Abstract
Dasatinib is a multitargeted tyrosine kinase inhibitor (TKI), recommended for chronic myeloid leukemia (CML) in chronic, accelerated and blastic phases that are resistant or intolerant to imatinib. The recognition of BCR-ABL gene and corresponding protein with deregulated tyrosine kinase (TK) activity as crucial for malignant transformation in CML, led to synthesis of the small-molecule drugs designed to interfere with Bcr-Abl TK activation. The first TKI introduced into clinical practice in 1998, was imatinib mesylate, which became the first choice drug in chronic phase CML. However, approximately 20%-25% of patients initially successfully treated with imatinib, develop resistance to this drug, mainly because of selection of clones expressing mutant forms of BCR-ABL with impairing imatinib binding. Dasatinib is 325-fold more potent than imatinib against cell expressing wild-type BCR-ABL and showing activity against most imatinib-resistant Bcr-Abl mutants. Several clinical trials demonstrated that dasatinib is effective and generally well tolerated in imatinib resistant or intolerant CML and represents a promising therapeutic option for these patients.
Keywords
Introduction
The characteristic cytogenetic abnormality underlying CML is the Philadelphia chromosome (Ph), which is the result of the reciprocal translocation between chromosomes 9 and 22. This translocation causes the Abelson gene to fuse with the breakpoint cluster region of the BCR gene.1,2 The BCR-ABL oncogene activates many transduction pathways responsible for the proliferation and survival of hematopoietic cells, that is crucial for their leukemic transformation.3,4 Deregulated Bcr-Abl TK activity is the molecular marker for CML and the target for the new agents introduced to therapy. The first TKI, introduced into clinical practice in 1998, was imatinib mesylate, which became the first choice drug in chronic phase CML (CP CML).5–8 Despite the significant improvements in the outcome for CML patients treated with imatinib, resistance to this agent is an important clinical problem. The absence of response to imatinib treatment known as primary resistance occurs in about 3% of CP CML patients. Patients that achieved a certain level of response (hematological, cytogenetic and molecular) and than have lost it (that occurred in 20% of CP CML patients), can be described as having secondary (acquired) resistance.9–12 The risk of resistance is much higher in patients in advanced phases of CML (accelerated and blast phase, AP CML and BP CML).9–12 The leading cause of acquired resistance to imatinib is reactivation of Bcr-Abl kinase activity due to kinase domain mutations that decrease the sensitivity to imatinib. The presence of mutations can affect residues directly involved in drug binding, impair the ability of the kinase to undergo the conformational changes required for imatinib binding, or favor the active conformation of the kinase, to which imatinib is unable to bind.13–15 Other proposed mechanisms of resistance include: amplification of BCR-ABL gene and overexpression of oncoprotein, activation of BCR-ABL independent pathways, such as members of Src kinases family, binding of imatinib to α1-acid glycoprotein (AGP), increased drug efflux through the multidrug resistance gene, clonal evolution of the disease and the presence of population of quiescent stem cells inherently insensitive to imatinib.13,14,16–21 Thus, there has been interest in developing new agents that are able to overcome imatinib resistance. The second-generation TKIs–-dasatinib, nilotinib and bosutinib have been constructed and provided new therapeutic option. Among them dasatinib is the first approved in the US and European Union for all stages CML patients resistant or intolerant to imatinib.
