Abstract
BACKGROUND:
Epithelial ovarian cancer is a highly lethal gynecological malignancy. Accurate and cost-effective predictive tools to estimate the prognosis of patients with epithelial ovarian cancer before treatment are currently lacking.
OBJECTIVE:
The purpose of this study was to evaluate the prognostic significance of pretreatment serum levels of carcinoembryonic antigen (CEA) and carbohydrate antigen-125 (CA-125) in primary epithelial ovarian cancer.
METHODS:
Between 2008 and 2016, 326 patients with a diagnosis of primary epithelial ovarian cancer were retrospectively reviewed. We attempted to identify an optimal cut-off value of CEA to predict survival using ROC curve analysis. Cox regression univariate and multivariate analyses were used to evaluate prognostic factors.
RESULTS:
The optimal cut-off value of CEA was 2.6 ng/mL. In univariate and multivariate analyses, FIGO stage and pretreatment CA-125 and CEA levels significantly predicted progression-free and overall survival. The 5-year progression-free survival rate for patients with both a CEA level
CONCLUSIONS:
In addition to traditional prognostic factors, a pretreatment serum CEA level
Introduction
Epithelial ovarian cancer is a common gynecological malignancy in developed countries, and is one of the leading causes of death from female malignancy due to difficulty in making an early diagnosis, a high recurrence rate, and because it has usually spread beyond the ovaries at the time of diagnosis [1]. Past studies have reported that changes in perioperative serum carbohydrate antigen-125 (CA-125) level are strongly associated with relapse in patients with advanced ovarian cancer who received optimal debulking surgery [2]. Currently, accurate and cost-effective predictive tools for the prognosis of epithelial ovarian cancer before any treatment are lacking, which may lead to a delay in diagnosis and worse outcomes.
The carcinoembryonic antigen (CEA) family is a superfamily of glycoproteins with a carboxyl terminal providing linkage to cell membranes [3]. Most CEAs in healthy people are located in colon and play a role in cell adhesion, signal transduction and regulation [4]. It is thought that tumor cells produce this protein more and altered in posttranscriptional regulation [5]. Previous studies have shown that the level of CEA can be used to assess the prognosis and monitor recurrence in patients with colorectal cancer, and also that it can predict the response to surgical and chemotherapy treatment [6, 7]. Moreover, pretreatment CEA level has been shown to be an independent factor for prognosis and survival in breast cancer, colorectal cancer, gastric cancer, and non-small cell lung cancer [8, 9].
Nevertheless, few studies have investigated the prognostic significance of CEA level in patients with epithelial ovarian cancer, and the results have been controversial. In one meta-analysis, Guo and colleagues [10] reported that the combination of serum CA-125, CA-199 and CEA had a relatively high sensitivity and specificity for the diagnosis of epithelial ovarian cancer. In 2015, Sagi-Dain and colleagues found that a higher CEA level could be a promising tool to differentiate metastatic tumors from primary ovarian malignancy, and in the diagnosis of a mucinous histology [11]. In 2018, Lin and colleagues [12] investigated the prognostic significance of preoperative serum CEA level in small sample of patients with a primary mucinous histology of ovarian cancer, and they found that elevated preoperative serum CEA level may indicate a poorer prognosis.
To date, powerful evidence about the prognostic significance of pretreatment serum CEA level in patients with epithelial ovarian cancer is lacking. Therefore, in this study, we aimed to evaluate the prognostic value of pretreatment levels of CEA and CA-125 in patients with primary epithelial ovarian cancer.
Material and methods
Study population
This retrospective cohort study included patients with primary epithelial ovarian cancer treated at Kaohsiung Chang Gung Memorial Hospital between 2008 and 2016. All patients had a histopathologically confirmed diagnosis of epithelial ovarian cancer and pretreatment measurements of serum CEA and CA-125. Data acquired from medical records included age, FIGO (International Federation of Gynecology and Obstetrics) stage, histologic type, tumor size, and surgical debulking status. Optimal debulking status was defined as a residual tumor
All patients were followed up every 3 months in the first 2 years after completing primary treatment, and the interval was extended to every 3–6 months in the following 3 years and then to every 6–12 months thereafter. Details of clinical symptoms and pelvic examinations were recorded, and serial chest X-rays and computed tomography scans (if necessary) were arranged. Serum CEA level was analyzed on ADVIA Centaur XP with principle method of chemiluminescence immunoassay (CLIA). Architect CA-125 II assay, a chemiluminescent microparticle immunoassay (CMIA) was used for quantitative determination of serum CA-125. Both tumor markers were measured during every follow-up visit.
