Abstract
Background:
Pediatric acute myeloid leukemia (AML) has poor prognosis and high rate of relapse and mortality, and exploration of new treatment options is still critically needed.
Objectives:
To summarize the outcome of our new treatment strategies for pediatric AML, which is characterized by dual induction and acute lymphoblastic leukemia (ALL) elements consolidation.
Design:
Retrospective, single-arm study.
Methods:
From July 2012 to December 2019, an intensive chemotherapy protocol was used for newly diagnosed children with AML, which contains dual induction, three courses of consolidations based on high-dose cytarabine, and two courses of consolidations composed of high-dose methotrexate, vincristine, asparaginase, and mercaptopurine (ALL-like elements). Blasts were monitored by bone marrow smears at intervals, and two lumbar punctures were performed during chemotherapy. We retrospectively analyzed the efficacy and safety of this study. The last follow-up was on 26 May 2023.
Results:
A total of 70 pediatric AMLs were included. The median age at diagnosis was 6.7 (0.5–16.0) years. The median initial WBC count was 23.74 × 109/L, 11 of whom ⩾100 × 109/L. After dual induction, there were 62 cases of complete remission (CR), 5 cases of partial remission, and 3 cases of nonremission. The CR rate was 88.57%. The median follow-up time was 5.8 (0.2–9.4) years, the 5-year overall survival was 78.2% ± 5%, the event-free survival (EFS) was 71.2% ± 5.6%, and the cumulative recurrence rate was 27.75%. The 5-year EFS of patients with initial WBC < 100 × 109/L (n = 59) and ⩾100 × 109/L (n = 11) were 76.4% ± 5.7% and 45.5% ± 15% (p = 0.013), respectively. A total of 650 hospital infections occurred. The main causes of infection were respiratory tract infection (26.92%), septicemia (18.46%), stomatitis (11.85%), and skin and soft-tissue infection (10.46%).
Conclusion:
This intensive treatment protocol with dual induction and ALL-like elements is effective and safe for childhood AML. Initial WBC ⩾ 100 × 109/L was the only independent risk factor in this cohort.
Trial registration:
It is a retrospective study, and no registration on ClinicalTrials.gov.
Keywords
Introduction
Acute myeloid leukemia (AML) is a group of diseases characterized by clonal proliferation of primitive or naïve myeloid cells due to defective differentiation of hematopoietic stem and progenitor cells. Over the past few decades, the outcomes for pediatric AML have improved significantly but remain unsatisfactory when compared to childhood acute lymphoblastic leukemia (ALL). The event-free survival (EFS) and overall survival (OS) of several large international cooperative groups varied from 50–60% to 65–75%, respectively.1 –6 Relapse and treatment-related mortality (TRM) are the main causes of treatment failure. 7 After intensive chemotherapy, 30–40% of children still experience relapse, 8 and the 5-year OS of relapsed AML is less than 40%.8,9 High-intensity chemotherapy regimens increase the risk of TRM, the incidence of which ranged from 7.8% to 13.8%.10 –13 Therefore, novel treatment is still urgently needed for pediatric AML.
It is clear that pediatric AML is a disease distinguishable from adult AML with regard to the gene mutation spectrum. As a result, treatment strategies differ between adults and children. The treatment of adult AML consists mainly of remission induction and post-remission therapy, which contains chemotherapy, targeted therapies, and hematopoietic stem cell transplantation (HSCT). 14 In terms of induction, a combination of anthracycline for 3 days and standard-dose cytarabine (Ara-C) for 7–10 days (‘7 + 3’ therapy) 15 are recommended. An alternative method to intensify standard induction regimen is the addition of a purine analog such as fludarabine or cladribine. In addition, more therapeutic approaches, such as molecularly targeted inhibitors like FLT3 inhibitors, IDH1 inhibitors, and immunotherapies such as monoclonal antibodies and CAR-T therapies, are gradually used in clinical treatment. In pediatric AML patients, ‘7+3’ therapy is also the most traditional treatment, but it is limited by the safety and efficacy of the previously described. Therefore, several treatment strategies have been explored to improve the outcome of AML in different collaborative groups. One is to increase the dosage of chemotherapy drugs. The results of EORTC + GIMEMA 16 showed that the high-dose cytarabine regimen could achieve higher remission and survival rates than the standard-dose regimen. ECOG-ACRIN 17 confirmed that high-dose daunorubicin (DNR) can significantly benefit AML patients under 50 years of age with NPM1 and DNMT3A mutations. Another strategy is to up-front the second-line drugs to the first line. MRC-AML15 18 using fludarabine in newly diagnosed patients with AML and suggested that FLAG-Ida could improve complete remission (CR) and decrease the cumulative recurrence rate (CRR) after the first course of treatment, but significant myelosuppression occurred after two courses of treatment. In addition, new drugs developed in recent years, such as gemtuzumab ozogamicin, can reduce the recurrence risk of FLT3/ITD-positive children, 19 and the BCL2 inhibitor Veneclax combined with intensive chemotherapy has also been confirmed to achieve a higher remission rate in newly diagnosed adult AML patients during induction. 20 The schemes mentioned above have demonstrated advantages in improving EFS or OS; however, their disadvantages, such as excessive doses, high TRM, expensive costs, and poor accessibility, restrict their widespread application, especially in areas with limited medical resources.