Chemical Structure, Mechanism of Action and Pharmakodynamic Properties
Dasatinib (BMS-354825, Bristol-Myers Squibb) N-(2chloro-6-methylphenyl)-2-(6-(4-(2-hydroxyethyl)piperazin-1-yl)-2-methylpyrymidin-4-ylamino)thiazole-5-carboxamide (Fig. 1) is a dual v-src sarcoma viral oncogene homolog (Src) family kinases (SFKs) and Abl kinase inhibitor, 325-fold more potent than imatinib against cell expressing wild-type BCR-ABL. 22 Bcr-Abl protein activates many signal transduction pathways including Ras/Raf/mitogen activated kinase (MAPK), phosphatidylinositol 3 kinase (PI3-K), STAT5/Janus kinase, c-Jun NH2-terminal kinase/stress activated protein kinase, nuclear factor -κB, CRC oncogene-like protein/focal adhesion kinase, Src family kinases and Myc.23–25 (Fig. 2) It leads to uncontrolled cell proliferation, genetic instability, reduced apoptosis, and weakening cellular adhesion resulting in the malignant expansion of BCR-ABL positive cells. In vitro studies using Src kinase inhibitors revealed that SFKs play an important role in Bcr-Abl signal transduction and have demonstrated that inhibition of SFks results in growth arrest and induction of apoptosis in Ph+ CML cell lines. 26 Therefore the inhibition of SFks by dasatinib may contribute to the efficiacy of this agent in Bcr-Abl independent imatinib resistance. Dumka et al demonstrated that dasatinib activated p38 MAPK pathway and upregulated mixed lineage kinase 3, MKK3/6, MSK1, and Mapkapk2. 27 Such dasatinib-dependent activation of p38 MAPK and its effectors plays a critical role in the generation of antileukemic responses, since pharmacological inhibition of p38 or siRNA-mediated knockdown of its expression reverse dasatinib-mediated apoptosis, cell cycle arrest, and anti-proliferative effects. p38 MAPK inhibition also reversed dasatinib-induced suppression of CML patient-derived leukemic colony-forming units progenitor growth in vitro, as well as BCR/ABL expressing KT-1 cell-derived leukemic progenitor growth. 27
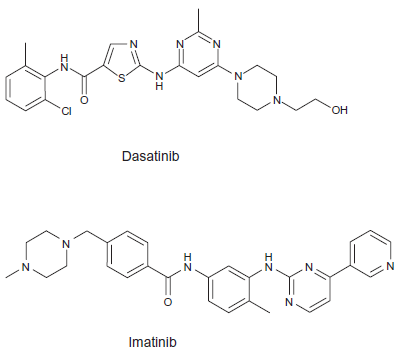
Chemical structure of imatinib and dasatinib.
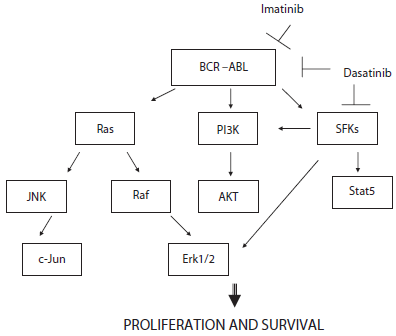
Schematic BCR-ABL related signaling cascade.
Dasatinib in contrast to imatinib can bind to both the active and inactive conformation of the Abl kinase domain.28,29 Analysis of the Abl kinase domain crystal structure in complex with dasatinib may indicate that Abl conformational requirements are less stringent for dasatinib than for imatinib. 30 Moreover, because for dasatinib contacts with P-loop are not so important, it is not involved in critical interactions with many of the mutated residues. It may explain activity of dasatinib against wide range of imatinib-resistant Bcr-Abl mutants, with exception of insensivity to T315I mutation and relative insensivity to F317 L.22,29,31 The crystal structure reveals that a critical interaction is made directly between dasatinib and Threonine315 of the hinge region and explains the lack of activity against T315I mutant. 30
Shah et al revealed, that orally administered dasatinib, significantly prevented disease progression and prolonged survival in mice with myeloproliferative disorder caused by native or M351T Bcr-Abl transformed hematopoietic cells. 29 No clinically significant improvement in survival of mice harboring the T315I mutation was detected. 29 To assess the safety and efficacy of dasatinib on human bone marrow progenitors, Shah et al tested the compound using in vitro colony-forming-unit (CFU) assay. 29 They found that dasatinib at a concentration of 5 nM did not inhibit growth of bone marrow progenitors isolated from healthy volunteers, but did inhibit by 60% to 80% the growth of bone marrow progenitors isolated from CML patients with either imatinibsensitive (nonmutant Bcr-Abl) or imatinib-resistant (M351T) disease. In addition to inhibiting of Abl kinase, dasatinib is potent inhibitor of Src family kinases: Fyn (IC50 0.2 nM), Lck (IC50 1.1 nM), Src (IC50 0.55 nM), Yes (IC500.41 nM). 