Abstract
PURPOSE:
Insulin like growth factor receptor 1 (IGF-1R) has been documented to play a key role in radiation response, thereby offering an attractive drug target to enhance tumor sensitivity to radiotherapy. Here, we investigated wether knockdown of IGF-1R can sensitize colorectal cancer (CRC) cell lines to radiation.
MATERIAL AND METHODS:
Human colon carcinoma SW480 and HT-29 cells were transfected with specific small interference RNA (siRNA) to mediate IGF-1R depletion. The expression of IGF-1R mRNA and protein among transfected and untransfected cells was detected by Western blot analysis. Changes in cell proliferation and radiosensitivity were evaluated by the clonogenic survival assay. NVP-ADW742, an IGF-1R inhibitor, in combination with radiation was studied. RAD51, a measure for homologous recombination repair, and 53BP1, a maker for non-homologous end-joining (NHEJ), were determined by immunofluorescence for double-strand breaks (DSB) repair pathways. Cell cycle was also examined in the IGF-1R knockdown and IGF-1R-inhibited cells.
RESULTS:
CRC cell lines were selectively sensitized to radiation after siRNA-mediated IGF-1R depletion. NVP-ADW742 efficiently increases cancer cell response to radiation. Furthermore, initial formation of RAD51 foci after IR, and 53BP1 foci were significantly reduced in IGF-1R-depleted or with IGF-1R Inhibitor CRC cell lines. Lastly, IGF-1R-depleted or with IGF-1R Inhibitor caused more G2 phase cell arrest.
CONCLUSION:
Our findings demonstrate that depletion of IGF-1R lead to an increase in radiosensitivity in CRC.
Introduction
Colon cancer is one of the most malignant tumors in humans [1, 2]. For the clinical treatment of rectal cancer, neoadjuvant radiotherapy is currently one of the most important auxiliary treatment methods, which eliminates subclinical sites by shrinking tumors, thereby reducing local recurrence, and improving sphincter retention rate and survival rate [3]. Because of the drug resistance of tumors, the treatment effects of neoadjuvant radiotherapy are challenging [4]. Thus, effectively improving the sensitivity of tumor radiotherapy is of utmost importance, and is of great significance to the clinical treatment of rectal cancer.
Many human cancers, including lung, breast and colorectal cancers, express insulin like growth factor receptor 1 (IGF-1R), which is a transmembrane tyrosine kinase [5, 6]. In previous studies, it was shown that overexpression of IGF-1R promotes the proliferation, survival, and differentiation of uncontrolled cells, thereby activating IGF-1R to promote cell survival and block apoptosis. Furthermore, the interference with IGF-1R inhibited cancer cell growth [7]. In addition, IGF-1R has been shown to be closely associated with radiation response and tumor radiosensitivity [8]. The expression of IGF-1R was increased in colon cancer cells that were exposed to radiation. Through in-depth studies, it was shown that IGF/IGF-1R signals play a role by activating a series of signal transduction events involving DNA damage repair [9]. Therefore, IGF-1R classified as part of the radiation protection mechanism. Based on this, IGF-1R has been considered to have strong tumor sensitivity to anticancer radiotherapy, and may be a potential clinical drug target.
The purpose of this study was to investigate the role of the IGF-1 system in colon cancer cellular responses to radiation, and to evaluate the effect of IGF on the expression of DNA repair genes to eventually increase the clinical applications of IGF-1-based approaches.
