Abstract
Colorectal cancer (CRC) has become the third most common cause of cancer-related deaths. CRC occurs because of abnormal growth of cells that can invade other tissues and cause distant metastases. Researchers have suggested that aberrant microRNA (miRNA) expression is involved in the initiation and progression of cancers. However, the key miRNAs that regulate the growth and metastasis of CRC remain unclear. The circulating miRNAs from BALB/c mice with CRC CT26 cell implantation were assayed by microarray. Then, Mus musculus (house mouse) mmu-miR-762 mimic and inhibitor were transfected to CT26 cells for analysis of cell viability, invasion, and epithelial-mesenchymal transition (EMT), cell cycle, and regulatory molecule expression. Human subjects were included for comparison the circulating Homo sapiens (human) has-miR-762 levels in CRC patients and control donors, as well as the patients with and without distant metastasis. The result for miRNA levels in mice with CRC cell implantation indicated that plasma mmu-miR-762 was upregulated. Transfection of mmu-miR-762 mimic to CT26 cells increased cell viability, invasion, and EMT, whereas transfection of mmu-miR-762 inhibitor decreased the above abilities. Cells treated with high-concentration mmu-miR-762 inhibitor induced cell cycle arrest at G0/G1 phase. However, mmu-miR-762 did not cause apoptosis of cells. Western blot analysis showed that mmu-miR-762 mimic transfection upregulated the expression of Wnt-1 and
Keywords
Introduction
Colorectal cancer (CRC) is epithelial in origin and localized to the large intestine and rectum. CRC has been ranked among the top five most common cancers worldwide. Because of the aging population, westernization of diet, excessive intake of fat, reduced intake of dietary fiber, and other factors, the incidence of CRC continues to increase and has become more prevalent in younger people [1]. The most common treatment for CRC is surgery to remove the tumor before metastasis occurs, which shows a very high cure rate. Moreover, combinations of chemotherapy drugs such as 5-fluorouracil, camptothecin, bevacizumab, and cetuximab are effective for CRC treatment [2]. However, metastasis is the cause of death in most CRC cases; depending on the tumor stage, liver and lung metastases occur in 20–70% and 10–20% of cases, respectively [3]. It has been reported that approximately 50% of patients with CRC develop tumor metastases which are associated with very poor prognosis and treatment efficiency [4].
MicroRNAs (miRNAs) are non-translated RNAs that regulate gene expression by degrading mRNA or inhibiting translation [5]. This endogenous mechanism is common to animals, plants, and viruses. miRNAs have been shown to be involved in many biological processes, such as embryonic development, inflammation, cell cycle regulation, cell differentiation, apoptosis, and tumor metastasis [6]. In recent years, studies have shown that the occurrence of cancer and tumor formation are closely related to miRNA regulation of transcriptional gene expression [7] and miRNAs can be used as indicators for diagnosing cancer [8]. For example, the miRNAs involved in the development of CRC and other cancers include miR-17-5p [9], miR-21 [10], miR-31 [11], miR-92 [12], miR-198 [13], and miR-203 [14]. These miRNAs can be used as biomarkers for cancer diagnosis diagnose or targeted therapy.
Therefore, identifying miRNAs that regulate the growth of CRC and clarifying the regulation of tumor formation and metastasis may reveal disease-specific biomarkers to detect the occurrence or prognosis of cancers. These results would also provide an important foundation for the research and development of advanced treatments in new target therapy strategies. In our previous miRNA array analysis in animal experiments, we detected some miRNAs that may be involved in the growth of CRCs. Among these, plasma Mus musculus (house mouse) mmu-miR-762 was found to be upregulated in CRC CT26 cell-transplanted BALB/c mice, suggesting that miR-762 is a potential biomarker for CRC growth regulation. CT26 cells are the CRC line which have been reported with the aggressive and high metastatic activity. Therefore, we assessed the effects of mmu-miR-762 on the viability, colony formation, invasion, cell cycle, and regulatory molecules of CRC CT26 cells by transfection with miRNA mimics and inhibitors. Finally, blood samples were collected from patients with CRC, and the effects of Homo sapiens (human) has-miR-762 in control subjects and cancer cases were compared. These results by analysis in CRC cell line, mouse model, and human subjects would provide insight into the role of miR-762 in regulating CRC. Through this study, it will be used as a reference for future application of miR-762 as a potential biomarker, and even for evaluating future development of new drugs to CRC treatment.
