Abstract
BACKGROUND:
CA125 level normalization at different chemotherapy cycles has been reported to be a prognosticator in advanced epithelial ovarian cancer.
OBJECTIVE:
In the present study, we investigated whether the time (in days) to CA125 normalization or nadir during treatment could be used as markers to predict survival.
METHODS:
Patients with FIGO stage III–IV epithelial ovarian cancer treated with cytoreductive surgery followed by adjuvant chemotherapy between 2008 and 2016 were enrolled in this retrospective study. Clinicopathological characteristics, changes in CA125 level during treatment, and survival outcomes were analyzed. Time-dependent receiver operating characteristic curve analysis was used to determine the optimal cut-off values of the time to normalization and time to nadir of CA125 levels to predict survival. Univariate and multivariate Cox regression analysis were used to examine the impact of each variable on survival.
RESULTS:
A total of 106 patients were included in the analysis. The optimal cut-off values for the time to normalization and nadir for predicting survival were 60 and 194 days, respectively. In Kaplan-Meier survival analysis, CA125 level normalization
CONCLUSIONS:
Normalization of CA125 level within 60 days after cytoreductive surgery followed by adjuvant chemotherapy in patients with advanced ovarian epithelial cancer could be used as a marker to predict survival.
Keywords
Introduction
The serum level of cancer antigen 125 (CA125) [1] is a widely used biomarker for the diagnosis [2], evaluation of therapeutic response [3, 4, 5, 6, 7, 8], and detection of the recurrence of epithelial ovarian cancer (EOC) [9, 10, 11]. All histologic types of EOC, including the most common serous cell subtype, mucinous tumors, and clear cell tumors are associated with elevated CA125 levels before treatment [12, 13]. Around 75% of patients diagnosed with EOC have advanced stage disease (International Federation of Gynecology and Obstetrics [FIGO] IIb-IV) with a 5-year survival rate of around 29% [2, 14]. The standard treatment approach for advanced stage EOC includes cytoreductive surgery (CRS) and postoperative adjuvant chemotherapy [15] or neoadjuvant chemotherapy (NACT) followed by interval CRS [16, 17, 18].
Previous studies have extensively explored the use of CA125 level in predicting survival outcomes in patients with advanced EOC, including single values (e.g., pretreatment, time to nadir, or time to normalization) [19, 20, 21] and kinetic parameters such as percentage decrease and half-life of CA125 during chemotherapy [5, 6]. Most of these studies have reported that normalization of CA125 level at the first, second, or third chemotherapy cycle is a good prognosticator. However, previous studies have shown a wide range of timing of chemotherapy initiation from as fast as 1 week to a delay of more than 6 weeks after CRS [22]. Therefore, CA125 normalization at the third cycle may represent a wide range of times depending on the study. No previous study has investigated the best cut-off day of the time (in days) required for CA125 reaching normal or nadir to predict survival in advanced EOC. Furthermore, differences in the statistical methods used and inconsistent results in the previous studies highlight the need for an accurate and reliable marker to predict the prognosis of EOC before and during treatment [4, 11, 23, 24]. Therefore, the aim of this study was to determine whether the time to CA125 level normalization (
Materials and methods
Study population
In this retrospective study, we retrieved the medical records of patients with FIGO stage III–IV EOC treated with primary CRS followed by adjuvant chemotherapy between January 2008 and December 2016 at Kaohsiung Chang Gung Memorial Hospital. The inclusion criteria were: 1. all histological types of EOC; 2. advanced ovarian cancer disease defined as FIGO stage III–IV; 3. patients who underwent six or more cycles of platinum-based adjuvant chemotherapy after primary CRS; 4. patients who had serial CA125 values before and during every cycle of adjuvant chemotherapy; and 5. patients who underwent clinical and imaging studies during follow-up or when being evaluated for recurrence. The exclusion criteria were: 1. patients with a normal CA125 level (
Data collection and definition
The following clinical data were retrieved from the medical records of our hospital: age of the patient, pathologic characteristics of the tumor (histologic type and grade), FIGO stage at primary diagnosis, preoperative level of CA125, and CA125 levels after the first, second, third, and sixth cycle of adjuvant chemotherapy. A normal CA125 level was defined as
Data of the primary surgery were based on operative records documented by the surgeon. A residual tumor
The treatment date was defined as the date of undergoing primary CRS. Preoperative serum CA125 values were measured within 2 weeks before the treatment date, and usually 1 day before the CRS. The time to normalization/nadir of CA125 level (in days) was calculated from the treatment date until the date that a normal/nadir CA125 level was documented. During adjuvant chemotherapy, CA125 levels were measured prior to each chemotherapy cycle, which was around 1–3 days before starting chemotherapy. The date of last follow-up was defined as the last medical record of a patient visit to either the gynecologic clinic or any other medical department in our hospital.
