Abstract
Colorectal cancer is the third most frequently diagnosed cancer and the combination of radiation with capecitabine has been shown to achieve only 15% to 25% of pathologic complete response. This study aimed to investigate the effect of MLN4924, a potent small molecule inhibitor of SKP1-Cullin-F-box proteins E3 ubiquitin ligases, as a novel radiosensitizing agent in colorectal cancer cells. Indeed, we found that MLN4924 effectively sensitized colorectal cancer cells to radiation with a sensitivity-enhancement ratio of 1.61 for HT-29 cells and 1.35 for HCT-116 cells. Mechanistically, MLN4924 significantly enhanced radiation-induced G2/M arrest, apoptosis, and DNA damage response through accumulation of p27. Knockdown of p27 via small interfering RNA partially inhibited MLN4924-induced radiosensitization, indicating a causal role played by p27. Our study suggested that MLN4924 could be further developed as a novel radiosensitizing agent against colorectal cancer.
Introduction
Colorectal cancer is the third most frequently diagnosed cancer and the third leading cause of cancer death in the United States. 1 Neoadjuvant chemoradiation (CRT) followed by total mesorectal excision is the standard of care for patients with locally advanced rectal cancer (LARC). However, the 5-year disease-free survival and overall survival were about 75% and 65%, respectively. 2 Indeed, the combination of radiation with capecitabine has been shown to achieve only 15% to 25% of pathologic complete response and 50% to 60% of downstage. 3 Thus, new combinational therapies are urgently needed.
The SKP1-Cullin-F-box protein (SCF) E3 ubiquitin ligases, consisting of an adaptor protein SKP1, a scaffold protein cullin, a substrate receptor F-box protein, and a RING protein RBX1 or RBX2, are responsible for the ubiquitination of ∼20% of all ubiquitinated proteins for targeted degradation by proteasome.
4
–6
MLN4924, a potent small molecule inhibitor of NEDD8 (neural precursor cell-expressed developmentally downregulated protein 8)-activating enzyme (NAE), has demonstrated inhibitory activity against SCF E3 ligases via inhibiting cullin neddylation. By inactivating SCF E3 ubiquitin ligases, MLN4924 caused the accumulation of a number of SCF E3 substrates to induce apoptosis, autophagy, and senescence, thus inhibiting the tumor growth both
Most recently, MLN4924 was found to act as a novel radiosensitizing agent by causing accumulation of several substrates of SCF E3 to enhance radiation-induced DNA damage, G2/M arrest, and apoptosis in pancreatic and breast cancer cells. 11,12 Here we extended our MLN4924 radiosensitization study to colorectal cancer cells and reported that MLN4924 is an effective radiosensitizing agent against colorectal cancer cells via a p27-dependent mechanism.
Materials and Methods
Compound
MLN4924 was synthesized by Medchem Express and was dissolved in dimethyl sulfoxide to make a 10-mmol/L stock solution and kept at −20°C before use.
Cell Culture
Colorectal cancer lines, including HCT-116, HT-29, SW620, and SW480, were purchased from Biochemistry and Cell biology Institute of Shanghai, Chinese Academy of Sciences, within 3 months of experiments. HCT-116 and HT-29 were cultured in McCoy’s 5A medium and SW620 and SW480 were cultured in Leibovitz’s L-15 medium both containing 10% fetal bovine serum. Cells were maintained at 37°C in a humidified atmosphere containing 5% CO2. The experiments were carried out in the exponential phase of growth.
