Abstract
BACKGROUND:
The incidence of lung cancer in Yunnan area ranks firstly in the world and underlying molecular mechanisms of lung cancer in Yunnan region are still unclear. We screened a novel potential oncogene CYP2S1 used mRNA microassay and bioinformation database. The function of CYP2S1 in lung cancer has not been reported.
OBJECTIVE:
To investigate the functions of CYP2S1 in lung cancer.
METHODS:
Immunohistochemistry and Real-time PCR were used to verify the expression of CYP2S1. Colony formation and Transwell assays were used to determine cell proliferation, invasion and migration. Xenograft assays were used to detected cell growth in vivo.
RESULTS:
CYP2S1 is significantly up-regulated in lung cancer tissues and cells. Knockdown CYP2S1 in lung cancer cells resulted in decrease cell proliferation, invasion and migration in vitro. Animal experiments showed downregulation of CYP2S1 inhibited lung cancer cell growth in vivo. GSEA analysis suggested that CYP2S1 played functions by regulating E2F targets and G2M checkpoint pathway which involved in cell cycle. Kaplan-Meier analysis indicated that patients with high CYP2S1 had markedly shorter event overall survival (OS) time.
CONCLUSIONS:
Our data demonstrate that CYP2S1 exerts tumor suppressor function in lung cancer. The high expression of CYP2S1 is an unfavorable prognostic marker for patient survival.
Introduction
Lung cancer is one of the most leading cause of cancer death in both males and females worldwide [1]. The incidence of lung cancer in Yunnan ranks firstly in the world [2]. Although a great achievement has been made in the treatment of lung cancer in the past decades in Yunnan regions, the 5-year overall survival rate statistics underling successful surgery hover around 20%. Because most patients were already in the advanced stage when they were diagnosed with lung cancer, and missed the best opportunity for surgery and chemotherapy [3, 4, 5, 6]. The latest research suggests that inhalation of mineral dust containing arsenic and radon progeny is the main cause of lung cancer in Yunnan region, but the underlying molecular mechanism is still to be elucidated [7]. Thus, there is an imperative need to identify the driver gene that causes specific mutations in lung cancer patients in Yunnan area, in order to realize the early detection and prevention of lung cancer, and to find a new target for the treatment of NSCLC.
To explore the molecular mechanism of lung cancer in Yunnan region, a comparative study of absenteeism spectrum differentially expressed in lung cancer and adjacent normal tissues was performed using Affymetrix U133 plus 2.0 array mRNA microassay. We found that CYP2S1 is the most differentially expressed in cancer and para-cancer tissues. The Kaplan-merier plot analyze revealed that high expression of CYP2S1 correlated with poor prognosis in lung cancer patients, which suggested that it plays a vital role in lung cancer progression in Yunnan area.
CYP2S1 encodes a member of the cytochrome P450 superfamily of enzymes and locates at 19q13.2 and is mainly expressed in the endoplasmic reticulum [8]. The CYP2S1 expression in different tissues has been investigated in previous studies, which was mainly expressed in the trachea, lung, stomach, small intestine, and spleen, and with low expression in the liver tissues [9]. The biology function of CYP2S1 in previous studies was described as involved in endogenous metabolism as well as in the detoxification of xenobiotics, including environmental toxins and small molecule drugs [10]. In colorectal cancer, the depletion of CYP2S1 can enhance colorectal cell proliferation and mediate 2-(3,4-Dimethoxyphenyl)-5-Fluorobenzothiazole (GW-610, NSC 721648) sensitivity in colorectal cancer and breast cells [11]. But the function of CYP2S1 in lung cancer has not been reported.