Statistical analysis
All statistical analyses were performed using IBM SPSS software (version 20). Receiver operating characteristic (ROC) curves and areas under the ROC curves (AUCs) were used to analyze CEA. We attempted to identify an optimal cut-off value of CEA to predicting survival by determining the point with a maximal Youden index value on the ROC curve. The cut-off value for an elevated CA-125 level (
Clinical characteristics of the 326 patients with primary epithelial ovarian cancer
Clinical characteristics of the 326 patients with primary epithelial ovarian cancer
Clinical characteristics of the patients with respect to preoperative CEA level (ng/mL)
A total of 562 primary ovarian cancer patients were treated during the study period, of whom 201 with no pretreatment CEA or CA-125 data and another 35 with a histological diagnosis other than primary epithelial ovarian cancer were excluded. The remaining 326 patients were included and analyzed, and the median follow-up period was 42.5 months (range, 1–132 months). The clinicopathological characteristics of the patients including age, early (I and II) and advanced (III and IV) FIGO stage, histologic type (serous, mucinous, clear cell, endometrioid or others), tumor size (
In univariate analysis, age, FIGO stage, histology type, surgical debulking status, pretreatment CA-125 and CEA level were significantly associated with PFS and CSS respectively. Tumor size was significantly associated with PFS (
Univariate and multivariate analysis of factors related to PFS and CSS
Univariate and multivariate analysis of factors related to PFS and CSS
Progression-free survival (PFS) curves of epithelial ovarian cancer patients related to pretreatment CEA (ng/mL) and CA-125 (U/mL) levels.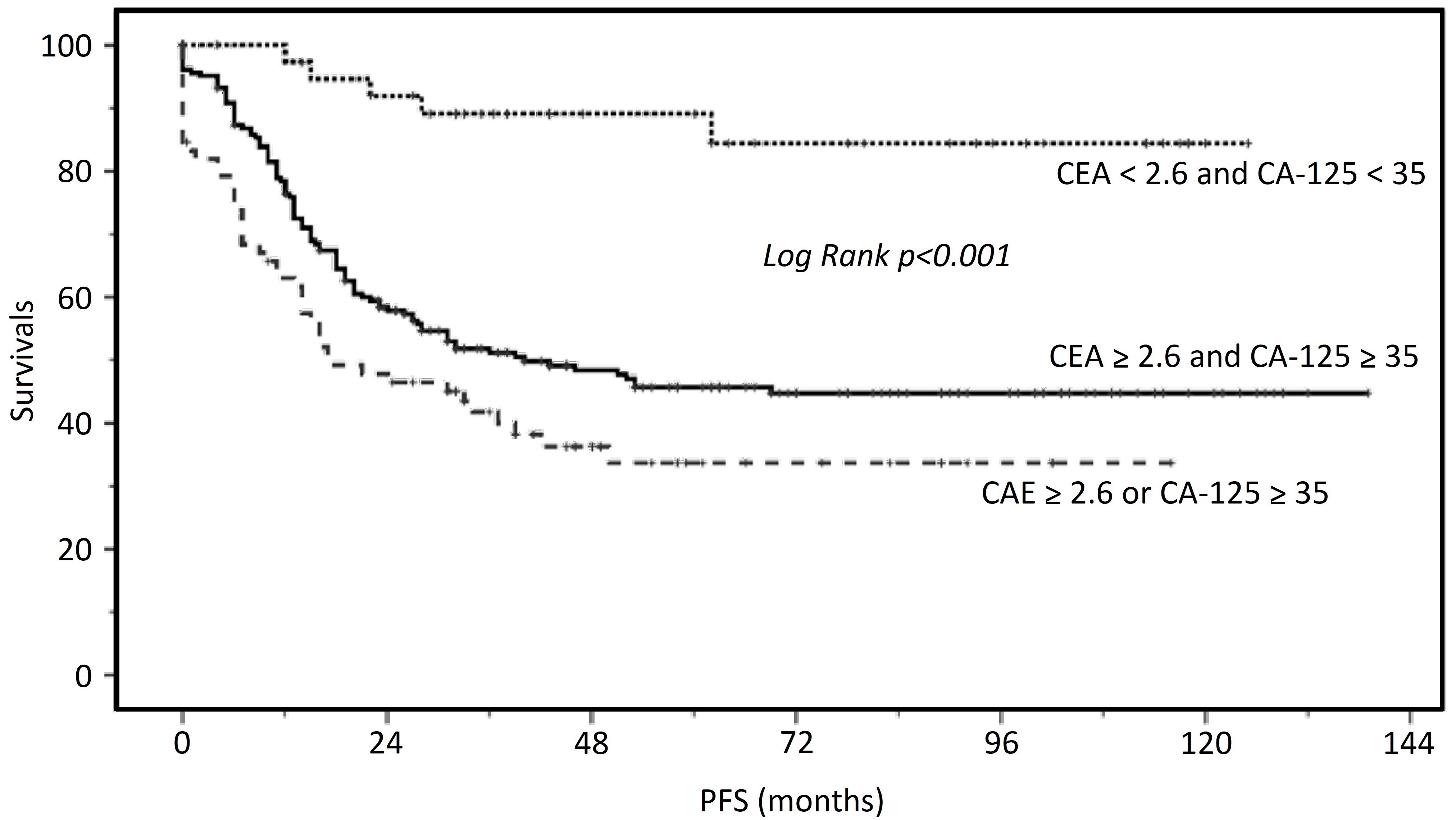
Cancer specific survival (CSS) curves of epithelial ovarian cancer patients related to pretreatment CEA (ng/mL) and CA-125 (U/mL) levels.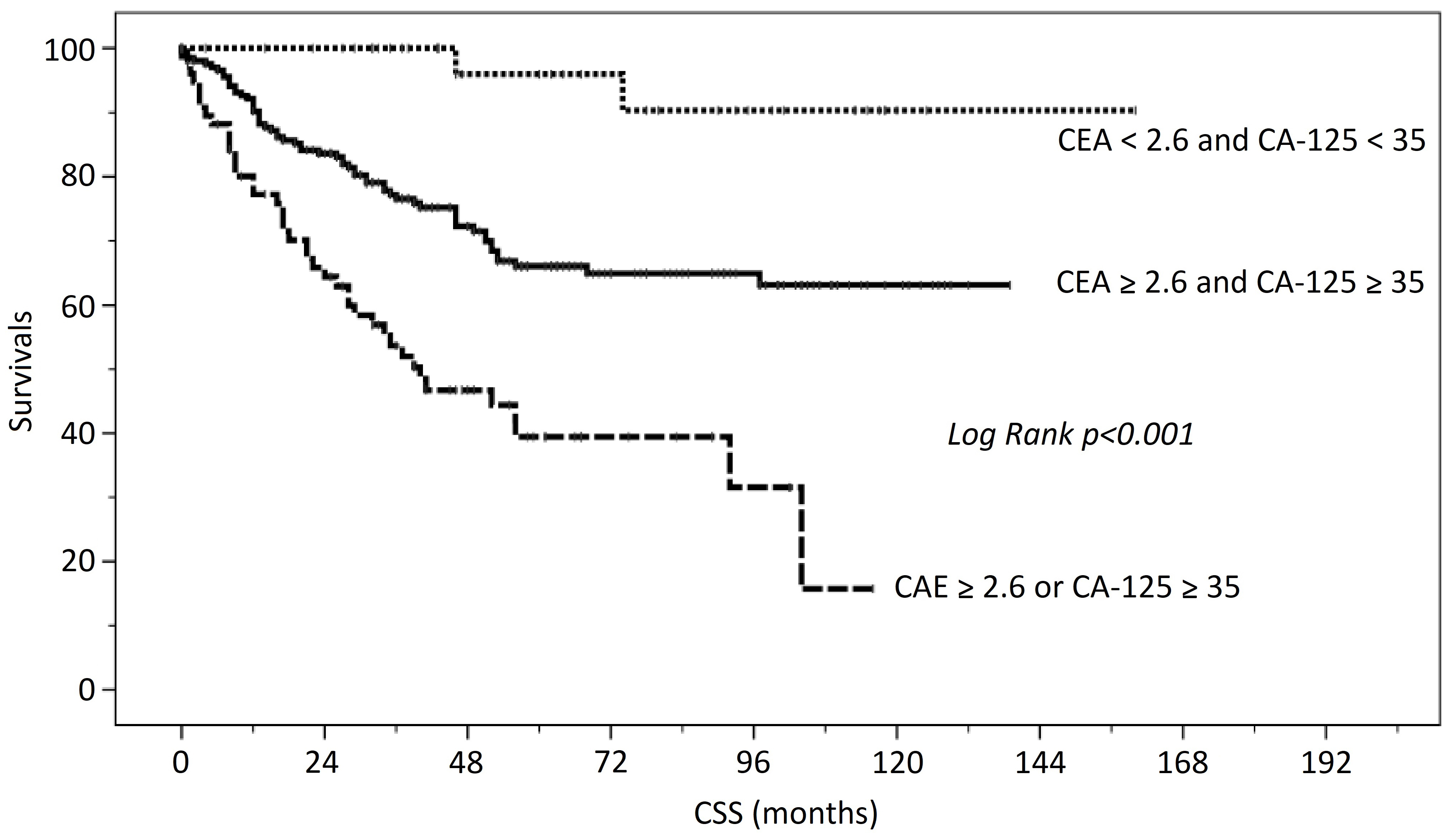
As shown in Fig. 1, the PFS was correlated to pretreatment CEA and CA-125 levels. The 5-year PFS rate for patients with both a CEA level