Materials and methods
Cells and cell culture
CT26 cells are undifferentiated CRC cell lines produced in N-nitroso-N-methylurethane-induced BALB/c mice and possess aggressive and high metastasis activities. The cells were purchased from American Type Culture Collection (ATCC, Manassas, VA, USA). CT26 cells were cultured in RPMI-1640 medium (Gibco, Grand Island, NY, USA) supplemented with 2 mM L-glutamine and 10% heat-inactivated fetal bovine serum (FBS; Gibco) at 37
Animal experiments and mouse miRNA arrays
BALB/c male mice (6–8 weeks old), obtained from the National Laboratory Animal Center (Taipei, Taiwan), were maintained in a pathogen-free environment and allowed free access to food and water. All animal experiments were performed according to the guidelines for the care and use of research animals (DHHS publ. NIH 85-23, revised 1996) and the animal use protocol had been reviewed and approved by the institute animal care and use committee (IACUC) of National Yang-Ming University, Taipei, Taiwan (Approval no. 100609). In order to rule out the variation interference caused by hormonal changes in female animals during the experimental period, this study used male mice for analysis. The mice were randomly divided into two groups: normal control (
Transfection of mmu-miR-762 mimic and inhibitor
CT26 cells were cultured in a petri dish for 4 h before transfection. miR-762 mimic and inhibitor (miR-Ribo
Cell viability assay
