Abstract
BACKGROUND:
Although Abelson (ABL) tyrosine kinase inhibitors (TKIs) have demonstrated potency against chronic myeloid leukemia (CML), resistance to ABL TKIs can develop in CML patients after discontinuation of therapy.
OBJECTIVE:
Glucose metabolism may be altered in CML cells because glucose is a key metabolite used by tumor cells. We investigated whether D-mannose treatment induced metabolic changes in CML cells and reduced CML growth in the presence of ABL TKIs.
METHODS:
We investigated whether D-mannose treatment induced metabolic changes in CML cells and reduced CML growth in the presence of ABL TKIs.
RESULTS:
Treatment with D-mannose for 72 h inhibited the growth of K562 cells. Combined treatment using ABL TKIs and D-mannose induced a significantly higher level of cytotoxicity in Philadelphia chromosome (Ph)-positive leukemia cells than in control cells. In the mouse model, severe toxicity was observed as evidenced by body weight loss in the ponatinib and D-mannose combination treatment groups.
CONCLUSION:
Our results indicate that metabolic reprogramming may be a useful strategy against Ph-positive leukemia cells. However, caution should be exercised during clinical applications.
Abbreviations
Introduction
Chronic myeloid leukemia (CML) is a myeloproliferative disorder caused by the breakpoint cluster-Abelson (BCR-ABL) oncogene and is characterized by the presence of the Philadelphia (Ph) chromosome within hematopoietic stem cells [1]. The BCR-ABL gene encodes the fusion oncoprotein BCR-ABL, which activates downstream molecules such as phosphatidylinositol-3-kinase and mitogen-activated protein kinase [2]. ABL tyrosine kinase inhibitors (TKIs) have improved the survival of patients with CML [3]. However, clinical cases of TKI-resistant or TKI-intolerant CML patients have also been reported [4, 5]. Moreover, ABL TKIs do not eliminate leukemia stem cells (LSCs), which may represent the most important event in leukemia relapse after TKI discontinuation [6]. Therefore, alternative strategies are required to improve the cure rate of patients with CML.
Because conventional forms of cancer therapy lack selectivity, the development of targeted therapies is one of the main focuses in anticancer research. Glycolysis is a universal pathway for glucose catabolism in normal animals and plants [7]. Facilitative glucose uptake by mammalian cells is mediated by a family of glucose transporters. The generation of adenosine triphosphate (ATP) from glucose is an essential eukaryotic cellular process [8, 9]. In contrast, metabolic programs are altered in cancer cells. Glucose is a key metabolite used by tumor cells, and tumor cells consume more glucose than normal cells. Tumor metabolism relies on glycolysis-mediated ATP production.
Previously, it was reported that administration of the simple sugar D-mannose, either alone or alongside chemotherapy, slows tumor growth in mice [10]. Because mannose can interfere with glucose to reduce tumor growth, we hypothesized that D-mannose can also reduce CML growth. Therefore, we decided to determine whether D-mannose treatment induced metabolic changes in CML cells and reduced CML growth in the presence of ABL TKIs.
Materials and methods
Ph-positive cell lines
The CML cell lines K562, KCL-22, and KU812 were obtained from the American Type Culture Collection (ATCC, Manassas, VA, USA). Another CML cell line, NCO2, was purchased from the JCRB Cell Bank (Ibaraki, Osaka, Japan). T315I mutant Ba/F3 cells were established previously [11]. All cell lines were cultured in Roswell Park Memorial Institute (RPMI) 1640 medium containing 10% fetal bovine serum (FBS) and maintained at 37
Reagents
Ponatinib was purchased from Selleck Chemicals (Houston, TX, USA), while imatinib was kindly provided by Novartis Pharma AG (Basel, Switzerland). Stock solutions of ponatinib were prepared in dimethyl sulfoxide (DMSO). Imatinib wa s dissolved in distilled water, aliquoted, and stored at
Cell proliferation assays
The culture medium was replenished with fresh glucose-free RPMI 1640 medium containing 11.11 mM D-mannose or 25 mM D-glucose. The cells were treated with the indicated concentrations of imatinib or ponatinib for 72 h, and viability was evaluated using the trypan blue exclusion assay or with Cell Counting Kit-8 (Dojindo Laboratories, Kumamoto, Japan), followed by measurement of absorbance at 450 nm. All experiments were performed in triplicate.
