Abstract
BACKGROUND:
Acute myeloid leukemia changes the bone marrow (BM) niche to support leukemia cells by modulating the stromal microenvironment. The aim is to assess Activin-A as a biomarker in acute myeloid leukemia (AML).
METHODS:
The level of Activin-A and CXCL-12 protein concentration levels in the plasma of bone marrow aspirate samples of eighty AML patients at diagnosis, after induction and at relapse were determined by ELISA.
RESULTS:
We found that Activin-A concentration levels was significantly up regulated in AML cases at diagnosis, and down regulated at complete remission and rise again at relapse (
CONCLUSION:
Activin-A level at diagnosis is a new simple easily assessed biomarker that could predict AML patient’s response to therapy as well as patient’s outcome.
Introduction
The incidence and mortality of leukemia account for 2.4–3.2% of all cancers Acute myeloid leukemia (AML) as the most common acute leukemia in adults, is characterized by the accumulation of immature and nonfunctional myeloid precursor cells in the blood and bone marrow [1].
The bone marrow (BM) represents a specific microenvironment characterized by a high concentration of cytokines and growth factors needed for hem-atopoiesis and necessary for homing of the leukemic cells, proliferation and survival [2]. The family of
Activin-A is necessary to maintain pluripotency and the subsequent differentiation of hematopoietic human pluripotent stem cells during human development. The Prolonged treatment of hematopoietic human pluripotent stem cells with Activin-A can induces definitive differentiation [4]. The mesenchyme bone marrow and the endothelium derived stromal cells express Activin-A and they are known to induce, maintain hemopoiesis, influence and support growth and survival of different types of leukemic cells [5].
Recent studies have reported that Activin-A is an important regulator of carcinogenesis as it can directly modulate cancer cell proliferation, migration and enhance tumor progression through the regulation the microenvironment of the tumor [6]. Previous study found that Activin-A production is strongly increased after direct contact with the leukemic cells and the bone marrow mesenchymal stem cells are important source of it, these data agrees with the theory of seed and soil indicating that the cancer cells can alter the microenvironment for their survival, migration and proliferation [6]. The Activin receptor II ligand trap ACE-011 is currently under investigation in a Phase II clinical trial on multiple myeloma [7]. On the other hand CXCL-12 is a chemokine found in both soluble form and membrane-bound form, Also widely distributed in the bone marrow microenvironment act as main ligand for CXCL-4 and can enhance proliferation and survival of chronic lymphocytic leukemia cells, also possess prognostic value in leukemia [8]. On the leukemic cells, CXCL-12 binds and stimulates their specific receptor CXCR-4, increases the ability of leukemic cells trafficking, homing and keeps these cells in close contact to stromal cells which promotes growth and anti-apoptosis [9]. The CXCL-12 expression is up regulated by A kinase interacting protein 1 (AKIP1), a molecular regulator of protein kinase A, is reported to act as a structural intracellular protein and is found in the cytoplasm, mitochondria and nucleus, and induce the activation of CXCL-1/CXCL-12 downstream oncogenic signaling pathway (Wnt
Recent studies showed that the AKIP1 has a role to the angiogenesis, lymph angiogenesis, tumorigenesis and invasiveness in many solid cancers, including gastric cancer, colorectal cancer and breast cancer, and it predicts worse survival in patients with cancer, suggesting that AKIP1 functions as an oncogene and may be an effective prognostic marker in these cancers [11].
Activin-A and CXCL-12 are hypothesised to be prognostic biomarkers in patients with AML. Thus, we conducted this study to explore the impact of Activin-A and CXCL-12 concentrations on the AML patient’s outcome.
Methods
This is a prospective observational cohort study included eighty patients with AML. The AML patients attending Oncology Center-Mansoura University (OCMU). were sequentially included in our study.
Blood and Marrow samples were collected after informed consent from controls or patients with AML according to protocols approved by the faculty of medicine of Mansoura university ethics committee.
Laboratory data of the studied group
Laboratory data of the studied group
Correlation of Activin-A with CXCL-12 plasma concentration levels in AML studied cases. There were significant negative correlations between both biomarkers (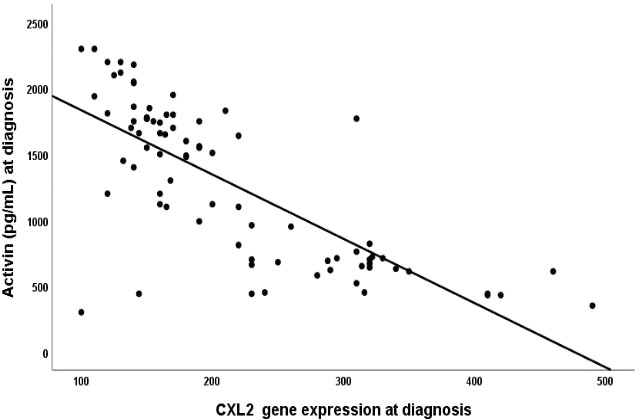
Adult patient (age
The enrolled subjects were subjected to the following detailed history and clinical examination, complete blood count (CBC), differential leukocytic count and blood film, Other routine laboratory tests, liver function tests, kidney function tests, BMA, biopsy, Flowcytometry, karyotyping, molecular analysis, Radiological investigation, and brain imaging.