Cultured CT26 cells were collected and their viability was assayed by the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT, Sigma, St. Louis, MO, USA) colorimetric method. Briefly, treated cells were collected, washed with PBS, and reacted with MTT reagent (Sigma) for 2 h at 37
Colony formation assay
Viable cells (10
Characters of control donors and colorectal cancer (CRC) patients
Characters of control donors and colorectal cancer (CRC) patients
Primers used in this study
Assays of cell invasion properties were performed using a modified Boyden chamber with polyethylene terephthalate filter inserts coated with a Matrigel matrix in 24-well plates containing 8-mm pores. Briefly, the cells were suspended in serum-free medium containing 0.5% bovine serum albumin. These cells were plated into the upper chamber followed by filling the lower chamber with the same medium with or without miR-762 mimic and inhibitor. Cells were incubated for 24 h, and then non-invading cells were gently removed. Cells on the upper side of the filter were carefully removed and cells invading the lower side were counted by microscopic examination (100X, Olympus). For quantification, 10% acetic acid (100
Cell cycle and sub-G1 assay
The treated CT26 cells were collected, washed with PBS, fixed, and permeated with ice-cold 70% ethanol overnight, followed by incubation with 0.1% Triton X-100, 0.2 mg/mL RNaseA (Sigma) and staining with 20
Prediction and western blot assay of regulatory pathways
The targeted genes regulated by miR-762 were predicted by the MIRDB (
Patients and data collection
