Abstract
BACKGROUND:
Homeobox A5 (HOXA5) is a member of the HOX protein family which is involved in several carcinogenesis pathways, and is dysregulated in many cancer types. However, its expression and function in human colorectal cancer (CRC) is still largely unknown.
OBJECTIVE:
This study aimed to evaluate HOXA5 expression in Tunisian patients with CRC in order to define new potential biomarker.
METHODS:
An immunohistochemical labeling using an HOXA5 antibody was performed on 85 formalin fixed paraffin embedded specimens from patients with CRC. Six normal colon mucosa cases were used as controls.
RESULTS:
HOXA5 expression showed a cytoplasmic staining in both tumor and stromal/endothelial cells. Loss or low HOXA5 expression was seen in tumor cells in 74/85 cases (87.06%) and in stromal/endothelial cells, in 77/85 (90.59%). In control group of normal colon mucosa HOXA5 was moderately expressed in all the cases. The abnormal expression, was significantly associated to lymph nodes metastasis in tumor cells (
CONCLUSION:
HOXA5 immunostaining results suggest the valuable role of this protein in colorectal carcinogenesis. Moreover, the association of lymph node metastasis to HOXA5 abnormal expression underlies its crucial role in colorectal cancer dissemination and prognosis.
Introduction
Colorectal Cancer (CRC) is currently the third most prevalent cancer worldwide [1, 2, 3, 4]. It is the most common digestive malignant tumor leading to death with a high mortality rate of 10.2% for both men and women [4]. In Tunisia, the CRC mortality rate is lower, estimated to 6.6% [4]. Metastases occur in about 50% of patients and around 25% of CRC patients present metastases at initial diagnosis [5]. Currently, the standard therapeutic protocol for non metastatic colorectal cancer is to combine surgery and chemotherapy. In metastatic CRC, the main option is a combination with targeted therapies using anti-Epithelial Growth Factor Receptor (EGFR) for patients with RAS wild type Status (exon 2, 3 and 4 of both KRAS and NRAS genes) or anti-Vascular Endothelial Growth Factor Receptor (anti-VEGFR) for patients with mutated RAS [6, 7]. Other EGFR pathway effectors could be mutated like BRAF (V600E mutation). This effector is not recommended for anti-EGFR therapy but only as a prognostic biomarker [7]. However, some patients develop therapeutic resistance which could be due to alterations in the signaling pathways that control the cell cycle, apoptosis, cell migration. Resistance could be promoted by current biotherapies targeting limited number of specific pathways. Therefore, other biomarkers involved in the regulation of several processes in carcinogenesis could be further relevant targets in oncotherapy.
Homeobox A5 (HOXA5), a member of homeobox genes which encode homeoproteins, acts as transcription factor implicated in regulating human embryonic development and adult stem cell differentiation by activating or suppressing downstream target genes. In addition, HOXA5 plays an important role in carcinogenesis by modeling the expression of various proteins involved in proliferation, apoptosis, angiogenesis, invasion and metastasis [8, 9]. HOXA5 gene can act as anti-proliferative and tumor suppressor by inhibiting cell proliferation and stimulating apoptosis [10, 11]. Aberrant expression and dysfunction of HOXA5 gene play an important role in the development of colorectal cancer as well as in leukemia and various malignant tumors, including lung cancer, breast, prostate, cervical and gastric cancer [11, 12, 13, 14, 15, 16, 17]. In addition, HOXA5 is involved in immune evasion and in transcriptional regulation of programmed death-ligand 1 (PD-L1) in melanoma cells [18]. It was reported that this gene plays a role in DNA repair mechanism in breast cancer cells through hMLH1 transcriptional regulation [19]. In colorectal cancer, high expression levels of HOXA5 were observed in patients with an improved probability of relapse-free survival [12]. In tumor tissue, it prevents invasion, angiogenesis and metastasis [21, 22, 23]. Moreover, in non-small cell lung carcinomas HOXA5 has the ability to suppress metastasis and is associated to prolonged survival [20].
In colorectal cancer, additional studies are required to elucidate the contribution of HOXA5 at cellular level.
Our study aims to evaluate HOXA5 expression in colorectal cancer in Tunisian patients in order to define a new potential biomarker.
Material and methods
Material
Eighty five Formalin Fixed Paraffin Embedded (FFPE) tissue samples of colorectal cancer were included. Six tissue specimens from normal colon mucosa cases were used as controls. Specimens were collected from the Department of Human and Experimental Pathology at Institut Pasteur de Tunis between 2016 and 2018. Clinical and pathological related data have been collected from pathological reports and patient medical records. All patients have undergone RAS testing for the determination of the RAS status. Mutation assessed were exons 2, 3 and 4 of KRAS and NRAS (Table 1).