In this study, we characterized a novel oncogene CYP2S1 in lung cancer. We found CYP2S1 is up-regulated in lung cancer tissues when compared with normal cancer tissues, and the high expression of CYP2S1 has an adverse effect on patient overall survival. Down-regulated of CYP2S1 expression in lung cancer cells could markedly suppress the ability of cells proliferation and invasion in vitro, and in vivo, knockdown of CYP2S1 significantly inhibited lung cancer cells growth. GSEA analysis showed that the gene affected by the CYP2S1 down-regulation was enriched in E2F target pathway that involving in cell cycle progression, which interpreted the reason of CYP2S1 knockdown suppressed lung cancer cells proliferation and invasion. These results revealed that CYP2S1 played an important role in the lung cancer progression.
Materials and methods
Tissue samples collected and cells culture
A total of 52 fresh primary lung adenocarcinoma tissues and 46 normal lung tissues were obtained from the Third Affiliated Hospital of Kunming Medical University (Kunming, China). Clinical protocols were approved by the Ethics Committees of the Third Affiliated Hospital of Kunming Medical University, and patients provided informed consent. All patients underwent surgery plus lymph node dissection. Patients with relapsed disease or those who have received radiation, chemotherapy or preoperative biotherapy were excluded from this study to avoid any changes in tumor marker determination due to the effect of the treatment. Patients diagnosed with multiple primary cancers in other organs or tissues were also excluded.
In the present study, the A549 cells were obtained from the American Type Culture Collection (Manassas, VA, USA), A549 cells were maintained in Dulbecco’s modified Eagle’s medium (DMEM; Gibco; Thermo Fisher Scientific, Inc., Waltham, MA, USA) supplemented with 10% fetal bovine serum (FBS; Shanghai ExCell Biology, Inc., Shanghai, China) 2 mM L-glutamine (21,051, Invitrogen, USA), 100 U/mL penicillin (P3032, Sigma-Aldrich, USA), and 100 mg/mL streptomycin (WB11000, Sigma-Aldrich, USA). All cells were incubated in a humidified chamber with 5% CO
Immunohistochemistry
The tissues were fixed in 10% formalin within 48 h at room temperature and embedded in paraffin. Paraffin sections (4
Construction of lentiviral shRNA vector and cell infection
Lentiviral short-hairpin RNA (shRNA) constructs targeting CYP2S1 were purchased from TranSheepBio (Shanghai, China). Lentiviral particles carrying full-length hsa-CYP2S1 vector and their flanking control (NC) were constructed. Subsequently, A549 cells were infected with lentiviral vector as described in our previous study. The transient transfection of plasmid was performed as described previously.
Cell migration and invasion assays
The present study used transwell assay to detect cell migration, 1
Cell proliferation assays
Cell numbers were calculated using the hemocytometer, and a total of 300 transfected cells were seeded in a six-well plate and maintained in medium with volume of 1 mL containing10% FBS. This medium in plate was replaced every 3 days. After14 days, absorb and discard the supernatant culture medium, cells were fixed with 4% paraformaldehyde and stained using a 0.1% crystal violet solution. Visible stained colonies were counted and used to evaluate cell proliferation.
