Abstract
BACKGROUND:
Long non-coding RNA-growth arrest specific transcript 5 (lncRNA-GAS5) plays a suppressive role in activated hepatic stellate cells (HSCs). LncRNAs could circulate in the blood in a cell-free form and serve as promising biomarkers for various human diseases. Herein, we investigated the feasibility of using serum GAS5 as a biomarker for liver fibrosis in chronic hepatitis B (CHB) patients and whether promoter methylation was responsible for GAS5 down-regulation.
METHODS:
Serum GAS5 levels were quantified using quantitative real-time PCR in CHB patients and healthy controls. GAS5 promoter methylation was examined in LX-2 cells and cirrhotic tissues.
RESULTS:
Compared with the sera from healthy controls, lower GAS5 levels were found in the sera from CHB patients. Receiver operating characteristic curve analysis indicated that serum GAS5 had a significant diagnostic value for liver fibrosis in CHB patients. Serum GAS5 negatively correlated with HAI scores as well as ALT values in CHB patients. GAS5 was additionally reduced in cirrhotic tissues, associated with its hypermethylation promoter. In LX-2 cells, transforming growth factor-
CONCLUSION:
We demonstrate that epigenetically-regulated serum GAS5 could serve as a potential biomarker in CHB patients. Loss of GAS5 is associated with DNMT1-mediated promoter methylation.
Introduction
It has been estimated that 248 million people worldwide are infected with hepatitis B virus (HBV), which is the main cause of cirrhosis and even hepatocellular carcinoma (HCC) [1]. There are still 93 million chronic HBV carriers or 7.8% of the population in China, even if the application of HBV vaccine [2]. Chronic HBV infection, responsible for the main viral hepatitis-related death, results in a heavy healthcare burden worldwide. Patients with chronic HBV infection is characterized by progressive liver fibrosis and inflammation. Therefore, effective early detection of liver fibrosis is important for the control and treatment of hepatic fibrosis progression in patients with chronic hepatitis B (CHB).
Up to now, liver biopsy is still the gold standard for assessing the existence and staging of liver fibrosis. However, the application of biopsy is often hampered by its invasiveness, possible complications and potential sampling errors [3, 4]. Therefore, we urgently need to find a simple non-invasive serum marker to evaluate disease progression in CHB patients.
Evidence is accumulating that circulating RNAs such as microRNAs (miRNAs) and long non-coding RNAs (lncRNAs) in peripheral blood serum, plasma and cells are promising biomarkers for various human diseases [5]. Tsui et al. recently reported that circulating miRNAs in plasma or serum are used to determine the progression and prognosis of human diseases [6]. For example, serum miR-124 has been found to serve as a new biomarker for liver necroinflammation in CHB patients [7]. Interestingly, circulating lncRNAs may serve as a diagnostic biomarker in liver fibrosis. We previously found that serum lincRNA-p21 is a novel biomarker in CHB patients [8]. However, seldom studies are performed on exploring the roles of circulating lncRNAs in liver fibrosis.
LncRNAs represent a group of transcripts that great than 200 bp in length with no or little translational potential [9]. Increasing evidence suggests that lncRNAs participate in the regulation of gene expression at the level of epigenetics, transcription and post-transcription [10]. Lines of evidence reveals that lncRNAs play a pivotal role in cell apoptosis, cell proliferation, cell differentiation, and organismal homeostasis [11, 12, 13]. Moreover, abnormalities of lncRNAs can also lead to human diseases, including cancers [14]. In organ fibrosis, our previous study showed that growth arrest specific transcript 5 (GAS5) contributes to the suppression of liver fibrosis via miR-222 and p27 [15]. However, whether serum GAS5 is a potential biomarker for liver fibrosis remains unclear. This study aimed to explore the clinical significance of the expression of GAS5 in the serum of CHB patients. We also investigated the relationship between GAS5 expression reduction and its promoter methylation.
Materials and methods
Patient selection
Serum samples were derived from 518 CHB patients attending in the First Affiliated Hospital of Wenzhou Medical University from 2007.1 to 2017.12. No antiviral treatment was performed in all patients and liver biopsy was applied for liver staging and grading. Meanwhile, we recruited 420 healthy controls (no previous liver medical history). Inclusion criteria for CHB patients were patients with positive serum hepatitis B surface (HBs) antigen as well as serum HBV DNA for more than half a year. Exclusion criteria were: (i) younger than 16 years old, (ii) infected with human immunodeficiency virus (HIV), (iii) liver damage from other causes, such as hepatitis C virus (HCV), alcoholism, autoimmune hepatitis, or drug intake, (iv) serious systemic disease, (v) pregnancy and lactation [16]. In another study, 10 healthy controls and 10 CHB patients with cirrhosis were selected for partial hepatectomy or liver biopsy, who were also from the First Affiliated Hospital of Wenzhou Medical University from 2017.01 to 2017.03. The applications of liver biopsy and/or the typical appearance of the liver by abdominal ultrasound and/or computed tomography scan were used to diagnose liver cirrhosis. The study was approved by the ethics committee of the First Affiliated Hospital of Wenzhou Medical University (Wenzhou, China), and informed consent forms signed by all patients were obtained. All procedures were implemented in accordance with the current international general standards, the human experiment standards of the Ethics Committee of the First Affiliated Hospital of Wenzhou Medical University, and the 1975 Declaration of Helsink revised in 2008.
