Abstract
Accumulating evidence validates that aerobic glycolysis is involved in chemotherapy resistance in human malignant tumors. In the present study, we explored the role of exosome-delivered circular RNA DLGAP4 (circDLGAP4), a novel identified circRNAs, in the chemoresistance of neuroblastoma (NB) cells. Our study demonstrated that doxorubicin-resistant cells expressed higher HK2, accompanied with enhanced glycolysis. In addition, circDLGAP4 was validated to act as a sponge for the HK2-targeting miR-143. As a molecular cargo, exosomes were found to deliver circDLGAP4 from doxorubicin-resistant cells to the sensitive cells. Functionally, exosomal circDLGAP4 enhanced glycolysis and drug resistance via regulating miR-143 and HK2 in NB cells. Consistently, upregulation of HK2 induced by circDLGAP4 or miR-143 inhibitors produced the similar malignant transformation in NB cells. However, knockdown of circDLGA P4 could reversed the drug resistance in the recipient cells. In conclusion, these findings demonstrate that exosome-delivered circDLGAP4 promotes the glycolysis, proliferation, and invasion of sensitive NB cells by regulating miR-143 and HK2, providing a novel link between drug resistance and circDLGAP4/miR-143/HK2 axis in drug-resistant NB.
Introduction
Neuroblastoma (NB) is one of the most common childhood malignancies, ranking the third of cancer-related mortality among children. Although significant advances for NB therapy have been achieved in the past decades, the five-year overall survival in high-risk patients is still less than 50%[1, 2]. Studies have showed that half of those patients did not respond sufficiently to the common therapeutics agents, especially to doxorubicin (DOX) therapy. The mechanism underlying the drug resistance is complex and multifactorial[3, 4]. Therefore, it is urgent to elucidate the mechanism of action that contribute to the chemoresistance, which may be of great significance for treatment of NB.
Aerobic glycolysis, or Warburg effect, refers to the phenomenon in which tumor cells consume large amounts of glucose to produce lactate even in the presence of abundant oxygen[5]. Aerobic glycolysis enables cancer cells abnormally proliferate, invade, and metastases. by providing necessary energy. Accumulating evidence validates that aerobic glycolysis is a critical feature of malignancies, and therapeutic options against key regulators of aerobic glycolysis are promising tool for cancer therapy[6]. Among many players involved in glycolysis, HK2/hexokinase-II plays a predominant role in glucose metabolism[7]. Various studies reveal that HK2 plays an important role not only in glycolysis but also in cancer cell survival. As a matter of fact, HK2 is highly expressed in most cancers[8]. Previously, it has been reported that HK2 expression and its activity levels were notably elevated in NB metastatic patients, suggesting that targeting HK2 may be a novel therapeutic strategy against this cancer[9]. Recently, it has been demonstrated that inhibition of key regulators (e.g., HK2)-mediated aerobic glycolysis is a promising tool against NB progression[10].
Until now, the mechanism remains unknown with regards to the chemoresistant transformation from drug-sensitive cells during tumor progression. Exosomes, extracellular vesicles with 30
In the present study, the expression pattern of HK2 was found to be elevated in chemoresistant NB cells, accompanied with higher glycolysis. Additionally, circDLGAP4 was predicted to modulate miR-143/HK2 axis in doxorubicin-resistant NB cells. Finally, our study demonstrated that exosomes from drug-resistant cells delivered circDLGAP4 to drug-sensitive cells and induced potent glycolysis and drug resistance via miR-143/HK2 axis in NB cells.
Materials and method
Cell culture
NB SH-SY5Y cells, obtained from the cell bank of the Chinese Academy of Sciences (Shanghai, China), were cultured in DMEM supplemented with 10% fetal bovine serum (Invitrogen, Carlsbad, CA, USA). The doxorubicin-resistant SH-SY5Y cell line (SH-SY5Y/Dox) was established via gradual exposure of the parent cells to increasing concentrations of doxorubicin. All cells were cultured in a humidified incubator at 37
CCK-8 assay for chemosensitivity
Cell viability was measured by using a Cell Counting Kit-8 (CCK-8, Dojindo, Japan) according to the manufacturer’ protocol. To be brief, SH-SY5Y or SH-SY5Y/Dox cells at the density of 2
Cell transfection
SH-SY5Y or SH-SY5Y/Dox cells were transfected with the indicated siRNA or recombinant plasmids using Lipofectamine 2000 (Invitrogen, Carlsbad, CA, USA) according to the manufacturer’s instructions. Cells were washed with PBS, and the medium was replaced by complete medium 4–6 h after transfection.