Western Blot Analysis
Methods for Western blot analysis have been described previously. 11 Briefly, after being blocked, the membranes were incubated with specific primary antibodies against CDT1, WEE-1, NOXA, BIM-EL, CHK1, CHK2 (Santa Cruz Biotechnology, China), ORC-1, p21, p27 (BD Biosciences, China), and β-actin (Hua’an Biotech, Inc, China) at 4°C overnight. The membranes were washed with Phosphate buffer solution tween (PBST) 3 times, for 10 minutes each. Protein bands were visualized using an enhanced chemiluminescence detection system (Thermo Scientific, China)
Radiation Exposure and Clonogenic Assay
Cells were pre-seeded into 60-mm plastic Petri dishes at a certain density and then irradiated with 6-MV X-rays, using a linear accelerator (Varian Medical Systems, Palo Alto, California) at 3 Gy/min. A standard colony formation assay was used to obtain survival curves for the tested cells. Colorectal cancer cells were plated onto 60-mm plastic Petri dishes to generate 30 to 100 colonies per dish. The cells were irradiated at a series of dose levels (0, 2, 4, 6, and 8 Gy). The cells were then incubated for 10 to 14 days to allow colonies to develop. The colonies were fixed with 4% paraformaldehyde and stained with 0.1% crystal violet (100% methanol solution) before being counted. Irradiations were performed in the Experimental Irradiation Core of the Fudan University Shanghai Cancer Center.
Small Interfering RNA Knockdown of p27
Expression of p27 was knocked down with the small interfering RNA (siRNA) oligonucleotide. The target sequences of p27 messenger RNA were selected to suppress
Fluorescence-Activated Cell Sorting Analysis
Cells were treated with MLN4924 or exposed to radiation alone or in combination. Cells were harvested 24 hours post radiation and analyzed by flow cytometry.
Statistical Analysis
All experiments were repeated at least 3 times independently. Data are means ± standard deviation. Statistical comparisons of the average values of 2 groups were performed using the Student
Results
Sensitivity of Colorectal Cancer Lines to MLN4924 as a Single Agent
We first determined the sensitivity of 4 colorectal cancer lines, HCT-116, HT-29, SW620, and SW480 to radiation alone. We observed a heterogeneous response. The most sensitive cell line was HCT-116 and the most resistant cell line was HT-29 (Figure 1).Therefore, we estimated the IC50 (Half growth inhibition concentration) of MLN4924 in HT-29 and HCT-116. In a standard clonogenic survival assay with MLN4924 in the culture medium for 24 hours, MLN4924 caused a dose-dependent inhibition of colony formation with an IC50 of 50 to 100 nmol/L in HT-29 and HCT-116 (Figure 2). Thus, MLN4924 is a potent inhibitor of cell proliferation and survival in colorectal cancer cells.

Four colorectal cancer cell lines showed a heterogeneous sensitivity to radiation. The most sensitive cell line was HCT-116 and the most resistant cell line was HT-29.

HT-29 and TCT-16 were treated with various concentrations of MLN4924 for 24 hours and then MLN4924 was washed out. Cells were cultured in MLN4924-free medium for additional 10 to 14 days, allowing colony formation.
MLN4924 Sensitized Colorectal Cancer Cells to Radiation
We therefore used 50 nmol/L of MLN4924 in our radiosensitization experiment. Cells were pretreated with MLN4924 (50 nmol/L) for 24 hours and followed by radiation at different doses of up to 8 Gy. MLN4924 was washed out 24 hours post radiation, and cells were cultured in MLN4924-free medium for an additional 10 to 14 days, allowing colony formation. Under this condition, we found that MLN4924 effectively sensitized HT-29 and HCT-116 to radiation with a sensitivity-enhancement ratio (SER) of 1.61 and 1.35, respectively (Figure 3). Thus, we conclude that MLN4924 is a potent radiosensitizer against colorectal cancer cells.

Radiosensitization by MLN4924: Cells were seeded in 60-mm dishes and treated with MLN4924 and radiation as indicated. The colonies with more than 30 cells were counted after 10 to 14 days. Surviving fraction was calculated as the proportion of seeded cells following irradiation to form colonies relative to that of untreated cells (mean ± SD, n = 3). SER was calculated as the ratio of the mean inactivation dose under untreated control conditions divided by the mean inactivation dose after MLN4924 treatment. SD indicates standard deviation; SER, sensitivity-enhancement ratio.