The protocols for obtaining clinical samples and information were approved by the Institutional Review Board of National Taiwan University Hospital Yunlin Branch (No. 201611007RINB). Written informed consent to participate in this study was obtained from all of the individuals. This study collected blood samples from CRC patients (
Statistical analysis
The results were expressed as the mean
Array assay of plasma miRNA in BALB/c mice with CT26 cell implantation. (A) Animal experiment process. CT26 cells were suspension in sterile phosphate-buffered saline (PBS) and injected subcutaneously (s.c.) with 2 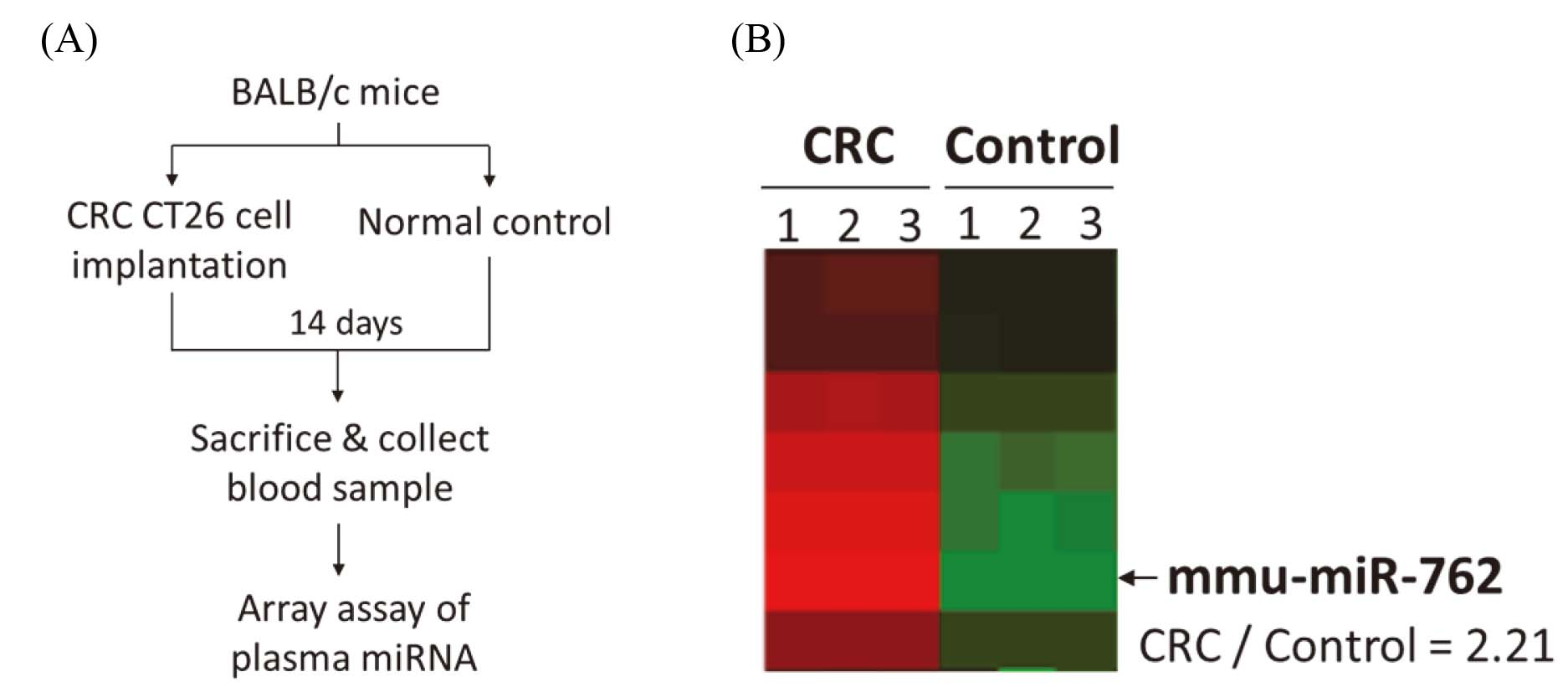
(A) Viability assay, (B) Morphological observation (200X), (C) Colony assay of CT26 cells transfected with mimic and inhibitor of mmu-miR-762. Cells transfected with mmu-miR-762 mimic (150 nM) increased cell viability and colony number, while mmu-miR-762 inhibitor (50 nM and 150 nM) decreased viability and colony formation by MTT assay. CT26 cells transfected with mimic and inhibitor of mmu-miR-762 did not show altered morphology under an inverted microscope view. The results were expressed as the mean 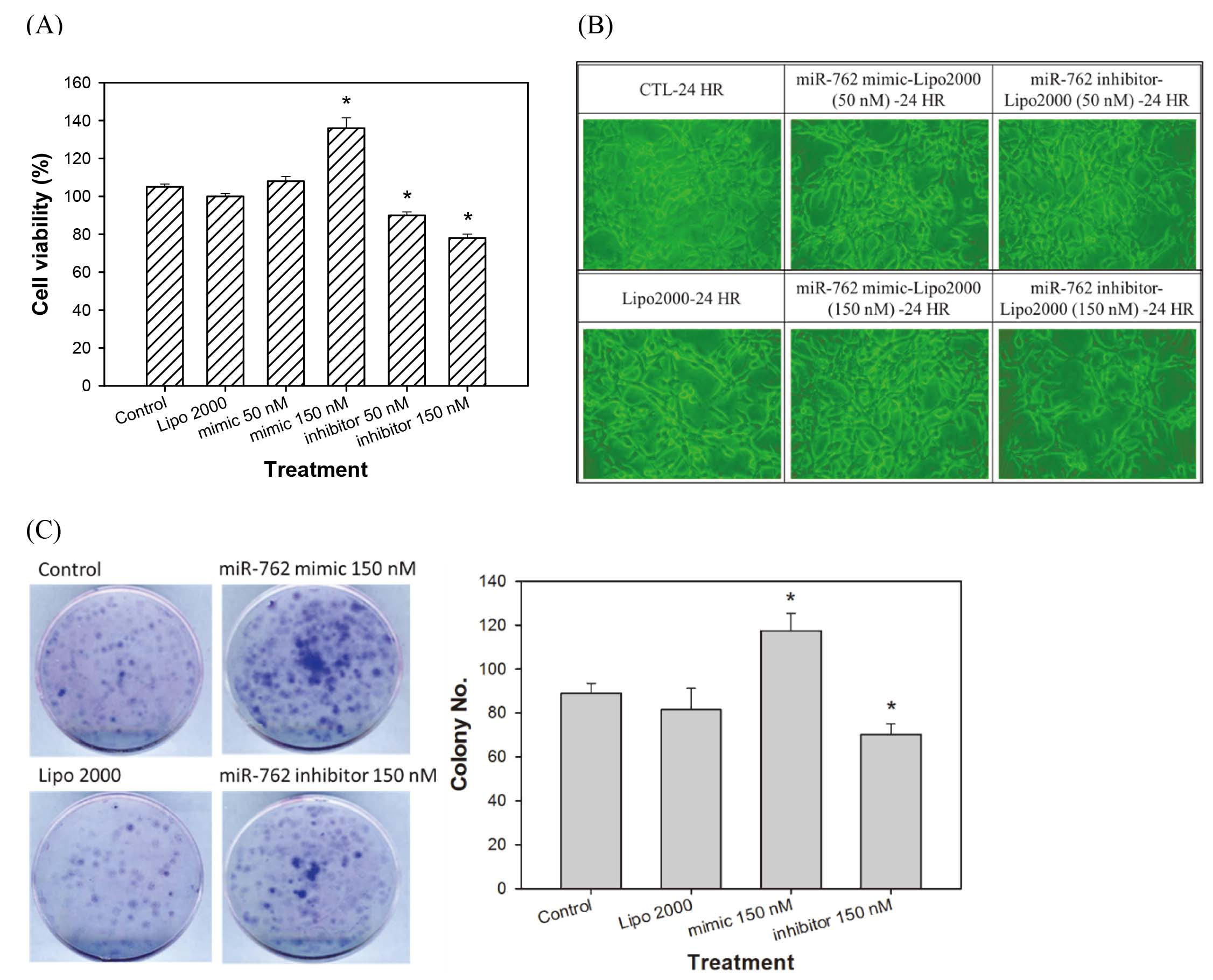
Invasion assay of CT26 cells transfected with mimic and inhibitor of mmu-miR-762. (A) Morphological observation of treated cells (100X), (B) Percent of invasive cells, (C) Expression of vimentin and E-cadherin by western blot analysis. Cells transfected with mmu-miR-762 mimic (150 nM) showed increased cell invasion ability, while mmu-miR-762 inhibitor (50 nM and 150 nM) decreased cell invasion. The results were expressed as the mean 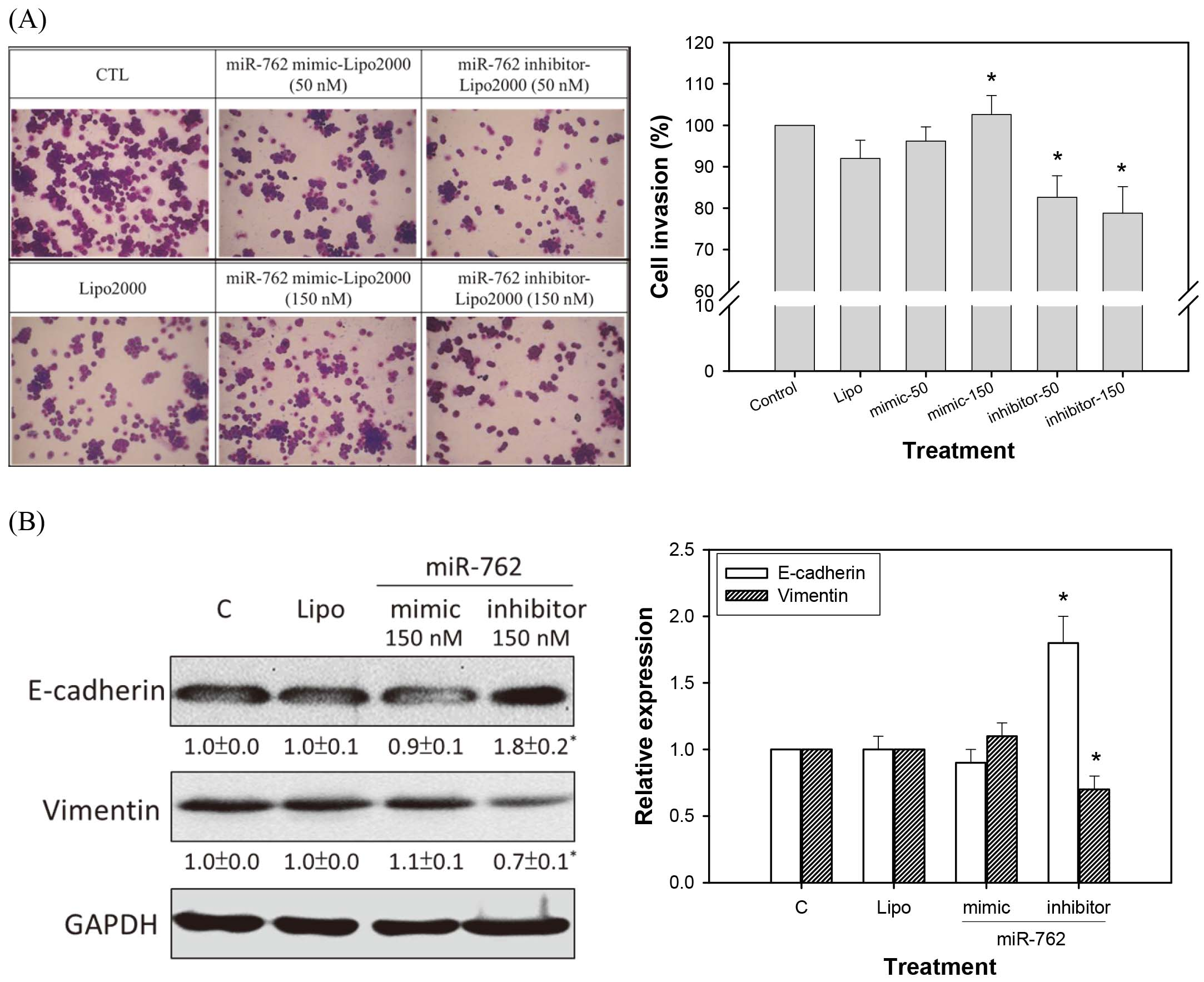