HOXA5 expression and clinicopathological features
HOXA5 expression and clinicopathological features
Written informed consents were obtained from all participants. Ethical approval according to the Declaration of Helsinki Principles was obtained from the Biomedical Ethics Committee of Institut Pasteur de Tunis (2017/6C/I/Cancer colorectal/V1).
Immunohistochemistry
Representative slides stained with Hematoxylin and Eosin (HE) were reviewed by a pathologist to select CRC tumoral areas for analysis and epithelial normal areas from controls and tumoral adjacent tissue.
For the immunohistochemistry (IHC) technique we proceeded first by the optimization step on normal colon biopsies to set the right concentration and the incubation time for the used antibody (anti-HOXA5 polyclonal antibody (clone H125 Santa Cruz Biotechnology). We preceded then in the same condition for all samples including external controls (normal biopsies) to ensure the same sensitivity of the technique. As an internal control, we considered the labeling of inflammatory and normal epithelial cells to eliminate false negatives.
Tissue sections of 3
Based on the immunostaining intensity, the distribution and the proportion of HOXA5 positive cells in the epithelium, HOXA5 expression was scored on a scale from 0 to 3 as following: 0: no immunostaining (loss); 1: less than 10% positive cells or weak intensity libeling (low); 2: 10 to 50% positive cells with moderate intensity libeling (moderate); 3: more than 50% positive cells with high intensity (high) [21].
Statistical analysis
SPSS 20.0 software (IBM, Chicago, IL, USA) was used for all statistical analyses. The
Results
The clinical data of the 85 CRC included in our series showed that the patients were distributed in 51 men and 34 women with an age ranging from 22 to 87 years (mean age 58.63). Patients were categorized by age into groups based on age at onset of colon cancer: young adults (ages 18–49 years), middle-aged adults (ages 50–64 years), and old adults (aged older than 65 years). 16.5% of patients were young adults. The control biopsies were from patients aged 21 to 70 years with a mean age of 35.33.
Immunohistochemical micrographs showing the differential expression of Homeobox A5 (HOXA5) in normal colon mucosa, and metastatic colorectal adenocarcinoma. (A) Normal colon mucosa with moderate HOXA5 expression (score 2) (magnification, X400). (B) The moderate HOXA5 staining in CRC versus adjacent normal mucosa (magnification, X200). (C) Colorectal cancer sample with high HOXA5 expression (score 2) (magnification, X200). (D) Colorectal cancer with low HOXA5 expression (score 1) (magnification, X400). (E) CRC with loss of HOXA5 expression (score 0) (magnification, X400).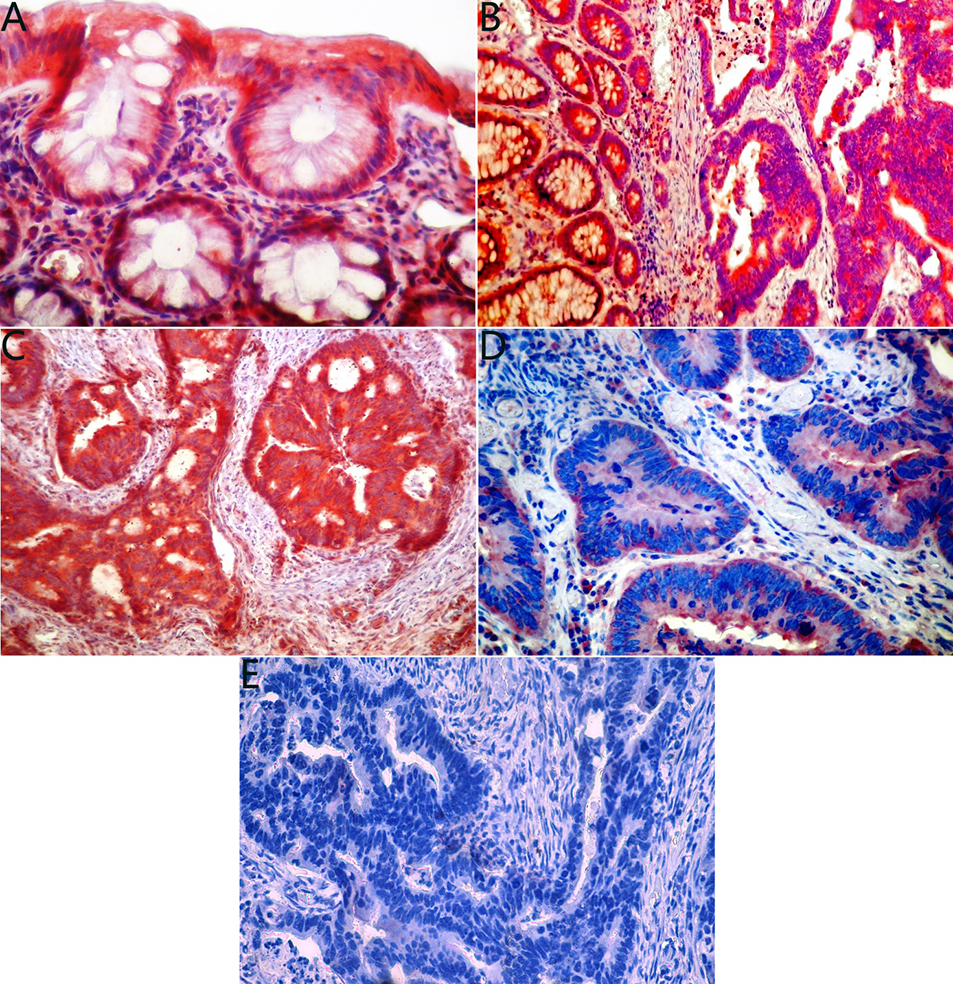
Association between HOXA5 expression in tumor and stromal/endothelial cells and clinicopathological features