siRNA sequences
siRNA sequences
A 16-gauge Menghini needle was used for liver biopsy. Following the advice of the physicians, the sample length of each liver biopsy case is not less than 2.0 cm. Next, these samples were treated with 4% formalin fixing, paraffin-embedding and hematoxylin-eosin staining. The results were evaluated by experienced liver pathologists [17]. Finally, at least 8–10 portal tracts in samples are required to admit patients. The Ishak scoring system was used to assess histological activity index (HAI) and fibrosis stage (F0
Blood sampling
Blood samples were taken from all CHB patients during liver biopsy. Once obtained, the blood samples were centrifuged at 3400 g for 7 min under the normal conditions. Next, the serum was obtained and further centrifuged at 12000 g at 4
Cell culture
The human LX-2 cell strain was obtained from JENNIO Biological Technology (Guangdong, China). Cells were cultured in DMEM containing 10% fetal bovine serum, 100 U/ml penicillin G sodium salt and 100 U/ml streptomycin sulfate (Gibco, Carlsbad, CA, USA), and incubated at 37
CCK-8 assay
Cells were plated in 96-well plates at a density of 1
RNA interference analysis
TGF-
Quantitative real-time PCR (qRT-PCR)
Total RNA was isolated from cell or tissue samples using the miRNeasy Mini Kit (Qiagen, Valencia, CA, USA). Next, total RNA was reversed into cDNA using the ReverTra Ace qPCR RT kit (Toyobo, Osaka, Japan). Then, SYBR Green real-time PCR Master Mix (Toyobo, Osaka, Japan) was used to measure the expression of the target gene using