Exosome isolation, staining, and characterization
Exosomes were extracted from SH-SY5Y or SH-SY5Y/Dox cells or serum samples using an ExoQuick precipitation kit (System Biosciences, CA, USA), according to the manufacturer’s instruction. Briefly, the culture medium or serum was centrifuged at 3,000
Glucose uptake and lactate production assay
SH-SY5Y or SH-SY5Y/Dox cells were seeded in 12-well plates at 3
Colony formation assay
Cells in different groups were seeded on 60 mm dish and cultured for 14 days. Subsequently, colonies were stained with crystal violet (0.1%), and the number of surviving colonies was counted under a microscopy.
Transwell assay
The cell invasion potential was determined utilizing transwell inserts (BD Bioscience) pre-coated with Matrigel (BD Bioscience). About 1
RNA immunoprecipitation (RIP) assay
Biotin-labelled miR-143 and control probes were synthesized by Sangon Biotech (Shanghai, China), and transfected into HEK293T cells. After 24 h, cells were harvested, fixed by 1% formaldehyde, and incubated with streptavidin dynabeads (Invitrogen, USA). The precipitated RNA was analyzed using quantitative real time PCR (qRT-PCR) for RNA identification. The experiment was replicated at least three times.
Quantitative real-time PCR (qRT-PCR)
