Abstract
Background:
Fucus vesiculosus-derived fucoidan, a multifunctional bioactive polysaccharide sourced from marine organisms, exhibits a wide range of therapeutic properties, including its anti-tumor effects. While previous research has reported on its anti-cancer potential, limited studies have explored its synergistic capabilities when combined with other natural bioactive ingredients. In this current study, we present the development of an integrative functional beverage, denoted as VMW-FC, which is composed of a fucoidan complex (FC) along with a blend of various herbal components, including vegetables (V), mulberries and fruits (M), and spelt wheat (W).
Objective:
Colorectal cancer (CRC) remains a significant cause of mortality, particularly in metastatic cases. Therefore, the urgent need for novel alternative medicines that comprehensively inhibit CRC persists. In this investigation, we assess the impact of VMW-FC on CRC cell proliferation, cell cycle dynamics, metastasis, in vivo tumorigenesis, and potential side effects.
Methods:
Cell growth was assessed using MTT and colony formation assays, while metastatic potential was evaluated through wound healing and transwell migration assays. The underlying signaling mechanisms were elucidated through qPCR and western blot analysis. In vivo tumor formation and potential side effects were evaluated using a subcutaneous tumor-bearing NOD/SCID mouse model.
Results:
Our findings demonstrate that VMW-FC significantly impedes CRC proliferation and migration in a dose- and time-dependent manner. Furthermore, it induces sub-G1 cell cycle arrest and an increase in apoptotic cell populations, as confirmed through flow-cytometric analysis. Notably, VMW-FC also suppresses xenograft tumor growth in NOD/SCID mice without causing renal or hepatic toxicity.
Conclusion:
The integrative herbal concoction VMW-FC presents a promising approach for inhibiting CRC by slowing proliferation and migration, inducing cell cycle arrest and apoptosis, and suppressing markers associated with proliferation (Ki-67, PCNA, and CDKs) and epithelial-mesenchymal transition (EMT) (Vimentin, N-cadherin, and β-catenin).
Keywords
Introduction
Cancer is one of the deadliest diseases that leads to immense medical and economic burdens. Among all cancer types, colorectal cancer (CRC) ranks as the third most commonly diagnosed cancer and is the second leading cause of cancer death worldwide in 2020. 1 Despite advances in therapeutic approaches like surgery, chemotherapy, radiation therapy, and immunotherapy, which may improve survival rates, fatality remains high, especially in metastatic cases. Hence, developing novel alternative treatments to comprehensively inhibit CRC is an urgent issue. Recently, traditional Chinese medicine (TCM) has been attracting attention for its moderate curative effects with fewer side effects. 2 Furthermore, combined herbal formulations have been shown to be more effective and to exhibit fewer side effects than individual herbs or drugs.3,4
Fucoidan is a naturally derived polysaccharide complex. It is mostly composed of l-fucose and sulfate groups and is commonly derived from the cell walls of various species of brown seaweeds, including Fucus vesiculosus.5,6 It also contains other monosaccharides such as glucose, uronic acid, mannose, galactose, xylose, rhamnose, arabinose, and xylose.7,8 Recently, many studies have reported the multiple biological activities of fucoidan, including anti-tumor, 9 antioxidant, 10 anticoagulant, 11 antiviral, 12 anti-inflammatory 13 and immunoregulatory 14 effects, revealing it as a promising candidate for treating different diseases. Though previous studies have reported its anti-cancer effects, only very limited studies have investigated its synergistic potential when combined with other bioactive natural ingredients like vegetables and fruits. Previously, we developed an integrative functional cocktail beverage comprising vegetable juice (V), mulberry and fruits (M), spelt wheat (W), and fucoidan complex (FC) as VMW-FC, which exhibited significant inhibitory effects on oral cancer. 15 However, given the critical incidence and mortality rates of CRC worldwide, the effects of VMW-FC on CRC and its related mechanisms remained unclear. Therefore, we aimed to explore the anti-cancer potential of VMW-FC on CRC in both in vitro and in vivo studies.
The detailed bioactive functions of each component in our VMW-FC formulation are elaborated in the discussion section (Table 1). In the in vitro study, we evaluated the growth ability and metastatic potential of CRC cells after treatment with various concentrations of VMW-FC. The effects of VMW-FC on the cell cycle and apoptosis were further investigated. Subsequently, to observe its in vivo anti-tumor effect, animals were administered VMW-FC via oral gavage following CRC inoculation. Additionally, after VMW-FC treatment, we isolated the liver and kidney of mice to assess potential renal and hepatic toxicity. Furthermore, we explored the signaling mechanisms underlying the effects of VMW-FC by determining key genes and proteins involved in proliferation, migration, apoptosis, and cell cycle arrest.