Method
Cell lines
Human CRC cell lines HT-29, SW480 were obtained from the Institute of Cell Biology, Chinese Academy of Science (Shanghai, China). We confirm that informed consent was obtained from all subjects. HT-29 and SW480 cells were maintained in RPMI 1640 medium (Thermo) supplemented with 10% fetal bovine serum (FBS), 1% essential amino acids, and antibiotics. Cells were cultivated at 37
Transfection with siRNA
IGF-1R siRNA and inverted control duplex were designed as described [10]. and were purchased from GeneParma Company (Suzhou, China). The sequences of IGF-1R siRNA duplex are: sense strand, 5
Western blot analysis
Cells transfected with siRNA and control cells were lysed in mammalian cell lysis buffer (10 ml of 1
Cell cycle
Cells were seeded in 6-well plates and treated with linsitinib (0.4 nM) for 1 h before IR (4 Gy), and then, cells were harvested for another 24 h after IR. Cell cycle distribution was analyzed by propidium iodide (PI) staining and flow cytometry. Briefly, after 24 h of IR, cells were collected, gently washed with cold PBS containing 2% FBS, fixed in 70% cold ethanol, and stored at
Plating efficiencies of IGF-1R knock-down cell lines from the clonogenic assay
Plating efficiencies of IGF-1R knock-down cell lines from the clonogenic assay
At 48 h after transfection, cells were plated at a cell density of 2
Immunofluorescence analysis
At 48 hours after siRNA transfection, cells were harvested, reseeded in chamber slides, and irradiated 4 hours later. At indicated times after irradiation, cells were washed, fixed, permeabilized, and incubated with blocking buffer. Primary antibodies including anti-RAD51 (Calbiochem, PC130-100UL), anti-cyclin B1 (EMD Millipore, 05-373), and anti-53BP1 (Abcam, ab21083) were added to the cells and incubated overnight. Secondary antibodies anti-mouse Alexa Fluor 488 (Invitrogen, A11017) and anti-rabbit Cyanine Cy3 (Jackson, 111-165-008) were applied together with 1
Statistical analysis