Total RNAs from cells were extracted using Trizol RNA kit (TaKaRa, Osaka, Japan) according to the manufacturer’s protocol. Then, RNA samples were reversely transcribed cDNA, and subject to qRT-PCR analysis utilizing miScript SYBR Green PCR Kit (TaKaRa, Osaka, Japan). The expression levels were indicated genes was determined using the 2-
Bioinformatics analysis
Target prediction of miR-143 was conducted with the algorithms from TargetScanHuman 7.2 (
Western blot analysis
Whole cells were lysed in 1
Statistical analyses
Experimental data were expressed as mean
Expression profile of HK2 and circDLGAP4 in NB cells. (A) Cells were treated with different doses of doxorubicin for 48 h, and cell viability was determined by CCK-8 assay. (B) Western blot analysis of HK2 expression in doxorubicin-sensitive (SH-SY5Y) and resistant SH-SY5Y cells (SH-SY5Y/Dox). (C) The relative glucose uptake and lactate production in SH-SY5Y and SH-SY5Y/Dox cells. (D) Bioinformatic analysis of putative binding regions between HK2 and miR-143. (E) TEM and NTA identification of human serum exosomes. (F) Western blot analysis of markers for exosomes. (G) qRT-PCR analysis of the expression levels of circDLGAP4 and miR-143 within serum exosomes in the serum of doxorubicin-sensitive (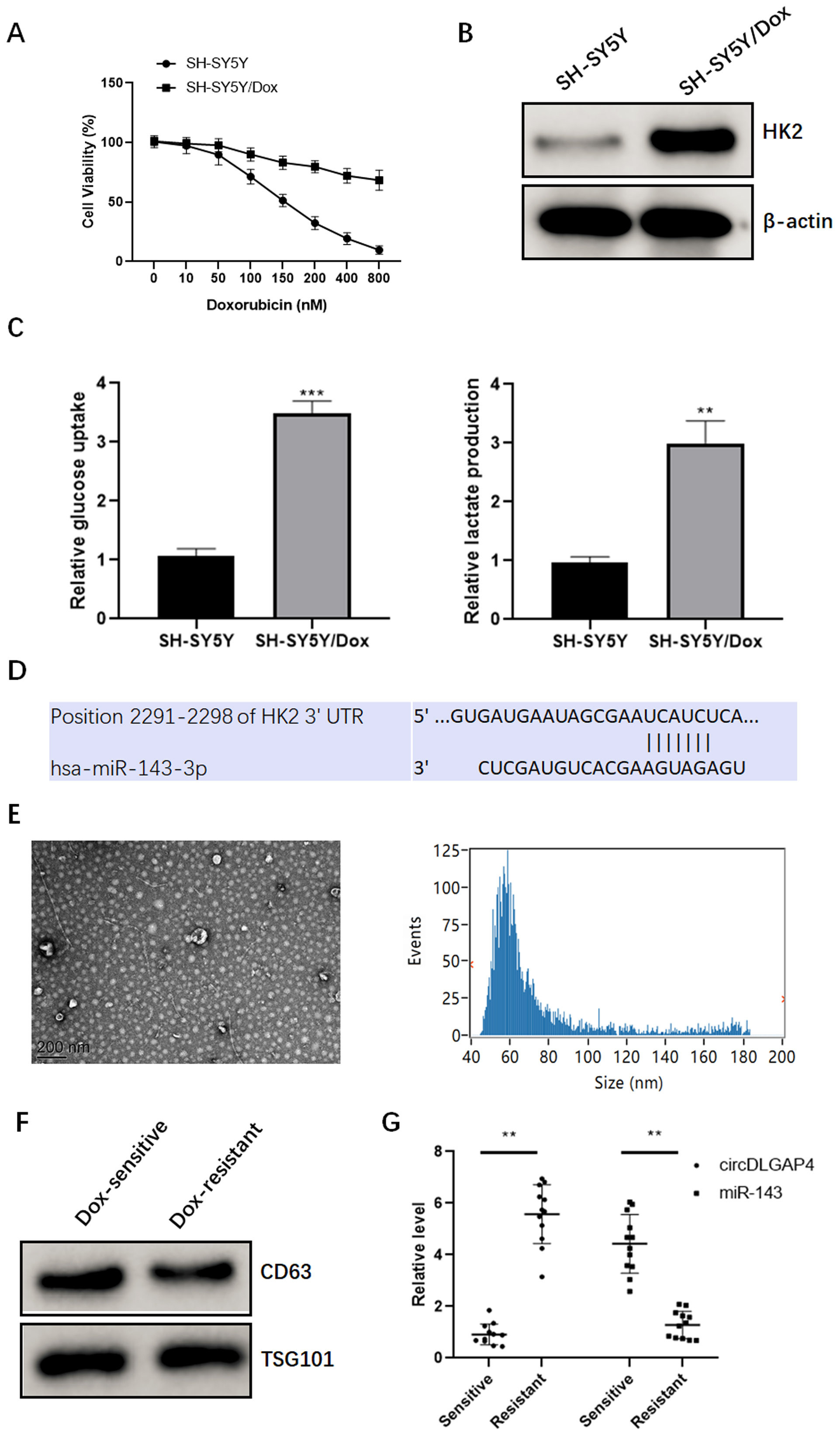
Chemoresistant cell-derived exosomes deliver circDLGAP4 to the sensitive NB cells. (A) TEM and NTA identification of exosomes from SH-SY5Y and SH-SY5Y/Dox cells. (B) Western blot analysis of markers for exosomes from SH-SY5Y and SH-SY5Y/Dox