Bioactive Functions of the Herbal Ingredients Involved in VMW-FC Functional Beverage.

Materials and Methods
Cell Culture
The CRC cell lines, namely HT-29, SW480, and SW620, were procured from the American Type Culture Collection (Manassas, VA, USA). These cell lines were maintained in
RPMI-1640 medium (Gibco, 23400-021) supplemented with 10% fetal bovine serum (FBS, Corning, 35-010-CV) and 1% antibiotic-antimycotic solution (Corning, 30-004-CI). Cultures were kept in a humidified incubator set at 37°C with 5% CO2.
VMW-FC Preparation
The VMW-FC cocktail beverage was developed by Enhance Biotechnology Development Co., Ltd., Taipei, Taiwan. VMW-FC comprises approximately: 20% vegetable fermented broth, which includes carrot (Daucus carota), spoon cabbage (Brassica chinensis L.), komatsuna (Brassica rapa var. perviridis), broccoli (Brassica oleracea var. italica), and sweet potato leaves (Ipomea batatas (L.) Lam); 10% fermented broth from mulberries and other fruits, specifically mulberry (Morus australis), guava (Psidium guajava), passion fruit (Passiflora edulis), noni (Morinda citrifolia), and pineapple (Ananas comosus); 65% fermented broth of spelt wheat (Triticum spelta); 5% fucoidan powder extracted from Fucus vesiculosus, with water being used as the solvent. The broths were fermented individually before being combined. All the ingredients in VMW-FC were sterilized. Furthermore, the particle size of the fucoidan was ensured to be <300 µm.
Cell Viability
The cell viability was assessed using the MTT assay. Initially, 2000 cells from the HT-29, SW480, and SW620 CRC cell lines were seeded per well in 96-well culture plates in triplicate. These plates were then incubated overnight at 37°C. The following day, the CRC cells were treated with different concentrations of VMW-FC (0, 2, 4 mg/ml) and incubated for another 48 hours. Subsequently, 100 μl of a 0.5 mg/ml solution of methylthiazolyldiphenyl-tetrazolium bromide (Sigma-Aldrich, M5655) was added to each well and incubated for 4 hours. This solution was then replaced with 100 μl of dimethyl sulfoxide (DMSO) (Sigma-Aldrich, D5879). The optical density (O.D.) was measured at 595 nm using an ELISA plate reader (Multiskan RC Microplate Reader, Thermo).
Colony Formation Assay
To evaluate colony-forming ability, 500 cells from each CRC cell line were seeded in 10 cm culture dishes and incubated at 37°C overnight. Following incubation, the cells were treated with VMW-FC at concentrations of 0, 2, and 4 mg/ml and incubated for an additional 2 weeks, with the medium being replaced every 3 days. After this treatment period, the medium was discarded, and the cells were rinsed twice with PBS. The cells were then fixed with 10% formalin (Macron, H121-08) for 30 minutes at 4°C. Subsequently, the cells were stained with a 0.1% w/v solution of crystal violet dye (Fisher Scientific, C581-25) and left at room temperature for an hour. The crystal violet dye was then discarded, and the plates were rinsed with double-distilled water. Colonies consisting of at least 50 cells and visible to the naked eye were counted using a colony counter (Suntex, colony counter 570).
Wound-Healing Assay
To assess cell migration using a wound-healing assay, CRC cells were seeded in 6-well plates at a density of 1.5x105 cells per well and incubated at 37°C overnight. Once the cells reached approximately 90% confluence, a wound was created in the cell monolayer using a 100 -μl pipette tip to ensure a straight and consistent gap. The cells were then gently washed with PBS to remove any non-adherent cells. Following this, the cells were treated with VMW-FC at concentrations of 0, 2, and 4 mg/ml and incubated for either 24 or 48 hours. At each of these time points, images of the wound were captured from 3 randomly selected areas using an Olympus IX-71 inverted microscope. The extent of wound closure was subsequently quantified by measuring the migrated area and results were normalized to their respective control groups.
Transwell Migration Assays
In vitro cell migration ability was evaluated by utilizing 8 μm BD Falcon cell culture insert (BD Biosciences, NJ). CRC cells were seeded in the upper compartment of each chamber at a density of 1 × 105 cells/well with 500 μl of serum-free medium, while the lower compartment was filled with 1 ml of medium enriched with 10% FBS. Following 48 hours of incubation at 37°C, cells that did not migrate through the membrane were scraped off from the upper surface. Cells that had migrated to the reverse side of the membrane were stained with 0.1% crystal violet. Representative images of migrated cells were then captured using an Olympus IX71 microscope (Tokyo, Japan). For quantification, the crystal violet-stained cells were dissolved in 10% acetic acid (Merck, 695092), and the resulting absorbance was measured using an ELISA plate reader (Multiskan RC Microplate Reader, Thermo) at 570 nm. Migration rates were then normalized to the respective control groups.
