Abstract
BACKGROUND:
Both Fms-like tyrosine kinase 3-internal tandem duplication (FLT3-ITD) and Additional Sex Comb-like 1 (ASXL1) mutations are frequent and early genetic alteration events in acute myeloid leukemia (AML) patients. These genetic alterations may be associated with an unfavorable prognosis.
OBJECTIVE:
Up to our knowledge, this is the first study performed to evaluate the clinical implication and prognostic significance of FLT3-ITD and ASXL1 mutations and their coexistence on the outcome of Egyptian AML patients.
METHODS:
Our study included 83 patients with AML who were subjected to immunophenotyping and detection of FLT3-ITD and ASXL1 gene mutation by polymerase chain reaction (PCR) and real-time PCR, respectively.
RESULTS:
FLT3-ITD and ASXL1 mutations were detected in 20.5% and 18.1% of AML patients respectively. Seven patients (8.4%) had co-expression of both genes’ mutations. FLT3-ITD mutation was significantly higher in younger age, higher WBCs count and poor cytogenetic risk patients (
CONCLUSION:
FLT3-ITD and ASXL1 gene mutations or their coexistence can predict a poor prognosis in AML patients.
Abbreviations
Introduction
Acute myeloid leukemia (AML) is a complex disease, in which somatic mutations, coexisting competing clones and disease evolution over time are characteristics [1]. AML can be grouped into morphologically or genetically-defined subtypes. In the 1970s, the French-American-British (FAB) criteria divided AML into subtypes, M0 through M7 (Table 1), based on the origin of leukemic cells and maturity of the cells. It is based on the appearance of the leukemia cells under the microscope after routine staining, as well. Subtypes M0 through M5 all start in immature forms of white blood cells. M6 starts in very immature forms of red blood cells, while M7 starts in immature forms of cells that make platelets [2].
Acquired genetic aberrations play an important role in the development of AML. They are found in approximately 55% of adult AML and have high prognostic significance [3]. AML chromosomal aberrations include t(8:21)(q22;q22), inv(16)(p13q22), t(16;16)(p13;q22), or translocations generating PML-RARA fusion transcripts (APL) collectively referred to as core-binding factor (CBF) AML [4]. Molecular genetic markers include NPM1, FLT3, CEPBA and MLL. Epigenetic factors include DNMT34, TET2. Proteomic biomarkers include PTP, PTK and PIP [5].
Fms-like tyrosine kinase 3 (FLT3) is a proto-oncogene located on chromosome 13q12. It belongs to the receptor tyrosine kinase (RTK) family that plays a major role in hematopoietic cell survival, proliferation and differentiation [6]. The most common form of FLT3 mutation is an internal tandem duplication (ITD) of the juxtamembrane (JM) domain-coding sequence that promotes ligand independent auto-phosphorylation and constitutive activation of the receptor [7, 8]. The length of the ITD varies from 3 to
Characteristics of the different subtypes of AML according to FAB criteria (Bennett et al., 1985)
Characteristics of the different subtypes of AML according to FAB criteria (Bennett et al., 1985)
AML: acute myeloid leukemia; FAB: French-American-British.
Additional Sex Comb-like 1 (ASXL1) gene is located on chromosome 20q11. ASXL1 interacts with multiple epigenetic regulators. Thus regulates the transcription of several target genes, including Hox genes [9]. Non-mutant ASXL1 interacts with EZH2, EED and SUZ12, the main components of the polycomb repressive complex (PRC) 2 to help PRC2 functions [10]. ASXL1 interacts with OGT and HCFC1, as well [11]. In addition, ASXL1 functions as a coactivator or repressor of retinoic acid receptors in a cell-specific manner. Moreover, ASXL1 represses the expression of a subset of adipogenic genes, most of which are peroxisome proliferator-activated receptor (PPAR)-gamma targets. So, it is a PPAR-gamma corepressor [12].
ASXL1 is a frequently mutated gene in myeloid malignancies including myelodysplastic syndrome (MDS) and AML [13]. The vast majority of ASXL1 mutations are deletions, insertions, or substitutions resulting in stop codon mutations and truncation of the ASXL1 protein [14]. Knockdown or deletion of ASXL1 induces MDS-like symptoms, suggesting loss-of-function effects of ASXL1 mutations. The majority of ASXL1 mutations in myeloid malignancies are frameshift or nonsense mutations localized near the 5’ end at the last exon, exon 12. Thus, it escapes nonsense-mediated decay of messenger RNA and generates C-terminal truncated mutations. The mutant ASXL1 gains novel functions to form a hyperactive complex [15]. In addition, these mutations are always heterozygous, leaving one non-mutant allele intact.