cells. (C) qRT-PCR analysis of the expression levels of circDLGAP4 and miR-143 within exosomes from SH-SY5Y and SH-SY5Y/Dox cells. (D) PKH26-labeled SH-SY5Y/Dox cell exosomes were endocytosed into SH-SY5Y cells. (E and F) qRT-PCR quantification of the expression levels of circDLGAP4 and miR-143 in SH-SY5Y cells treated with SH-SY5Y/Dox derived exosomes with or without circDLGAP4 siRNA. (G) Western blot analysis of the expression levels of HK2 in SH-SY5Y cells treated with SH-SY5Y/Dox derived exosomes with or without circDLGAP4 siRNA. 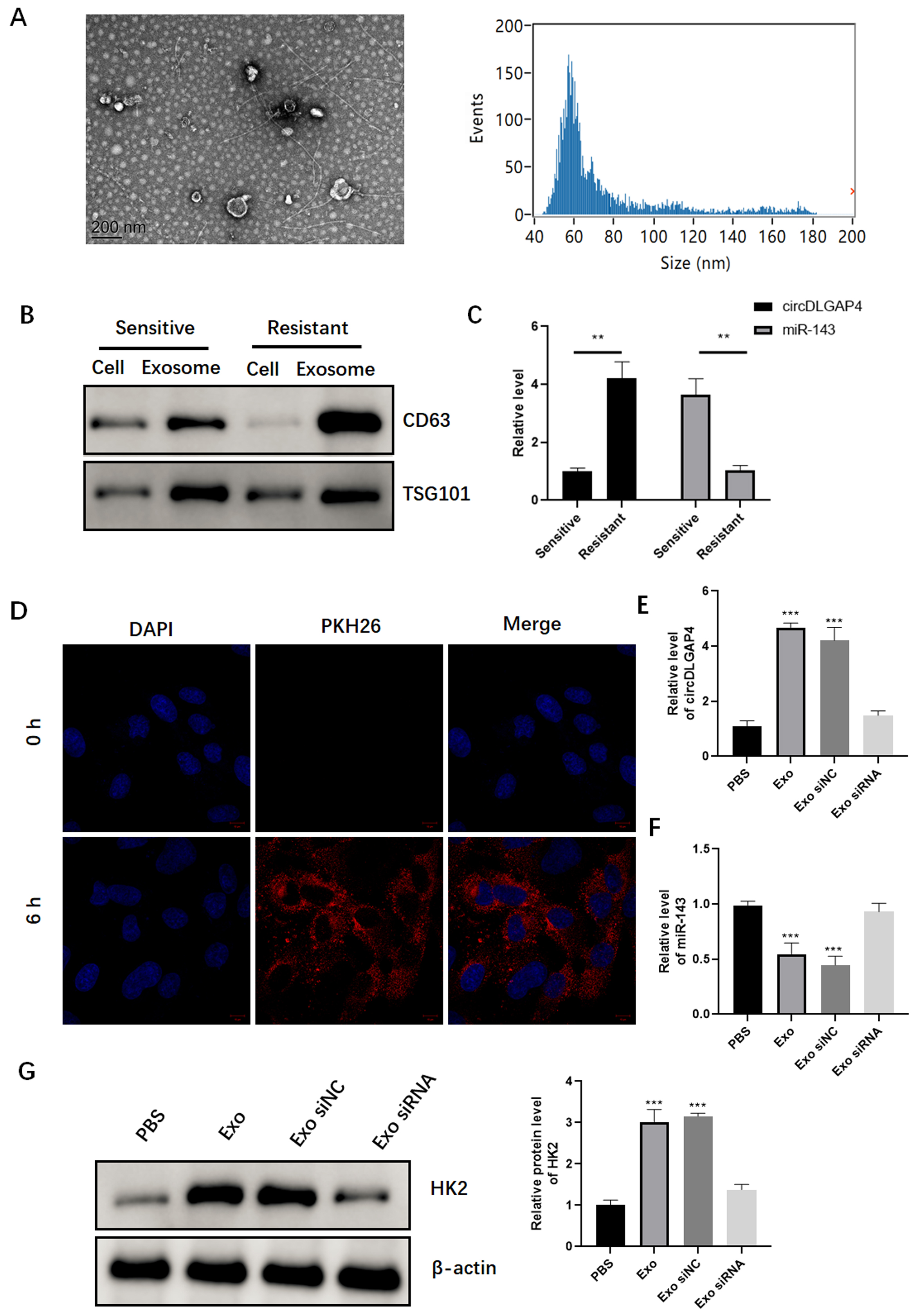
Expression profile of HK2 and circDLGAP4 in NB cells
Firstly, CCK-8 assays showed that the viability of SH-SY5Y cells significantly decreased upon exposure to doxorubicin, whereas doxorubicin-resistant SH-SY5Y cells (SH-SY5Y/Dox) only slightly decreased (Fig. 1A). To explore the role of HK2 in doxorubicin resistance in NB, we firstly evaluated its expression in doxorubicin-resistant SH-SY5Y cells (SH-SY5Y/Dox). Data from western blot assays showed that the expression of HK2 was significantly upregulated in drug-resistant cells compared to that in drug-sensitive cells (Fig. 1B). Interestingly, the relative glucose uptake and lactate production (Fig. 1C) were dramatically robust in doxorubicin-resistant SH-SY5Y cells, representing a stronger glycolysis. Subsequently, bioinformatics analysis revealed the 3’ UTR of human HK2 