Caspase 3/7 activity
Before administering the drug, Ph-positive leukemia cell lines were seeded onto 24-well plates and cultured for 24 h at 37
Cytotoxicity assay
The medium was replaced with fresh RPMI 1640 medium containing 11.11 mM D-mannose or 25 mM D-glucose, and the cells were incubated with the indicated concentration of imatinib for 72 h. In some experiments, d-mannose was administered for 24 h. The cytotoxic effects on leukemia cells were evaluated based on lactate dehydrogenase (LDH) release using the Cytotoxicity LDH Assay kit with water-soluble tetrazolium (WST) salt (Dojindo Laboratories) according to the manufacturer’s protocol. The amount of LDH released from dead cells was measured using an EnSpire Multimode Plate Reader (PerkinElmer, Waltham, MA, USA).
ATP assay
Intracellular ATP levels were determined using the “Cell” ATP assay reagent Ver.2 kit (TOYO B-Net, Tokyo, Japan) according to the manufacturer’s instructions. Luminescence was measured using an EnSpire multimode plate reader.
Enzyme-linked immunosorbent assay (ELISA)
Intracellular glucose,
Colony assay
Colony assays were performed according to the manufacturer’s instructions. K562 cells were seeded in a 24-well plate (1
In vivo assay
Animal experiments were performed with the approval of the Institutional Animal Care and Use Committee of Tokyo Medical University, and carried out in accordance with institutional procedures, national guidelines, and relevant national laws covering animal protection. In this study, humane endpoints were designated to reduce the severity and/or duration of pain and distress experienced by the animals. BALB/c nude mice were maintained under specific pathogen-free conditions. Sixteen mice were housed in a climate-controlled laboratory, and kept under a fixed 12 h light/12 h dark cycle at 22
Statistical analysis
All presented data were analyzed using Prism 8 (GraphPad) or the Excel software. Student’s
Results
Changes in glucose metabolism in CML cells
Glucose is a major source of energy for living organisms. Glucose transport across the plasma membrane of mammalian cells is mediated by members of the glucose transporter/solute carrier 2A (GLUT/SLC2A) family [12]. Because oncogenic signals can increase glucose uptake and drive aerobic glycolysis, we first investigated the expression of glucose transporter genes in CML cells. Gene expression data were analyzed using the Gene Expression Omnibus (GEO) database. In CML samples, the expression levels of SLC2A3 (GLUT3) and SLC2A5 (GLUT5) were higher than those in normal samples (GSE13159). In contrast, SLC2A1 (GLUT1), SLC2A2 (GLUT2), SLC2A6 (GLUT6), SLC2A8 (GLUT8), and SLC2A9 (GLUT9) levels were lower in CML cells than in normal samples (Fig. 1A). Thus, the expression pattern of glucose transporter genes in CML samples differed from that in normal cells.