Patients received standard protocol of AML therapy with 7
Plasma samples were collected from the BM of AML patients before start of therapy as well as controls who referred for bone marrow aspiration and found to be free. The plasma samples were preserved at
Statistical methods
The collected data were revised, coded, tabulated and introduced to a PC using Statistical package for Social Science (IBM Corp. Released 2017. IBM SPSS Statistics for Windows, Version 25.0. Armonk, NY: IBM Corp.). Data were presented and suitable analysis was done according to the type of data obtained for each parameter. Student T Test was used to assess the statistical significance of the difference between two study group means. Mann Whitney Test (U test) was used to assess the statistical significance of the difference of a non-parametric variable between two study groups. Chi-Square test was used to examine the relationship between two qualitative variables. Wilcoxon signed rank sum test was used to assess changes in parameters over time. Logistic regression analysis was used for prediction of risk factors, using generalized linear models. Kaplan-Meier test was used for survival analysis and the statistical significance of differences among curves was determined by Log-Rank test. Cox regression analysis was performed to identify factors that might jointly have a significant influence on survival. A
Comparison of serum Activin-A and CXCL12 concentrations levels between studied groups
Comparison of serum Activin-A and CXCL12 concentrations levels between studied groups
Activin levels is higher at AML diagnosis as compared to control levels (
Comparison of levels of Activin-A and CXCL12 at diagnosis in patients’ subgroups classified according to induction remission response, outcome and cytogenetic risk
Eighty adult AML with 37.1 years mean age were included in this study. They were forty-nine males and thirty-one females. Ten healthy control of matched age and gender, laboratory data for patients and control are presented in Table 1.
Correlating Activin-A with CXCL-12 gene expression in AML studied cases revealed a significant negative correlation between both markers, (
Activin-A concentration levels were noticed significantly higher at diagnosis as compared with the control levels (
The Activin-A concentration levels at diagnosis were found significantly higher in AML subgroup of patients who did not achieve complete remission; died and those had high cytogenetic grade as compared with those who achieved complete remission; alive; and subgroup of patients had low/intermediate cytogenetic grade (
In order to do risk assessment of the investigated parameters; univariate analysis was done using the following parameters: age, gender, BM blasts, cytogenetic grade, Activin-A, and CXCL-12 levels at diagnosis to predict AML patient’s outcome. The significant parameters in the univariate analysis were introduced in multivariate analysis. The results of multivariate analysis revealed that the significant predictors of CR are Activin-A concentration levels at diagnosis with OR 1.006 (CI: 1.002–1.010) (
Cox regression analysis for prediction of non-remission
Cox regression analysis for prediction of non-remission
OR, odds ratio; CI, confidence interval.
Cox regression analysis for prediction of relapse
OR, odds ratio; CI, confidence interval.
Cox regression analysis for prediction of OS
HR, hazard ratio; CI, confidence interval.
The survival analyses of the studied AML patients were evaluated by Kaplan-Meier curves. The analysis revealed that AML patients had high Activin-A levels (Fig. 2a) and those who had low CXCL-12 levels (Fig. 2b) had shorter OS as compared to the remaining group (
Kaplan-Meier plot of the OS probability in the high and low Activin-A groups. High Activin-A plasma concentration is associated with short OS as compared to patients have low Activin-A concentration (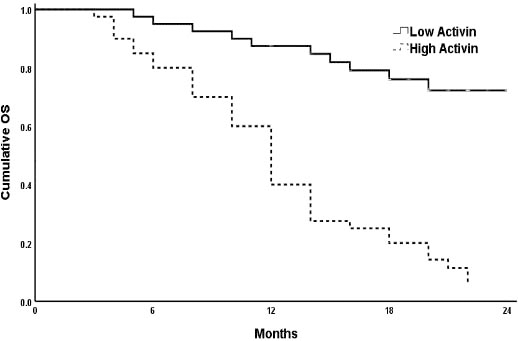
Impact of CXCL-12 concentration levels at AML diagnosis on OS. Low expression is associated with shorter OS (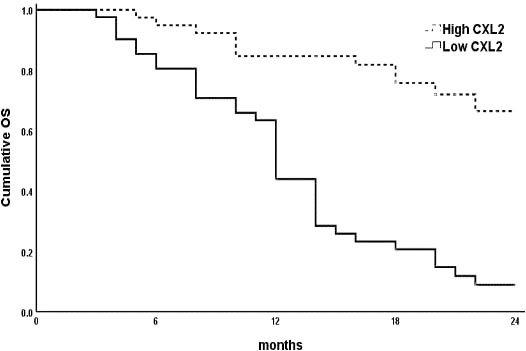
In acute leukemia the malignant cells infiltrating the bone marrow can alter and interfere with the microenvironment and create a malignant niche [12]. It has been documented that Activin-A protein and CXCL-12 have both direct and indirect effect on the malignant microenvironment. Therefore, they have a pro-tumoral role in many type of cancer [13].