harbored a putative binding site of miR-143 (Fig. 1D). Previous study reported that circDLGAP4 acts as a molecular sponge for miR-143[14]. Herein, the Starbase online software showed binding sites between circDLGAP4 and miR-143 (
Chemoresistant cell-derived exosomes transports circDLGAP4 to the sensitive cells
Exosomes are capable to transmit intercellular signals and might facilitate the drug-resistant phenotype. We isolated exosomes from SH-SY5Y sensitive and resistant cells to doxorubicin, both with a diameter of about 60 nm (Fig. 2A). The exosome markers CD63 and TSG101 were also detected in doxorubicin-sensitive and -resistant SH-SY5Y cells (Fig. 2B). The expression level of circDLGAP4 within exosomes was notably increased in the resistant cells derived exosomes, whereas miR-143 was obviously decreased in exosomes compared with those from the sensitive cells (Fig. 2C). In order to evaluate the intercellular transportation of exosomes, we stained exosomes from SH-SY5Y/Dox cells with PKH26 and cocultured with SH-SY5Y cells. Immunofluorescence analysis revealed that the stained exosomes from doxorubicin-resistant cells were endocytosed into the recipient cells (Fig. 2D). Consistently, endocytosis of exosomes from resistant cells significant enhanced circDLGAP4 while suppressed miR-143 in the recipient cells, which was reversed by knock down of exosomal circDLGAP4 (Fig. 2E and F). Additionally, the