ABI 7500 system software. All the primers in this study were shown as follows (GAS5: forward 5’-CTTCTGGGCTCAAGTGATCCT-3’, reverse 5’-TTGTGCCATGAGACTCCATCAG-3’; alpha-1(I) collagen (Col1A1): forward 5’-CCCCGAGGCTCTGAAGGT-3’, reverse 5’-GCAATACCAGGAGCACCATTG-3’; GAPDH: forward 5’-GCACCGTCAAGGCTGAGAAC-3’, reverse 5’-TGGTGAAGACGCCAGTGGA-3’). GAPDH, as an internal control, was used to standardize the relative abundance of GAS5 and Col1A1 mRNA. The expression level of GAS5 in sera was calculated using
DNA extraction and methylation analysis
Genomic DNA was extracted from cells and liver tissues using a mammalian genomic DNA extraction kit (Beyotime, Shanghai, China), as described previously [19]. The prediction of GAS5 CpG island was found in UCSC Genome Browser. Sodium bisulfite-treated genomic DNA was used for PCR. The GAS5 primers for PCR were 5’-AGAGTAGAACTGCGGAGAAGC-3’ and 5’-TAGCGACAGGTAAAGGATACACT-3’. The bisulfite-sequencing analysis was performed as described previously [20].
Patient characteristics
Patient characteristics
Data were analyzed using SPSS 13.0 (IBM, Armonk, NY). The Mann-Whitney test or Kruskal-Wallis test was performed to determine the significance of serum GAS5 levels. Receiver operating characteristic (ROC) curves were generated to assess the diagnostic potential of serum GAS5 via calculation of the area under the ROC curve (AUC), sensitivity and specificity according to standard formulas. Differences between multiple groups in LX-2 cells were evaluated using one-way analysis of variance. Data were considered significant at