Gene expression profile of the SLC2A gene family in CML cells. (A) Gene expression data were analyzed. Validation of the SLC2A family of genes by GEO data (GSE13159) comparing the CML group (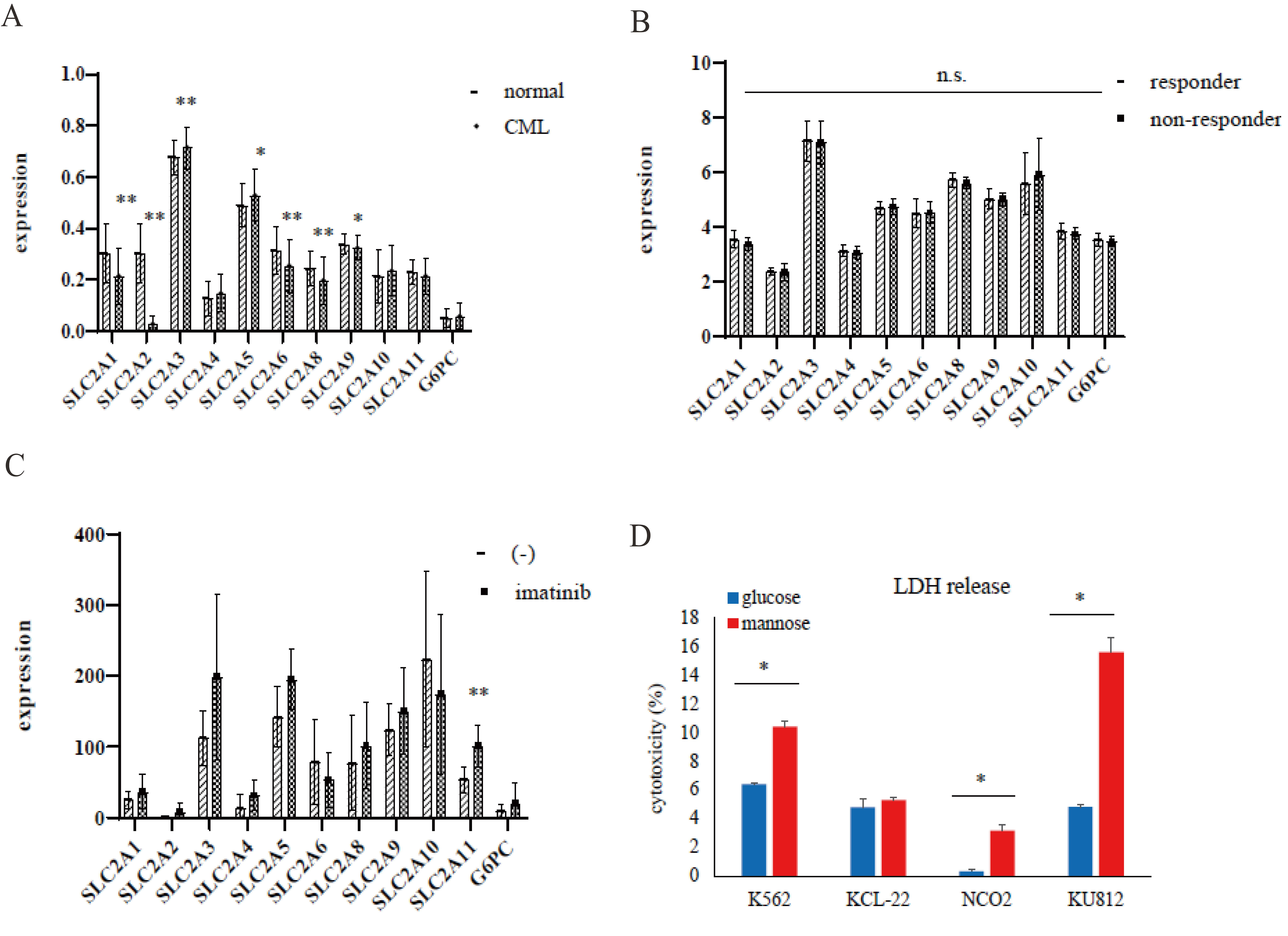
Newly diagnosed chronic-phase CML patients without a major cytogenetic response (MCyR) after 12 months of imatinib therapy have a high risk of progression [13]. We hypothesized that imatinib-resistant cells would exhibit altered glucose transporter gene expression. Thus, we analyzed gene expression in imatinib-resistant samples. From the GSE14671 data, expression levels of the SLC2A gene family were not different between imatinib-sensitive and imatinib-resistant samples (Fig. 1B). A previous report indicated that imatinib ameliorated endoplasmic reticulum stress and induced regression of type 2 diabetes in chronic leukemia patients [14]; therefore, we hypothesized that imatinib may alter glucose transport. We then investigated SLC2A gene expression in samples after imatinib treatment. From the GSE12211 data, we found that the expression levels of SLC2A11 in patient samples were elevated 7 days after imatinib treatment (Fig. 1C). These results indicate that imatinib treatment altered glucose metabolism in CML cells. Lactic acid was initially considered an indicator of glycolytic flux [15]. A lactic acid release assay was performed on the CML cell lines, and D-mannose treatment was found to induce LDH release in the CML cell lines K562, NCO2, and KU812. In contrast, no statistically significant differences were found in KCL-22 cells (Fig. 1D).