protein level of HK2 was also enhanced in the recipient cells cocultured with exosomes derived from SH-SY5Y/Dox cells (Fig. 2G). Taken together, these data suggest that exosomes could transport circDLGAP4 and regulate miR-143/HK2 axis between the resistant and sensitive NB cells.
Chemoresistant cell-derived exosomes facilitate malignant alterations in the sensitive cells. The relative glucose uptake (A) and lactate production (B) in SH-SY5Y cells treated with SH-SY5Y/Dox derived exosomes with or without circDLGAP4 siRNA. Colony formation (C and D) and invasive (E and F) capabilities of SH-SY5Y cells treated with SH-SY5Y/Dox derived exosomes with or without circDLGAP4 siRNA. 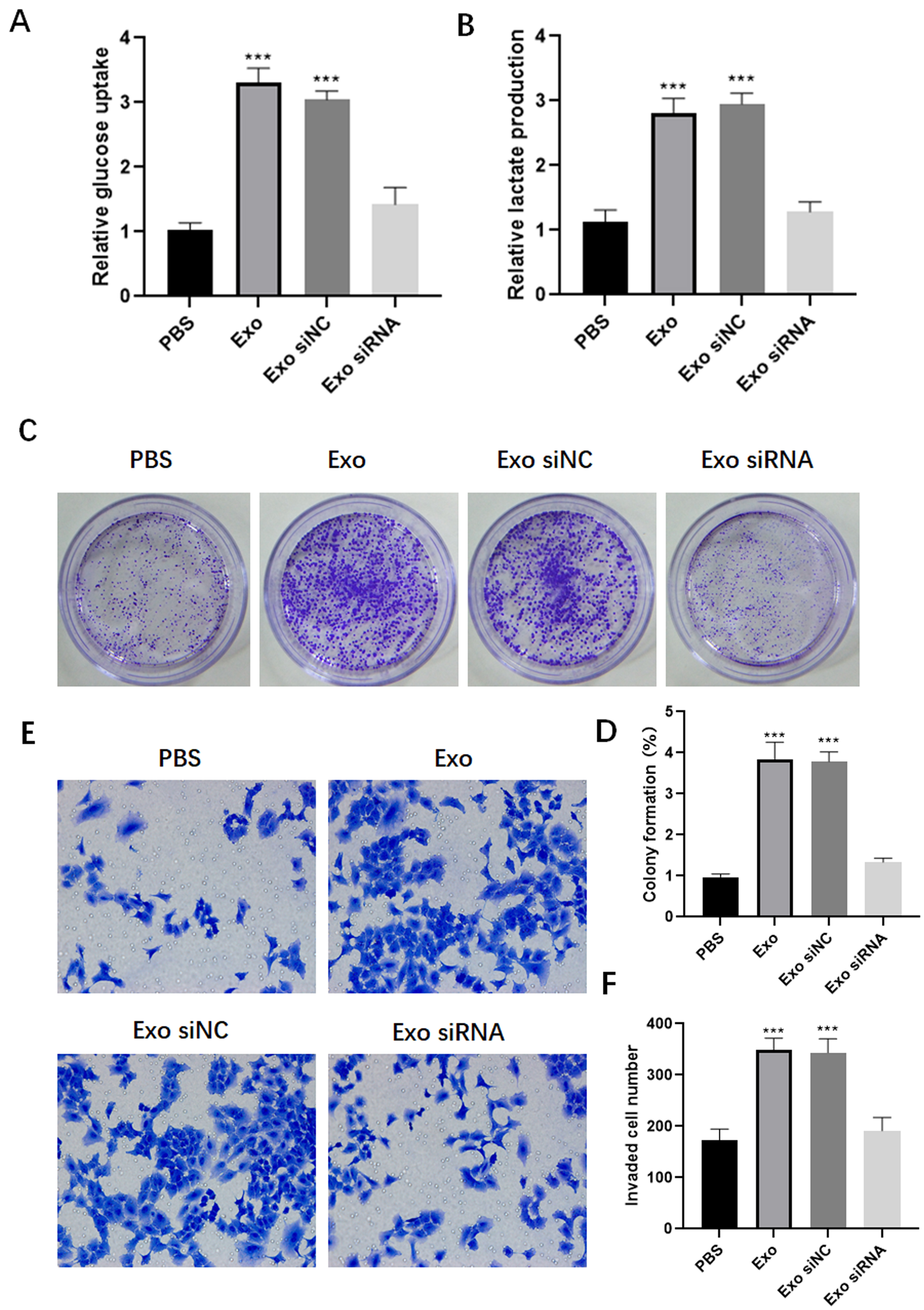
Furthermore, we determined whether exosome-mediated transfer of circRNAs could confer to functional alterations in the recipient cells. After coculture with exosomes from SH-SY5Y/Dox cells, the recipient SH-SY5Y cells exhibited increased glucose uptake and lactate production by doxorubicin (Fig. 3A and B). Additionally, incubation with exosomes from the resistant cells enhanced the colony formation (Fig. 3C and D) and invasion (Fig. 3E and F) capabilities of the recipient cells. Nevertheless, knockdown of exosomal circDLGAP4 abrogated such effects. These findings reveal that exosomes derived from the resistant cells confer to malignant transformation to the recipient NB cells.