Prior to this study, there was lack of powerful evidence about the prognostic significance of pretreatment serum CEA level in patients with epithelial ovarian cancer. Our data showed that a pretreatment serum CEA value
The prognostic value of CEA has been investigated in some studies, however the results have been controversial. In 2017, Nomelini et al. reported that the CEA level was significantly higher in patients with non-serous neoplasm, but that it was not statistically a prognostic factor for ovarian cancer [16]. A recent study from Beijing demonstrated the prognostic significance of preoperative serum CEA level in patients with primary mucinous ovarian carcinoma in univariate analysis but not in multivariate analysis [12]. In other previous small group studies, serum CEA level has been reported to be higher in patients with the mucinous type of ovarian cancer, and then to characteristically return to a normal level within 8 weeks after optimal debulking surgery. In these studies, the disappearance or a persistently low level of serum CEA during treatment course was associated with a good prognosis [17, 18, 19]. In addition to the serum level of CEA, the Danish ‘MALOVA’ ovarian cancer study found that the protein expression level of CEA was an independent prognostic factor for epithelial ovarian cancer, and that a higher proportion of mucinous tumors were CEA-positive compared with other histologic types [20].
Although past studies have revealed a more specific role of elevated CEA value in patients with the mucinous type of ovarian cancer, our data showed a similar percentage of patients with the serous, endometrioid, and mucinous types of ovarian cancer (20.7%, 19.5% and 25.3%, respectively) had a CEA level elevation when a more precise cut-off value of 2.6 ng/mL was used according to ROC curve analysis. To redefine a cut-off value for CEA levels is necessary since the median value of CEA in our study cohort is very low (median