Effect of D-mannose in Ph-positive leukemia cells. (A, B) Cells were cultured in RPMI 1640 medium supplemented with 11.11 mM D-mannose or 25 mM D-glucose and incubated with the indicated concentration of imatinib or ponatinib for 72 h. Cell growth was evaluated with Cell Counting Kit-8. 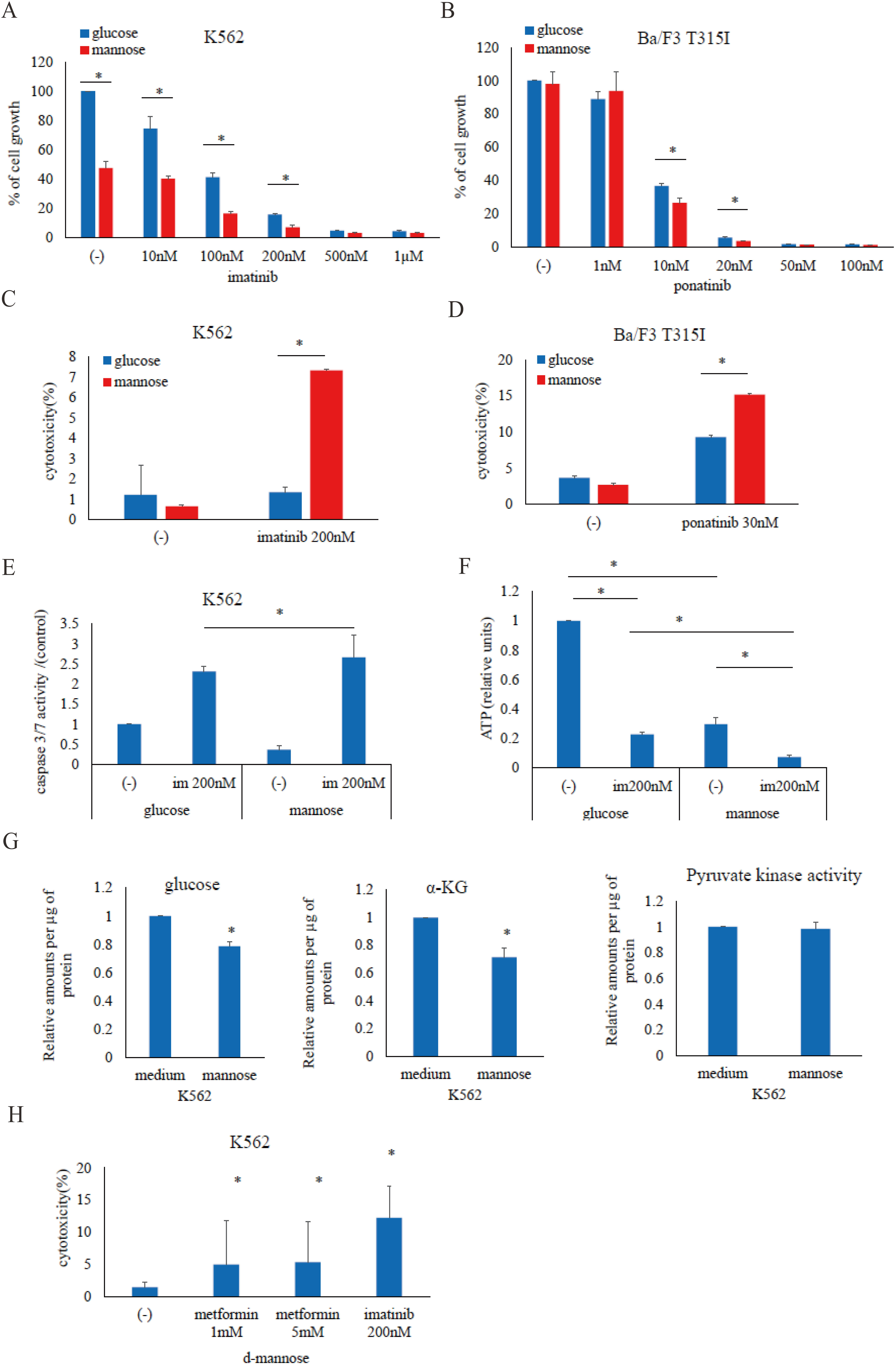
D-mannose is a natural, bioactive monosaccharide that is structurally related to glucose. First, we examined the effect of D-mannose on the Ph-positive leukemia cell line K562. Treatment with D-mannose (11.11 mM) for 72 h significantly reduced the proliferation of K562 cells compared to treatment with 25 mM D-glucose (Fig. 2A). Co-culture with D-mannose and imatinib induced dose-dependent inhibition of cell growth compared to that in the D-glucose treatment group. Next, we investigated the T315I mutant Ba/F3 cell line. Treatment with 11.11 mM D-mannose did not directly inhibit Ba/F3 T315I cells. However, co-culture with D-mannose and ponatinib inhibited cell growth compared to that in the D-glucose and ponatinib-treated cells (Fig. 2B). LDH is released into the culture medium when the cell membranes are damaged. Therefore, the LDH assay was performed as another indicator of Ph-positive leukemia cell cytotoxicity. Cellular cytotoxicity increased in the presence of D-mannose, imatinib, or ponatinib (Fig. 2C and D). Glycolysis produces metabolites for energy production. We next examined whether D-mannose and imatinib induced apoptosis in K562 cells through caspase activation. We observed that caspase 3/7 activity was elevated with D-mannose and imatinib treatment (Fig. 2E), indicating that apoptosis occurred. ATP analysis is used extensively to monitor energy stasis in a broad range of cell types and tissues [16]. Measurements in K562 cells revealed that D-mannose and imatinib treatment reduced intracellular ATP levels compared to D-glucose and imatinib treatment (Fig. 2F). Because D-mannose impairs the growth of CML cells, we next examined glucose metabolism in K562 cells. We found that intracellular glucose and
D-mannose inhibited colony formation in K562 cells
A colony formation assay was performed to identify the ability of D-mannose to inhibit the growth of K562 cells. The colony formation assay revealed that D-glucose-treated cells exhibited an increase in colony number (Fig. 3). In contrast, D-mannose-treated cells showed a decrease in the number of colonies. Co-treatment with 500 nM imatinib and glucose partially inhibited colony formation in K562 cells. We also found that co-treatment with imatinib and D-mannose considerably lowered the proliferation rate of K562 cells (Fig. 3).