Radiosensitization by MLN4924 is Attributable to Enhanced G2/M Arrest and Induction of Apoptosis
To determine the nature of MLN4924 radiosensitization, we performed cell cycle profile of HT-29 and HCT-116 treated with MLN4924, radiation, or MLN4924 in combination with radiation using fluorescence-activated cell sorting analysis and found that MLN4924 remarkably enhanced the radiation-induced G2/M arrest in both colon cancer lines (Figure 4A and C). In addition, MLN4924 increased the radiation-induced subG1 apoptotic population (Figure 4B and D). These results suggested that radiation-induced disruption of cell cycle progression and apoptotic cell death can be further enhanced by MLN4924.
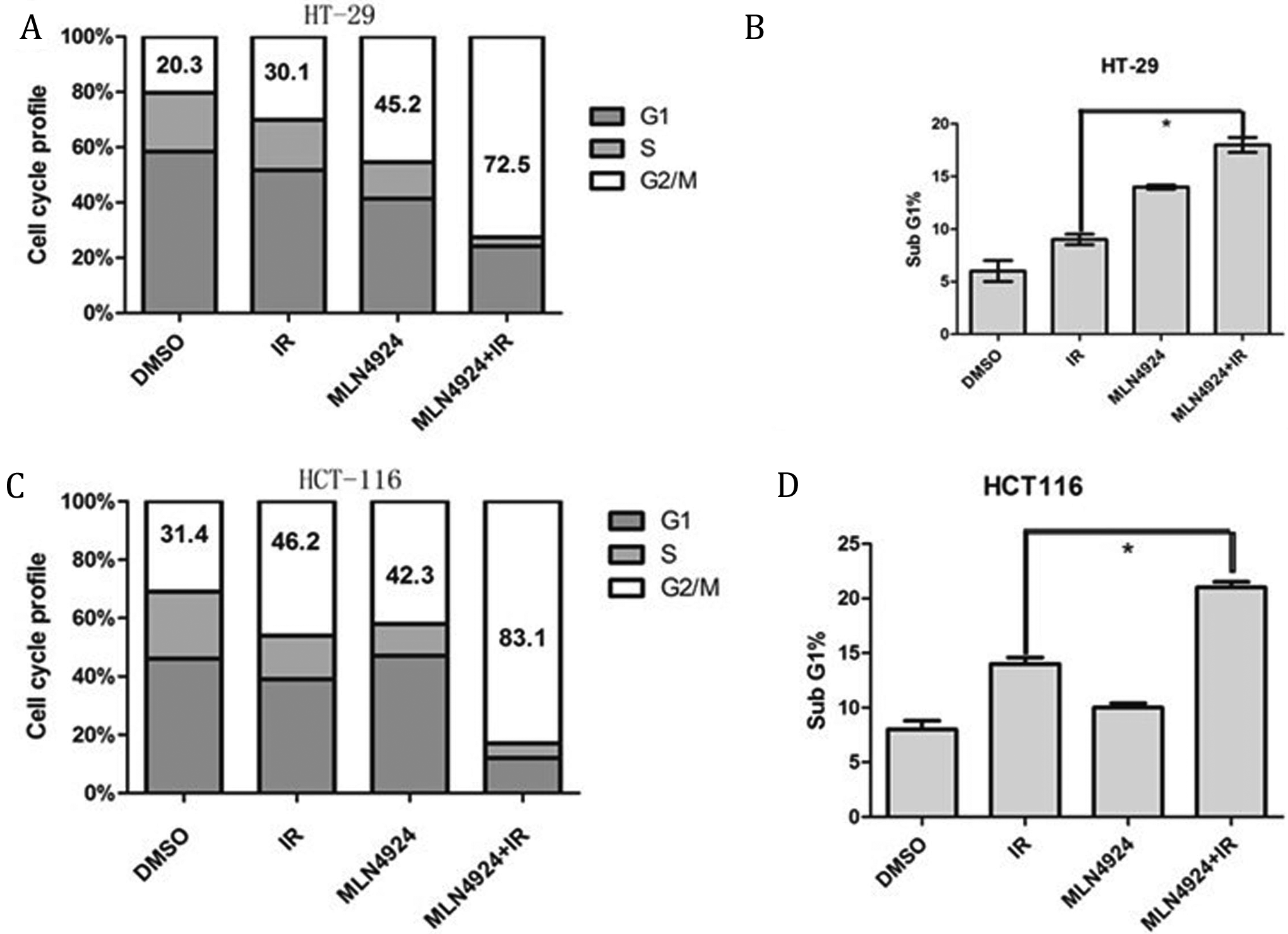
MLN4924 alters cell cycle progression and induces apoptosis. Cells were treated with MLN4924 at 50 nmol/L for 24 hours, alone or in combination with radiation (6 Gy), followed by FACS analysis for cell cycle distribution (A and C) and sub-G1 population for apoptosis (B and D). Shown is mean (A and C) or mean ± SD (B and D; n = 3): *
MLN4924 Had Effect on Radiation-Induced DNA Damage Response
Since the major cellular effect of ionizing radiation is to cause DNA damage and trigger the DNA damage response, we, therefore, examined whether MLN4924 treatment would enhance radiation-induced DNA damage. We determined the DNA damage response upon MLN4924-radiation treatment by measuring phosphorylation of CHK1 and CHK2 and found that the levels increased after single treatment, the combination treatment caused a further increase in both colon cancer lines (Figure 5). Thus, MLN4924 had a role in enhancing radiation-induced DNA damage response, which could contribute to its radiosensitizing effect.

MLN4924 radiation triggers DNA damage response. Subconfluent HT-29 and HCT-116 cells were treated with MLN4924 (50 nmol/L) or radiation (6 Gy) alone or in combination for indicated time periods of up to 24 hours, followed by immunoblotting (IB) analysis using indicated antibodies.