Cell Cycle Analysis
Before cell cycle analysis, the CRC cells were trypsinized, washed with PBS and fixed with 75% ethanol (Honeywell, 32221). Next, the cells were stained with 500 µl of RNase A (0.2 mg/ml, Sigma-Aldrich, 10109142001) and 500 µL of propidium iodide (0.02 mg/ml, Sigma-Aldrich, 11348639001), then incubated for 30 minutes in the dark. The cell suspension was filtered with a 40 μm cell strainer (Falcon, 352340) to exclude any cell aggregates. The cell cycle analysis was then conducted by flow cytometer (SONY SA3800), and 10 000 events for each sample were recorded. Before running each sample for flow cytometry, cell suspension were again well-pipetted. Data acquisition and analysis were done using SA3800 software version 2.0, and the percentages of each cell cycle phase were determined and quantified.
Apoptosis Detection
To determine apoptosis levels post VMW-FC treatment, we utilized the Annexin V-PE/7-AAD Apoptosis Detection Kit (Elabscience, E-CK-A216) in accordance with the manufacturer’s guidelines. CRC cells were exposed to VMW-FC at concentrations of 0, 2, and 4 mg/ml for a period of 48 hours. After treatment, the cells were harvested and labeled with Annexin V-PE/7-AAD for a duration of 15 minutes in the dark. Apoptosis quantification and analysis of the stained cells were conducted using the SONY SA3800 flow cytometer, with data interpretation performed with SA3800 software version 2.0.
Real-Time Polymerase Chain Reaction (qPCR)
For quantitative analysis of gene expression via real-time polymerase chain reaction (qPCR), CRC cells from each treatment group were harvested and total RNA was extracted utilizing the Total RNA Mini Kit (Geneaid, RB050), following the manufacturer’s guidelines. This RNA was subsequently converted to complementary DNA (cDNA) via reverse transcription-PCR (RT-PCR) using the RevertAid H minus first strand cDNA synthesis kit (Thermo scientific, K1632, USA). The qPCR was executed on the ABI 7300 real-time detection PCR system (Applied Biosystems, USA). Relative gene expression was computed utilizing the 2−ΔCt or 2−ΔΔCt methods, referencing calibration samples in each set. Specific primer sequences employed for each target gene in the qPCR are as follows:
Ki67—Forward: ACGCCTGGTTACTATCAAAAGG;
Reverse: CAGACCCATTTACTTGTGTTGGA
PCNA—Forward: CCTGCTGGGATATTAGCTCCA;
Reverse: CAGCGGTAGGTGTCGAAGC
CDK2—Forward: CCAGGAGTTACTTCTATGCCTGA;
Reverse: TTCATCCAGGGGAGGTACAAC
CDK4—Forward: ATGGCTACCTCTCGATATGAGC;
Reverse: CATTGGGGACTCTCACACTCT
CDK6—Forward: GCTGACCAGCAGTACGAATG;
Reverse: GCACACATCAAACAACCTGACC
Bcl-2—Forward: GGTGGGGTCATGTGTGTGG;
Reverse: CGGTTCAGGTACTCAGTCATCC
Bax—Forward: CCCGAGAGGTCTTTTTCCGAG;
Reverse: CCAGCCCATGATGGTTCTGAT
β-actin—Forward: CATGTACGTTGCTATCCAGGC;
Reverse: CTCCTTAATGTCACGCACGAT
Western Blot
After treatment with VMW-FC, cells were harvested and lysed using 1X RIPA Lysis Buffer (Millipore, CA92590, USA) supplemented with Protease Inhibitor Cocktail (Millipore, USA). To ensure complete lysis, the samples were sonicated with the Ultrasonic Liquid Processors XL-2000 (Misonix). The cell lysate was then centrifuged at 12 000g and 4℃ for 30 minutes. The supernatant containing proteins was collected, and its concentration was determined using the Bradford protein assay. For electrophoresis, 20 µg of each protein sample was loaded. Post-electrophoresis, proteins were transferred onto a PVDF membrane (Merck Millipore, IPVH00010) and blocked with 5% BSA (Cytiva, SH30574.02) in PBS. The membrane was probed with the following primary antibodies: cyclin A2 (Cell Signaling Technology #4656, 1:1000), cyclin B1 (Cell Signaling Technology #12231, 1:1000), cyclin D1 (Cell Signaling Technology #2978, 1:1000), CDK2 (Cell Signaling Technology #2546, 1:1000), CDK4 (Cell Signaling Technology #2546, 1:1000), CDK6(Cell Signaling Technology #13331, 1:1000), vimentin(Cell Signaling Technology #3932, 1:1000), N-cadherin (Cell Signaling Technology #13116, 1:1000), E-cadherin (Cell Signaling Technology #4065, 1:1000), β-catenin (Cell Signaling Technology #8480, 1:1000) and β-actin (Millipore #MAB1501, 1:10 000). After primary antibody incubation, the membrane was probed with secondary antibodies: anti-rabbit IgG (Cell Signaling Technology #7074, 1:5000) or anti-mouse IgG (Cell Signaling Technology #7076, 1:5000). Protein bands were visualized using an enhanced chemiluminescent (ECL) kit (Bionovas, #FL0010-0170) and images were captured using the MultiGel-21 imaging system (TOPBIO, Taiwan).