To evaluate disease-related outcomes, disease progression was defined as newly developed lesions confirmed by imaging studies following the detection of elevated CA125 levels. PFS and OS were presented in months, and calculated from the treatment date to the date of disease progression or death due to any cause, respectively.
Flowchart describing the study population selection.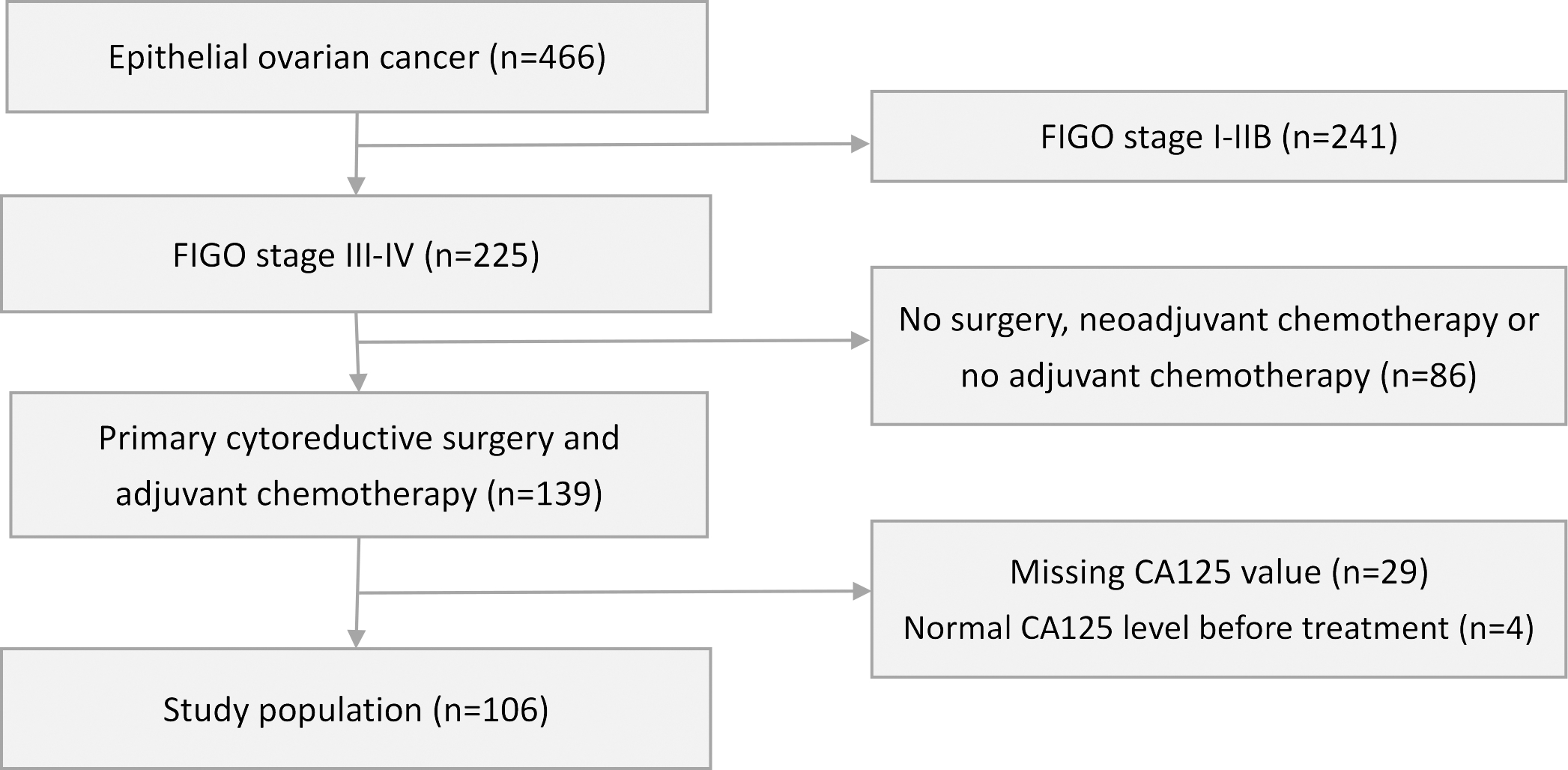
Univariate and multivariate Cox regression analyses were used to investigate the effects of each clinicopathological factor and CA125-related factor on survival. The CA125-related variables (preoperative CA125 level, CA125 levels after the first, second, third and sixth chemotherapy cycles, time to normalization, and time to nadir) were determined after completing a literatures review. The cut-off CA125 values we used after the first (35 u/mL), second (35 u/mL), third (35 u/mL), and sixth (10 u/mL) chemotherapy cycles were based on the results of earlier studies which showed statistical significance in terms of PFS and OS [3, 6, 7, 9, 25, 26, 27, 28, 29, 30]. We used time-dependent receiver operating characteristic (ROC) curve analysis to identify the optimal cut-off value of preoperative CA125 level, and the optimal cut-off value (in days) for the time to CA125 normalization and time to nadir of CA125 in predicting survival. Univariate Cox regression analysis was performed first with each clinicopathological factor and CA125-related factor, and statistically significant variables were identified. The variables reaching statistical significance were then entered into multivariate Cox proportional hazards analysis to identify the most significant independent prognostic factors for PFS and OS. Actuarial rates of survival were estimated using the Kaplan-Meier method, and statistical differences between groups were examined using the log rank test. All statistical analyses were performed using MedCalc and SPSS software (SPSS Inc., Chicago, IL, USA). All
Results
Patient characteristics