The value of serum GAS5 in the diagnosis of liver fibrosis in CHB patients. (A) Ct values of serum GAS5 levels in CHB patients (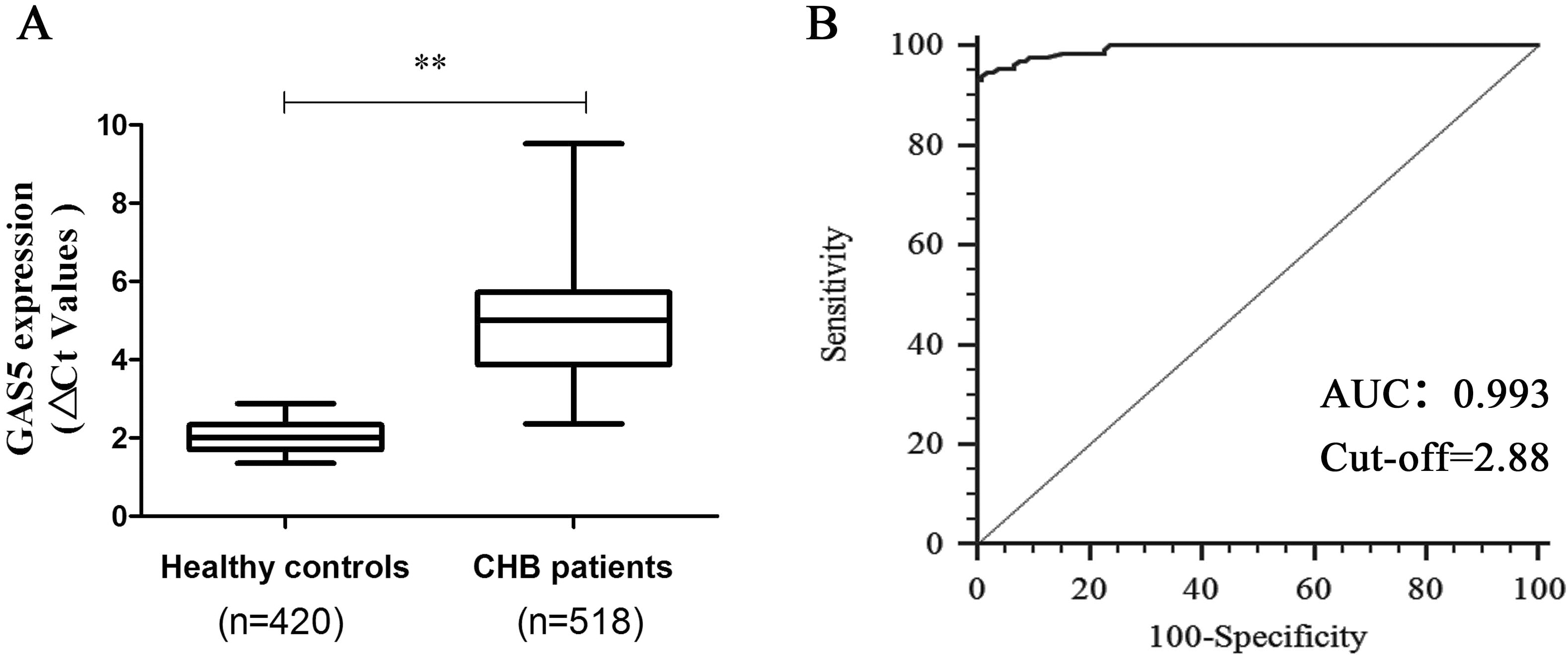
Serum GAS5 is correlated with necroinflammation in CHB patients with liver fibrosis. (A) Ct values of serum GAS5 levels in patients with F0-F2, F3-F4 or F5-F6 stages. (B) Ct values of serum GAS5 levels in CHB patients with HAI score 2–5, HAI score 6–8 and HAI score 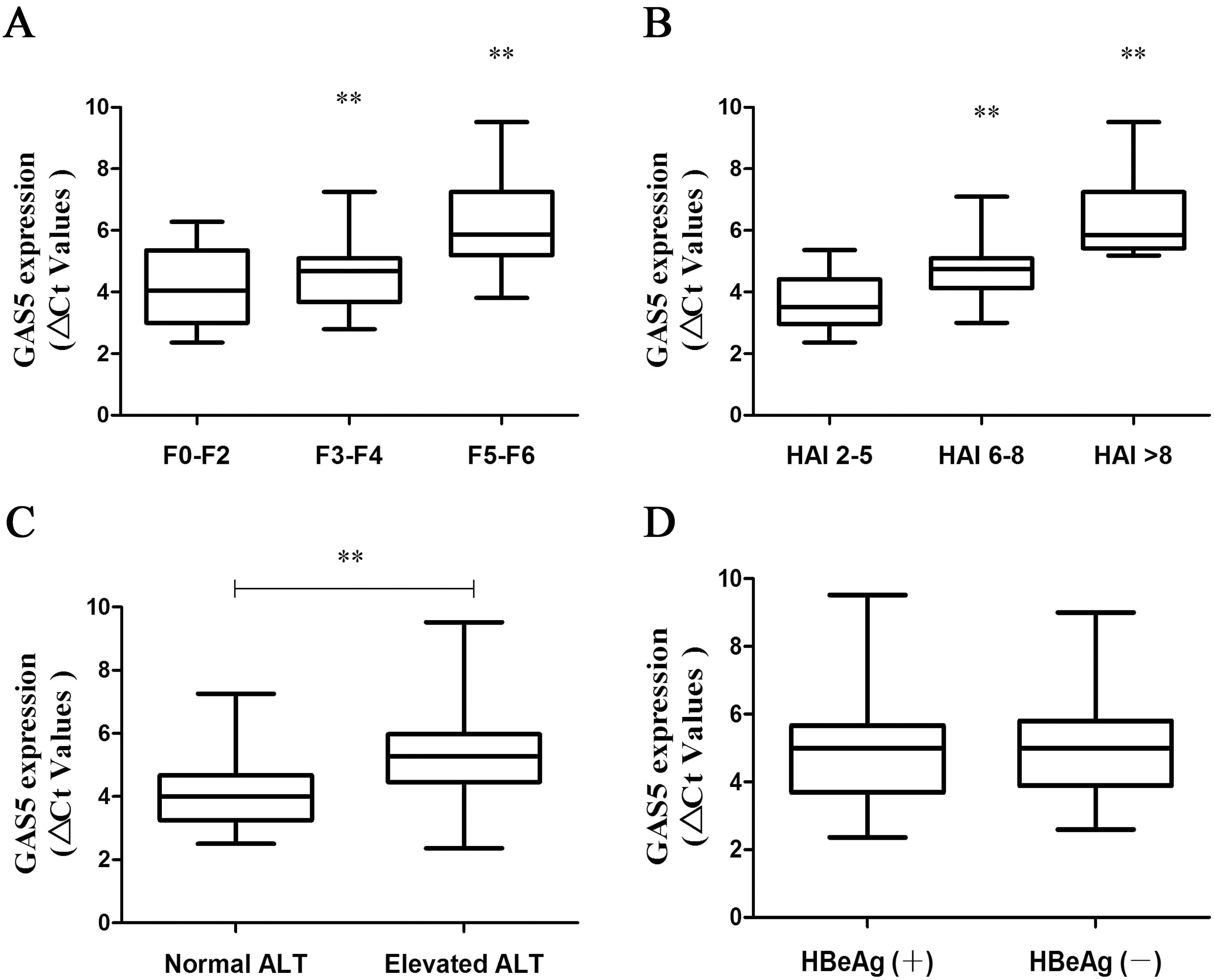
GAS5 expression and its promoter methylation status in patients with cirrhosis. (A) Schematic representation of a CpG island in the GAS5 promoter region. Gray block indicate a CpG island and there are 20 CpG sites in this CpG island. (B) The methylation of GAS5 was detected by bisulfide sequencing in the liver tissues from patients with cirrhosis (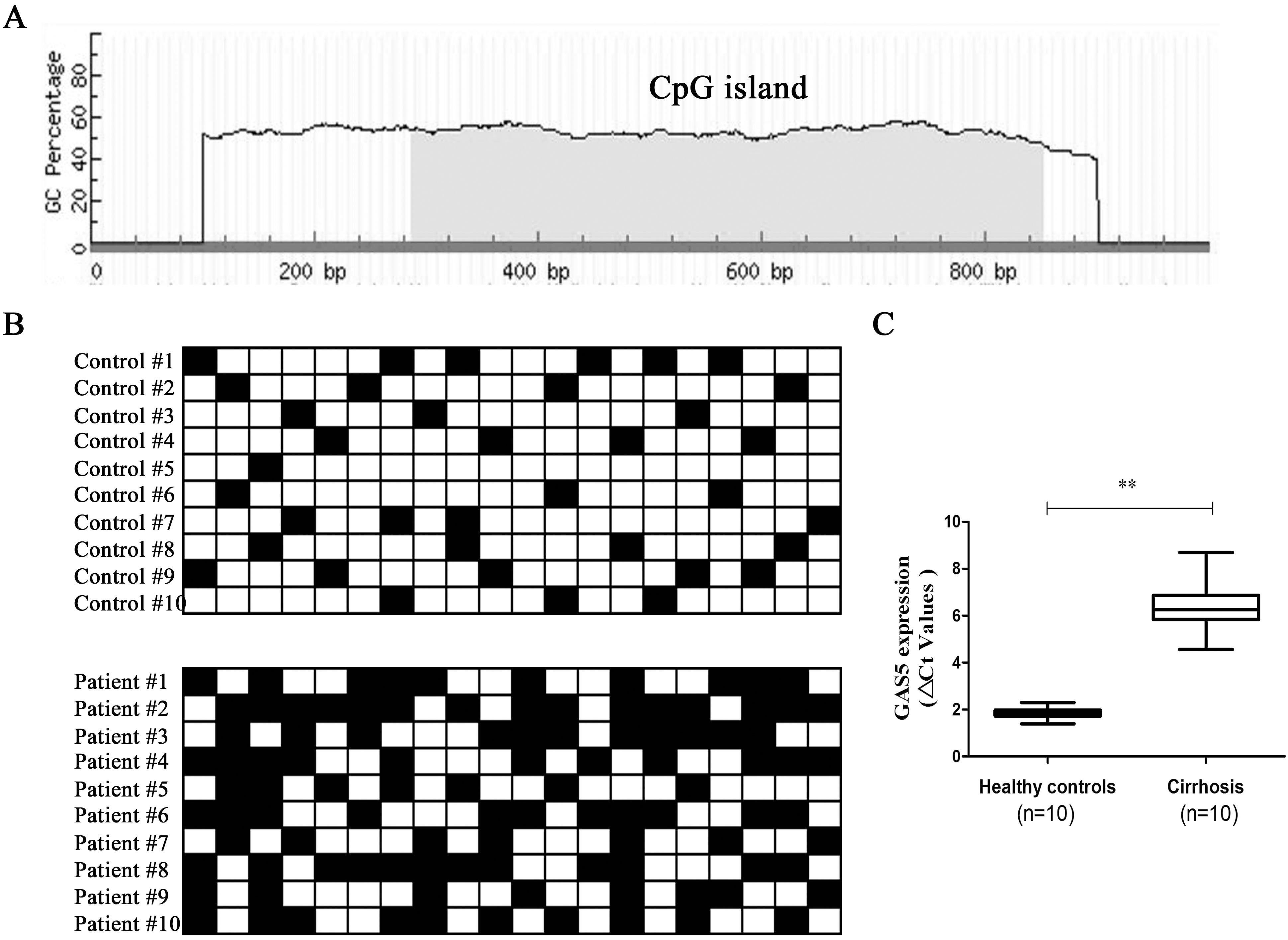
The value of serum GAS5 in the diagnosis of liver fibrosis in CHB patients
Due to down-regulation of GAS5 has been found in human cirrhotic tissues and rat fibrotic liver tissues [15], whether GAS5 was dysregulated in sera of CHB patients and the diagnostic value of serum GAS5 for liver fibrosis were explored. Then, 518 CHB patients and 420 healthy controls were recruited in this study (Table 2). It was confirmed that there were no significant differences in age between CHB patients and healthy controls (
Serum GAS5 is correlated with necroinflammation in CHB patients with liver fibrosis