MLN4924 Caused Accumulation of SCF E3 Ligase Substrates, Which is Responsible for G2/M Arrest and Apoptosis
We found that as expected, the levels of cell cycle regulators, such as p21, p27, and WEE1; DNA licensing proteins CDT1 and ORC1; and apoptosis inducer NOXA but not BIM-EL increased substantially upon treatment with MLN4924 but not radiation (Figure 6). Unlike what were observed in pancreatic and breast cancer cells, 11,12 the MLN4924-radiation combination did not further increase the levels of CDT1, WEE1, or P21 but did caused a further increase of p27.Thus, p27 is the only SCF E3 ligase substrate, among all tested substrates, that was induced by either MLN4924 or radiation, leading to a higher level of accumulation upon combination in both colon cancer lines.

MLN4924 induces accumulation of SCF E3 ligase substrates. Subconfluent cells were treated with MLN4924 (50 nmol/L) or radiation (6 Gy) alone or in combination for 24 hours, followed by immunoblotting (IB) analysis using indicated antibodies.
Partial Rescue of G2/M Arrest, Apoptosis, and Radiosensitization by siRNA Knockdown of p27
Given the fact that MLN4924 enhanced G2/M arrest and apoptosis by radiation and caused radiosensitization in HT-29 cell, and that p27 was the only SCF E3 substrate whose accumulation by MLN4924 or radiation was further enhanced by the combination. We reasoned that if p27 was critical to MLN4924-induced enhancement of radiation effects, we should be able to abrogate these effects, at least in part by their simultaneous knockdown. Upon p27 knockdown (Figure 7A), degree of apoptosis was reduced from 19.8% to 14.2% (Figure 7B), and the G2/M arrest was partially reversed from 67.7% to 53.9% (Figure 7C), indicating that p27 accumulation contributes at least in part to substantial apoptosis and G2/M arrest. More importantly, MLN4924-induced radiosensitization was remarkably inhibited upon p27 knockdown with SER value reduced from 1.53 to 1.23 (Figure 7D). These results clearly suggest a causal effect of p27 accumulation (upon SCF E3 inactivation by MLN4924) on MLN4924 radiosensitization in HT-29 cell.