A total of 106 patients met the inclusion criteria and were included in this retrospective study. The process of patient selection is shown in Fig. 1. The patients’ characteristics and CA125-related variables are presented in Table 1. The median age of the patients was 53 years (range 37–81 years). Most patients (65%) had FIGO stage IIIC disease at primary diagnosis, and the most common tumor histology was the serous type (61.3%), followed by the endometrioid type (15.0%). Fifty-three (50%) patients had grade 3 tumors. Eighty-five (80.2%) patients achieved optimal debulking after primary CRS, and the other 21 patients (19.8%) had residual tumors
Characteristics of the study population
Characteristics of the study population
CTx, chemotherapy; CA125, cancer antigen 125; CRS, cytoreductive surgery; FIGO, International Federation of Gynecology and Obstetrics.
We used time-dependent ROC curve analysis to define the optimal cut-off values of preoperative CA125 level and time to nadir and time to normalization of CA125 level in predicting survival. The optimal cut-off values and areas under the curve (AUCs) were as follows: pre-operative CA125 level: 430 u/mL (AUC: 0.521); time to nadir: 194 days (AUC: 0.556); time to normalization: 60 days (AUC: 0.737) (Figure-supplement). The mean preoperative CA125 level was 1875 u/mL, and 45 (42.5%) patients had a preoperative CA125 level
Impact of CA125-related variables on survival
Univariate Cox regression analysis of clinicopathological and CA125-related factors were done, and each variate was divided into two subgroups to compare its impact on survival (Table 2). The results revealed that a CA125 level
Univariate Cox regression analysis of clinicopathological characteristics, and CA125-related prognostic factors for progression-free survival (PFS) and overall survival (OS)
Univariate Cox regression analysis of clinicopathological characteristics, and CA125-related prognostic factors for progression-free survival (PFS) and overall survival (OS)
CI, confidence interval; HR, hazard ratio; CTx, chemotherapy; CA125, cancer antigen 125; OS, overall survival; PFS, progression-free survival; CI, confidence interval; HR, hazard ratio.