Expression of mmu-miR-762 in BALB/c mice implanted with CRC CT26 cells
Figure 1A shows the animal experiment flow of BALB/c mice implanted with CRC CT26 cells, and plasma samples from each group were used for miRNA array analysis. Based on the animal experimental design, differentially expressed miRNAs for each comparison were determined by clustering analysis. Standard selection criteria for identifying differentially expressed miRNAs were established at log2
(A) Apoptosis sub-G1, (B) Cell cycle assay of CT26 cells transfected with mimic and inhibitor of miR-762. Neither mmu-miR-762 mimic nor inhibitor changed the sub-G1 levels of CT26 cells analyzed by a FACScan flow cytometer. The cell cycle arrest at G0/G1 phase when CT26 cells treated with 150 nM mmu-miR-762 inhibitor. The results were expressed as the mean 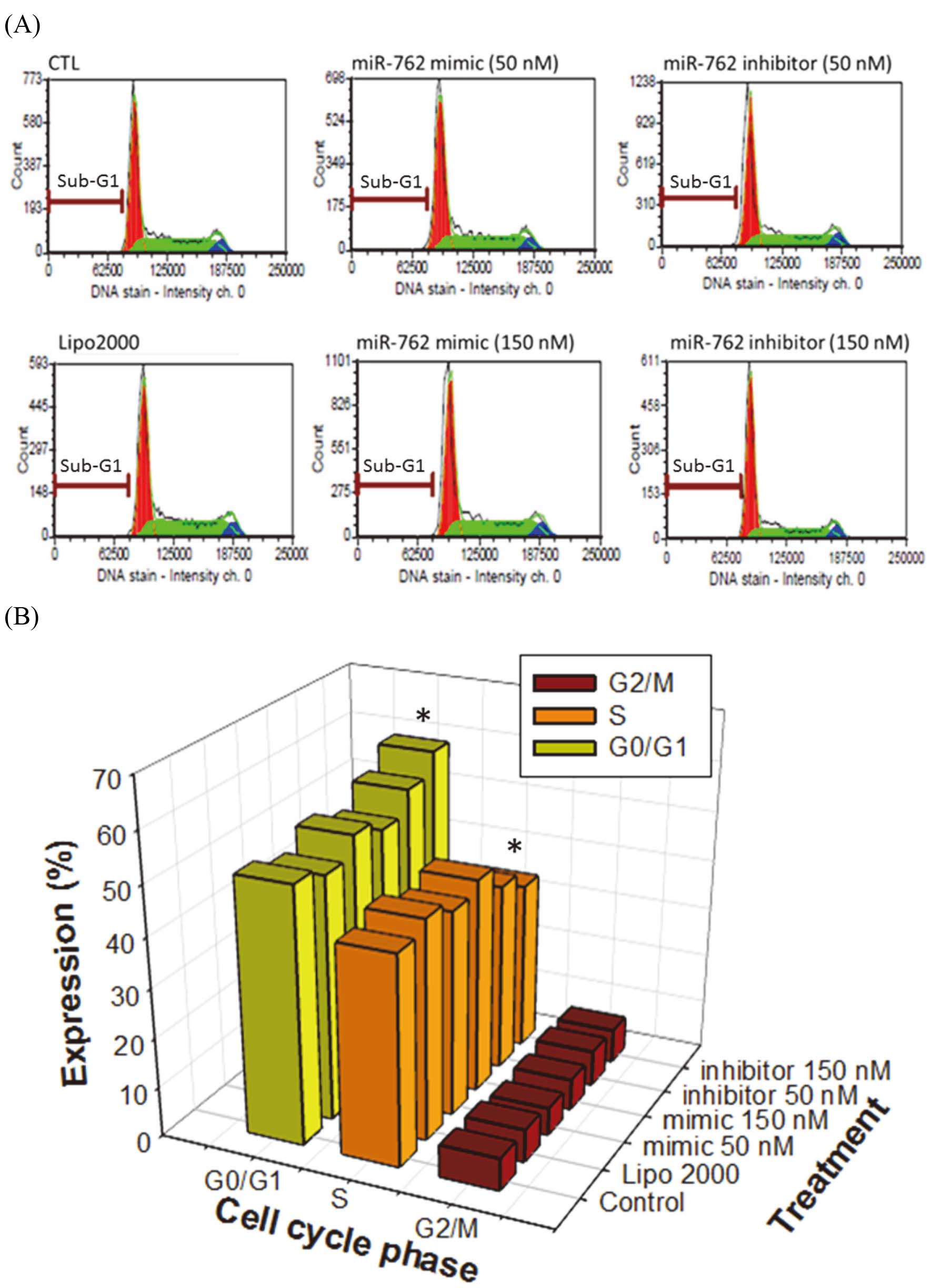
As shown in Fig. 2A, cells transfected with mmu-miR-762 mimic showed increased cell viability, where-as those transfected with mmu-miR-762 inhibitor showed decreased viability. However, transfection with the mimic and inhibitor of mmu-miR-762 did change the morphology of CT26 CRC cells (Fig. 2B). CT26 cells transfected with mmu-miR-762 mimic and inhibitor also showed increased and decreased colony formation, respectively, suggesting that mmu-miR-762 upregulated cell proliferation (Fig. 2C).