33 A consequence of the inhibition of Src family kinases, is, that dasatinib inhibits the proliferation of many human tumor cell lines which do not express Bcr-Abl, such as PC-3 (prostate, IC50 5-9 nM), MDA-MB-211 (breast, IC50 10-12 nM), WiDr (colorectal, IC 50 38-52 nM) and consequently parallel tumor xenografts models.32,33
Dasatinib is also a potent inhibitor of c-kit (IC50 13 nM), PDGFRβ (IC50 28 nM), EPHA2 (IC50 17 nM), HER1 (IC50 180 nM) and p38MAP (IC50 100 nM) kinases.32,33
Pharmacokinetics
Oral bioavailability of dasatinib in mammals ranged from 14% to 34%. Dasatinib has high intrinsic permeability in Caco-2 cells, however, the efflux ratio was approximately two-fold indicating that it may be a substrate for an intestinal efflux transporter. However, in vivo studies in P-glycoprotein (P-gp) knockout mice versus wild-type mice showed no difference in the amount of dasatinib remaining unabsorbed in the gastrointestinal tract, suggesting that P-gp may not be responsible for the incomplete bioavailability. 34 In contrast to imatinib, effective dasatinib uptake may occur without hOCT1 (human organic cation transporter 1), which can be one of the mechanisms overcoming imatinib resistance. 35 In a study of the effect of food on the oral bioavailability of dasatinib 54 healthy adult subjects received a single dose of dasatinib 100 mg after an overnight fast or within 10 min after ingestion of a low-fat meal. It was found that the meals had no significant impact on bioavailability. 36
In vivo brain distribution studies showed that the central nervous system (CNS) distribution of dasatinib is limited, with the brain-to-plasma concentration ratios less than 0.12 in mice. 37 Lagas et al demonstrated that brain accumulation of dasatinib is restricted by P-gp and breast cancer resistance protein (ABCG2) and can by enhanced by the dual P-gp and ABCG2 inhibitor. 38 The results presented by Chen et al confirmed that these transporters play a significant role in limiting the CNS delivery of dasatinib and may have direct implications in the treatment of CNS leukemia. 37 They found that selective inhibitors of P-gp and ABCG2 restored the intracellular accumulation of this drug.
Dasatinib is metabolized by the liver, mainly by the CYP3A4 enzyme system and in patients with severe hepatic disfunction dosage adjustment is recommended. Drugs capable of influencing CYP3A4 system would be expected to alter the effectiveness of dasatinib. The compound is eliminated mainly via the faeces. In vivo systemic plasma clearance values of dasatinib were 62, 26, 25, and 34 ml/min/kg in mouse, rat, dog, and monkey, respectively. Scaling of in vitro hepatocyte and liver microsomal data gave reasonably good predictions of in vivo clearances across all species. Percent distribution in blood cells ranged from 43% in mouse to 57% in dog. Dasatinib showed high volumes of distribution (>3l/kg) and high serum protein binding values (>90%) in all four species tested. 34
The Influence on Lymphocytes Function