According to the fibrosis scores, all patients were classified as three groups including low-score group (0–2), medium-score group (3–4) and high-score group (5–6). Next, the association between serum GAS5 and fibrosis stages was investigated. Clearly, serum GAS5 level was decreased in advanced fibrosis stages (Fig. 2A), indicating that serum GAS5 may be a potential biomarker in CHB patients. To explore the relation between serum GAS5 and liver damage as well as inflammation, we correlated the serum GAS5 expression with HAI scores. Notably, reduced serum GAS5 was observed in CHB patients in a HAI score-dependent manner (Fig. 2B). In line with it, serum GAS5 was down-regulated in patients with elevated ALT when compared with those with normal ALT (Fig. 2C), indicating that serum GAS5 expression was associated with inflammation and liver damage. Further studies were performed to confirm whether serum GAS5 expression is associated with HBV replication. As shown in Fig. 2D, there was no significant change in serum GAS5 expression between HBeAg (
Down-regulated GAS5 is associated with its promoter methylation
Recently, it has been demonstrated that aberrant methylation could be found in lncRNA promoters and responsible for its expression down-regulation [21]. Using bioinformatics analysis, a CpG island was observed in GAS5 promoter (Fig. 3A). Then, GAS5 methylation as well as its expression was determined in another cohort of CHB patients with cirrhosis (
GAS5 expression and its promoter methylation status in TGF-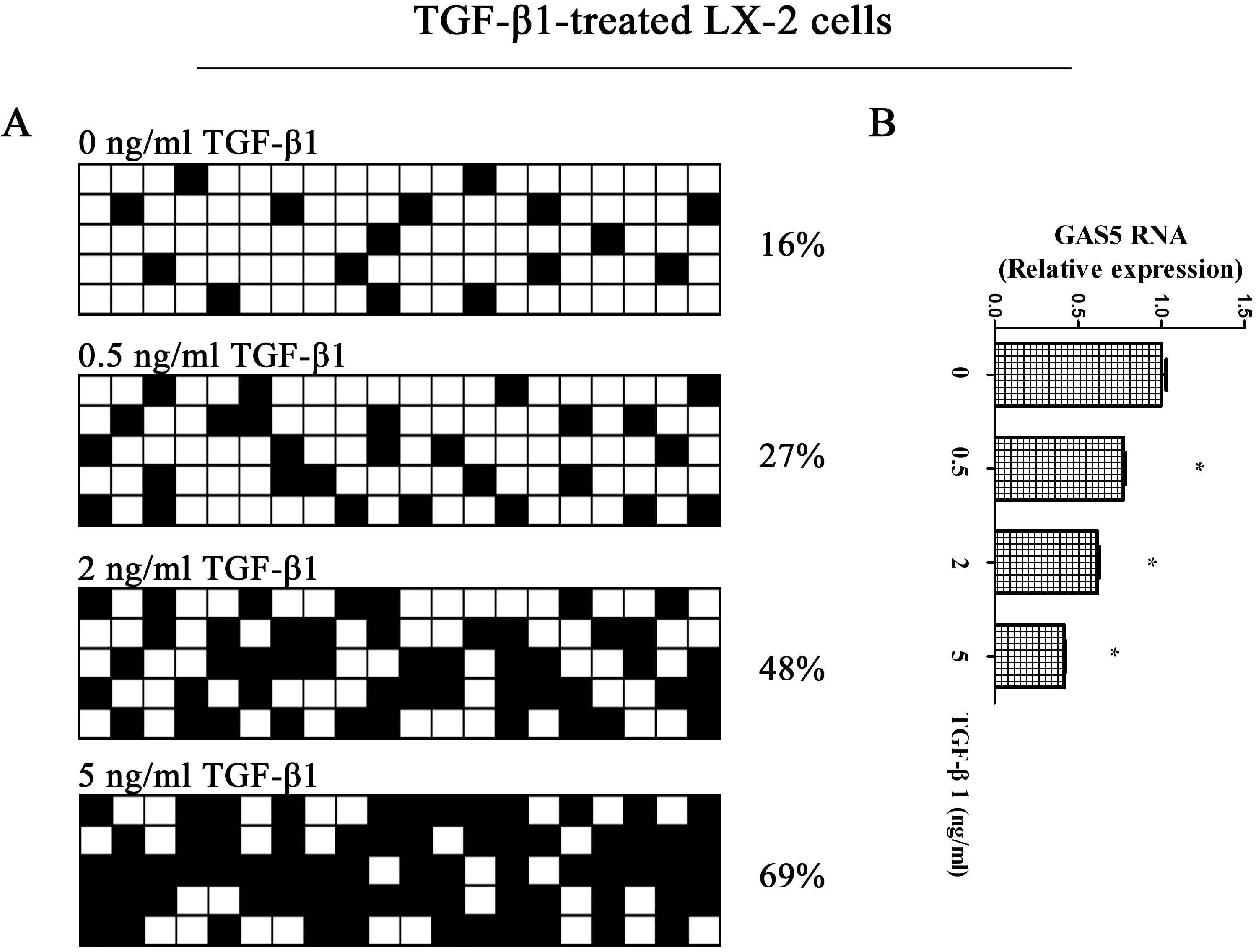