circDLGAP4 regulates chemosensitivity of NB cells via miR-143/HK2 axis. (A) qRT-PCR analysis of HK2 mRNA and circDLGAP4 immunoprecipitated with miR-143. Subsequently, SH-SY5Y cells were transfected with circDLGAP4, HK2 overexpressing (OE) plasmids, miR-143 inhibitors, and their controls. (B) qRT-PCR analysis of miR-143 in each group. (C) Western blot analysis of the expression levels of HK2 in each group. Determination of the relative glucose uptake (D) and lactate production (E). Colony formation (F and G) and invasive (H) capabilities in each group. 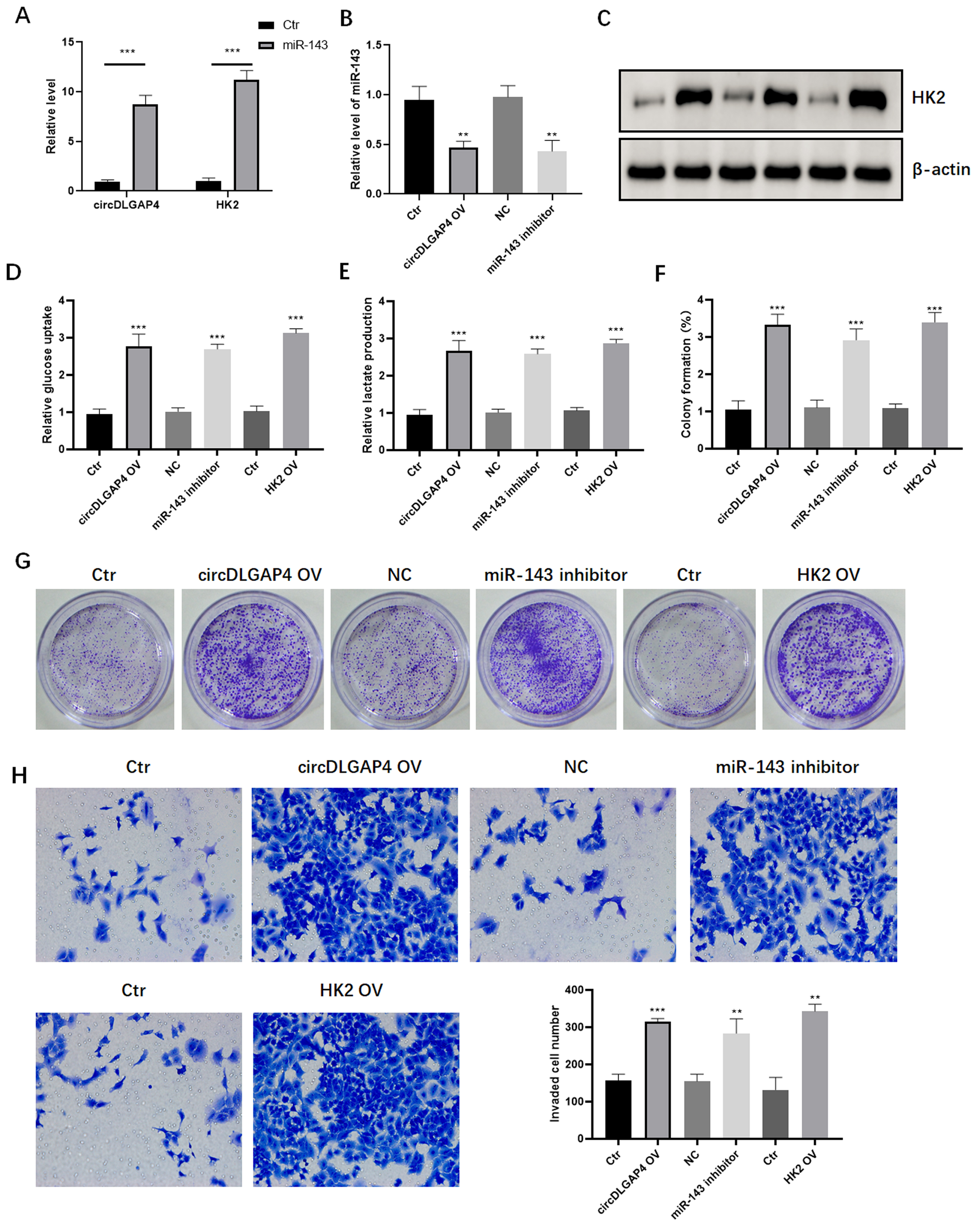
To explore the potential interaction among circDLGAP4, miR-143 and HK2, RNA pulldown using biotin-labeled miR-143 was performed. Consequently, the HK2 mRNA and circDLGAP4 were significant enriched by miR-143 compared with the control group (Fig. 4A). Moreover, circDLGAP4, HK2 overexpressing (OE) plasmids, and miR-143 inhibitors were transfected into the sensitive SH-SY5Y cells. We detected a significant reduction of miR-143 following overexpression of circDLGAP4 (Fig. 4B). Western blot analysis revealed that HK2 was dramatically elevated after transfection with circDLGAP4, HK2 OE plasmids, and miR-143 inhibitors (Fig. 4C). Consistently, upregulation of HK2 enhanced the relative glucose uptake and lactate production (Fig. 4D and E). Moreover, elevated expression of HK2 conferred to the malignant transformation of SH-SY5Y cells, as evidenced by enhanced colony formation (Fig. 4F and G) and invasion (Fig. 4H). Collectively, these findings demonstrates that circDLGAP4 could modulate miR-143/HK2 axis and contribute to chemoresistance in NB cells.