In Kaplan-Meier survival analysis, a time to CA125 level normalization
Kaplan-Meier survival curves for progression-free survival and overall survival according to CA125 level after the third cycle of chemotherapy (2A), after the sixth cycle of chemotherapy (2B), and normalization within 60 days (2C).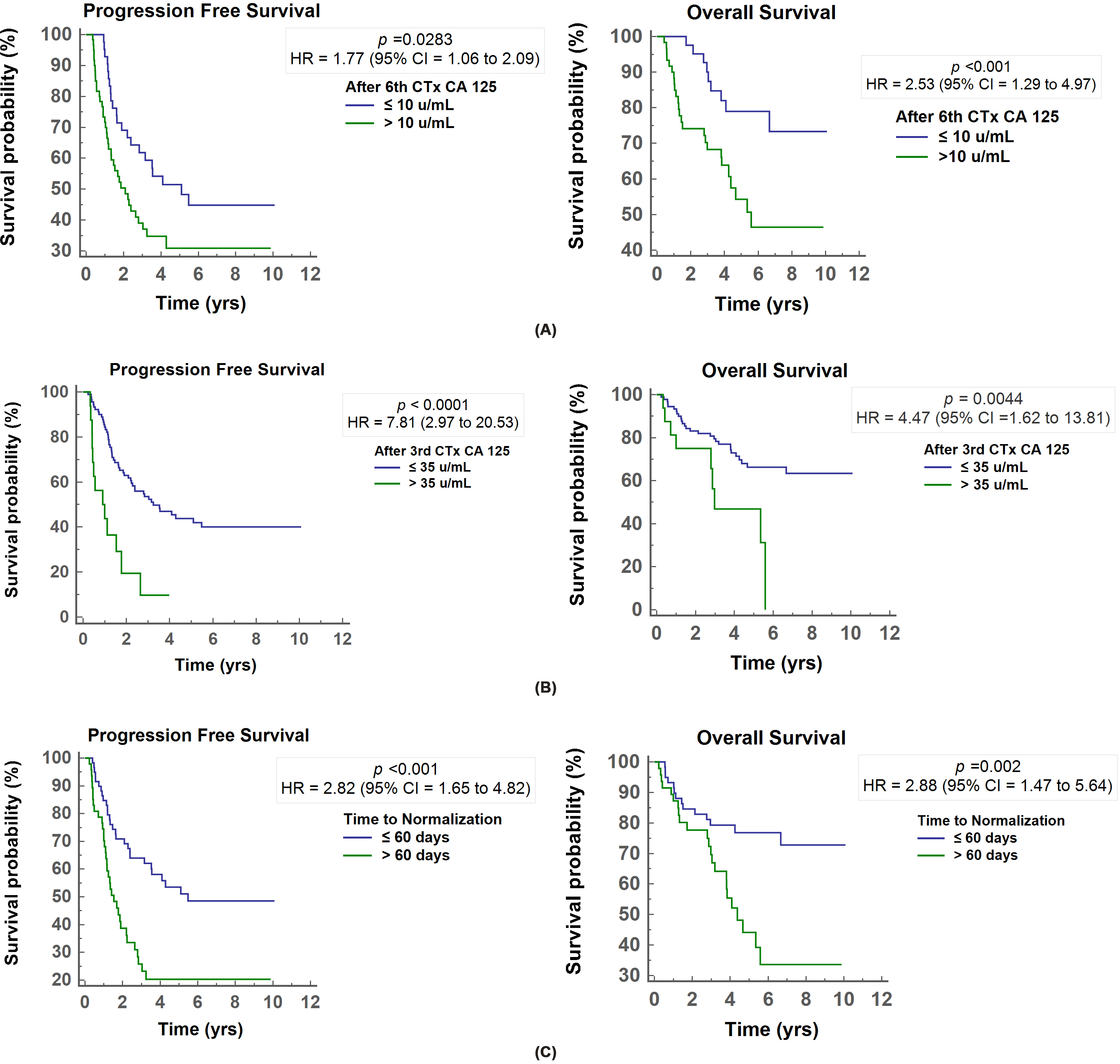
To determine the most significant prognostic factors for OS and PFS, we performed multivariate Cox proportional hazards analysis with statistically significant variables in the univariate analysis and factors which have been reported to have clinical importance on survival outcomes in previous studies [13, 24, 25, 27, 29, 31], including CA125 values after the first, second, third and sixth cycle of chemotherapy, time to normalization (days), tumor grade and residual tumor size after debulking. The results of multivariate analysis showed that only CA125 normalization time
Multivariate Cox regression analysis (backward method) of prognostic factors for progression-free survival (PFS) and overall survival (OS). Multivariate association was adjusted by CA125 after the first, second, third, sixth cycle of chemotherapy, time to normalization (days), tumor grade, and residual tumor size
OS, overall survival; PFS, progression-free survival; CI, confidence interval; HR, hazard ratio; CTx, chemotherapy; CA125, cancer antigen 125.