Invasion of CRC CT26 cells transfected with mmu-miR-762 mimic and inhibitor
As shown in Fig. 3A, cells transfected with mmu-miR-762 mimic showed increased cell invasion ability, while the mmu-miR-762 inhibitor decreased cell invasion. This suggests that mmu-miR-762 regulates specific genes related to CRC cell invasion. Additionally, mmu-miR-762 mimic transfection to CT26 cells increased vimentin and decreased E-cadherin expression, suggesting that mmu-miR-762 might upregulate EMT (Fig. 1B).
Cell cycle of CRC CT26 cells transfected with mmu-miR-762 mimic and inhibitor
Figure 4A shows that both the mmu-miR-762 mimic and inhibitor did not change sub-G1 levels in CT26 cells, suggesting that mmu-miR-762 does not regulate cell apoptosis. Moreover, cells treated with high concentration (150 nM) of mmu-miR-762 inhibitor caused cell cycle arrest at G0/G1 phase (Fig. 4B).
Signal molecule expression in CT26 cells transfected with mmu-miR-762 mimic and inhibitor
Figure 5A lists the predicted pathways that may be regulated by mmu-miR-762. The calcium signaling pathway, endocytosis, MAPK signaling pathway, and Wnt signaling pathway may be related to mmu-miR-762 regulation in CT26 cells.
Wnt/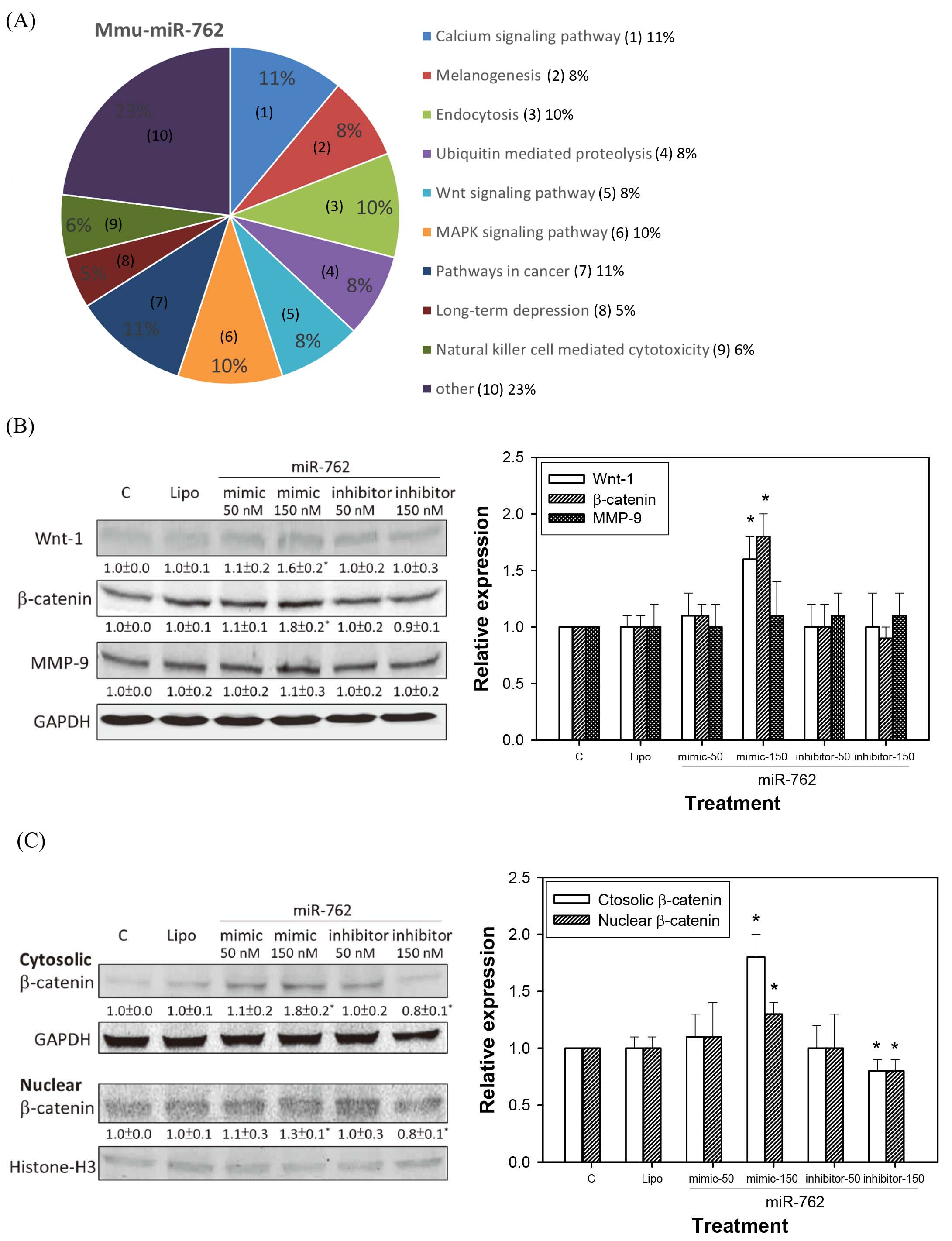
(A) Serum has-miR-762 expression in CRC patients and in control donors. Serum has-miR-762 expression in CRC patients (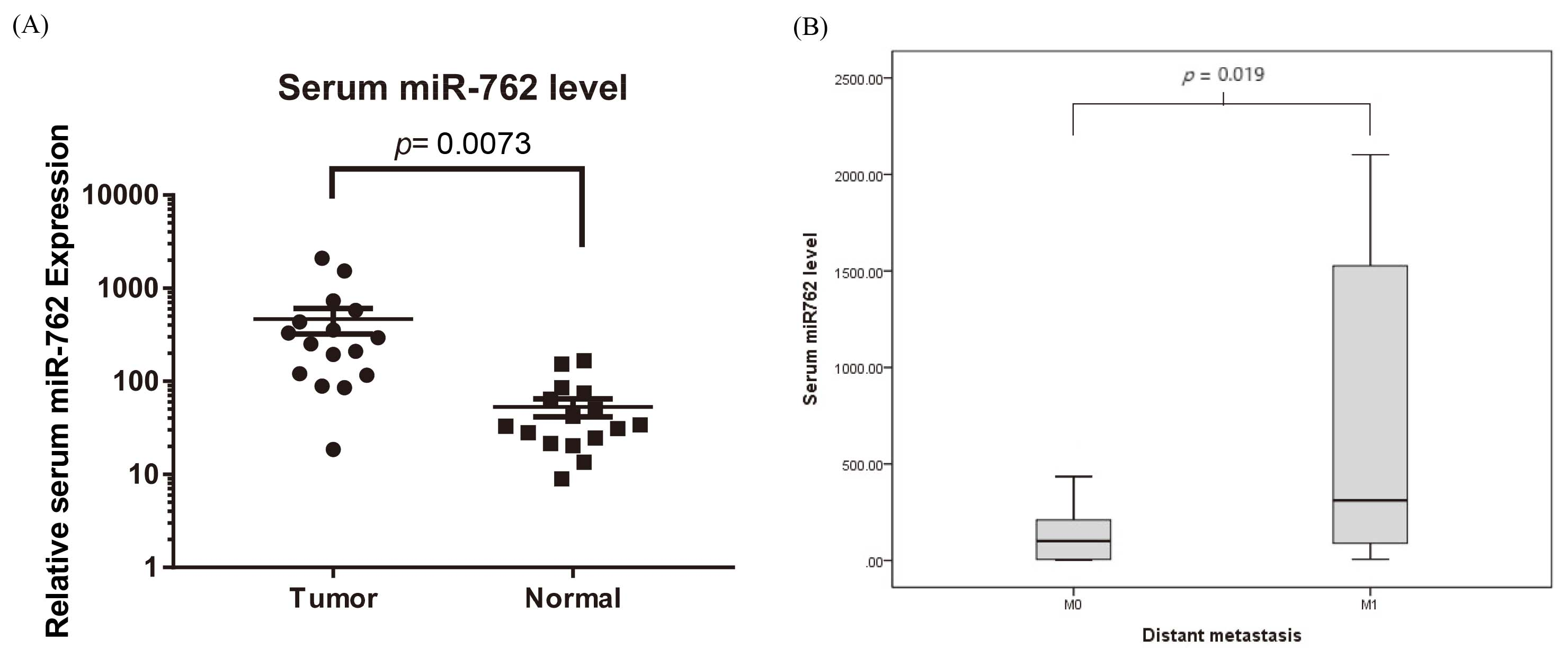
Western blot analysis showed that the mmu-miR-762 mimic increased the expression of Wnt-1 and
The human subject solicitation were performed form April to October, 2017. Table 1 shows the basic characters of control donors (
Discussion
miRNAs are a group of endogenous, small (18–25-nucleotide long), and non-coding RNA molecules that regulate the expression of specific mRNAs by either translational inhibition or mRNA degradation [5]. More than 50% of miRNAs are reported to be in cancer-associated genomic break points and function as tumor suppressors or oncogenic molecules [6]. Among the cancers, most CRCs form from the normal mucosa through the adenoma stage, which is accompanied by numerous gene mutations. Our study has screened circulating mmu-miR-762 expression in mice with CRC CT26 cell implantation. Further study demonstrate that CT26 cells transfected with mmu-miR-762 mimic promoted the cell viability and invasion/EMT of