CR: Complete response to treatment, PR: partial response, RR: refractory response, RFS: relapse-free survival.
No significant difference was found between the two groups concerning any of the studied parameters (
Relation between high CD105 and high CD38 patients and basic characteristics of studied cases
Relation between high CD105 and high CD38 patients and basic characteristics of studied cases
Correlation between CD105 and CD318 markers with other laboratory data
CD318 expression was positively correlated with CD105 expression (
Response to treatment and patient outcomes among high and low CD318 and CD105 groups
CR, RR, and PR were detected in 46.9%, 34.4%, and 18.8% in the high CD318 expression group and 81.7%, 6.1%, and 12.1% in the low CD318 expression group, respectively, with a significant difference between the two groups regarding CR (
CR was significantly different between patients with high CD105 expression (76.5%) and those with low CD105 expression (51.6%) (
Kaplan-Meier overall survival analysis of studied cases according to CD105 expression. The mean time of survival for patients with high CD105 was 13.3 months and 22.7 months for patients with low CD105.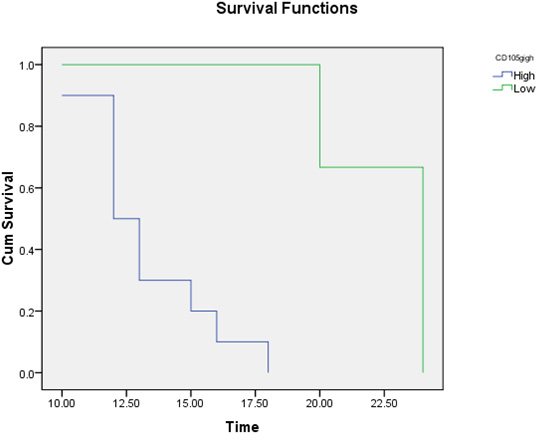
Kaplan-Meier overall survival analysis of studied cases according to CD318 expression. The mean time of survival for patients with high CD318 was 13.9 months and 24 months for patients with low CD318.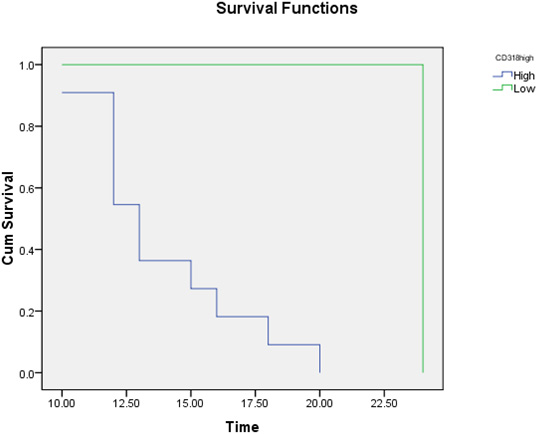
After a follow-up period of up to 24 months, the mean survival time was 13.9 and 13.3 months, and the number of deaths was 13 and 12 patients in the high CD318 and CD105 expression groups, respectively. The mean survival time was 24 and 22.7 months, and the number of deaths was 2 and 3 patients in the low CD318 and CD105 expression groups, respectively. There were significant differences between the groups (
Survival study of CD318 and CD105 by log-rank test
Survival study of CD318 and CD105 by log-rank test
CD318 and CD105 overexpression has been correlated with poor OS in different solid tumors [19, 20, 21, 22, 23, 24, 25, 26]. Their prognostic role in AML is not far fully evaluated. This study assessed the significance of CD318 and CD105 as novel prognostic markers in AML and their impact on patient outcomes. This study found that 49.3% and 47.7% of the patients showed high CD318 and CD105 expression, respectively. These results were similar that of Heitmann et al., who detected high CD318 protein expression in 42.8% of AML patients [28], and Kauer et al., who detected high CD105 protein expression in 51.6% of AML patients [29]. Our study is different from the previous two studies in the combined study of the two markers together from gene expression view for the first time.
Our study found that FAB M2 subtype, poor cytogenetic risk, secondary cause and WBC count were significantly increased, but favorable cytogenetic risk and platelet count were significantly decreased in high CD318 AML patients compared to low CD318 patients. CD318 was also significantly correlated with CD105, WBC count, platelet count, BM blasts, and PB blasts. These results are supported by Heitmann et al., who detected a significantly higher percentage of poor cytogenetic risk, M2 FAB subtype, and secondary AML patients in the CD318 high expression group. In contrast, they detected that the WBC count was significantly increased in the CD318 low expression group [28].
In this study, WBC count, BM blasts, CD318 expression, poor cytogenetic risk, and secondary AML were significantly higher, but platelet count and favorable cytogenetic risk were significantly lower in patients with high CD105 expression compared to those with low CD105 expression. The percentage of patients with the FAB M2 subtype was increased in the CD105 high expression group compared to the low CD105 expression group, but no significant difference was detected. CD105 was significantly correlated with CD318, WBC count, platelet count, BM blasts, and PB blasts. Following these results, Kauer et al. detected a significant difference between patients with high and low CD105 protein expression in AML concerning WBC count, poor cytogenetic risk, and FAB classification, but no significant difference was found in platelet count [29]. Similar results were detected by Abd El-Ghany et al., who found a significant correlation between CD105 with WBC count and BM blasts but no significant correlation with platelet count [33]. In contrast, Märklin et al. detected significant differences in CD105 expression regarding FAB classification and NCCN risk score but no significant difference between primary and secondary AML concerning CD105 expression, after allogenic stem cell transplantation [30].