Xenograft experiments
A total of 24 BALB/c nude mice were purchased from Experimental Animal Center of Kunming Medical University and divided into two groups and were housed in pathogen-free cages (
GEO and TIMER data analysis
In the present study, we used GSE19188 to analyse the CYP2S1 expression between tumor tissues and normal tissues, and the data was released from GEO Database (
Gene set enrichment analysis (GSEA)
The RNA-seq enrichment analysis was performed with random combination number of 1000 and false discovery rate (FDR)
The expression of CYP2S1 from lung cancer tissues in Yunnan
The expression of CYP2S1 from lung cancer tissues in Yunnan
Relative CYP2S1 expression in lung cancer tissues and its clinical significance. a. CYP2S1 expression in lung cancer tissues and para-cancer tissues analyzed using GEO database (GSE19188), CYP2S1 expression was up-regulated in lung cancer tissues. b. CYP2S1 expression was significance higher in lung cancer tissues compared with para-cancer tissues analyzed using TCGA database. c. The Real-time PCR results showed that CYP2S1 expression was significantly increased in lung cancer cells than that in normal lung cells. d. Positive expression of CYP2S1 (up) and negative expression of CYP2S1(down) in lung cancer tissues. e. Positive expression of CYP2S1 (up) and negative expression of CYP2S1 (down) in para-cancer tissues. f. Patients with high levels of CYP2S1 expression showed reduced survival times compared with patients with low levels of CYP2S1 expression (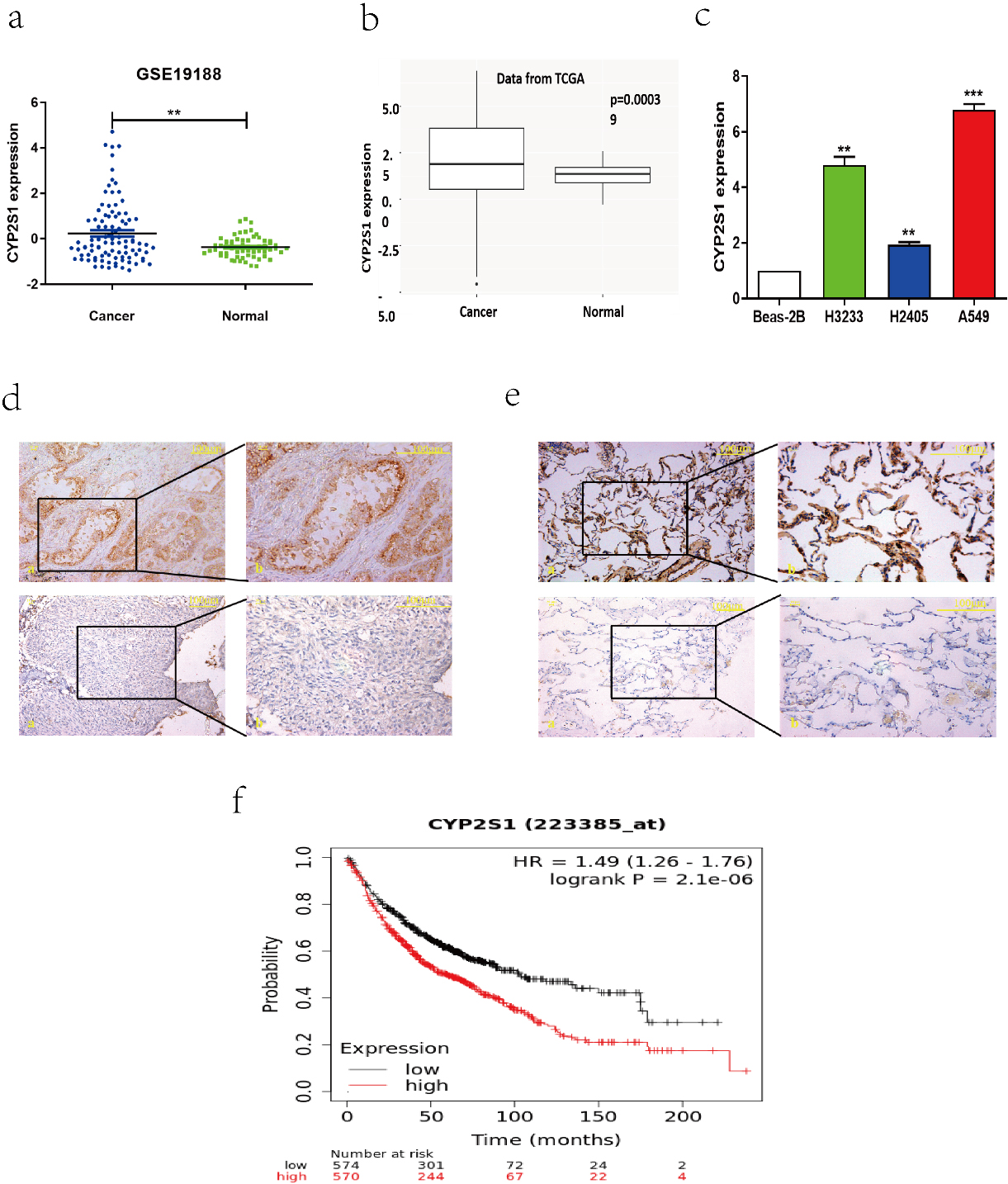
CYP2S1 inhibits lung cancer cells growth and metastasis in vitro. a. CYP2S1 knockdown significantly inhibited cell growth in A549 cells detected by cell counting assay. b. Knockdown of CYP2S1 in A549 cells reduced cell migration in vitro by Transwell assay. Student’s 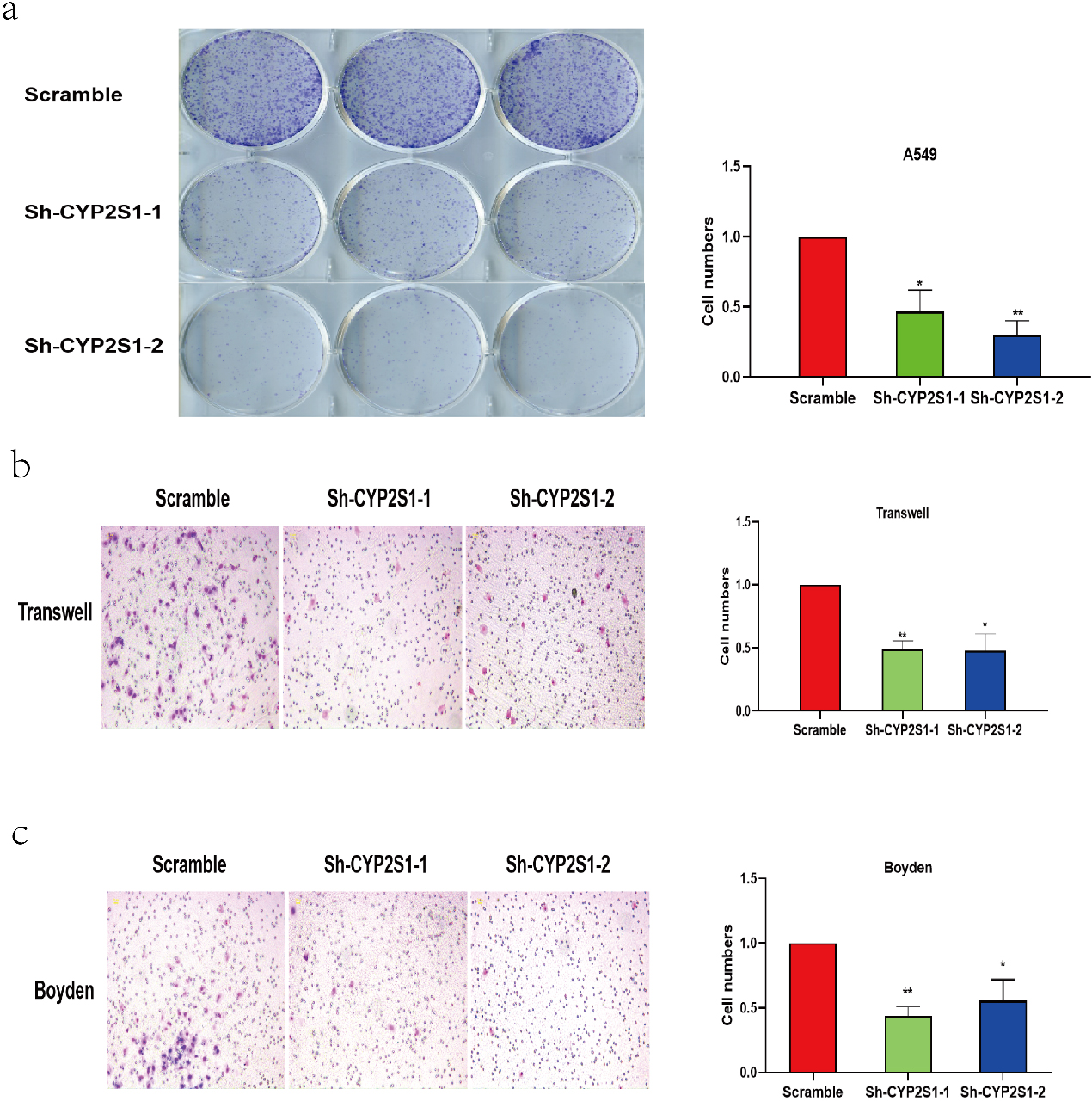