Animal Studies
The animal protocol (Approval number LAC-2021-0582) was approved by the Institutional Animal Care and Use Committee (IACUC) of Taipei Medical University of Taiwan. The 6-week old female non-obese diabetic/severe combined immunodeficiency (NOD/SCID) mice were purchased from BioLASCO., Taiwan. The sample size of 3 mice per group was determined valid based on a power analysis. Mice were maintained in a pathogen-free environment with access to autoclaved food and water. The administered concentration of VMW-FC for mice was 960 mg/kg/day, which was converted and calculated from the recommended daily dose for human (78 mg/kg/day), according to the equivalent doses converting guidance of US FDA. 34 To assess the in vivo anti-tumor effects of VMW-FC, we subcutaneously inoculated 1 × 106 HT-29 cells into the right flank of each mouse. After 2 weeks, when the tumor volumes reached 100 ± 30 mm 3 , 500 µl VMW-FC were given every day by oral gavage to treatment groups, whereas the control group was fed with water. The tumor volumes were measured twice a week, and were calculated by using the following formula: Volume = Length × Width 2 /2. The outliers were detected and excluded using interquartile range (IQR) method. The IQR represents the range between the first (Q1) and third quartiles (Q3), calculated as IQR = Q3 – Q1. Data points that fall below (Q1 – 1.5 IQR) or above (Q3 + 1.5 IQR) are typically considered outliers.
Further, after the treatment, mice were sacrificed to excise the tumors, and their liver and kidney were isolated, sectioned and performed hematoxylin and eosin (H&E) staining, for evaluating the possible renal- or hepato-toxicity of VMW-FC.
Statistical Analysis
The sample sizes of all data were at least n = 3, unless otherwise indicated. All the experiments were performed with biological replicates in both in vitro and in vivo. The statistical significances were analyzed through 2-way ANOVA (Tumor volume), One-way ANOVA (≥3 experimental groups) and student t-test (2 experimental groups). All statistical analyses were performed using GraphPad Prism 8. *, P < .05, **, P < .01 and ***, P < .001.
Results
Fucoidan Complex-Based Functional Beverage (VMW-FC) Inhibits Colorectal Cancer (CRC) Cell Proliferation and Cell Viability
To investigate the anti-CRC effects of VMW-FC, 3 different CRC cell lines (HT-29, SW480, and SW620) were chosen for subsequent experiments, using treatment doses of 0, 2, and 4 mg/ml. Firstly, the effects of VMW-FC on cancer cell growth were evaluated through cell counting, MTT assay, and colony formation assay. Our results revealed that VMW-FC significantly inhibits cell number (Figure 1A) and cell viability (Figure 1B) in a dose-dependent manner, in which all the cell lines reached IC50 at concentrations ranging from 2 to 4 mg/ml. Similarly, the ability to form colonies was notably inhibited in all treatment groups, as observed in the colony formation results (Figure 1C) along with their respective quantification (Figure 1D). These results demonstrated the strong inhibitory effects of VMW-FC on CRC cell growth.

Effects of VMW-FC on CRC cell growth abilities. CRC cell lines (HT-29, SW480, SW620) were treated with 0, 2 and 4 mg/ml of VMW-FC for 48 hours, the (A) Cell number, (B) Cell viability and (C) colony formation along with its (D) plating efficiency were obtained through cell counting, MTT assay and colony formation assay, respectively. All data were presented as mean ± SD unless otherwise stated. *, ** and *** indicate P < .05, P < .01 and P < .001, respectively using one-way ANOVA, compared to control (0 mg/ml) group.