Germline mutations of ASXL1 are identified in patients with Bohring-Opitz syndrome, which is characterized by severe developmental disorders [16]. Studies using ASXL1-depleted human hematopoietic cells and ASXL1 knockout mice have shown that deletion of ASXL1 protein leads to impaired hematopoiesis and accelerates myeloid malignancies via loss of interaction with PRC2 proteins [10, 17, 18].
There was a controversy in the results of the previous studies that evaluated the prognostic effect of ASXL1 mutation [19, 20]or FLT3-ITD [21, 8]. Moreover, a few studies evaluated the prognostic effect of the combination of both mutations [22, 23]. So, we tried to investigate the impact of either gene’s mutations and the coexistence of both mutations on the outcome of Egyptian AML patients for the first time.
Experimental subjects
This prospective cohort study included 83 AML patients admitted to the Clinical Hematology Unit, Internal Medicine Department and Medical Oncology Department, Faculty of Medicine, Zagazig University Hospitals. The study was performed in collaboration with the Clinical Pathology and Medical Biochemistry and Molecular Biology Departments, Faculty of Medicine, Zagazig University during the period from August 2017 to December 2020. Patients were diagnosed according to the WHO 2016 criteria [24]. All patients with severe co-morbidities, M3 subtype and pregnant females were excluded from the study. Informed written consent was taken from each patient. All procedures were following the standards of the IRB of the Faculty of Medicine, Zagazig University.
Cytogenetic analysis was done on bone marrow (BM) cells collected from all AML patients at the time of diagnosis. BM specimens were collected on potassium ethylene diamine tetra-acetic acid (K-EDTA) (1.5 mg/mL) for morphologic, immunophenotypic, and molecular analysis.
Flow cytometric immunophenotyping for standard acute leukemia panel
Immunophenotyping for acute leukemia was performed using the standard panel of FITC/PE/PerCP-labeled monoclonal antibodies: (CD45, CD34, CD38, HLA-DR, MPO, terminal deoxynucleotidyl transferase [TdT], CD64, CD14, CD2, CD3, CD5, CD7, CD19, CD79a, CD20, CD22) (Becton Dickinson (BD), Cal, USA). Antigens were scored positive using a cut-off value of
Polymerase chain reaction (PCR) for FLT3-ITD
For qualitative analysis of FLT3-ITD mutations, genomic DNA was isolated using a QIAamp Mini Kit (Qiagen). A 360-bp fragment of the FLT3 gene was amplified using primers 11F (5’ CATTGTCGTTTTAACCCTGCTA 3’) and 12R (5’ ATATTCTCGTGGCTTCCCAG 3’). The PCR reaction was performed using a mixture containing 150 ng of DNA, 10 uL of master mix (Invitrogen) and 25 pmol of the forward and reverse primers. Initial denaturation at 95
Molecular detection of ASXL1 gene mutatuion
For detection of the AXSL1 mutation, genomic DNA was amplified with a 9 G primer forward (5’ ATCGGAGGGGGGGGGT 3’) and reverse (5’ AGCTCTGGACATGGCAGTTC 3’) synthesized by Sangon Biotech (Beijing, China) that contain a repeating guanine base on the expected site for mutation producing a mutant ASXL1 that so-called ASXL1 c.1934dupG according to the method previously described by Yannakou et al. In brief, PCR was performed on a total volume of 25
Results
Conventional cytogenetic analysis is mandatory in the evaluation of suspected AML. Favorable cytogenetic risk is detected in inversion (16) or translocation (16;16), translocation (8;21) or translocation (15;17). Intermediate cytogenetic risk is detected in trisomy 8, abnormal chromosome 5 or 7, inversion 16 with complex karyotype or 3 combined abnormalities. Poor cytogenetic risk is detected in complex (
We investigated 83 newly diagnosed AML patients. They included 44 males (53%) and 39 females (47%) with a male to female ratio (1.1:1). The mean age was 58.1
Demographic and basic characteristics among the studied groups
Demographic and basic characteristics among the studied groups
CR: Complete response to treatment, PR: partial response, RR: refractory response, RFS: relapse-free survival.
Patients with FLT3-ITD mutation had significantly younger age, higher WBCs count, higher BM blasts and lower hemoglobin content (
Relation between FLT3-ITD and basic characteristics of AML patients
Relation between FLT3-ITD and basic characteristics of AML patients
Relation between ASXL-1 and basic characteristics of AML cases
Patients with ASXL1 mutation had significantly older age, higher WBCs count and lower BM blasts (
Comparison between patients with FLT3-ITD mutation and those with ASXL1mutation
Patients with FLT3-ITD had younger age, higher WBCs count, lower hemoglobin, higher BM blasts (
Relation between mutant ASXL-1 gene, mutant FLT-3 gene and co-expression of both among studied cases
Relation between mutant ASXL-1 gene, mutant FLT-3 gene and co-expression of both among studied cases
Survival analysis of AML cases according to FLT3-ITD and ASXL1 gene mutation
In patients with FLT3-ITD mutation, age was significantly lower than patients with ASXL1 mutation and lower than patients with co expression of both mutations (
Survival study of AML patients with FLT3-ITD and ASXL1 mutations
The mean time of survival of patients with FLT3-ITD and ASXL1 mutations were 10.3 and 11.1 months respectively compared to non-mutant types (21.01 and 20.7 months respectively). The percent of RFS of patients with FLT3-ITD and ASXL1 mutations were 33.3% and 50% respectively, compared to their non-mutant types (63.6% and 62.7% respectively) (Table 6 and Figs 1 and 2).