Colony formation assay of K562 cells. Cells were treated with 11.11 mM D-mannose or 25 mM D-glucose, followed by 500 nM imatinib for 14 days. Colonies per dish were photographed using a digital camera and counted using an EVOS™ FL Digital Inverted Fluorescence Microscope. The quantification graph displaying colony formation and representative images from three independent sets of experiments are shown. Scale bar: 1,000 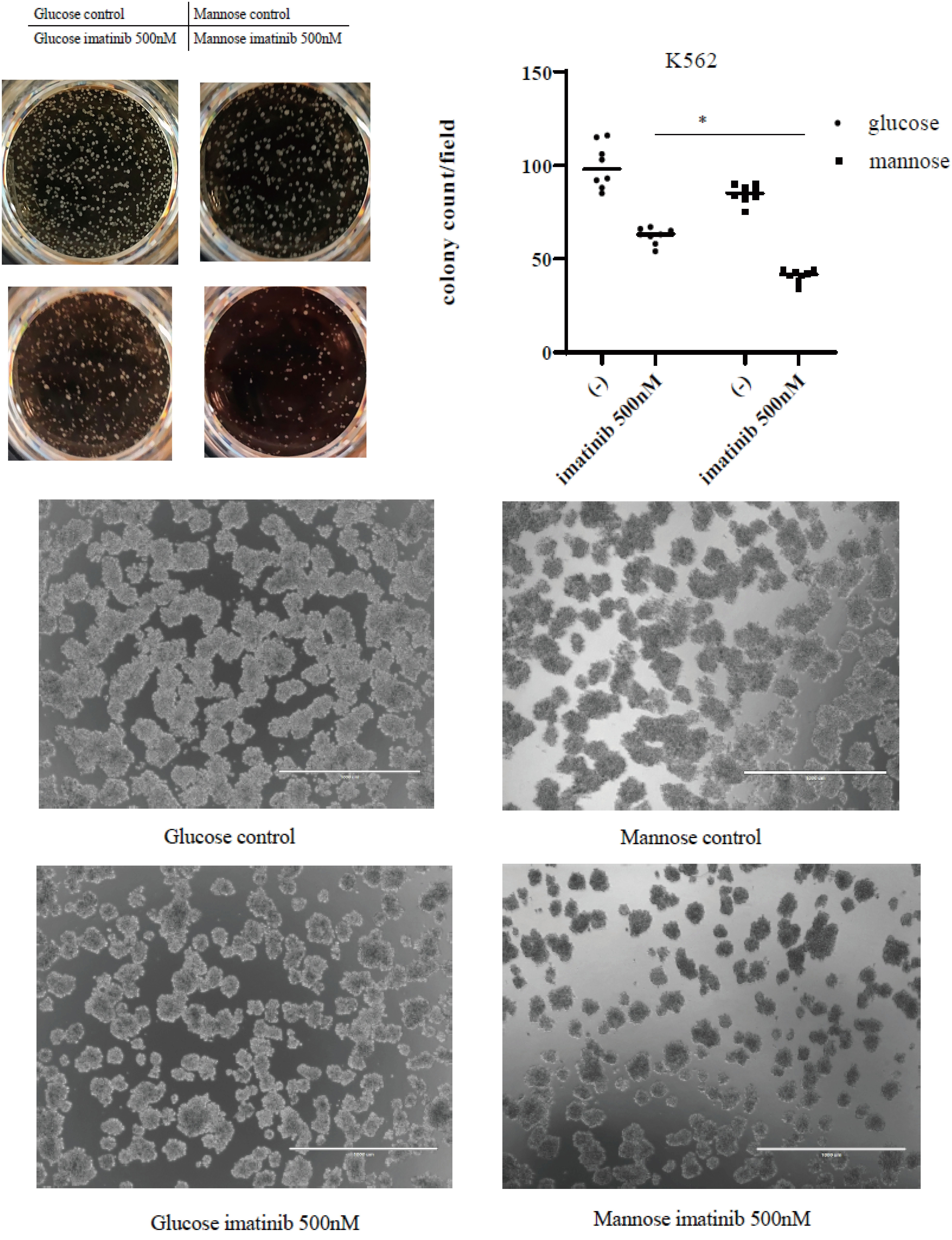
To test whether D-mannose could affect leukemia growth in vivo, 1
Effects of ponatinib and D-mannose in a mouse model. (A) Tumor volumes in mice treated with ponatinib and/or D-mannose (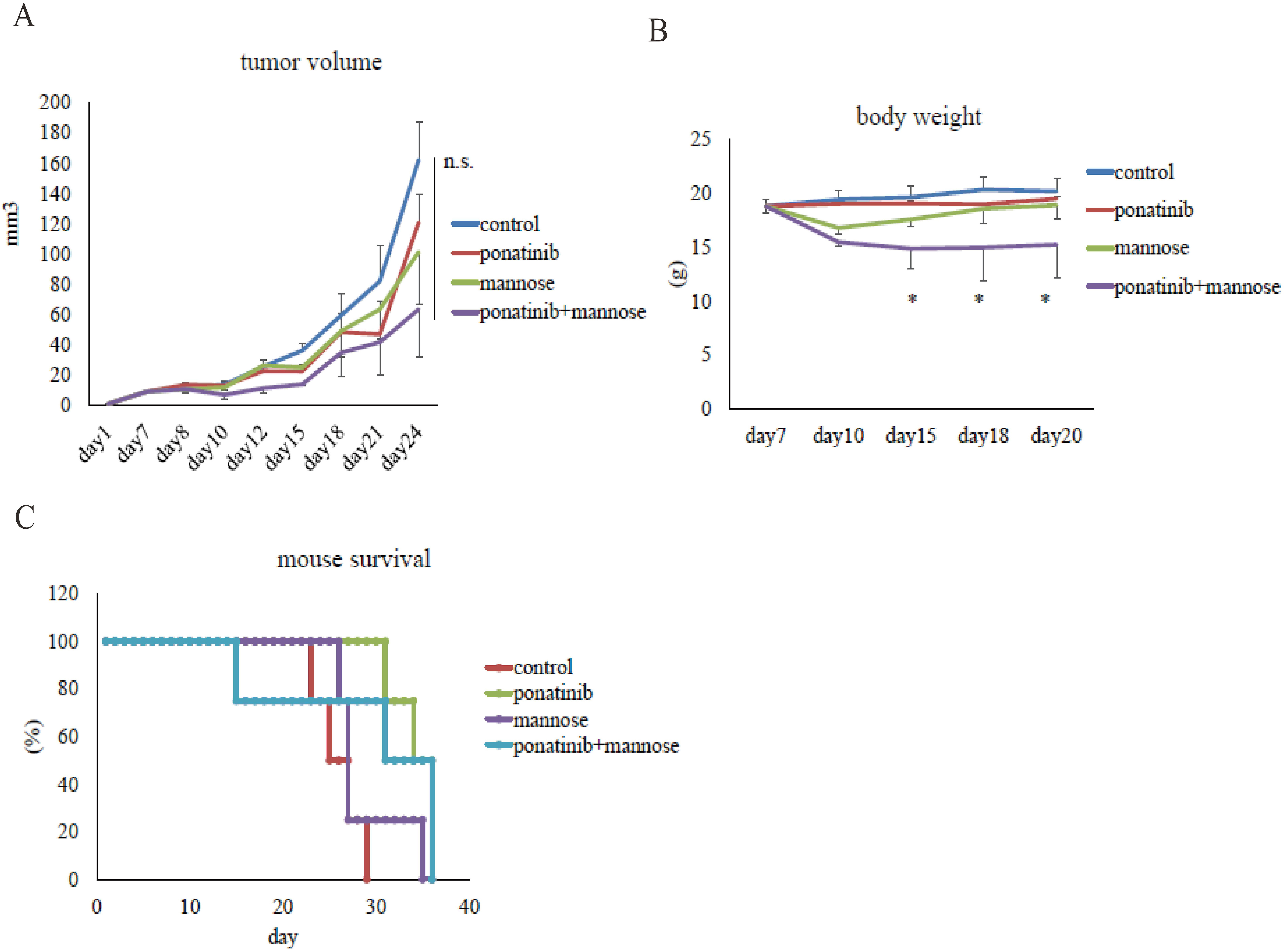
The clinical approach to CML has changed dramatically since the development of TKIs against BCR-ABL kinases. Clinically, treatment with ABL TKIs must be based on CML risk, comorbidities, and patient expectations. However, half of the patients will eventually fail due to intolerance or resistance to first-line treatment, with many of them requiring a third or even further line of therapy [19]. ABL TKI-resistant patients and LSCs are also problematic [4, 5]. To overcome the ABL TKI resistance in LSCs, which are present in patients with a deep molecular response and are certainly the origin of relapses [6], the development of targeted glucose metabolism systems is one of the main focuses in anticancer research because many types of cancers have high metabolic requirements and use reprogrammed metabolism to support cellular activities [8, 9]. We confirmed that high rates of glucose transport via SLC2A family members, especially SLC2A3 and SLC2A5, are required to satisfy the high metabolic demands of CML cells (Fig. 1A). We also confirmed that D-mannose treatment-induced LDH release in CML cell lines (Fig. 1D).