Dasatinib as Src kinases inhibitor may influence the function of T-cells. Weichsel et al demonstrated that the drug inhibited T-cell activation, proliferation, cytokine production, and degranulation at therapeutically relevant concentrations. 39 This inhibitory effect was mediated by the blockade of early signal transduction events and was not due to loss of T-cell viability. CD4+ cells were more sensitive than CD8+ cells and naïve T cells more sensitive than memory T cells. Fei et al observed that dasatinib inhibited proliferation and function of CD4+ CD25+ regulatory T cells (Tregs) and CD4+ CD25+ cells. 40 The mechanisms of this inhibition included arrest of cells in the G0/G1 phase of cell cycle, down-regulation of the transcription factor forkhead box P3, glucocorticoid-induced tumour necrosis factor receptor and the cytotoxic T lymphocyte associated protein 4 as well as inhibition of signaling events through Src and nuclear factor kappaβ. Fraser et al analysed the influence of dasatinib on viral antigen-specific murine CD4+ and CD8+ cells and cytolytic activity of NK cells. 41 They observed that dasatinib inhibited antigen-specific proliferation of murine CD4+ and CD8+ transgenic T cells in vitro and in vivo. Endogenous antigenspecific helper T-cell recall responses, induction of T-cell-mediated cytotoxicity and the ability of NK cells to eliminate MHC class I-deficient cells in vivo were also inhibited. In contrast to these findings was observation of Kim et al that dasatinib may stimulate the NK and NK/T cell lineage. 42 They identified eight patients with peripharal blood large granular lymphocytosis (LGL) among 18 patients treated with dasatinib. The development of LGL lymphocytosis was found to be significantly associated with an improved response to dasatinib therapy in terms of complete cytogenetic response (CCyR) (p = 0.001), major molecular response (MMoR) (p = 0.001) and complete molecular response (CMoR) (p = 0.003). Moreover, NK cells from the case that developed LGL lymphocytosis had greater cytotoxic effect than those of normal healthy donors, which may explain the better disease control in that group of patients. Similarly Mustyoki et al identified 22 patients on dasatinib with increased peripheral blood lymphocytosis. 43 Fifteen patients had a cytotoxic T-cell and seven patients had an NK-cell phenotype. All T-cell expansions were clonal. Interestingly in majority of patients (18 of 22 patients) lymphocytosis preceded adverse effects, such as colitis and pleuritis. Moreover in pleural effusion and colon biopsy samples cytotoxic T cells were also detected.
By inhibiting immunoregulatory kinases, dasatinib may induce a reversible state of aberrant immune reactivity which may have the important clinical implications.
Clinical Development
Phase I Study
In a phase I study dasatinib was administered in CML patients resistant or intolerant to imatinib, at doses ranging from 15 to 240 mg per day. 44 A total of 84 patients were enrolled in the study: 40 with CP CML, 11 with AP CML, 23 with myeloid BP CML, and 10 with lymphoid BP CML or Ph-positive ALL. Complete hematologic response (CHR) and major cytogenetic response (MCyR) in patients with CP CML were observed in 92% and 45% respectively. In the group of patients with AP, 45% achieved CHR and 27% MCyR. Among patients with BP CML or Ph+ ALL, CHR was observed in 45% of patients and MCyR in 49%, but nearly all of these patients had a relapse within six months. Responses occurred among all BCR-ABL genotypes, with the exception of T315I mutation.
Phase II Studies
Dasatinib has also been examined in phase II studies including imatinib-resistant and imatinib-intolerant patients in CP, AP and BP CML.45–47 These are START studies (Src/ABL Tyrosine Kinase Inhibition Activity Research Trials) including START-C (chronic phase), START-A (accelerated phase), START-B and START-L (myeloid and lymphoid blast crisis) (Table 1). In all groups of patients dasatinib demonstrated high percentage of hematologic and cytogenetic responses. Importantly, comparable responses were achieved by patients carrying Bcr-Abl mutations conferring imatinib resistance and patients without mutations.
Clinical trials of dasatinib in CML patients.