Kaplan-Meier DF survival analysis of studied cases according to mutation in ASXL-1 gene.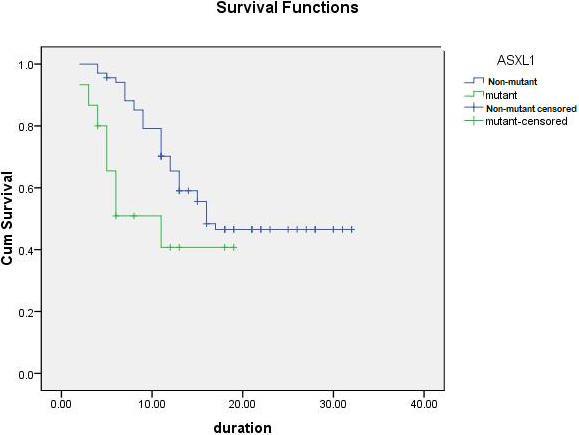
Kaplan-Meier DF survival analysis of studied cases according to FLT-3 gene mutation.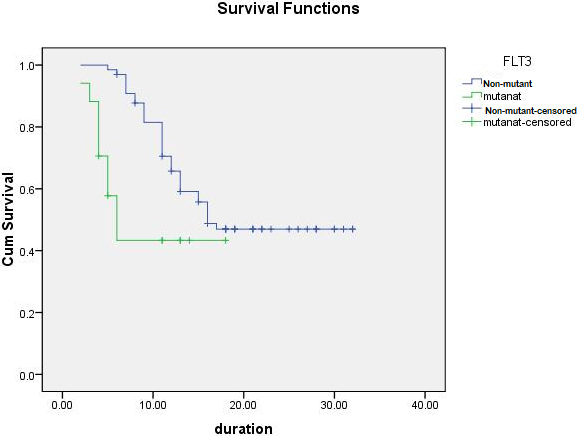
Age, WBCs count, secondary AML and poor cytogenetic risk were significant predictors for FLT3-ITD mutation among AML patients (
Multivariate analysis of significant predictors of FLT3-ITD and ASXL1 mutations
Multivariate analysis of significant predictors of FLT3-ITD and ASXL1 mutations
CI: confidence interval.
Univariate and multivariate Cox-regression analyses for survival
HR: hazard ratio, 95% CI: 95% confidence interval.
FLT3-ITD and ASXL1 mutations were found to be independent risk factors for OS in AML patients in univariate and multivariate analyses (
Discussion
In this work, we tried to evaluate the role of FLT3-ITD and ASXL1 genes’ mutation in the prognosis of AML patients with trial to demonstrate the impact of coexistence of both genes’ mutation on the outcome of patients. We detected FLT3-ITD mutation in 17 patients (20.5%) and ASXL1 gene mutation in 15 patients (18.1%). These results were in accordance with each of Awad et al. and Wang et al. who detected FLT3-ITD mutation in 18.5% of their patients [29, 30], Kandel et al. who detected it in 21.4% [31], and Schnittger et al. who detected ASXL1 gene mutation in 17.2% of their patients [32]. However, Prats-Martín et al. found ASXL1 gene mutation in 31% of myelodysplasia-related changes AML patients [33].
In our study, patients with FLT3-ITD mutation were significantly younger than patients with ASXL1 gene mutation and were younger than patients with co-expression of both mutations. Besides, age was a significant predictor for FLT3-ITD mutation, and it was an independent univariate risk factor for RFS. Several studies supported our results [24, 27, 34]. Other previous studies supported that ASXL1 mutations in AML were more common in older patients [30, 31]. In our study, we detected no significant difference between patients with FLT3-ITD mutation or those with ASXL1 gene mutation as regard gender. On the other hand, Juliusson et al. detected that females more often had FLT3-ITD mutation [34] while Paschka et al. found that ASXL1 gene mutations were more associated with the male gender [35]. This difference may be attributed to the difference in ethnicity and the sample size.