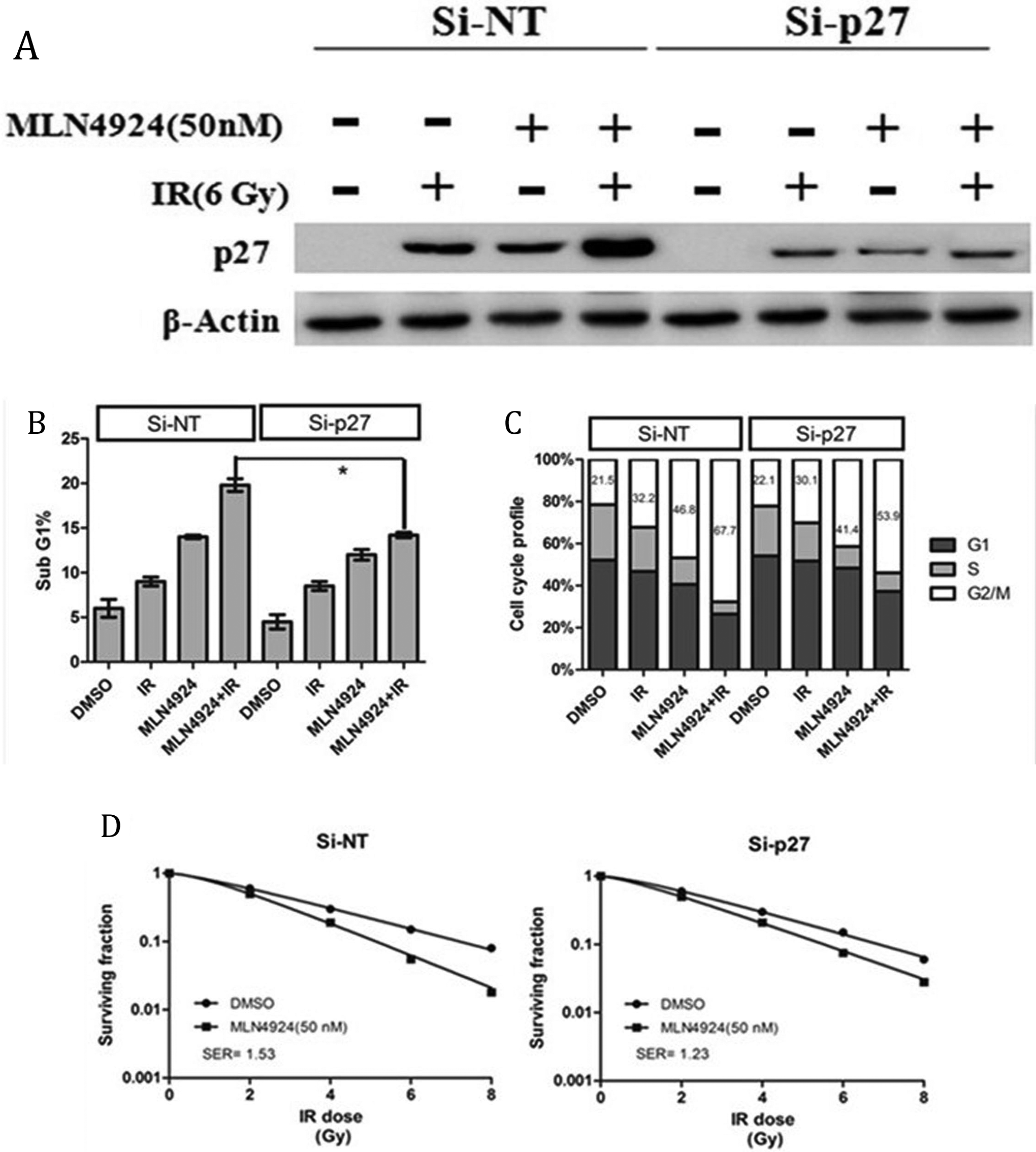
Radiation-enhancing activity of MLN4924 is inhibited at least in part by siRNA knockdown of p27. HT-29 cells were transfected with siRNA oligonucleotides targeting p27. Forty-eight hours later, one portion of the cells was subjected to Western blotting (A), the other portion was analyzed by FACS for sub-G1 apoptotic population (B, mean ± SD, n = 3) or cell cycle profile (C, mean number shown, n = 3), and still other portion was plated for clonogenic assay (D, mean ± SD, n = 3): *
Discussion and Conclusion
Colorectal cancer is the third most frequently diagnosed cancer and the third leading cause of cancer death in the United States. 1 The standard of care for patients with LARC is neoadjuvant CRT followed by total mesorectal excision, and treatment response to neoadjuvant chemoradiotherapy is an early surrogate marker and correlates with clinical outcomes. 14 However, the combination of radiation with capecitabine has been shown to achieve only 15% to 25% of pathologic complete response and 50% to 60% of downstage. 3 Thus, development of radiosensitizing agents against colorectal cancer is in high demand for the improvement of therapeutic outcome. To pursue this end, we estimated the effect of MLN4924 in sensitizing the colorectal cancer cells.
In this study, we showed here that MLN4924 is a potent radiosensitizing agent against colorectal cancer cells. Mechanistic study revealed that MLN4924 enhanced radiation-induced G2/M arrest, apoptosis, and DNA damage response. Our rescue experiment demonstrated that MLN4924-induced radiosensitization was inhibited upon p27 knockdown with SER value reduced from 1.53 to 1.23 (Figure 6D). These results clearly suggest a causal effect of p27 accumulation (upon SCF E3 inactivation by MLN4924) on MLN4924 radiosensitization in colon cancer cells. On the other hand, incomplete rescue of MLN4924-triggered radiosensitization by p27 knockdown might be attributable to incomplete p27 elimination (Figure 6A) and accumulation of other yet to be identified SCF E3 substrates.