Interestingly, St. Jude 21 proposed the use of methotrexate (MTX) combined with asparaginase to treat relapsed or refractory childhood AML, and the remission rate could reach 20%. In adult studies, asparaginase was demonstrated to be used in combination with other conventional chemotherapeutic agents for the treatment of elderly patients with refractory AML, 22 suggesting that asparaginase may be effective for the treatment of AML and proposing that asparaginase may induce autophagy by promoting apoptosis and inhibiting AML cell growth. MTX is also considered one of the effective drugs in the treatment of acute monocytic leukemia due to its property of inducing cell growth arrest and apoptosis, and it has been shown that MTX, followed by asparaginase administration, produces a synergistic effect. 23 These drugs are widely used in children with ALL, and the side effects are relatively well controlled compared to high-dose Ara-C-based regimens. Therefore, we wondered whether these drugs could improve efficacy in newly diagnosed AML children. Therefore, two courses of ALL-like element regimens, including asparaginase and MTX were added to the intensive protocol based on traditional high-dose cytarabine and anthracycline since 2012. In this article, we reviewed the clinical characteristics, treatment outcomes, and chemotherapy-related complications and analyzed factors associated with prognosis.
Method
General information
This study was a multicenter, retrospective, single-arm study that enrolled newly diagnosed AML patients in three centers in Guangdong Province (the First Affiliated Hospital of Sun Yat-sen University, Huangpu District of the First Affiliated Hospital of Sun Yat-sen University, and Huizhou Central People’s Hospital). All children with newly diagnosed AML age at diagnosis ⩽16 years old and treated in our collaboration groups from July 2012 to December 2019 were enrolled. Prior to treatment, all children underwent routine blood, bone marrow cell morphology, immunological typing by full panel flow cytometry (FCM), fusion genes including AML1/ETO, PML/RARa, MLL rearrangement, and CBFβ/MYH11 by fluorescence in situ hybridization and multiplex nested polymerase chain reaction. FLT3/ITD mutation was tested by PCR-conjugated agarose gel electrophoresis. Common AML prognostic gene mutations, such as EVI1, c-kit, NPM1, and CEBPA mutations, were detected by multiplex nested PCR. Chromosome karyotype analysis was also included. In this study, only morphology non-CR at the end of induction was considered as high-risk group and transferred to HSCT. 24 This study followed the Helsinki Declaration and was given official approval by the Ethics Committee.
Exclusion criteria
Patients were excluded if they (a) had PML/RARa-positive leukemia, MDS-AML, secondary Acute Myeloid Leukemia (sAML), genetic syndrome, and Trisomy 21 syndrome with AML, and (b) gave up treatment within 7 days of induction therapy initiation because the aim of this study was to evaluate the outcomes of our treatment protocol including dual induction therapy (described in detail below).
Treatment regimens
The detailed treatment protocol is shown in Figure 1, and specific dosages of intrathecal injection are shown in Supplemental Table S1. In summary, this intensive protocol was composed of a dual induction, three courses of consolidation based on traditional high doses of Ara-C and anthracycline, and two courses of ALL-like regimens containing HD-MTX and asparaginase. In double induction, we used DNR, Ara-C, and etoposide (VP16) for the first 6 days, and after an interval of 4 days, we used equal dosages of Ara-C and VP16 for the next 3 days. The cumulative amounts of chemotherapy drugs were as follows: Ara-C 40.5–43.5 g/m2, anthracycline 350 mg/m2 (converted according to DNR), and VP-16 1.05 g/m2.