The hypermethylation of GAS5 is inhibited by 5-Aza. (A) The methylation of GAS5 was detected by bisulfide sequencing in TGF-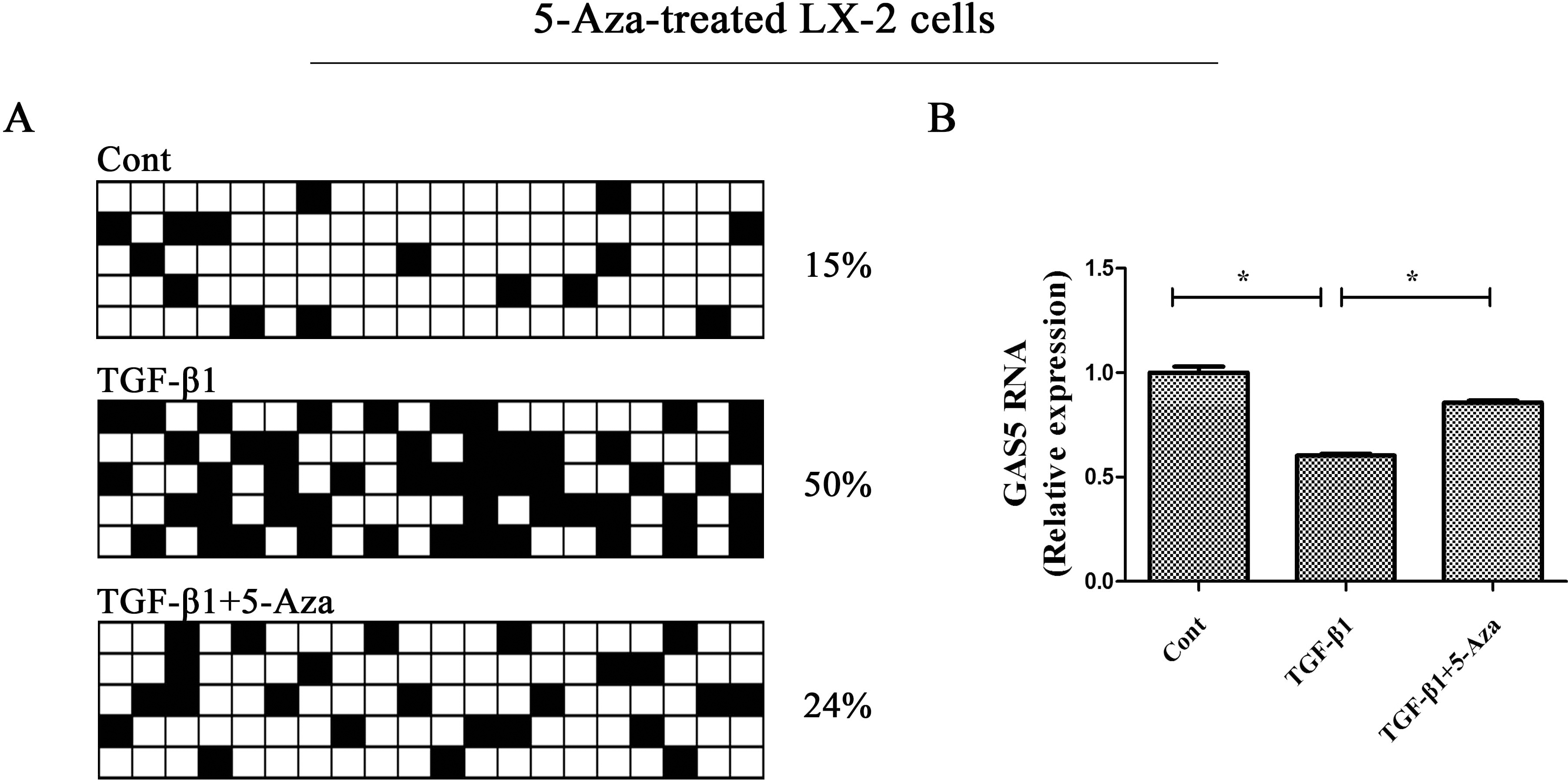
Demethylation of GAS5 inhibits HSC activation. LX-2 cells were treated with TGF-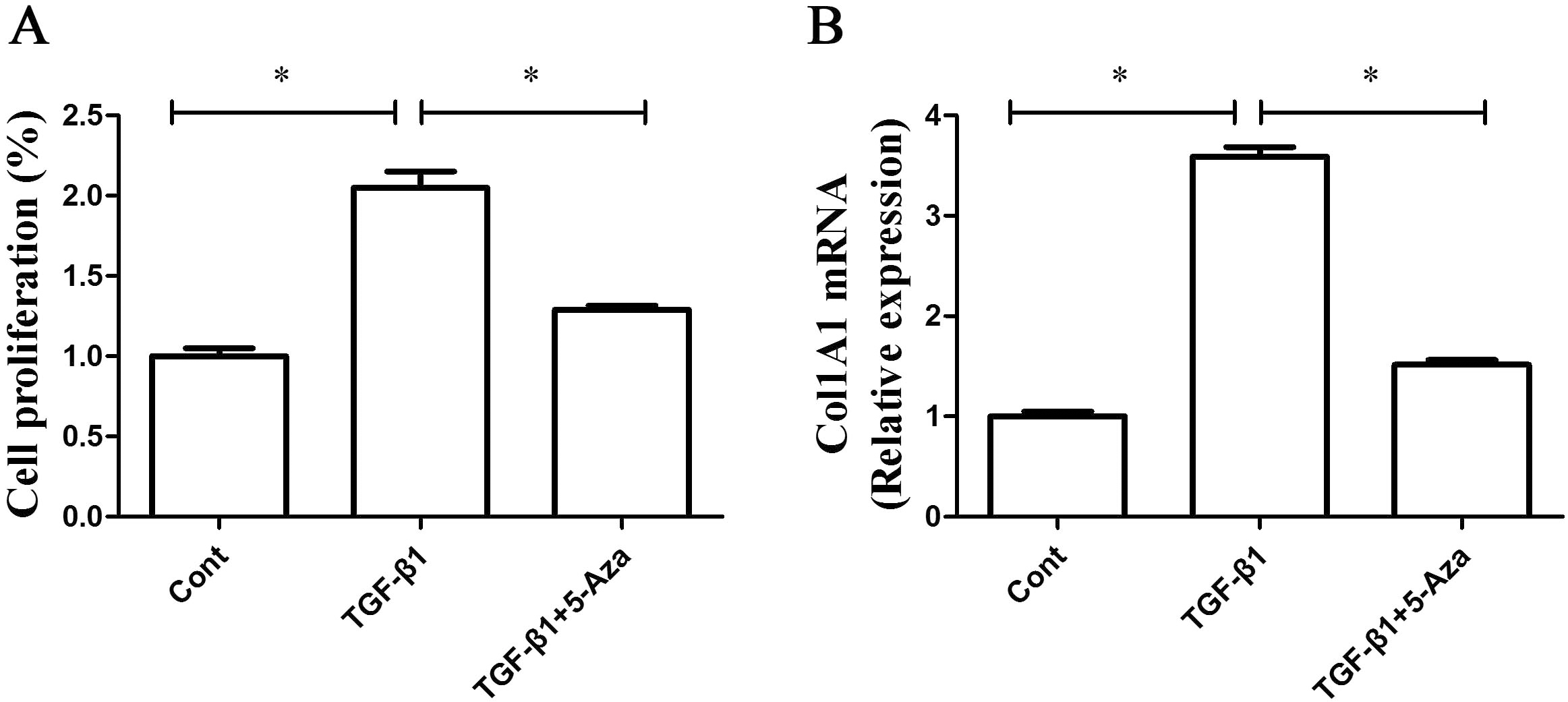
DNMT1 participates in the methylation of GAS5. LX-2 cells were treated with TGF-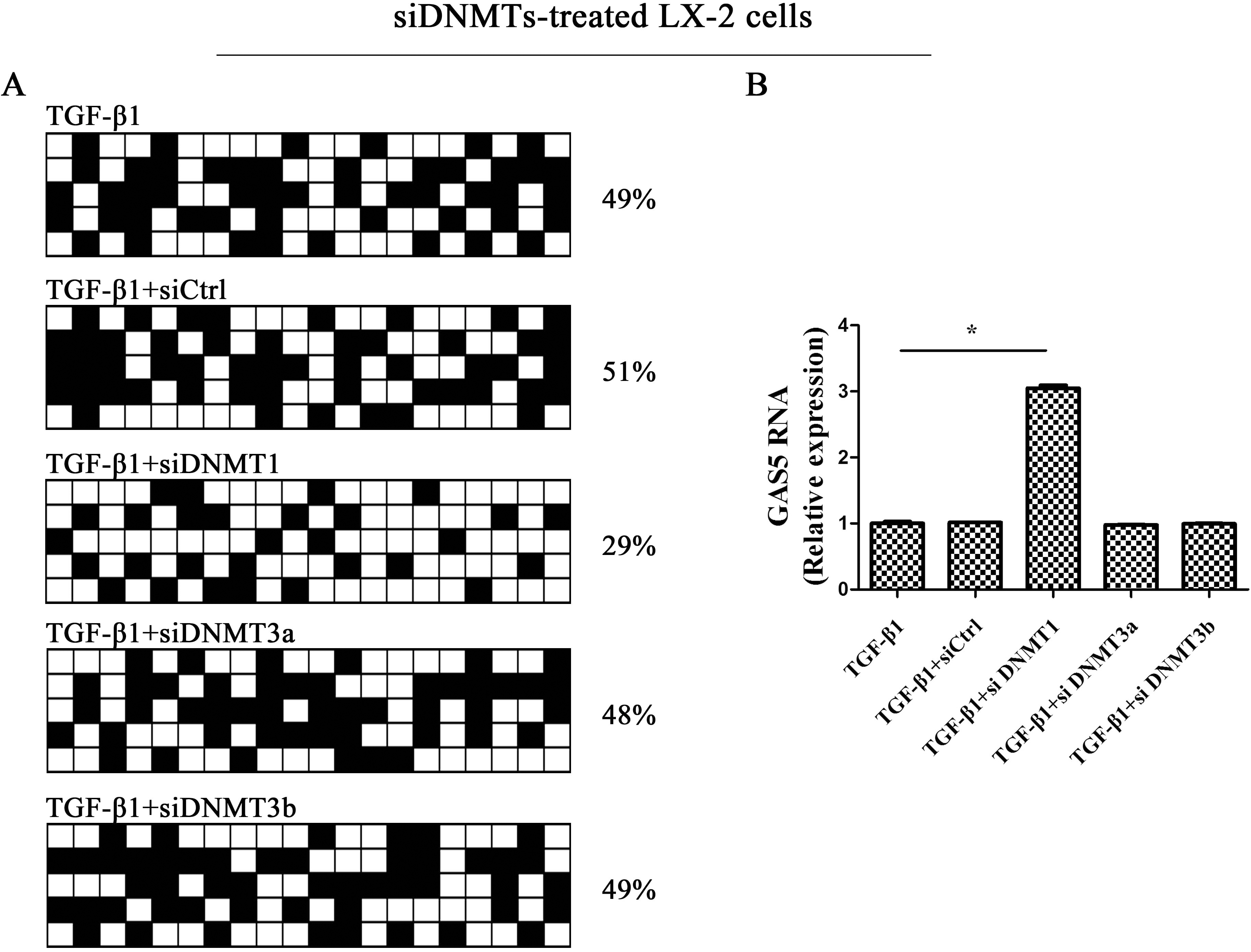
Whether the restoring of GAS5 expression by DNMT inhibitor contributes to the suppression of activated HSCs was further examined. As indicated by Fig. 6A, TGF-
DNMT1 participates in the methylation of GAS5
Mammalian DNA is dominantly methylated in the C-5-position of complimentary CpG bp DNMTs, including DNMT1, DNMT3a and DNMT3b [22]. To further determine which members of DNMTs are involved in the methylation of GAS5, GAS5 expression and methylation status were examined in cells with DNMT1 siRNA, DNMT3a siRNA or DNMT3b siRNA. It was found that TGF-
Discussion
In this study, we demonstrated that down-regulated serum GAS5 levels in CHB patients may be a promising biomarker of liver fibrosis. Loss of serum GAS5 levels was correlated with necroinflammation in CHB patients with liver fibrosis. Interestingly, liver GAS5 levels were also reduced in the cirrhotic tissues, which was consistent with the changes in serum GAS5 levels during liver fibrosis. We interfered that serum GAS5 may be from the release from the liver, but it needs more validation in future. It is well known that activation of HSCs is a key event in liver fibrosis. GAS5 was obviously reduced in TGF-