VMW-FC Reduces CRC Cell Migration Capability
Metastatic potential is a crucial characteristic and a leading factor that promotes the lethality of cancer, where the 5-year survival rates for localized and regional CRC are 90.6% and 71.7% respectively, but dramatically drop to 13.7% for distant CRC. 35 In the current study, the effect of VMW-FC on cancer metastasis was determined through wound healing and transwell migration assays. The wound was created by scratching the plate once it reached 90% confluency, followed by VMW-FC treatment (0, 2, and 4 mg/ml) for 24 and 48 hours. Our results exhibited that VMW-FC retarded cell migration in all 3 cell lines (Figure 2A), which were further confirmed by their respective quantified results (Figure 2B). In alliance with this result, the transwell migration capability was also inhibited in a dose-dependent manner. The results showed fewer migrated cells in the treatment groups compared to the control (Figure 2C), and this was validated through their relative quantification (Figure 2D).
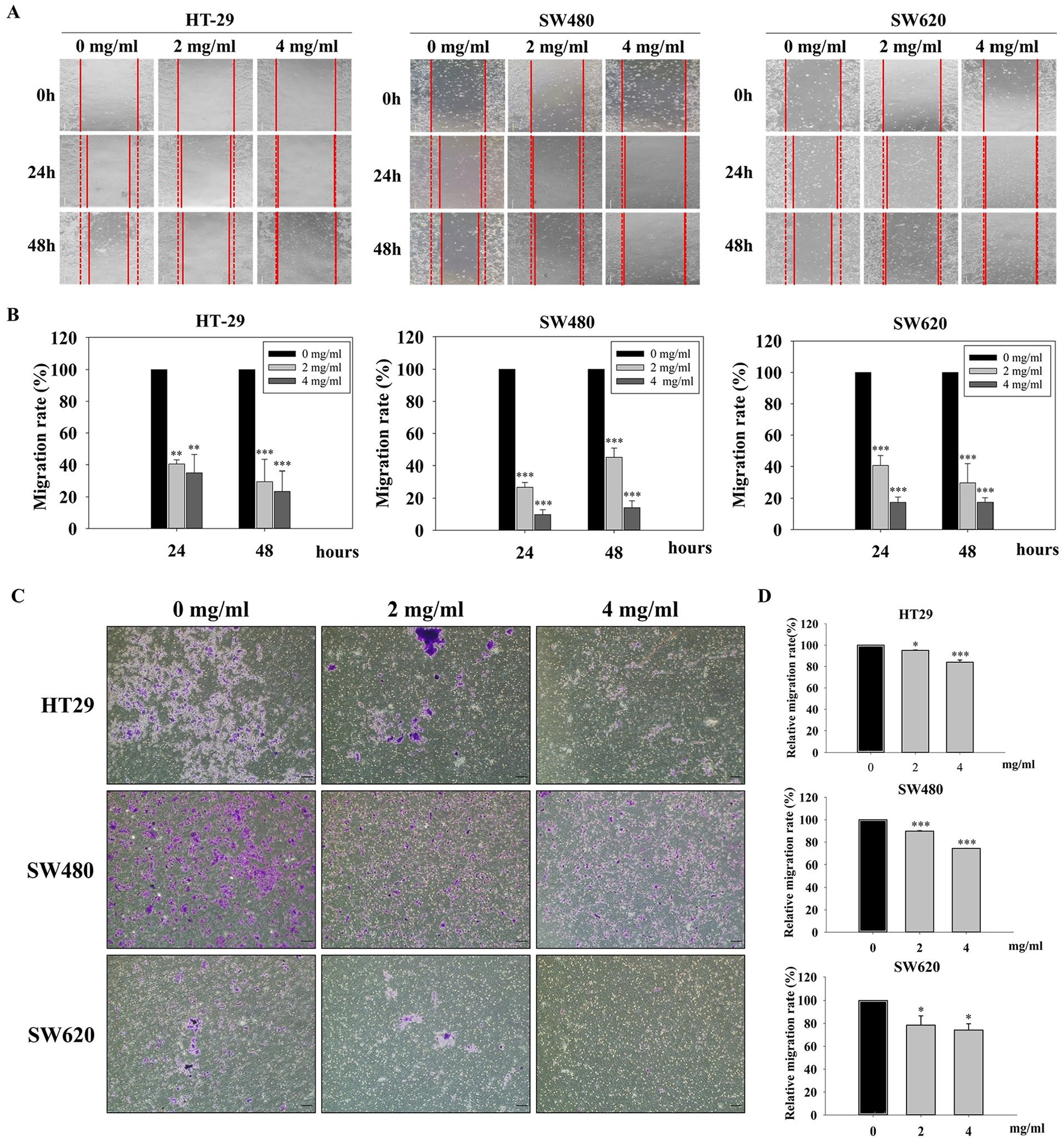
Effects of VMW-FC on CRC metastatic potential. The metastatic potential was investigated by wound healing assay and transwell migration assay. (A) Cell migration area were determined after VMW-FC treatment for 24 and 48 hours, and (B) the quantitative migration rate were calculated and normalized to the control (0 mg/ml) group. (C) The migrated cells in the transwell assay were stained with crystal violet, and were later dissolved in alcohol for its (D) quantification. All data were presented as mean ± SD unless otherwise stated. *, ** and *** indicate P < .05, P < .01 and P < .001, respectively using one-way ANOVA, compared to control (0 mg/ml) group.