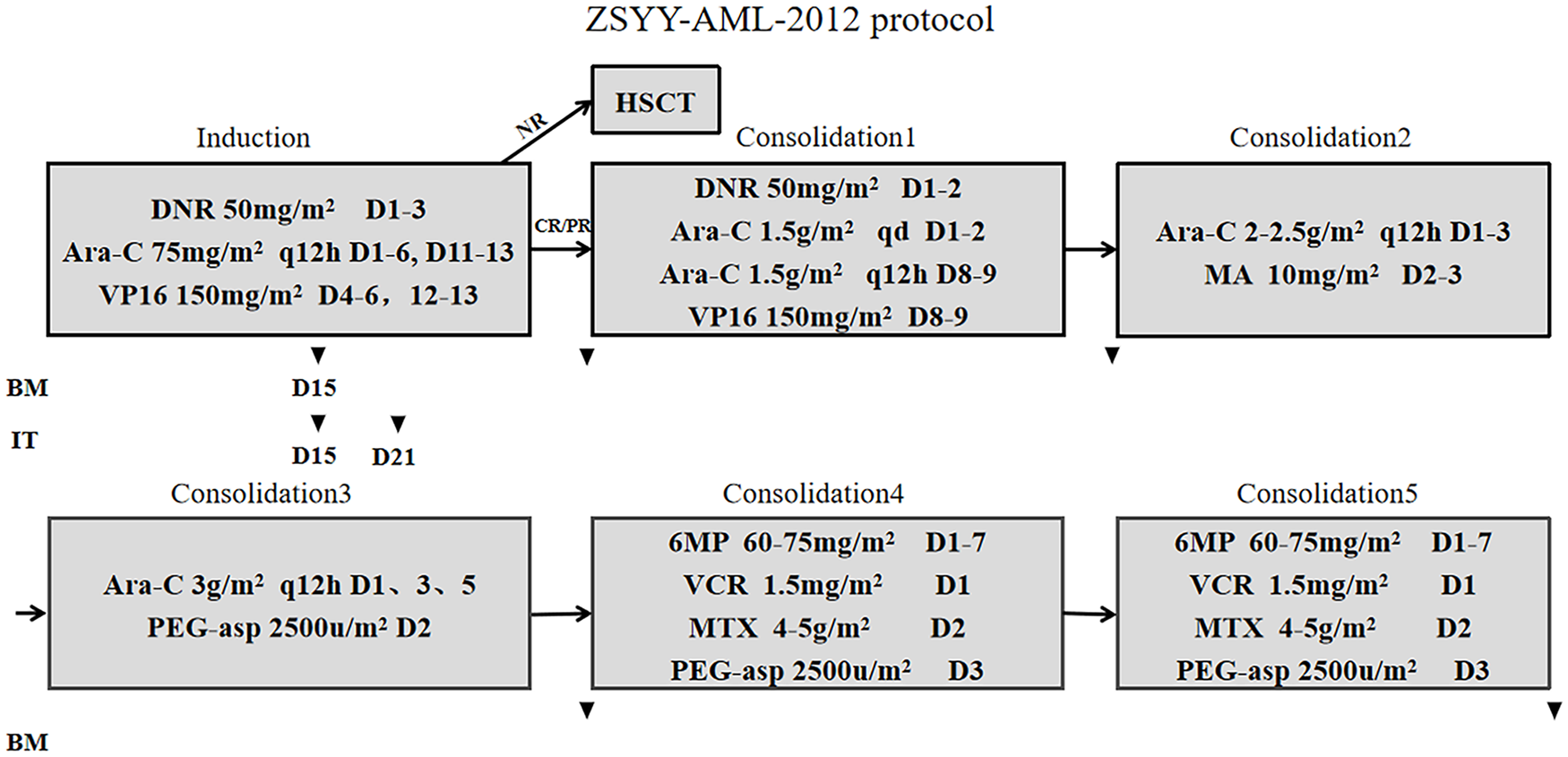
ZSYY-AML-2012 protocol. BM is usually performed on the 15th day of induction and after the first, fourth, and fifth consolidation. On the 15th day of induction, we only check the examination of marrow smear while we also check for FCM, positive fusion gene, or mutation after consolidation. IT, intrathecal injection of cytarabine and dexamethasone, is done on the 15th and 21st day of chemotherapy.