All methods were carried out in accordance with relevant guidelines and regulations. Statistical analysis was performed using SPSS 17.0 software. Comparison of the mean values was performed by Student’st-test and one-way analysis of variance (ANOVA). Colony data were analyzed using a linear-quadratic model showing the dependence of the logarithm of cellular survival on dose. The interaction between IGF-1R depletion and the radiation dose response was described as a slope modifying effect of the linear term of the linear-quadratic model (Procedure GLM, SAS/STAT 14.1, SAS Institute Inc. Version, Cary, NC, USA).
Results
IGF-1R depletion and proliferation in colorectal cancer cell lines
Western blot analysis was performed to evaluate the expression of IGF-1R protein. We confirmed that SW480 and HT-29 cells expressed IGF-1R. In our study, siRNA was used to knockdown IGF-1R in both SW480 and HT-29 cell lines. The effect of siRNA was confirmed by Western blot analysis (Fig. 1A).
Effect of IGF-1R knockdown on radiosensitivity and cell proliferation. A: The expression levels of IGF1R were examined by Western blots; B: Survival curves for IGF-1R depleted and control in CRC cell lines; C: Proliferation of cells. At day 0, 0.5 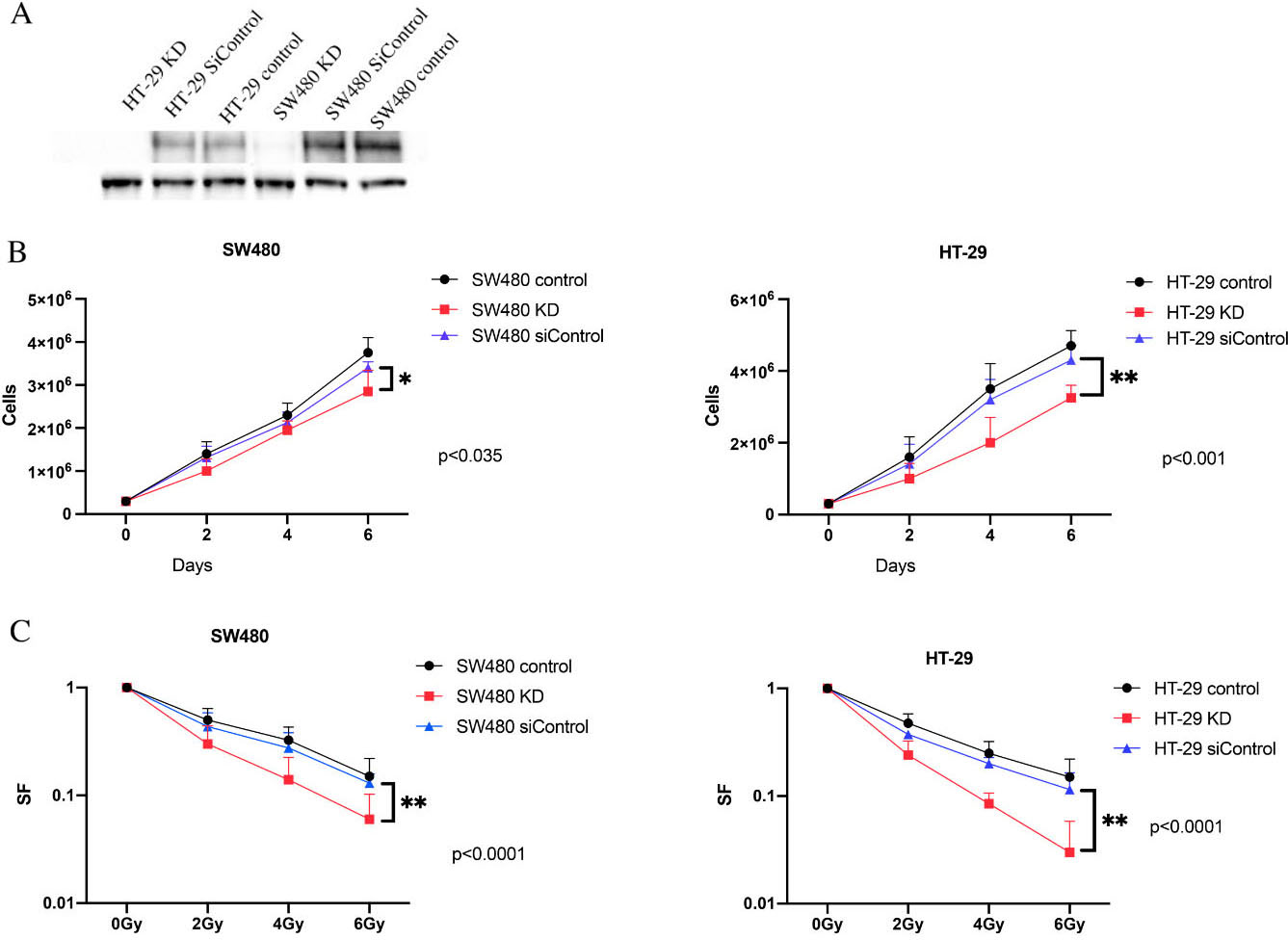
IGF-1R inhibitor NVP-ADW742 enhances the sensitivity of colorectal cancer cell lines SW480 and HT-29 cells. A: Survival curves for treatment with Radiation and Radiation plus NVP-ADW742 in SW480 cell lines; B: A: Survival curves for treatment with Radiation and Radiation plus NVP-ADW742 in HT-29 cell lines; Results from 3 independent experiments are shown, with 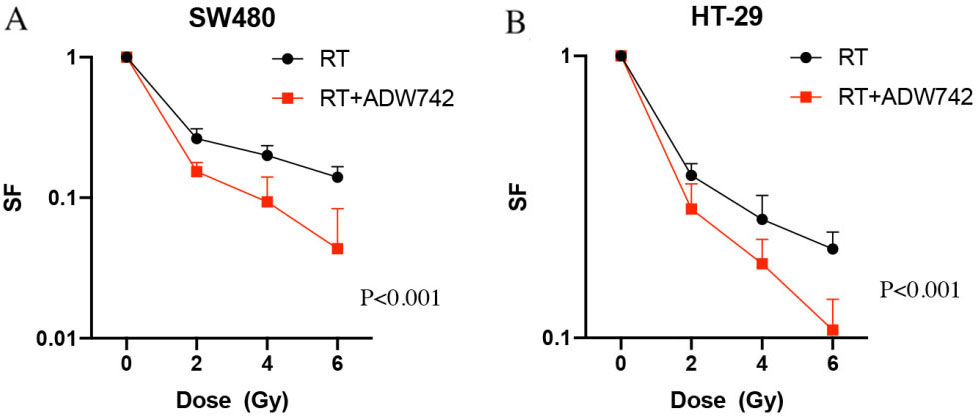
To evaluate the effect of IGF-1R knockdown on proliferation, we compared the short-term proliferative potential of SW480 and HT-29 cells after depletion of IGF-1R. Our data showed that knockdown of IGF-1R significantly inhibited proliferation in CRC cell lines (Fig. 1B). Furthermore, colony formation assays were performed using control and IGF-1R knockdown cells to evaluate the impact of IGF-1R knockdown on long-term survival. Downregulation of IGF-1R resulted in a significant decrease in plating efficiency in all cell lines (Table 1).