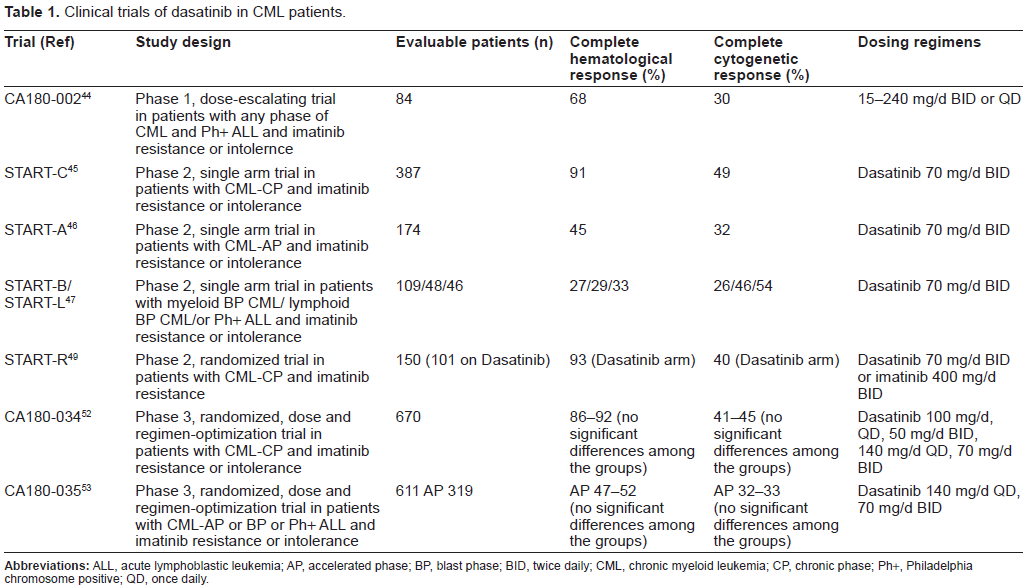
A phase II clinical study of dasatinib in patients with imatinib-resistant or-intolerant CP CML involved 186 patients. 45 They received dasatinib at dose 70 mg twice daily. At 8-months’ follow-up, dasatinib induced notable responses, with 90% and 52% of patients achieving CHR and MCyR. Responses were long lasting: only 2% of patients achieving MCyR progressed or died. Dasatinib also induced molecular responses, reducing median BCR-ABL/ABL transcript ratios from 66% at baseline to 2.6% at 9 months.
The promising results were also observed in AP CML with imatinib-resistance or intolerance.46,48 Patients with imatinib-resistant (n = 161) or-intolerant (n = 13) AP CML received dasatinib 70 mg orally twice daily. At a median follow-up of 14.1 months (treatment duration, 0.1 to 21.7 months), CHR and CCyR were attained by 45% and 32% of patients, respectively. Responses were achieved irrespective of imatinib resistance or intolerance, prior stem-cell transplantation, or the presence of prior Bcr-Abl mutation. The 12-month PFS and OS rates were 66% and 82%, respectively.
Phase II clinical trial of dasatinib in patients with BP CML involved 116 patients (74 with myeloid BP and 42 with lymphoid BP). 47 At 8 months’ follow up, dasatinib induced CHR in 27% and 29% of myeloid BP and lymphoid BP patients and CCyR in 26% and 46% of these patients respectively.
Of great interest are results of START-R study, a randomized Phase II trial, comparing dasatinib (70 mg twice daily) with high dose of imatinib (400 mg twice daily) in imatinib-resistant CML patients.49,50 At a minimum follow-up of 2 years, dasatinib demonstrated higher rates of CHR (93% vs. 82%; P = 0.034), MCyR (53% vs. 33%; P = 0.017), and CCyR (44% vs. 18%; P = 0.0025). At 18 months, the MCyR was maintained in 90% of patients on the dasatinib arm and in 74% of patients on the high-dose imatinib arm. MMoR rates were more frequent with dasatinib than with high-dose imatinib (29% vs. 12%; P = 0.028). The estimated PFS also favored dasatinib (unstratified log-rank test; P = 0.0012). However it should be underlined that approximately two-thirds of the patients in the study would have received 600 mg of imatinib per day before entry. This may at least in part explained the worse results in the imatinib arm. On ASH Meeting 2008 the preliminary results of phase II trial to study efficiacy and safety of dasatinib in patients with previously untreated CP-CML were presented. 51 Overall, 44/45 (98%) evaluable patients achieved CCyR. The CCyR rate at 3, 6 and 12 month compares favorably to that observed in historical controls treated with imatinib 400 mg or 800 mg daily. MMR was achieved in 12/35 (34%) at 12 month and 12/25 (48%) at 18 month. These results indicated that frontline therapy with dasatinib may result in higher response rates compared with standard-dose imatinib and that those responses may be achieved faster.