CRC cells. A similar result was also seen in human subjects. Expression of mmu-miR-762 in circulation was significantly higher in clinical CRC patients than in control donors. We subsequently confirmed that mmu-miR-762 significantly increased in distant metastasis of CRC, resulting in poor prognosis. Our study of the biological roles of miR-762 in CRC development revealed that Wnt/
miR-762 is upregulated in radiation-induced tumors in mice and may affect the pathways involved in apoptosis in this context [17]. Previous studies reported abnormal miRNA expression in numerous cancers, and miR-762 overexpression has been reported in breast cancer and ovarian cancer, where it promoted cancer cell growth and metastasis [18, 19]. A previous study also demonstrated an association between miR-762 expression and oral carcinogenesis [20]. However, no studies have examined the expression and function of circulating miR-762 in CRC. In this study, CRC cell line with mmu-miR-762 transfection was frequently up-regulated the cell viability and EMT, as well as has-miR-762 detected in serum extracted from CRC patients. Overexpression of mmu-miR-762 enhanced CRC cell proliferation and invasion. The ability of miR-762 to promote cell proliferation and invasion was confirmed by both overexpression and down-regulation experiments
To explore the molecular mechanisms by which miR-762 enhances CRC cell growth and invasion, Wnt/
Although miR-762 is overexpressed in several cancers, no studies have investigated miR-762 in CRC. In this study, real-time PCR analysis of human subjects confirmed that the expression of serum has-miR-762 in CRC patients was much higher than that in control donors. There was also an association between serum has-miR-762 expression and CRC patients with distant metastasis. miR-762 exerted its regulatory effects on cell proliferation and invasion/EMT, and accompanied by affecting the Wnt/
To confirm that human miR-762 can regulate Wnt pathway as well as mouse miR-762, we used the TargetScan microRNA target prediction which algorithm is considered the conservation among species. The target prediction results were listed of both human hsa-miR-762 target and murine mmu-miR-762 target prediction, demonstrating that the Wnt ligands may be regulated by miR-762 in both human and murine. In human, has-miR-762 