Efficacy evaluation and follow-up endpoints
Morphological CR was defined as myeloid blasts ⩽5%, PR was defined as myeloid blasts ranging from 5% to 20%, and nonremission (NR) was defined as bone marrow blasts ⩾20%. Morphological relapse was defined as primordial cells >5% after remission. OS was defined as the time from the date of diagnosis to the child’s death from any cause or the date of last follow-up. EFS was defined as the time from the date of diagnosis to the first event or the last follow-up date, where events included leukemia recurrence, death from any cause, and secondary tumor. NR and transplantation were not considered events but served as follow-up cutoffs. TRM referred to deaths associated with therapy that were not related to other causes, such as disease progression or recurrence.
Statistical analysis
The Kaplan-Meier method was used to estimate OS and EFS. Mean ± SD was used to describe normally distributed variables, while skewed variables were expressed as median (range). The comparison between categorical variables was evaluated by the chi-square (X2) test. Cox regression analysis was used to analyze the risk factors for recurrence, and the chi-square test was used to analyze chemotherapy-related complications. p < 0.05 was considered statistically significant. All statistical analyses were conducted using SPSS Statistics 25.0.
Results
Population baseline characteristics
From July 2012 to December 2019, a total of 90 newly diagnosed AML cases were admitted to three hospitals, of whom 19 with PML/RARa-positive AML and 1 with inherited syndrome were excluded. No patient gave up treatment and withdrew from the study since the treatment initiation. Seventy cases were finally included in the study: 53 cases in the First Affiliated Hospital of Sun Yat-sen University, 8 cases in Huangpu District of the First Affiliated Hospital of Sun Yat-sen University, and 9 cases in Huizhou Central People’s Hospital (Figure 2). Of the 70 children with AML, 41 were male and 29 were female. The median age was 6.7 (0.5–16.0) years, and the median WBC count in peripheral blood was 23.74 × 109/L (1.23–304.4) at initial diagnosis. There was 1 case of M1 in the morphological type, 28 cases of M2, 2 cases of M4, 3 cases of M4eo, and 36 cases of M5. AML1/ETO, CBFβ/MYH11, MLL rearrangement, and FLT3/ITD mutation were positive in 22, 5, 12, and 3 cases, respectively. There were four cases of CEBPA double mutation, four cases of EVI1 high expression, two cases of c-kit mutation, one case of NPM1 mutation, and one case of TLS/ERG (Table 1).

Flowchart of patients enrolled in the 2012 AML protocol. All 70 patients shown above were included in the analysis.
Patient and disease characteristics.

Response rate of induction therapy and overall response rate
Bone marrow evaluation was performed in all 70 children at the end of induction when blood counts recovered, indicating that 62 cases were CR, 5 cases were PR, 3 cases were NR, and the remission rate of dual induction was 88.57%. One case died of severe infection during consolidation 1, and one case was transferred to HSCT according to parent’s requirements. Among the five cases of PR, all continued the same chemotherapy and achieved CR after consolidation 1, but two cases died after relapse, and three cases were still alive at last follow-up. Among the three cases of NR, one case achieved CR, one case achieved PR, and one case sustained NR, all of whom were alive after hematopoietic stem cell transplantation. Therefore, the total CR rate of this intensive protocol was 97.14% at the end of consolidation 1, and there were only 4 of 70 cases (5.7%) received HSCT in CR1 in this cohort. The detailed patient flowchart is depicted in Figure 2.
Follow-up and prognosis
The last follow-up was on 26 May 2023. The median follow-up for all patients was 5.8 (0.2–9.4) years. During the follow-up, 18 patients had bone marrow recurrence, 16 of whom were CR at the end of induction. The median recurrence time was 1.0 (0.5–2.0) years. Nine cases received HSCT in CR2, and 4 of them were alive at the follow-up. In total, 14 patients died after recurrence. The 5-year EFS was 71.2% ± 5.6% [Figure 3(a)], and the 5-year OS was 78.2% ± 5% [Figure 3(b)]. The CRR of our protocol was 27.75% [Figure 3(c)]. In order to better observe the prognosis of our protocol, we further grouped the patients according to WBC count, morphologic typing, and molecular genetics, in which the combination of FLT3/ITD, MLL arrangement, and TLS/ERG considered as poor prognostic markers. The 5-year EFS of patients with initial WBC < 100 × 109/L (n = 59) and initial WBC ⩾ 100 × 109/L (n = 11) were 76.4% ± 5.7% and 45.5% ± 15%, respectively [p = 0.013, Figure 3(d)], and the 5-year OS were 82.7% ± 5% and 54.5% ± 15%, respectively (p = 0.022). However, the 5-year EFS rates of FAB morphology subtype M2 (n = 28) and M5 (n = 36) were 78.6% ± 7.8% and 66.7% ± 8.2% [p = 0.261, Figure 3(e)], respectively. Moreover, the 5-year EFS of positive AML1/ETO was 77.3% ± 8.9%, and that of negative AML1/ETO was 68.2% ± 7% (p = 0.407). Unexpectedly, the presence or absence of combined poor prognostic genes did not appear to have an effect on survival [p = 0.492, Figure 3(f)].