Phase III Study
In open-label phase III trial, 670 patients with imatinibresistant or-intolerant CP CML were randomly assigned 1:1:1:1 among four dasatinib treatment groups: 100 mg once daily, 50 mg twice daily, 140 mg once daily, or 70 mg twice daily. 52 With minimum follow-up of 6 months marked and comparable hematologic (complete, 86% to 92%) and cytogenetic (major, 54% to 59%; complete, 41% to 45%) response rates were observed across the four groups. Time to and duration of cytogenetic response were similar, as was PFS (8% to 11% of patients experienced disease progression or died). Compared with the approved 70-mg twice-daily regimen, dasatinib 100 mg once daily resulted in significantly lower toxicity. Similary results were observed in phase 3 study of dasatinib 140 mg once daily versus 70 mg twice daily in patients with AP CML resistant or intolerant to imatinib. 53 Dasatinib in both scheme of dosing was equally effective but once daily regimen had an improved safety. The results of phase III studies are presented in Table 1.
Toxicity
In a phase I study a maximum tolerated dose of dasatinib was not determined, and no patient withdrew from the study as a result of toxic effects. 44 The most often side effect of dasatinib is myelosuppresion.44–47 In CP CML 48% of patients have grade 3-4 neutropenia and 46% grade 3-4 thrombocytopenia. Among non-hematological side effect the most often are nausea, diarrhea, rash, headache and fatigue, but they are rarely 3-4 grade. Fluid retention is the most troublesome non-hematological toxic effect of dasatinib. Bergeron et al reported 9 CML patients on dasatinib, who developed dyspnea, cough, and chest pain. 54 Of these nine patients, six had pleural effusions (all were exudates) and seven had lung parenchyma changes (four patients had both). Lymphocytic accumulations were detected in pleural and bronchoalveolar lavage fluids in all but one patient. After dasatinib interruption, lung manifestations resolved in all cases. Quintas-Cardama et al investigated the risk factors and management of pleural effusion associated with dasatinib therapy for CML after failure of imatinib. 55 Among 138 analyzed patients pleural effusion of any grade occurred in 48 (35%) and grade 3/4 in 23 (17%). According to phase of the disease this side effect was observed in 29% of those in CP, 50% in AP and 33% in BP. By multivariate analysis, history of cardiac disease, hypertension, and use of a twice-daily schedule (versus once daily) were identified as factors associated with development of pleural effusions. Interestingly in the study of Hammersmith group the independent risk factors of pleural effusion were a history of skin rash on imatinib and the presence of autoimmune disease. 56 This last observation may indicate the immune-mediated mechanism of the fluid retention. This hypotesis is also supported by observation of Mustyoki et al that in some CML patients treated with dasatinib clonal T-lymphocytosis preceded pleuritis and cytotoxic T-cells were detected in the pleural fluid. 43 It has been also suggested that fluid retention-related adverse events may be result of PDGFR and/or Src family kinases inhibition. 55
Management of pleural effusion usually included transient dasatinib interruption and supportive care with duretics. Short pulse of steroids is sometimes required. In rare cases of large effusion thoracentesis may be necessary.55,57,58
Shah et al demonstrated that compared with the approved 70-mg twice-daily regimen, dasatinib 100 mg once daily resulted in significantly lower rates of pleural effusion (all grades, 7% vs. 16%; P = 0.024) and grade 3 to 4 thrombocytopenia (22% vs. 37%; P = 0.004), and fewer patients required dose interruption (51% vs. 68%), reduction (30% vs. 55%), or discontinuation (16% vs. 23%). 52 This dose has become a recommended one in CP CML.