Discussion
Over the past decades, large amounts of studies have focused on the molecular and cellular mechanisms underlying the acquired chemoresistance, one of the main causes of chemotherapeutic treatment failure[2, 3]. For the first time, our study demonstrated that exosomal circDLGAP4 from drug-resistant cells could enhance glycolysis and drug resistance via miR-143/HK2 axis in the recipient cells, representing a potential therapeutic option for treatment of chemoresistant NB.
Malignant tumors usually manipulate aerobic glycolysis to consume glucose and generate lactate. It has been reported that chemoresistant cells exhibit potent glycolysis to maintain malignant phenotypes[16]. In NB cells, treatment with glycolysis inhibitor (3-bromopyruvate) significantly increased the cytotoxicity of doxorubicin, and decreased intracellular ATP and lactate production, thereby overcoming chemotherapy resistance in human neuroblastoma[17]. In the present study, we found that HK2, a key regulator of glucose metabolism, was upregulated in the doxorubicin-resistant cell line SH-SY5Y. Consistently, upregulation of HK2 in drug resistant cells exhibited potent glucose uptake and lactate production, suggesting that accelerated glycolysis may contribute to the chemoresistance in NB cells.
Previously, several circRNAs have been reported to modulate HK2 expression, such as circRNF20, circPRKCI, and circPVT1[18, 19, 20]. Moreover, the bioinformatic analysis and previous reports both showed that HK2 was a direct target of miR-143[20, 21, 22]. In our study, circDLGAP4 was predicted to act as the sponge of miR-143 to regulate HK2 expression. Recent studies have suggested that circDLGAP4 sponges miR-143 and miR-134 to upregulate different downstream genes (e.g., CREB), thereby playing a diverse role in human diseases[23]. However, it remains unknown about the role and its associated mechanism of circDLGAP4 in neuroblastoma. Exosomes are recently discovered carriers for intercellular signal transduction. Our study found that circDLGAP4 within exosomes from chemoresistant NB patients was significantly elevated, whereas miR-143 was downregulated in resistant cases. In addition, PKH26-stained exosomes from SH-SY5Y/Dox cells were endocytosed into the recipient cells, delivering circDLGAP4 and further modulating miR-143/HK2 axis. These finding suggest that exosomal circDLGAP4 from drug-resistant cells may be involved in the acquired drug resistance in NB cells via miR-143 and HK2.
Unraveling the mechanisms responsible for the acquired chemoresistance could provide experimental targets to developing novel strategies to increase chemosensitivity. Several studies have demonstrated that circDLGAP4 is involved in myocardial ischemia-reperfusion injury by sponging miR-143[24, 25]. Recently, circDLGAP4 has been found to promotes diabetic kidney disease by sponging miR-143 and targeting ERBB3/NF-
In conclusion, the present study demonstrated that exosomes derived from drug-resistant NB cells delivered circDLGAP4 to drug-sensitive cells, thereby increasing glycolysis and drug resistance by suppressing miR-143 and promoting HK2 expression. These findings demonstrated that targeting circDLGAP4 may be a novel treatment approach to reverse chemoresistance in NB.
Disclosure statement
No potential conflict of interest was reported by the authors.
Author contributions
Conception: Wei-qiang Tan and Li Yuan.
Interpretation or analysis of data: Xiao-yan Wu.
Preparation of the manuscript: Cheng-guang He.
Revision for important intellectual content: Shai-cheng Zhu.
Supervision: Ming Ye.
Data availability statement
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