Effect of CYP2S1 knockdown on tumor growth in nude mouse xenografts. a and b. Scramble or shCYP2S1 was stably transfected into A549 cells, which were injected in the nude mice, respectively. Tumor volumes were calculated after injection every 7 days. Bars indicate S.D. c. Tumor weights are represented as means of tumor weights 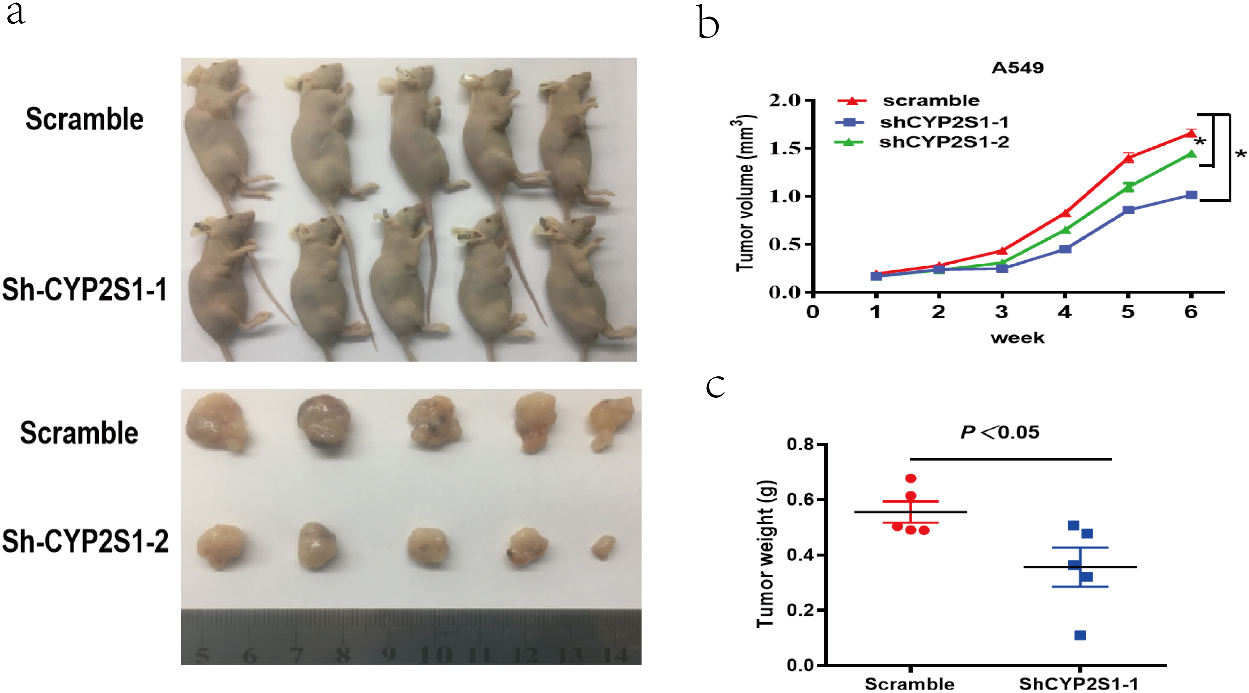
Analysis of gene enrichment affected by CYP2S1 expression. a. hierarchical clustering of gene transcripts expression after the knockdown of CYP2S1 in A549 cells, with three repeats. b and c. GSEA results showed that Knockdown of CYP2S1 affected genes were enriched in E2F targets pathway and G2M checkpoints pathway.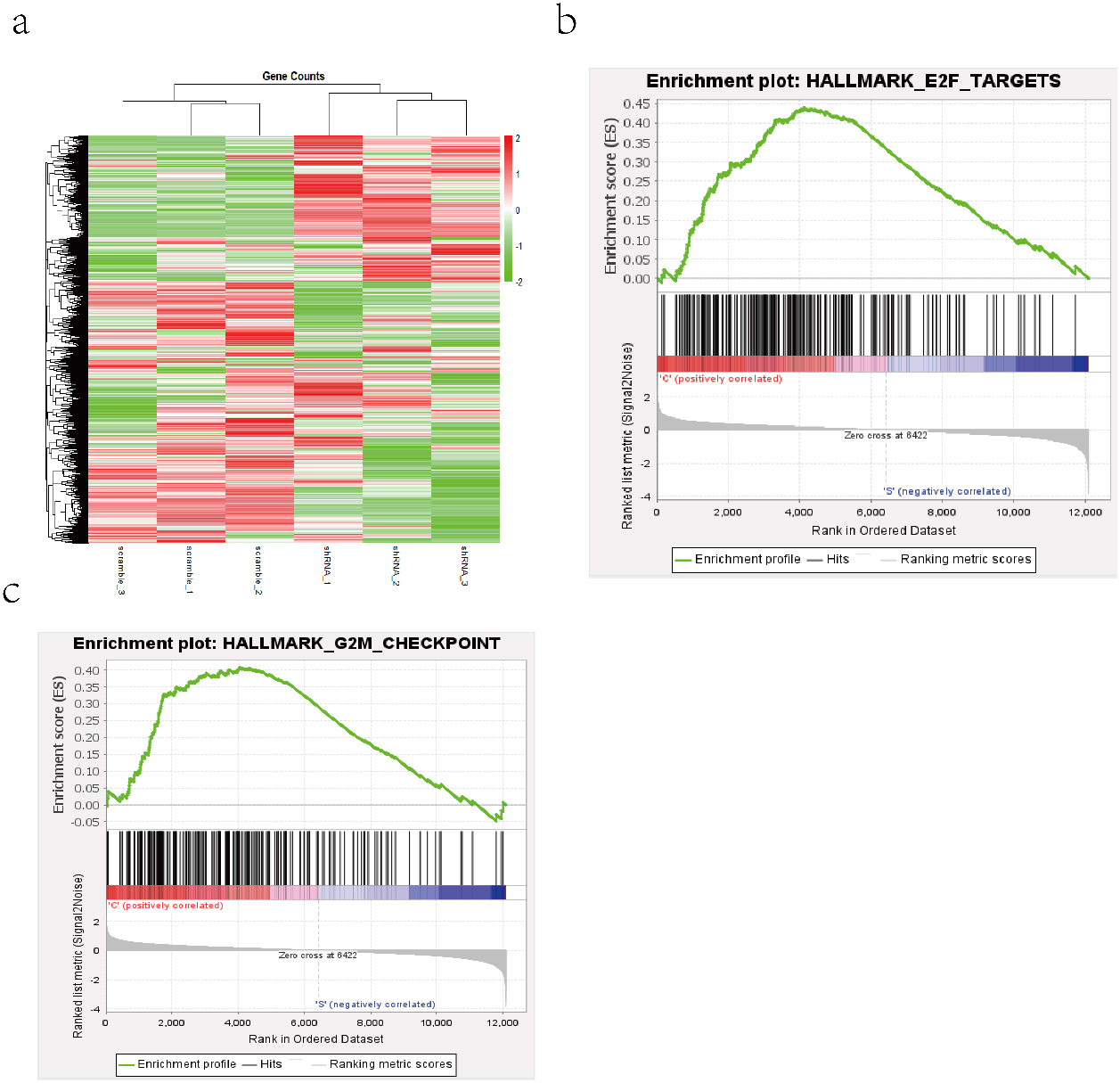
CYP2S1 is up-regulated in NSCLC tissues and cells
To explore the role of CYP2S1 in lung cancer, we first assessed the expression of CYP2S1 in lung cancer tissues and normal lung tissues using GEO and TCGA data base. The results showed that CYP2S1 mRNA expression was significantly up-regulated in lung cancer tissues when compared with normal lung tissues (Fig. 1a and b). We also examined the expression of CYP2S1 in lung cancer cells by Real-time PCR, our results showed that CYP2S1 was up-regulated in lung cancer cells (Fig. 1c). Next, we used immunohistochemistry assay to detect the CYP2S1 protein expression from 52 lung cancer tissues and 46 normal lung tissues in Yunnan region (Fig. 1d and e). We found that the proportion of positive expression of CYP2S1 in lung cancer tissues was up to 84.6%, while the positive proportion of CYP2S1 protein expression in normal lung tissues was just only 50%, and the difference was statistically significant (Table 1). The Kaplan-Meier survival analysis revealed that lung cancer patients with high expression of CYP2S1 exhibited negative correlation with overall survival times (Fig. 1f). The above results suggested that CYP2S1 could contribute for lung cancer progress.