Effect of IGF-1R knockdown on DSB repair signaling. A: Representative immunofluorescent pictures of RAD51 foci formation 4 hours after irradiation with 4 Gy and 53BP1 foci formation 4 hours after irradiation with 0.5 Gy in SW480 cells. B: Effect of IGF-1R knockdown or with IGF-1R inhibitor on RAD51 foci formation. As a measure of HR, RAD51 foci formation in SW480 and HT-29 cells 4 hours and 24 hours after irradiation with 4 Gy; C: Effect of IGF-1R knockdown or with IGF-1R inhibitor on 53BP1 foci formation. As a measure of NHEJ, 53BP1 foci formation was analyzed at 1 h (initial) and 24 h (residual) after irradiation. IGF-IRi: IGF-1R inhibitor, *: 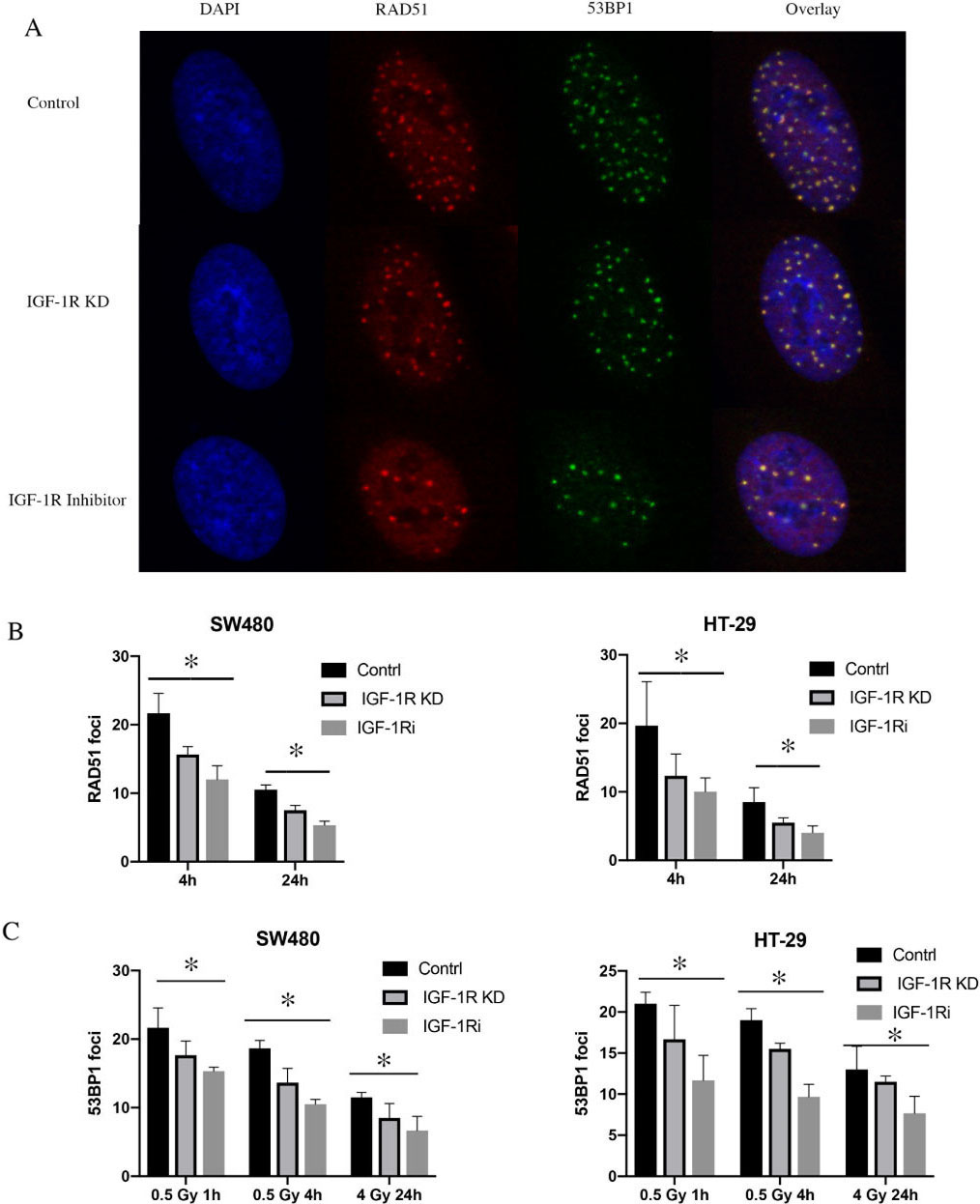
Effect of ARID1B knockdown on cell cycle progression. i: IGF-1R inhibitor.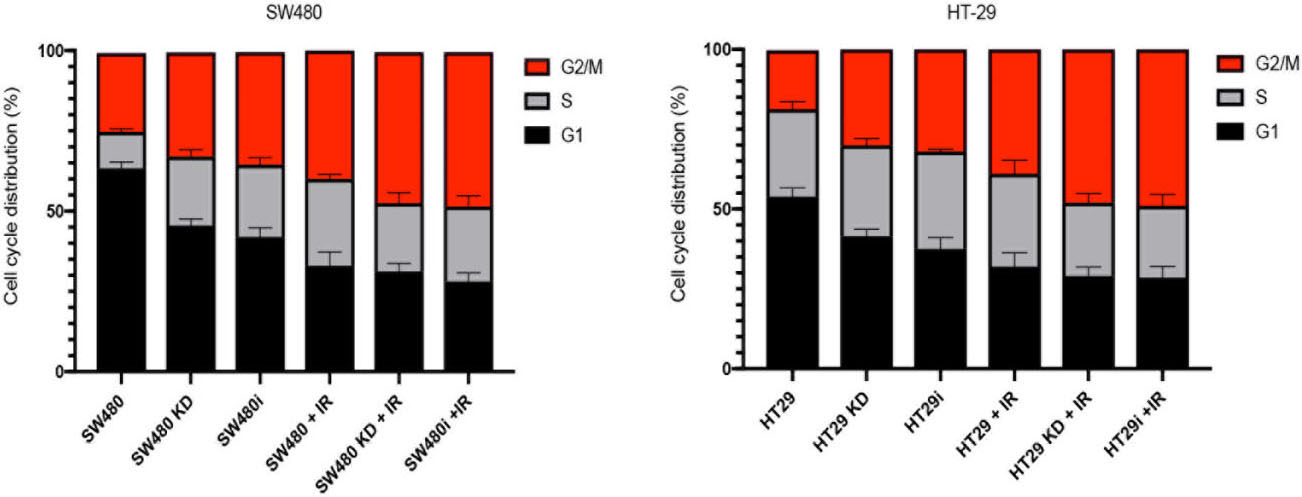
Taken together, the results from the short-term proliferation assay and the clonogenic survival assay revealed a significant inhibition in proliferation and clonogenic ability after IGF-1R knockdown.
To evaluate the impact IGF-1R knockdown on radiation sensitivity, colony formation assays were performed (Fig. 1C). Our data showed that IGF-1R knockdown significantly reduced the surviving fraction in CRC cells and the radiation sensitivity of all IGF-1R knockdown cell lines was increased over radiation dose levels of 2–6 Gy when compared to the control (