D-mannose is a simple sugar with a complex biosynthesis pathway and is used in therapies for genetic and acquired human diseases, or sometimes even for short periods to treat urinary tract infections [20]. Tumors use more glucose than healthy tissues do. Gonzalez et al. reported that mannose enhances the effects of chemotherapy, slowing the growth of pancreatic tumors, reducing tumor size, and even increasing the lifespan of some mice [10]. Pancreatic cancer cells exhibit deregulated cellular energetics, characterized by the “Warburg effect.” In this study, we also confirmed that intracellular glucose and
We found that D-mannose and imatinib combination therapy inhibited K562 cell growth and induced increased caspase 3/7 activity and cytotoxicity compared to in the control cells. Intracellular ATP levels have been implicated as cellular determinants of apoptosis both in vitro and in vivo. Because we demonstrated that intracellular ATP levels were reduced by D-mannose treatment, glucose metabolism may be important in maintaining CML cell growth. We found that co-treatment with D-mannose and metformin-induced cytotoxic effects against CML cells (Fig. 2H), and D-mannose treatment enhanced ABL TKI activity in vitro. Although D-mannose treatment can reduce cell growth and colony formation in K562 cells in vitro, D-mannose treatment cannot reduce tumor volume in vivo. Because we used different cell lines in this study, such as K562 and T315I mutant Ba/F3 cells, the sensitivity to D-mannose might be different in these cell lines.
In this study, we found that ponatinib and D-mannose treatments exhibited severe drug toxicity in mice, as evidenced by the weight loss and early mouse death observed in this study. The reason for body weight loss was not the exclusion of metastatic disease. However, body weight loss occurred several days after treatment with ponatinib and mannose, suggesting that it might be due to drug toxicity. Ponatinib is a tyrosine kinase receptor inhibitor that is used as a therapy for refractory CML positive for the Philadelphia chromosome [21]. CML patients who receive TKIs should be monitored for signs and symptoms of toxicity throughout the treatment period. Our study showed that body weight loss and early death were found in the mouse model, suggesting that the treatment of patients with cancer should be performed carefully in clinical practice.
Adverse effects of ABL TKIs have also been observed. Because impaired glucose metabolism induced by BCR/ABL inhibitors, especially nilotinib, has been reported in the FDA Adverse Event Reporting System and Japanese Adverse Drug Event Report database, ponatinib may also alter glucose metabolism and enhance the toxicity of D-mannose [22]. Patient education, support, monitoring, and adverse event management are required to clinically follow patients with CML. Cancer therapies can cause side effects, including cardiovascular events, and ABL TKIs are no exception. To improve the cure rate in CML patients, co-treatment with ABL TKIs and molecular target inhibitors may be important [23, 24]. Although D-mannose interferes with glucose metabolism and can potentially be used clinically, either alone or in combination with other forms of cancer therapy, the combination of D-mannose and ponatinib is toxic in vivo. Thus, it is imperative to use this strategy carefully when targeting glucose metabolism.
Author contributions
Conception: Seiichi Okabe
Interpretation and analysis of data: Seiichi Okabe, Yuko Tanaka, and Akihiko Gotoh
Preparation of the manuscript: Seiichi Okabe
Revision for important intellectual content: Seiichi Okabe, Yuko Tanaka, and Akihiko Gotoh
Supervision: Akihiko Gotoh
Footnotes
Acknowledgments
This work was partially supported by the High-Tech Research Center Project for Private Universities, a matching fund subsidy from the Ministry of Education, Culture, Sports, Science, and Technology (MEXT), the University-Industry Joint Research Project for Private Universities, and a matching fund subsidy from MEXT (Grant number: 17K07227). We thank the Tokyo Medical University Research Center and Animal Research Center for providing technical support.