(a) Kaplan-Meier curve of 5-year event-free survival among 70 pediatric patients; (b) Kaplan-Meier curve of 5-year overall survival among 70 pediatric patients; (c) The percentages of all patients with relapse; (d) Kaplan-Meier curve of 5-year event-free survival among 70 between WBC ⩾ 100 × 109/L group and WBC < 100 × 109/L group; (e) Kaplan-Meier curve of 5-year event-free survival among 70 between M2 group and M5 group; (f) Kaplan-Meier curve of 5-year event-free survival among 70 between poor prognostic marker and no poor prognostic marker group.
Analysis of risk factors for recurrence
To explore the risk factors for recurrence, a multivariate Cox regression analysis was performed, including sex, age, WBC count, FAB typing, and risk stratification, and the results showed that high WBC count (HR = 3.538, 95% CI: 1.321–9.476, p = 0.022) was the only independent risk factor for relapse or death in childhood AML in this group.
Chemotherapy-related infections
There were 650 cases of infection that occurred after chemotherapy (556 cases of induction and consolidation 1–3 and 94 cases of consolidation 4–5), and most of them can be controlled by anti-infective or symptomatic support. The percentage of TRM in this study was 1.43%. The main causes of infection were in the respiratory tract (26.92%), followed by septicemia (18.46%), stomatitis (11.85%), skin/soft-tissue infection (10.46%), and fever of unknown origin (FUO) (6.77%) (Table 2, the detail was depicted in Supplemental Table S2). When compared with induction and consolidation 1–3, this ALL-like regimen consolidation 4–5 had lower incidence of respiratory infections, sepsis, skin and soft-tissue infections, and FUO, and the differences were statistically significant (p < 0.05), but the incidence of stomatitis was not statistically significant (p = 0.157).
Infections profile.

Discussion
In this study, we presented the outcomes of the ZSYY-AML-2012 protocol, which is characterized by dual induction with timed-intensified chemotherapy and ALL-like elements in consolidation 4 and 5. The CR rate of all 70 patients at the end of induction was 88.57%, and 5-year EFS and OS were 71.2% ± 5.6% and 78.1% ± 5.2%, respectively, which preliminarily suggested that this regimen could be a promising treatment strategy for children with AML.
We first adopted a double induction scheme for childhood AML, which was used in adult AML before.25 –27 The CR rate achieved up to 88.57% without increasing the dosages. As we know, the traditional induction regimen for AML is ‘3+7’, 15 including anthracyclines and cytarabine, but the CR rate was usually only 40–80%. 28 To further improve the induction remission rate, different collaboration groups tried to find the best solution by adjusting the dose and type of anthracycline drugs or adding targeted drugs. The induction remission rate was 65–84% when VP-16 and other drugs were added in the NOPHO-AML trials, CCF-2891, and other studies.29 –32 In Europe, 17 high doses of Ara-C were used in induction, and the CR rate increased to 82.4%. However, the risk of TRM also increased due to the high intensity of chemotherapy; the introduction of effective novel therapies to the current standard AML therapy would be a key solution. Therefore, in addition to increasing dosages of drugs, examples of novel therapies of interest for pediatric AML are immunotherapies, cell signaling inhibitors against FLT3 (e.g. NCT03591510, NCT04293562), BCL2 (e.g. NCT05183035), and menin. 33 A long-term study 26 of AML in adults indicated that in terms of overall remission rate, there was no statistically significant difference between double induction and ‘3+7’ regimen, but the former required less salvaged course and did not have a higher mortality rate. Similar conclusions were likewise confirmed in AMLCG, 25 which showed double induction followed by consolidation and maintenance, proved a safe and effective strategy and a new way of delivering early intensification treatment for adult AML. However, the double induction regimen has not been clinically studied in pediatric AML. Therefore, the DAE-AE dual induction regimen was adopted in this protocol, in which the second stage of Ara-C and VP16 chemotherapy was given after an interval of 10 days at the beginning of the first stage of anthracycline plus Ara-C and VP16 chemotherapy, resulting in a final induced remission rate of 88.57%. Our preliminary results indicate that dual induction chemotherapy also has potential applications in pediatric AML.