Dasatinib therapy is associated with increased risk of bleeding, non related to thrombocytopenia. Quintas-Cardama et al observed impaired platelet aggregation on stimulation with arachidonic acid, epinephrine or both in 79%, 85% and 59% of patients on dasatinib. 59
The tolerance of dasatinib in patients in more advanced phases of CML, accelerated and blast phase is worse. In the group of patients in AP-CML, in START-A study the most frequent nonhematologic severe treatment-related adverse event was diarrhea (52%; Grade 3 to 4, 8%).46,48 Cytopenias were common, including grade 3 to 4 neutropenia (76%) and thrombocytopenia (82%). Pleural effusion occurred in 27% of patients (Grade 3 to 4, 5%). The results of phase 3 study of dasatinib 140 mg once daily versus 70 mg twice daily in patients with AP CML resistant or intolerant to imatinib demonstrated that dasatinib in both scheme of dosing is equally effective but once daily regimen has an improved safety. 53 In particular, significantly fewer patients in the once-daily group experienced a pleural effusion (all grades, 20% vs. 39%, p < 0.001). This dose has become a recommended one in AP CML.
Mutational Analysis
In the majority of CML patients the decreased efficacy of imatinib is due to point mutation in BCR-ABL gene. At present more than 50 mutation sites and more than 70 individual mutations have been found.10,11 Müller et al reported the results of mutational analysis performed in 1043 patients with CML-CP treated with dasatinib.60,61 402 patients (39%) had a BCR-ABL mutation, including 8% of 238 imatinibintolerant and 48% of 805 imatinib-resistant patients. 64 different BCR-ABL mutations were detected affecting 49 amino acids, with G250 (n = 61), M351 (n = 54), M244 (n = 46), F359 (n = 42), H396 (n = 37), Y253 (n = 26), and E255 (n = 25) most frequently affected. Dasatinib treatment in patients with or without a baseline BCR-ABL mutation, respectively, resulted in high rates of MCyR (56% vs. 65%), CCyR (44% vs. 56%), MMoR (33% vs. 45%), PFS (70% vs. 83%), and OS (89% vs. 94%). In general, high response rates and durable responses were observed in patients with different mutation types, including highly imatinib-resistant mutations in amino acids L248, Y253, E255, F359, and H396. As expected patients with a T315I mutation were resistant to therapy. Impaired responses were also observed with some mutations with a dasatinib IC50 greater than 3 nM (including Q252H, V299 L, and F317 L). Overall data from clinical trials suggest that dasatinib may be more effective vs. another second-generation TKI nilotinib for treating patients harboring P-loop mutations. Other mutations that are differentially sensitive to both TKIs include F317 L and F359I/V, which are more sensitive to nilotinib and dasatinib, respectively. 62
Jabbour et al proposed mutation scoring system to predict outcome in CML-CP with imatinib failure treated with second generation TKIs. 63 Among 169 patients with CML after imatinib failure, mutations were detected prior to TKi switch in 41 patients (48%) treated with dasatinib and 45 (52%) treated with nilotinib. IC50 values for each TKI-mutation pair were stratified into high, intermediate, low, or unknown sensitivity. For patient with CP-CML hematologic and cytogenetic responses correlated with mutation score. Patients with low and intermediate score had lower response rates and worse event free survival (EFS) and OS than those with sensitive mutations.
Hematopoietic Stem Cell Transplantation
Allografting still has an important place in the treatment of CML patients especially those resistant to available TKIs. Patients undergoing allogeneic hematopoietic stem cell transplantation (allo-HSCT) for CML are increasingly likely to have received a novel TKI after failing imatinib. The data regarding the possible effects of dasatinib on subsequent HSCT are limited. The reports regarded only small number of patients and indicated that dasatinib given before allo-HSCT do not negatively affect transplant engraftment and response rate, nor increase transplant-related toxicity.64,65
Conclusions
Dasatinib has proven to be highly effective alternate approach for CML patient imatinib resistant or intolerant. Across a series of phase II and III trials, dasatinib has demonstrated durable efficacy in patients with CML following resistance or intolerance to imatinib.
Disclosure
Research grant from BMS to Tadeusz Robak.