NVP-ADW742 enhances the sensitivity of colorectal cancer cell lines SW480 and HT-29 cells
NVP-ADW742 is novel IGF-I receptor (IGF-IR) kinase inhibitor. In our study, Colony formation assay showed that colony formation capacity of SW480 cells treated with NVP-ADW742 plus radiation showed a more pronounced decrease than that obtained for either NVP-ADW742 or radiation alone (Fig. 2A). Similar results were also obtained with HT-29 cells (Fig. 2B).
Depletion of IGF-1R inhibits DSB repair pathways in CRC cell lines
To elucidate the underlying mechanism of radiosensitization in IGF-1R knockdown cell lines, the RAD51 foci formation as a marker of HRR was determined. Following IGF-1R knockdown or with NVP-ADW742, an IGF-1R inhibitor, cells were irradiated with 0 and 4 Gy and stained at 4 and 24 hours after irradiation (Fig. 3A). RAD51 foci formation was evaluated in cells as a pathway for HRR DNA damage repair (Fig. 3B). Depletion of IGF-1R and with IGF-1R inhibitor significantly reduced the number of RAD51 foci at 4 hours and 24 hours after irradiation, compare with control in our cell lines (
Next, we investigated 53BP1 foci formation as a marker of non-homologous end-joining (NHEJ) activity following DNA damage. IGF-1R knockdown or with IGF-1R inhibitor affected 53BP1 foci formation in cell lines at 1, 4, and 24 hours following irradiation with 0, 0.5, and 4 Gy (Fig. 3C).