VMW-FC Induces the Accumulation of sub-G1 Phase in Cell Cycle
The cell cycle is an ordered sequence of events during cell replication that fundamentally controls cell proliferation, especially in rapidly proliferating cancer cells. After treating the cell lines with VMW-FC for 48 hours, the cell cycle was analyzed through flow cytometry. Notably, our results demonstrated a dramatic increase in the sub-G1 phase in all 3 cell lines in a dose-dependent manner, evident not only in the flow cytometric cell cycle histogram (Figure 3A) but also in its quantitative results (Figure 3B). This suggests that VMW-FC induces the discontinuous fragmentation of nuclear DNA, leading to the accumulation of the Sub-G1 phase, which is considered indicative of the apoptotic cell population.

Cell cycle analysis of the VMW-FC treated CRC cells. (A) The distribution flow chart of different cell cycle phases. (B) The quantification of sub-G1 phase of each cell lines. All data were presented as mean ± SD unless otherwise stated. *, ** and *** indicate P < .05, P < .01 and P < .001, respectively using one-way ANOVA, compared to control (0 mg/ml) group.
VMW-FC Promotes Apoptotic Population in CRC Cells
To determine whether VMW-FC inhibits CRC by inducing cell apoptosis, 7-Aminoactinomycin D (7-AAD)/Annexin V-PE double-staining was conducted and analyzed through flow cytometry. 7-AAD is a viability probe that distinguishes viable cells. On the other hand, Annexin V-PE is a sensitive probe used for identifying apoptotic cells. In this context, the Q4 and Q2 quadrants represent early and late apoptosis populations, respectively. Our flow cytometric apoptosis analysis revealed that VMW-FC induced both early and late apoptosis (Figure 4A). As a result, the total apoptotic cell population significantly increased, as seen in the quantified results (Figure 4B). These results are consistent with the increased sub-G1 phase observed in the cell cycle analysis, further confirming that VMW-FC inhibits CRC by promoting apoptosis.

Flow cytometric apoptosis assay of the CRC cells. (A) The representative flow scatterplots showing the conditions of each cell. Q1 to Q4 quadrants indicate the cell populations of necrosis, late apoptosis, healthy and early apoptosis respectively. (B) The quantification of the apoptosis population in each cell line. All data were presented as mean ± SD unless otherwise stated. *, ** and *** indicate P < .05, P < .01 and P < .001, respectively using one-way ANOVA, compared to control (0 mg/ml) group.
VMW-FC Attenuates In Vivo Tumor Growth Without Further Adverse Effects
Based on the aforementioned in vitro inhibitory effects of VMW-FC on CRC, we further examined its in vivo anti-tumor effects using a tumor-bearing mouse model. To establish this model, the HT-29 cell line was subcutaneously inoculated into NOD-SCID mice. VMW-FC was then administered through oral gavage when the tumors became visible (100 ± 30 mm 3 ) by week 2. The tumor size was measured twice a week, and all mice were sacrificed on day 43, with tumors surgically excised (Figure 5A). Compared to the control, the VMW-FC-treated group exhibited a remarkably reduced tumor size, as shown in the photo and quantified tumor volume (Figure 5B). The hepatic and renal toxicity of VMW-FC was further evaluated to assess its safety profile. In the hematoxylin and eosin-stained sections of the liver and kidney, no tissue deformation was detected in either the control or the VMW-FC-administered groups (Figure 5C).

Effects of VMW-FC on in vivo tumorigenesis and the tissue deformation for safety. (A) An illustration of experimental timeline of in vivo study. (B) Image of the excised tumors and their quantitative tumor volume. (C) Hematoxylin and Eosin (H&E) staining of the liver and kidney tissue sections for examining deformation. All data were presented as mean ± SD unless otherwise stated. *** indicate P < .001.