CYP2S1 regulated lung cancer cells proliferation and invasion
To confirmed the function of CYP2S1in lung cancer, the A549 lung cancer cells were infected with lentivirus carrying CYP2S1-shRNAs and negative control shRNA for 48–72 hours, respectively. After confirming the knockdown effect of CYP2S1, colony formation assays were conducted. The results showed that knockdown of CYP2S1 in A549 cell lines significantly inhibited the ability of cells growth when compared with that of control cells (Fig. 2a). Furthermore, Transwell assays demonstrated that knockdown of CYP2S1 markedly suppressed cell migration and invasion compared with the control cells (Fig. 2b and c).
CYP2S1 inhibited A549 cell proliferation in vivo
To further determine whether CYP2S1 has an effect on lung cancer cell proliferation in vivo, a nude mice xenograft assay was performed. The A549 cells stably transfected with sh-CYP2S1 or control vector were inoculated into nude mice. The data showed that matrigel substrate was completely absorbed 3 days after injection, and palpable tumors were detectable on day 7 after subcutaneous inoculation of the cells. The tumors formed in the sh-CYP2S1 group were markedly smaller than those in the control group during the whole period (Fig. 3a and b). Furthermore, the mean weight of tumor at the end of experiment was significantly lower in the sh-CYP2S1 group when compared with control group (Fig. 3c). These findings indicated that knockdown of CYP2S1 in A549 cells inhibited tumor growth in vivo.
CYP2S1 is involved in the cell cycle progression
As the above results showed that down-regulated of CYP2S1 expression can significantly inhibit A549 cells proliferation and invasion in vivo and in vitro, however, the mechanism is still unclear. We performed RNA-seq assay and used gene set enrichment analyses to analyse the microarray data. The GSEA output results data indicated that the gene sets was enriched in E2F targets and G2M checkpoint signaling pathway in the high expression of CYP2S1 group. The previous research indicated that the E2F family and G2M checkpoint signaling pathway plays a crucial role in the control of cell cycle. These results suggested that CYP2S1 influenced the ability of cell growth and invasion by participating in cell cycle progression.
Discussion
In this study, we first identified the roles of CYP2S1 in lung cancer progression. Our results presented that the expression of CYP2S1 in lung cancer tissues was significantly up-regulated than that in normal lung tissues from our clinical lung cancer samples and GEO as well as TCGA database. The overall survival analysis indicated that the patients with high expression levels of CYP2S1 have a poor prognosis. In vitro, CYP2S1 could markedly suppress lung cancer cells proliferation and invasion, and in vivo experiments, knockdown of CYP2S1 significantly inhibit lung cancer cells growth. The underlying mechanism was released from GSEA enrichment analysis, the output data of microassay was enriched in E2F targets pathway and G2M checkpoint pathway which participated in cell cycle regulation.