Induces cell cycle arrest cell cycle
To investigate whether Depletion of IGF-1R radiosensitizes colon cancer cells via redistributing cell cycle, we performed cell cycle assay on our colon cancer cells exposed to IR with or without IGF-1R knockdown/IGF-IR inhibitor.
Figure 4 showed that both SW480 and HT-29 cell with IGF-1R knockdown or with IGF-1R inhibitor lead to higher proportion of cells in phase G2, compared with control (
Our results suggest that the radiosensitization effects of Depletion of IGF-1R is due to the augmentation of damage by cycle arrest.
Discussion
Neoadjuvant radiotherapy has become an effective treatment to improve the outcome in patients with rectal cancer [11]. As the resistance to radiotherapy emerges as an important problem, developing novel strategies that enhance the radiosensitivity of cancer cells has become an urgent need. Many studies have focused on the overexpression of IGF-1R and have shown that under some conditions, which could mediate cancer cells’ evasion from apoptosis, thereby favoring tumor malignancy and resistance to radiotherapy.
In this study, we first examined the level of IGF-1R expression in colon cancer lines, which confirmed IGF-1R overexpression in the colon cancer cells used. In some studies, it was reported that IGF-1R expression positively correlated with the tumor resistance to radiotherapy [12]. The data from our colony formation assay revealed that IGF-1R knockdown sensitized CRC cells without significantly affecting the proliferation or survival faction, thereby highlighting the specific effect on radiosensitivity. In addition, we use NVP-ADW742, a novel IGF-I receptor (IGF-IR) kinase inhibitor, in our study to verify the radiosensitivity caused by inhibition of IGF-1R. As results, NVP-ADW742 could significan tly enhance the radiosensitivity in HT-29 and SW480 cells. This inhibitor results again confirm that depletion of IGF-1R could improve radiosensitivity in colon cancer treatment.
To further investigate the underlying mechanisms of radiosensitization, we investigated whether IGF-1R knock-down affected the formation of DSB repair foci. Since previous studies showed that IGF-1R inhibition involved both NHEJ and HRR [13], we detected RAD51 foci after irradiation, as a measure for homologous recombination repair, and 53BP1 foci as a maker for NHEJ. Consistent with data derived from the clonogenic survival assay, depletion of IGF-1R had a significant impact on the radiation induced RAD51 foci formation in CRC cell lines, and we confirmed this result by using IGF-1R inhibition. In addition, we detected 53BP1 foci formation, a marker of NHEJ, and showed that in IGF-1R knock-down cell lines or treated with IGF-1R inhibitor could decrease accessibility of 53BP1 to DNA lesions resulted in reduced NHEJ activity. Chitnis et al. showed that IGF-1R depletion enhanced radiosensitivity and impaired ATM kinase activity in murine melanoma cells [13], indicating that IGF-1R inhibition attenuated repair by NHEJ. Furthermore, Venkatachalam et al. showed that IGF-1R knock-down significantly down-regulated the mRNA levels of BRCA2 [14], a major protein involved in homologous recombination DNA repair. Taken together, our findings are consistent with previous data, and confirmed that IGF-1R attenuated repair by both NHEJ and HR.
In conclusion, our findings show that radiosensitivity in CRC cell lines can selectively be increased through IGF-1R depletion, thereby suggesting that IGF-1R may be a potential therapeutic target for increasing the radio-sensitivity in CRC.
Author contributions
Conception: Rui Zong and Shan Xu
Interpretation or analysis of data: Rui Zong, Shan Xu and XiaoMan Chen
Preparation of the manuscript: Rui Zong and JingJing Feng
Revision for important intellectual content: Shan Xu and JingJing Feng
Supervision: Shan Xu
Source of funding
None.
Ethical statement
The study was approved by the insititutional review board of Qingdao Central Hospital. All methods were carried out in accordance with relevant guidelines and regulations.
Footnotes
Acknowledgments
The authors thank the reviewers for their helpful comments on this article. The authors are also grateful to Mogo Editing for polishing and revising the language.
Conflict of interest
The authors have declared no conflict of interests.