VMW-FC Inhibits CRC Through Modulating Proliferation, Cell Cycle, Apoptosis and Migration-Related Biomarkers
To further understand the inhibitory signaling pathway of VMW-FC in CRC, we determined several dominant biomarkers that regulate cell proliferation, cell cycle, apoptosis, and migration at gene or protein levels. For gene expression levels, Ki67 and PCNA, markers of proliferation, 36 were significantly inhibited in the VMW-FC treatment groups, especially in the 4 mg/ml group (Figure 6A). Among the well-known cell cycle regulators, Cyclin-dependent kinases (CDKs), our results displayed that CDK2 and CDK4 were strongly inhibited in the VMW-FC treatment groups, while CDK6 showed a decreasing trend in a dose-dependent manner (Figure 6B). Among the Bcl-2 family proteins, which play a pivotal role in regulating cell apoptosis, 37 levels of the cytoprotective protein Bcl-2 significantly decreased while the pro-apoptotic protein BAX increased in the VMW-FC treatment groups (Figure 6C). Furthermore, we detected the protein expression of cell cycle-related markers, including cyclins and CDKs. Our results exhibited that all the cyclins (cyclin A2, B1, and D1) and CDKs (CDK 2, 4, and 6) were inhibited after VMW-FC treatment, especially in the 4 mg/ml groups (Figure 6D), consistent with our previous gene expression findings. Additionally, we determined the protein expressions of the epithelial-mesenchymal transition (EMT) markers, including vimentin, n-cadherin, e-cadherin, and β-catenin to validate its anti-migration function. As vimentin, n-cadherin, and β-catenin are mesenchymal markers, 38 their protein expressions were inhibited, while the epithelial marker e-cadherin increased in the VMW-FC treatment groups. These results revealed that VMW-FC attenuates CRC cell growth by inhibiting proliferation and cell cycle-related markers, and its anti-metastatic potential is evident from the inhibition of EMT markers.

The genes and proteins regulated by VMW-FC in CRC. After treated HT-29 with VMW-FC, several signaling biomarker expressions were determined, including (A) cell proliferation related gene expressions, (B) cell cycle-related gene expressions (C) cell apoptosis related gene expressions (D) cell cycle-related protein expressions, and (E) EMT related protein expressions. All data were presented as mean ± SD unless otherwise stated. *, ** and *** indicate P < .05, P < .01 and P < .001, respectively using one-way ANOVA, compared to control (0 mg/ml) group.
Discussion
The current study focuses on a fucoidan complex-based cocktail therapy for treating colorectal cancer (CRC). Although fucoidan has been studied in many previous researches, to the best of our knowledge, this is the first article that investigates its potential as a functional cocktail beverage for alternative CRC therapy. Notably, previous research has indicated that cocktail therapy can achieve better efficacy with fewer side effects compared to single-agent therapies. This approach is attracting more attention recently, especially for treating complex diseases like cancer.39,40 In this study, we combined the fucoidan complex with multiple natural bioactive ingredients. These include fermented broths of various vegetables such as spelt wheat (Triticum spelta), carrot (Daucus carota), spoon cabbage (Brassica chinensis L.), komatsuna (Brassica rapa var. perviridis), broccoli (Brassica oleracea var. italica), and sweet potato leaves (Ipomea batatas (L.) Lam). We also incorporated fermented broths of various fruits including mulberry (Morus australis), guava (Psidium guajava), passion fruit (Passiflora edulis), noni (Morinda citrifolia), and pineapple (Ananas comosus).
Within all the natural components, many have been proven possessing beneficial effects to human health (Table 1). Spelt wheat is rich in phenolic acids, which could exert strong antioxidative activities to scavenge or neutralize free radicals thereby minimizing oxidative damage, 16 the phytic acid and alkylresorcinols of which were also mentioned could control blood glucose levels, insulin sensitivity and hyperinsulinemia. 17 Carrots have been shown to reduce the risk of cancer and cardiovascular diseases due to its antioxidative and anti-inflammatory properties 18 ; a previous cohort study also indicated carrot intake is associated with decreased risk of CRC. 19 Spoon cabbage, komatsuna and broccoli are categorized as cruciferous vegetables, which are well-known of their health-benefit effects due to their abundance in various beneficial metabolites such as glucosinolates, S-methylcysteine sulfoxide and flavonoids, etc., which further contribute to cancer prevention.21,22 Sweet potato leaves have also been revealed rich in multiple bioactive compounds, which may improve oxidative stress, immune responses and cardiovascular diseases.23,24 Further, previous studies have demonstrated that mulberry extracts may inhibit different cancer types including colorectal, liver, gastric, lung, and thyroid cancer.25 -29 Other fruit ingredients (guava, passion fruit, noni and pineapple) have also been found effective for different cancer inhibition.30 -33 Therefore, to balance the possible side-effects and enhance the therapeutic efficacy of each component, we designated the vegetables (V), mulberry (M), fruits and spelt wheat (W) juice with fucoidan complex as an integrative functional beverage called VMW-FC.