CYP2S1,an extrahepatic cytochrome P450 (P450), shows marked individuality in constitutive and inducible expression, for example, CYP2S1 mRNA expression is increased by treatments for psoriasis, including retinoids and UV radiation, although endogenous substrates remain poorly characterized [9]. The previous studies have been reported that the levels of CYP2S1 mRNA were highest in human trachea, small intestine, stomach, spleen, lung and colon, the expression levels of CYP2S1 mRNA were low in kidney, liver, thymus, placenta and peripheral leukocytes, and were undetectable in human heart, brain and skeletal muscle [12, 13]. The protein expression levels of CYP2S1 in different tumor tissues and normal tissues has also been demonstrated, in ovarian cancer, the CYP2S1 expression was found to be similar in normal ovary and primary ovarian cancer samples, but was increased in metastatic ovarian cancer samples [14]. The CYP2S1 expression in colon cancer samples was higher than the normal colon samples, and the high expression of CYP2S1 was associated with poor prognosis [15]. In this study, we used immunohistochemistry to detect the expression of CYP2S1 in lung cancer tissues and adjacent tissues, our results revealed that the percent of positive samples of CYP2S1 expression in lung cancer tissues group is up to 84.6%, only 50% in the adjacent tissues group, and the difference was statistically significant. The CYP2S1 mRNA expression analysis results released from GEO data base were also according with our immunohistochemistry data, CYP2S1 mRNA expression was significantly up-regulated in lung cancer tissues than that in normal tissues in GSE19188. The kaplan Meier-plot suggested that the high expression of CYP2S1 has an association with poor prognosis in lung cancer. This suggested that CYP2S1 played a vital role in lung cancer progression.
To explore the role of CYP2S1 in lung cancer, we knocked down the CYP2S1 expression in A549 lung cancer cells, the results showed that CYP2S1 significantly inhibited the ability of A549 cell proliferation and invasion in vivo and vitro. This suggested that CYP2S1 played an oncogene role in lung cancer. To determine the underlying mechanism, we first knocked down the CYP2S1 expression in A549 cells and performed an RNA-seq microassay. Next, cluster enrichment analysis was carried out by GSEA, the output data results indicated that the gene sets were enriched in E2F targets signaling and G2M checkpoint pathway in the high expression of CYP2S1 group when compared with down-regulation group. The E2F family plays a crucial role in the control of cell cycle, and the dysregulation of cell cycle progress contributes to cancer progression [16, 17, 18]. The G2M checkpoint is essential for the maintenance of the cell cycle and is the last checkpoint to prevent cells with DNA damage from entering the mitosis phase [19]. Abnormalities in this checkpoint will promote cell cycle abnormalities, and promote tumorigenesis and malignant progression [20, 21]. This suggested that CYP2S1 may be involved in cell cycle progress to inhibit tumor cells proliferation and invasion. The GSEA results are according with our experiments results, but whether CYP2S1 plays its function through influencing cell cycle by E2F target gene pathway and G2M checkpoint pathway still needs to be further verified by experiments.
In summary, we systematically explored the function of CYP2S1 in lung cancer, our experiments and clinical sample analysis show that CYP2S1 is a potential target for lung cancer treatment.
Author contributions
Huan Guo, Baozhen Zeng were responsible for the conception. Liqiong Wang, Chunlei Ge participated in the interpretation or analysis of data. Xianglin Zuo were responsible for preparation of the manuscript, and participated in drafting and revision of the manuscript. Yan Li Wanbao Ding, Lei Deng, Jie Zhang, Xiaoling Qian, Xin Song, Peixian Zhang was responsible for supervision of the manuscript.
Funding
This work was supported by the Applied Basic Research Foundation of Yunnan Province Science and Technology Department& Kunming Medical University (2018FE001(-273)).
Ethics approval and consent to participate
The tissues were approved by the Ethics Committees of the Third Affiliated Hospital of Kunming Medical University. Informed consent was obtained from patients.
Competing interests
No competing interests.
Footnotes
Acknowledgments
Not applicable.