Serial measurements of CA-125 are routinely used in clinical practice to monitor treatment response during chemotherapy and recurrence during surveillance. However, the prognostic role of CA-125 in epithelial ovarian cancer has also been extensively surveyed. Many investigators have found that a high postoperative decline in serum CA-125 level was associated with an increased likelihood of achieving optimal primary cytoreduction [2, 22]. Thus, a perioperative decline in serum CA-125 could be used as an early biomarker to predict disease-specific survival in patients who undergo primary cytoreductive surgery for advanced stage disease. In our study cohort, we did not focus on changes in perioperative CA-125 level since the baseline characteristics of our patients were different to those of previous studies. As many as 40% of our patients had FIGO stage I disease, and only about half had a CA-125 level
There are several limitations to this study. First, this was a retrospective cohort study, and patients were excluded if data on pretreatment serum CEA level was not available. This may have caused selection bias. Second, the limited number of articles discussing the prognostic role of CEA in epithelial ovarian cancer may increase the difficulty in describing a clear pathogenesis. Pretreatment CEA level may carry prognostic information in different types of epithelial ovarian cancer, but itself is not strong enough to guide the treatment. Third, the predictive role of CEA value in different histological subtypes was unable to be analyzed due to the limited number of cases. Further multi-center validation studies with more cases may be needed to minimize selection bias and potential confounding factors such as difference in geographic distribution or other benign physiological status that would influence CEA level.
In conclusion, our results provide evidence that an elevated pretreatment serum CEA level
Footnotes
Acknowledgments
This work was supported by the research grant of Chang Gung Memorial Hospital [CMRPG8G0111-3]. We also thank the Biostatistics Center of Kaohsiung Chang Gung Memorial Hospital for assistance with the statistical analysis.