GAS5, characterized as a tumor suppressor, is often lowly expressed in various cancers [23, 24]. It has been reported that GAS5 is involved in many cellular processes such as cell proliferation, cell apoptosis, cell migration and invasion [25]. In addition, loss of GAS5 is often associated with poor prognosis in many cancers [26]. Recently, Li et al. found that GAS5 is down-regulated via aberrant methylation in breast cancer cells [27]. In gastric cancer, the reduction in GAS5 is associated with its promoter hypermethylation [28]. Our results showed an obvious methylation in GAS5 promoter in the cirrhotic tissues as well as activated HSCs, similar with the results in cancers [27, 28]. Notably, GAS5 was poorly expressed in the above tissues and cells. Further studies demonstrated that inhibiting DNMT1 contributed to the restoration of GAS5 expression. In sum, loss of GAS5 is associated with its hypermethylation status.
DNA methylation is an epigenetic mechanism that can regulate genetic performance without changing the DNA sequence [29]. Increasing evidence has shown that aberrant DNA methylation is often associated with the suppression of transcriptional activity and finally leads to the dysregulation of many vital biological processes such as fibrosis [30]. Generally, DNMTs, including DNMT1, DNMT3a and DNMT3b, are responsible for the regulation of the global patterns of DNA methylation. Among them, DNMT1 has been reported to maintain the methylation of newly replicated DNA [31]. In our study, it was found that only DNMT1, not DNMT3a or DNMT3b, was involved in the methylation of GAS5. Inhibiting of DNMT1 restored GAS5 expression, associated with the demethylation of GAS5, indicating that DNMT1 may be responsible for GAS5 methylation during liver fibrosis.