In present study, we demonstrated that a time to normalization of CA125 level within 60 days was the strongest and only prognostic factor for better PFS and OS in patients with advanced EOC treated with primary CRS followed by adjuvant chemotherapy. To the best of our knowledge, only one similar study has been published, however the authors enrolled patients with all FIGO stages for analysis, including 52.6% with stage I–II. The authors reported that CA125 normalization within 1 month was significantly associated with survival, however a major concern is that they did not include stage or residual tumor status, both of which are considered to be universally important prognostic factors, in the final multivariate Cox regression analysis [25].
CA125 level normalization by the third cycle of chemotherapy [24, 26, 27] and CA125 level
Based on earlier studies, although CA125 normalization patterns could provide prognostic information to identify patients at risk of platinum resistance and poor prognosis, the exact timing of CA125 measurements was not clearly described. Some reports have demonstrated that CA125 normalization by the third cycle of chemotherapy is a good prognosticator [24, 27], whereas other studies have reported after the third cycle [25]. Given that the timing of subsequent chemotherapy may be postponed due to poor compliance, unfavorable performance status, or treatment toxicity, the actual time interval between every adjuvant chemotherapy cycle may vary in different patients. These factors could lead to a large discrepancy in the time between CA125 measurement and the actual administration of chemotherapy in each cycle, which has not been well discussed in previous studies. The major difference between our study and the previous studies mentioned above is that we calculated the time to CA125 normalization in days, which is an easier and more reliable way to determine the prognosis in real-world clinical practice. When we analyzed all of the CA125-related variables together, we found that CA125 normalization within 60 days was better than normalization by chemotherapy cycle in predicting survival.
Our study has several limitations. First, the retrospective nature of our study design and potential patient selection bias may have impacted the results. In addition, patients who received NACT followed by interval debulking surgery were excluded from our study, and this group of patients may have more extensive disease. We only included patients who received primary CRS, and most of them presented with FIGO stage III (88.7%) at diagnosis. These patients were considered to be eligible to receive primary CRS, had a higher likelihood of achieving optimal debulking surgery, and may have benefited from surgery based on previous prospective randomized trials showing that patients with FIGO stage IIIC with metastatic lesions
Conclusion
In conclusion, this is the first study to measure the time to CA125 normalization in days rather than cycle number of adjuvant chemotherapy. CA125 normalization within 60 days can be considered as a checkpoint to evaluate survival outcomes in patients with advanced EOC and an elevated pretreatment CA125 level receiving primary CRS followed by adjuvant chemotherapy. If a patient is shown to have a poor prognosis based on this 60-day checkpoint, a higher potential of platinum-resistant disease should be considered, and early adjustments of the therapeutic plan, chemotherapy regimen, and follow-up interval may be necessary to improve outcomes. This concept can help physicians to provide more precise counseling to patients regarding their risk of persistent disease, recurrence, and the possible benefits of changing chemotherapy regimen and further maintenance therapy. Further prospective studies are required to optimize clinically significant cut-off values for the time to normalization of CA125 level for both OS and PFS endpoints.
Author contributions
Conception: L.H.
Interpretation or analysis of data: L.Y.C., L.H., F.H.C.
Preparation of the manuscript: L.Y.C.
Revision for important intellectual content: L.H., O.Y.C., W.C.H., F.H.C., T.C.C.
Supervision: L.H.
Supplementary data
The supplementary files are available to download from http://dx.doi.org/10.3233/CBM-210156.
Footnotes
Acknowledgments
We thank the Biostatistics Center of Kaohsiung Chang Gung Memorial Hospital for assistance with the statistical analysis.