P27 is a 198-amino acid protein whose gene maps to the short arm of chromosome 12 and was originally discovered in cells arrested by transforming growth factor α.
15
It is known as a negative regulator of the cell cycle. Using p27/Kip1 knockout mice, it has been demonstrated that p27-deficient mice show a more rapid clonal expansion.
16
In addition to the cell cycle regulation activity, p27/Kip1 has also been demonstrated to be involved in the regulation of cancer cell proliferation, cell differentiation, and apoptosis.
17
Chen
In clinic, low or absent p27 expression has been frequently observed in many human cancers, including breast, prostate, gastric, lung, and colorectal cancer (CRC). 19 –25 Moreover, downregulation of p27 was strongly associated with aggressive tumor biology and poor prognosis. 19 –25 In colorectal cancer, a number of studies agree that high levels of p27 expression are associated with a well-differentiated tumor histology and good overall survival, whereas low levels are associated with aggressive tumor behavior and poor clinical outcome. 13,26,27 In this study, accumulation of p27 by radiation or MLN4924 induced G2/M arrest and apoptosis. Thus, determination of p27 levels plays an important role in the treatment of colorectal cancer.
In summary, our study revealed radiosensitizing activity of MLN4924, a small molecule inhibitor of SCF E3, in colorectal cancer, and elucidated its mechanisms of action, which include accumulation of p27. Our study, therefore, provides an appealing piece of evidence for future development of MLN4924 as a novel radiosensitizing agent against colorectal cancer.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