In the current study; we found that Activin-A BM concentration was significantly up regulated in AML cases at diagnosis, and down regulated after CR when compared with control group. While at relapse the Activin-A levels increased again to the diagnosis levels. Similar findings were reported in a study that was done on forty four healthy donors and one hundred and eight patients with acute leukemia [6]. The source of elevated Activin-A at AML diagnosis was suggested to be from blast cells or from stromal cells. In this context; Previous study recorded that the bone marrow stromal cells express Activin-A in a constitutive manner, and have role in the regulation of cell migration and invasion in the context of several solid malignancies [14, 15].
Moreover, we found that the Activin-A levels were significantly higher in AML patient’s subgroup that resist induction of remission; relapsed during course, and died, as compared to the remaining patients subgroups. These findings are in accordance with many recent researches reporting that Activin-A increases the migration and invasive criteria of many tumors [14, 16]. In a previous study [6] that analyzing the effect of leukemic cells treatment with Activin-A revealed that this protein was able to significantly enhance cell motility and random movements even at lower concentrations. On the other hand, Activin-A can severely impair the CXCL-12 induced migration of healthy CD34+ cells which may be explained by the ability of Activin-A to increase calcium inside the leukemic cells resulting in higher rate of actin polymerization [6].
In the present study we observed that CXCL-12 concentrations level was significantly down regulated in AML cases at diagnosis when compared to control group. After CR it increased significantly to control levels and in relapsed patients the levels down regulated more than the diagnosis levels. This could be explained on the basis of findings in a previous studies which reported that the CXCL-12 reduction is one of the micro environmental alterations occurring in the leukemic BM, as seen in both leukemic patients and mice models [17] and that CXCL-12 reduction is usually associated with impairment of normal hematopoiesis [13].
In multivariate analysis, only high Activin-A and low CXCL-12 BM plasma levels were considered as independent predictors of non-remission in studied AML cases. This, could be explained on the basis of previous observation that the interference with the CXCR-4/CXCL-12 axis may be an effective way to mobilize blast cells which leading its escape from combination chemotherapy.
In our study we found that CXCL-12 protein levels are lower at AML diagnosis as compared with both non leukemic control and post induction therapy. Moreover, The CXCL-12 levels after induction of remission were similar to the levels seen in non-leukemic patients. Similar findings was reported in a previous AML study [12] and in adult acute lymphoblastic leukemia [19]; this lower serum level could not be explained by consumption of CXCL-12 by the leukemic cells or by an altered production capacity of MSC at the time of diagnosis [12]. Agarwal et al. [18] Assessed the effect of CXCL-12 provided by the microenvironment on the growth and survival of leukemia cells in an vitro study, they found that loss of CXCl-12 expression is dispensable for leukemia development.
Recent studies linked Activin-A with the facilitation of cell invasion and infiltration in several solid cancers like colorectal cancer, prostate cancer, breast cancer, non-small cell lung neoplasm [14, 15, 16, 20]. In addition to its main role in adapting the homing processes in the BM micro-environment, CXCL-12 is thought to be involved in the widespread invasion and infiltration of other organs because of its constitutive expression in extra medullary tissues such as liver, spleen, thymus, lung, kidney, and brain [21]. The study showed that Activin-A treated leukemic cells when in vivo injected they had the ability to reach its target organs and cause extramedullary lesion than the untreated cells. Furthermore, in vitro study showed that Activin-A significantly increased the ability of leukemic cells to pass through an extracellular matrix in response to CXCL-12. These data suggest a possible role of Activin-A and CXCL-12 in the promotion of leukemic cell invasiveness [6].
Conclusion
Activin-A level at diagnosis is a new simple easily assessed biomarker that could predict AML patient’s response to therapy as well as patient’s outcome.
Footnotes
Acknowledgments
The authors of this manuscript acknowledge the patients who participate in this study as well as the technicians in the clinical pathology Department Laboratories, Mansoura University.
Conflict of interest
The authors declare that there is no conflict of interest.
Author contributions
Salah Aref: Conception and supervision.
Lamiaa Ebrahim: Interpretation or analysis of data.
Shaimaa El-Ashwah: Interpretation or analysis of data.
Mohamed El Agdar: Laboratory work.
Mohamed Ayed: Preparation of the manuscript, interpretation or analysis of data, revision.