has predicted to suppress Wnt family such as 7B, 2B, 3A, 9A, 5A, 10B, 3, 7A, and 11. In mmu-miR-762, it has predicted to suppress Wnt ligands 9B, 7B, 6, 10B, 10A, 4, 3A, 7A, and 8. Among these, Wnt-7B, -3A, and -10B have the genetic conservation of miR-762 binding site between human and murine 3’-UTR. Although the miR-762 has certain different target specify among human and mice, they still share some intersection in Wnt signaling. In the on-going work, further analysis of the regulation of circulating miR-762 on cancer promotion and progression in CRC patients will be advanced elucidated.
Hou et al. reported that miR-762 can downregulate the expression of a tumor suppressor protein menin through a binding site in its 3’-UTR and consequently upregulate the Wnt cell signaling pathway to promote the development of ovarian cancer [19]. Our present study first revealed that circulating miR-762 expression may be useful as a biomarker for diagnosing CRC and higher serum miR-762 expression-caused distant metastasis of patients with CRC. While patients with high expression of miR-762 may have shorter survival times than those with low expression of miR-762, survival analysis could not be performed because of the relatively small sample size and short time. Furthermore, the associations between serum miR-762 and tumor size and lymph node invasion were not thoroughly investigated. Therefore, further large-scale studies are necessary to investigate whether serum miR-762 levels can be used as a predictive and a prognostic factor of patients with CRC.
Conclusions, CT26 cell line is a highly metastatic colon cancer which possesses aggressive and invasion activities. The present study first demonstrated that the circulating mmu-miR-762 levels in CT26 cell-implanted BALB/c mice were higher than those in normal mice determined by miRNA array analysis. Then, mmu-miR-762 inhibitor was used for
Footnotes
Acknowledgments
This work was supported by Grants NTUHYL106. C003 from the National Taiwan University Hospital Yunlin Branch, Yunlin, Taiwan.
Conflict of interest
No potential conflicts of interest are disclosed.
Author contribution
Liao HF and Chen CY conceived and designed the experiments, Chang WM, Chen YY and Lin YF performed the experiments, Lai PS, Liao HF and Chen CY analyzed the data and wrote the paper.