In our study, WBCs count was significantly higher in patients with FLT3-ITD mutation compared with those with ASXL1 gene mutation or patients with co-expression of both mutations. This was supported by Paschka et al. [35]. On the other hand, Kakosaiou et al. found that ASXL1 gene mutation was associated with higher peripheral leukocytosis [36]. In our work, WBCs count was a significant predictor of FLT3-ITD mutation and was a significant univariate independent risk factor for OS in AML patients. Lagunas-Rangel and Chávez-Valencia found that FLT3-ITD has been strongly associated with a bad prognosis, leukocytosis, high blast counts, increased risk of relapse and shorter OS [8]. In addition, de Jonge et al. demonstrated that high WBC count and FLT3-ITD/FLT3 ratio were important prognostic factors in patients with AML [21].
We found that AXL1 mutation was more associated with secondary type AML. This is supported by Paschka et al., Kakosaioua et al. and Prats-Martín et al. who found that ASXL1 mutation was more associated with secondary AML [35, 36, 33]. Secondary AML was found to be a significant predictor for ASXL1 mutation, and it was found to be an independant risk factor for OS in univariate and multivariate analyses in our patients. Different from our results, Fathi and Chen found that FLT3-ITD mutation was present in a quarter of patients with de novo AML [37].
In our study, ASXL1 mutation was more associated with intermediate cytogenetic risk while FLT3-ITD mutation was associated with poor one. These results are following Awad et al. and Prats-Martín et al. [29, 33]. Prats-Martín et al. stated that ASXL1 mutations have been reported in up to 35% of AML patients with intermediate-risk [33]. This is different from the results of Lin et al. who found that the ASXL1 mutation was more frequent in normal karyotype CN-AML patients and Tao et al. who found that FLT3-ITD was common in AML patients with normal karyotype [20, 38]. This discrepancy may be due to the different cytogenetic types in different races.
In our study, the poor cytogenetic risk was a significant predictor of FLT3-ITD mutation but the cytogenetic risk was not an independent risk factor for OS or RFS. FLT3-ITD and ASXL1 mutations were more associated with partial response to treatment compared to the non-mutant FLT3 and ASXL1. Both mutations were more associated with poor outcomes compared to the non-mutant types but there was no significant difference between both mutations regarding the outcome of the patients.
Concerning the possible role of gene mutations on the prognosis of AML, our survival study after a follow-up period of 33 months found the mean survival period was shorter and the number of relapses was more frequent and RFS were less common in FLT3-ITD and ASXL1 mutations compared to non-mutant types. Boddu et al. found that patients with FLT3-ITD have a high risk of relapse and a low cure rate [39]. Hand by Hand with these results, Wang et al. stated that FLT3-ITD mutation can predict prognosis in AML and it is associated with shortened RFS and OS [30].
Prats-Martín et al. concluded that ASXL1 mutation could be the worst outcome marker in de novo AML and they demonstrated that ASXL1 mutations were associated with inferior response rates and survival [33]. Poor prognostic role of ASXL1 and FLT3-ITD in AML was supported by Yu et al. [40] who confirmed that ASXL1 mutation is associated with poor prognosis in older patients who had a higher incidence of mutation, and Lyu et al. who found that FLT3-ITD was related to worse CR and OS. They also stated that FLT3-ITD may serve as an independent prognostic factor for OS in non-M3 AML patients [41]. Niparuck et al. had shown that FLT3-ITD can predict the outcome in patients with adult AML and showed poor RFS and OS [1]. Because of this bad prognosis of AML patients with FLT3-ITD mutation, Helbig et al. advised that these patients should proceed to allogeneic stem cell transplantation as soon as possible in the disease course [42].
ASXL1 and FLT3-ITD were independent predictors of OS and RFS but Hb, WBCs count, BM blast were independent predictors of OS. Dohner et al. found that older age
Conclusion
ASXL1 mutation, FLT3-ITD or co-existence can predict poor OS and RFS in Egyptian AML patients. Age, WBCs count, secondary AML and poor cytogenetic risk are significant predictors of FLT3-ITD and ASXL1 mutations which are independent risk factors for OS in these patients.
Limitations and recommendations
The results of this study are limited by the relatively small number of participants from a single center. Besides, patients with severe co-morbidities, M3 subtype and pregnant females were excluded from the study. Moreover, ethnic variation must be put into consideration as this study was performed on Egyptians only. Our results need to be confirmed by further prospective randomized controlled studies with an increased number of participants, ethnic variations, and multi-centric cooperation. From our results, we recommend the combination of ASXL1 mutation and FLT3-ITD as prognostic markers in AML patients.
Author contributions
Conception: Huda F. Ebian
Interpretation or analysis of data: Samia Hussein, Ahmad Embaby Haitham Mohamed, Tarek Khamis and Norhan A. Sabbah
Preparation of the manuscript: Huda F. Ebian and Samia Hussein
Revision for important intellectual content: Reham Sameh and Samia Hussein
Supervision: Huda F. Ebian and Sherin Elshorbagy
Funding
None.
Footnotes
Acknowledgments
None.
Conflict of interest
None.