Second, ALL-like elements, including HD-MTX and
Progress in supportive treatment also accounted for the improving outcomes for children with AML. Due to the high intensity of chemotherapy and severe myelosuppression, infection was one of the most common causes of death. 40 Infection could also lead to delayed or weakened chemotherapy and further affect the prognosis of the patients. Proper management of febrile neutropenia would reduce TRM, and the experience of our department was shared in a prior study. 41 Our better OS was mainly derived from lower TRM, whereas studies of pediatric AML showed up to 5% of all deaths during primary therapy of patients with AML are treatment related.2,42 In our study, only one person (1.42%) died during treatment. Myelosuppression and infection during the consolidation 4–5 in our study were significantly fewer than other regimens. Besides, the experienced management of febrile neutropenia in our center is another important reason for low TRM. 43
There were also some limitations in this study. First, the number of cases included in this cohort was small and not randomized controlled. Larger sample sizes and prospective randomized controlled studies are needed to confirm these conclusions. There was no statistically significant difference in 5-year EFS between AML1/ETO-positive and negative children in this study, which might also be related to the small sample size. Second, the evaluation criteria for treatment response in this study was based on morphology and only patients with NR after induction were upgraded to high risk and assigned to HSCT. Nowadays, the Minimal residual disease (MRD) is widely used to judge the prognosis and recurrence risk of AML. 44 In this study, moreover, among the five cases of PR at the end of induction, there were two cases that carried the blast cells of 6% and 7% and sustained CR without HSCT by parents’ informed consent, indicating false positives of CR and PR by morphology. In addition, the prevalence of FLT3/ITD in childhood AML was only 4.29% in this study, which was approximately 15% in most studies. A study showed that the positive rate of FLT3/ITD mutation was low in PCR-based sequencing compared to the next-generation sequencing-based method. 45 Therefore, in the subsequent SCCCG-AML-2020 protocol (ChiCTR2000038039), we performed Whole Exon Sequencing (WES) and RNA-seq for molecular genetic typing and MRD monitoring guided by FCM, quantification of Wilms’ Tumor 1 (WT1) and AML-related fusion genes to more accurately stratify treatment.
Conclusion
In conclusion, in this cohort of children with newly diagnosed AML from multiple centers and a long follow-up, we show that the CR at the end of dual induction was 88.57%, and the 5-year EFS and OS were 69% ± 6.0% and 78.1% ± 5.2%, respectively. An initial WBC count ⩾100 × 109/L was the only independent risk factor in this cohort. These results indicated that the intensive treatment protocol with dual induction and ALL-like elements is effective and safe for childhood AML, which is worth more clinical studies to confirm.
Supplemental Material
sj-docx-1-tah-10.1177_20406207241256894 – Supplemental material for Intensive chemotherapy with dual induction and ALL-like consolidation for childhood acute myeloid leukemia: a respective report from multiple centers in China
Supplemental material, sj-docx-1-tah-10.1177_20406207241256894 for Intensive chemotherapy with dual induction and ALL-like consolidation for childhood acute myeloid leukemia: a respective report from multiple centers in China by Jia-Nan Li, Yi-Jun Chen, Zhong Fan, Qiao-Ru Li, Liu-Hua Liao, Zhi-Yong Ke, Yu Li, Li-Na Wang, Cui-Yun Yang, Xue-Qun Luo, Yan-Lai Tang, Xiao-Li Zhang and Li-Bin Huang in Therapeutic Advances in Hematology
Supplemental Material
sj-docx-2-tah-10.1177_20406207241256894 – Supplemental material for Intensive chemotherapy with dual induction and ALL-like consolidation for childhood acute myeloid leukemia: a respective report from multiple centers in China
Supplemental material, sj-docx-2-tah-10.1177_20406207241256894 for Intensive chemotherapy with dual induction and ALL-like consolidation for childhood acute myeloid leukemia: a respective report from multiple centers in China by Jia-Nan Li, Yi-Jun Chen, Zhong Fan, Qiao-Ru Li, Liu-Hua Liao, Zhi-Yong Ke, Yu Li, Li-Na Wang, Cui-Yun Yang, Xue-Qun Luo, Yan-Lai Tang, Xiao-Li Zhang and Li-Bin Huang in Therapeutic Advances in Hematology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