Firstly, we investigated whether VMW-FC inhibits CRC (HT-29, SW480, SW620) cell growth ability. Our results revealed that VMW-FC exhibited significant inhibitory effects on cell proliferation, cell viability and colony forming ability in a dose-dependent manner, demonstrating its anti-proliferation characteristics in CRC. This result is in line with our previous study showing inhibited cell growth in oral cancer after VMW-FC treatment, 15 and further supported by other research indicating fucoidan could inhibit the proliferation activity of the HT-29 CRC cell line. 41 Next, VMW-FC was also found significantly effective in inhibiting cell migration in a time- and dose-dependent manner, disclosing its inhibitory property to cancer metastasis. Additionally, as the epithelial-mesenchymal transition (EMT) process is the key feature during cell metastasis, 42 we determined the EMT-related proteins and found that EMT-inducing proteins were inhibited (vimentin, N-cadherin and β-catenin) while the epithelial marker E-cadherin was increased. This is also in accord with other studies showing fucoidan could inhibit cancer metastasis through modulating the EMT pathway.43,44
Moreover, VMW-FC treatment significantly promoted sub-G1 phase accumulation in cell cycle analysis, impling DNA fragmentation during CRC cell death. We further confirmed that this cell death was initiated through the apoptotic pathway rather than the necrosis pathway by double-staining with Annexin V/7-AAD. This finding was also supported by the increased levels of the pro-apoptotic protein Bax and the decreased levels of the apoptosis-inhibiting protein Bcl-2. These results align with another study that showed fucoidan induces apoptosis in prostate cancer by upregulating Bax and downregulating Bcl-2. 45 Furthermore, as cyclins and cyclin-dependent kinases (CDKs) are key regulators of cell cycle in each phase, which directly regulate DNA replication and cell proliferation, 46 we discovered that VMW-FC inhibited multiple cyclins (Cyclin A2, B1 and D1) and CDKs (CDK2, 4 and 6), eventually causing cell cycle arrest as displayed in a cell cycle representative diagram (Figure 3A). Consistent with these findings, fucoidan has previously been shown to inhibit cyclin D1, cyclin E, CDK2, and CDK4 in HT29 cells, further inducing G1-phase cell cycle arrest. 47 Additionally, Ki-67 and PCNA, the classic markers of cell proliferation, 36 were also significantly inhibited following VMW-FC treatment. Notably, other study also revealed the inhibitory effects of fucoidan on Ki-67 and PCNA in hepatocellular carcinoma, leading to the suppression of cancer cell proliferation. 48
Last, in addition to in vitro studies, we also evaluated the effects of VMW-FC on in vivo tumorigenesis using a tumor-bearing mouse model. HT29 has been reported to possess characteristics of mature intestinal cells, such as enterocytes and mucus-producing cells, 49 making it more closely resemble the microenvironment of human colonic tissues. Therefore, we selected HT29 as the target cell line for our animal study. Our results showed a significant reduction in tumor size and volume in the VMW-FC treatment group compared to the control. Further, to address safety concerns, we observed tissue sections of the liver and kidney post-treatment. No morphological changes were detected, indicating minimal renal and hepatic toxicity in vivo.
However, the current research of VMW-FC effects on CRC, while promising, has several limitations. More colorectal cancer cell lines will be required to represent the full spectrum of CRC. The translation from the NOD-SCID mice model to humans needs more consideration. The cocktail nature of VMW-FC makes it challenging to pinpoint the specific ingredient responsible for the observed effects. Moreover, the potential effects on other cancers needs further investigation.
Conclusively, VMW-FC could effectively impede CRC with no further side-effects, through inhibiting cell cycle regulators, promoting pro-apoptotic protein, and deregulating the EMT-inducing markers (Figure 7), which underlined its potential as a novel cocktail therapy for CRC in the future.

The schematic VMW-FC regulated signaling pathway in colon cancer. The red colored arrows indicate upregulated and downregulated levels during VMW-FC treatment.
Footnotes
Acknowledgements
We are highly grateful to Enhance Biotechnology Inc., Taipei, Taiwan for providing fucoidan complex-based functional beverage.
Author Contributions
Conceptualization, C.-Y.W. and C.-H.C.; methodology, C.-Y.W., C.-H.C. and B.-Y.P.; validation, B.-Y.P., P.-C.C. and F.-C.T.; formal analysis, P.-C.C., F.-C.T. and Y.-H.D.; investigation, Y.-H.D. and W.T.; data curation, C.-Y.W., B.-Y.P.; writing—original draft preparation, C.-H.C. and P.-C.C.; writing—review and editing, F.-C.T. and W.-C.L.; visualization, Y.-H.D. and W.T.; supervision, W.-C.L. and W.-P.D.; project administration, W.-P.D.; funding acquisition, W.-P.D. All authors have read and agreed to the published version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by Ministry of Science and Technology Taiwan (MOST 110-2314-B-038-026); Stem Cell Research Center, Taipei Medical University (104-5600-001-400); Taipei Municipal Wanfang Hospital (111-wf-f-6) and Enhance Biotechnology Inc., Taipei, Taiwan.