Cytogenetic changes are the strongest prognostic factors for CR and OS in AML. AML is classified into favorable, intermediate, or adverse prognostic risk groups depending on their cytogenetic profile. The chromosomal rearrangements have a favorable prognosis. CN-AML with a mutated CEBPA or a mutated NPM1 in the absence of FLT3-ITD has a prognostic risk like that of AML with favorable cytogenetic changes. FLT3-ITD is associated with a poor prognosis. TP53 mutations, found in 2–8% of cases and frequently associated with unfavorable cytogenetic and a complex karyotype, are associated with a very poor prognosis and it is the single worst genetic prognostic factor. Mutated DNA methyl transferase gene (DNMT)3A is associated with a poor prognosis in CN-AML and adverse-risk AML. MLL, which encodes a histone methyltransferase, is associated with a poor prognosis in CN-AML. The prognostic impact of IDH-1/IDH-2 mutations is likely modified by co-occurring mutations. Among cases of FLT3-ITD-negative and NPM1-mutated CN-AML, IDH-1/IDH-2 mutations have been shown to improve OS [5].
On comparing between patients with high and low CD318 and CD105 concerning the response to treatment and patient outcomes, patients with RR were significantly increased in the high CD318 and CD105 groups. Dead patients were significantly increased in patients with high CD318 and CD105, as well. CR was significantly higher in patients with low CD318 and CD105. In agreement with these results, Kauer et al. found that CR after induction therapy occurred in a significantly higher number of cases in the CD105 low expression group than in the CD105 high expression group, in line with the observation that the CD105 high expression group comprised more poor cytogenetic risk [29]. Märklin et al. detected CR in 62% of the low CD105 expression group and 57% of the high CD105 expression group [30]. Number of patients with RFS were significantly higher in low CD318 expression group compared to high CD318 expression group
After a follow-up period of up to 24 months, the survival study revealed that the mean survival time in CD318 and CD105 was shorter in the high expression groups than the low expression groups, and the numbers of dead patients were higher in the high expression groups than the low expression groups. The results followed that of Märklin et al., who reported that low CD105 expression was significantly correlated with a better response to induction therapy, and high CD105 levels were associated with poor OS [30].
Kauer et al. stated that the association of CD105 with inferior OS was unclear and speculated that CD105 might have influenced the occurrence of treatment resistance and reduced the response [29]. Hand by hand with this study, Abd El-Ghany et al. detected that patients with positive CD105 expression had a significantly lower CR rate and a significantly higher relapse and death rate. They also found a significant decrease in both OS and DFS in the positive CD105 expression group [33]. Dourado et al. reported that CD105 was associated with the increased leukemogenic activity of blast cells, and this may decrease both OS and DFS [34].
Poor prognosis with high CD105 expression can be attributed to that CD105 induces the activation and proliferation of endothelial cells through the promotion of TGF-
In contrast, Heitmann et al. failed to detect a significant difference between high and low CD318 protein levels in AML concerning OS. However, when they grouped patients according to the applied treatment, they found that patients with high CD318 who received the best available alternative therapy showed increased survival, but those who received intense AML treatment and expressed low CD318 had longer OS [28]. They attributed this difference with treatment to the dependency of AML blasts on the promoter methylation of tumor suppressor genes [40], which is inhibited by hypomethylating agents [41]. Therefore, CD318 could be used as a marker for the degree of DNA methylation in AML [28].
In lung carcinoma, CD318 expression has been associated with the presence of genetic aberrations, such as Ras mutations [42]. CD318 expression is induced via HIF-2
Conclusion
CD318 and CD105 are upregulated in AML. They are significantly correlated with poor patient outcomes. Therefore, they can be useful novel prognostic markers in AML. Besides, a phase II clinical trial of TRC105 (anti-endoglin antibody) in adults with advanced/metastatic urothelial carcinoma was performed. Monoclonal antibodies are recommended in AML as they exert anti-tumor activity through direct antibody-dependent cytotoxicity or through the conjugation of cytotoxic agents to ensure targeted delivery of potent chemotherapy to neoplastic cells avoiding long-term myelosuppression with selectively target leukemic myeloid cells. This is considered a targeted delivery of potent chemotherapy to neoplastic cells.
Funding
None.
Author’s contributions
Conception: Huda F. Ebian and Samia Hussein.
Interpretation or analysis of data: Rasha L. Etewa, Dina R Issa, Amira S. Al-Karamany, Hanaa M. El Maghraby and Samia Hussein.
Preparation of the manuscript: Huda F. Ebian and Samia Hussein.
Revision for important intellectual content: All authors.
Supervision: Huda F. Ebian and Samia Hussein.
Footnotes
Acknowledgments
None.
Conflict of interest
None.