There are many advantages in this study. Firstly, epigenetically-regulated GAS5 could be found in liver fibrosis. Secondly, GAS5 methylation was mediated by DNMT1 in activated HSCs. Lastly, serum GAS5 may serve as promising biomarker for liver fibrosis.
In conclusion, serum GAS5 could serve as a potential biomarker in CHB patients for liver fibrosis. During liver fibrosis, GAS5 down-regulation is caused by DNMT1-mediated promoter methylation.
Footnotes
Acknowledgments
The project was supported by the National Natural Science Foundation of China (No. 81873576), the Medical Health Science and Technology Project of Zhejiang Provincial Health Commission (No. 2020RC081), Wenzhou Municipal Science and technology Bureau (No. Y20190076), the Key Laboratory of diagnosis and treatment of severe hepato-pancreatic diseases of Zhejiang Province (No. 2018E10008) and the project of Wenzhou Medical University Basic Scientific Research (No. KYYW201904).
Conflict of interest
The authors declare that they have no competing interests.
Authors’ contributions
Conception: Yong Guo and Jianjian Zheng.
Interpretation or analysis of data: Yong Guo, Chunxue Li, Rongrong Zhang, Yating Zhan, Jinglu Yu and Jinfu Tu.
Preparation of the manuscript: Yong Guo and Jianjian Zheng.
Revision for important intellectual content: Jianjian Zheng.
Supervision: Jianjian Zheng.