The majority of tumors were located in the distal colon 39 cases (45.88%). Histological classification showed moderately differentiated adenocarcinomas in 59 cases (69.41%). including one case with a mucinous component, poorly differentiated in 20 cases (29.41%) and mucinous carcinoma in 2 cases (2.35 %). Tumors with high grade (Poorly differentiated, mucinous and carcinomas with mucinous component) according to the WHO classification of digestive tumor 2010 were found in 23 cases (27.06%). Regarding tumor invasion stage, T4 was found in 30/85 tumors (35.3%), T3 in 51/85 samples (60%), T2 in 3/85 samples (3.53%) and only one samples (1.17%) was classified T1. Tumors confined to the bowel wall (T1, T2 and T3) were grouped in early stages, they represented 64.71% of all tumors. T4 was considered as advanced stage as it invaded adjacent colon tissues. Lymph node metastasis (LNM) was noted in 54 patients (63.53%). RAS testing was performed for all patients, 38 (44.7%) showed wild type status. Clinical, pathological and RAS testing features are summarized in Table 1.
Histological selection of the CRC cases showed that 54 samples included normal adjacent tissues. These areas were analysed with the tumor tissue in IHC. HOXA5 immunohistochemical study was performed in all the 85 CRC cases, in the 54 adjacent normal tissues and in the six normal colon mucosa controls. HOXA5 expression was observed in both tumor cells and stromal/endothelial cells. The immunostaining results showed that in all the control cases, HOXA5 protein was moderately expressed (score 2) in epithelial and stoma cells (Fig. 1). In tumor cells, HOXA5 expression was as follows: 18/85 negative cases (score 0) (21.18%), low expression (score 1) in 56/85 cases (65.88%) and 11/85 cases (12.94%) with moderate expression (score 2). Moreover, in stromal/endothelial cells, HOXA5 expression was absent (score 0) in 38/85 cases (44.70%), low (score 1) in 39/85 cases (45.88%) and moderate (score 2) in 8/85 cases (9.41%). The HOXA5 expression in adjacent normal tissues was the same as in tumor cells in all 54 cases. In comparison with the HOXA5 pattern in normal mucosa cases, score 0 and score 1 were considered as abnormal HOXA5 expression, score 2 was considered as a normal feature. HOXA5 abnormal expression was shown in tumor cells in 87.1% (74/85) of the CRC cases and in stromal/endothelial cells in 90.6% (77/85) of cases. The HOXA5 immunostaining features are illustrated in Fig. 1.
Among the 54 patients with LNM, abnormal HOXA5 expression was shown either in tumor cells in 81.5% (44/54) and in stromal/endothelial cells in 85.2% (46/54) of cases. LNM was significantly associated with the abnormal HOXA5 expression in stromal/endothelial cells (
Several gene expression studies using qRT-PCR have demonstrated that HOXA5 is involved in tumor suppression, invasion prevention, angiogenesis and metastasis, with a potential of wideranging significance for applications in oncology [13, 20, 21, 22, 24, 25, 26, 27, 28]. Tissue and cellular expression of HOXA5 protein could lead to better understanding its role in colorectal carcinogenesis and to assess its value as a potential prognostic marker. To the best of our knowledge, and in contrast to other malignant tumors immunohistochemical study of HOXA5 in colorectal cancer and its association with LNM was not previously performed.
In our study, we assessed the immunohistochemical HOXA5 protein expression in normal colon mucosa and in CRC samples. Our results showed that HOXA5 was normally expressed (score 2) in normal mucosa. In contrast, the protein was under expressed in CRC (score 0, score 1) in both tumor cells and stromal/endothelial cells, respectively in 74/85 cases (87%) and 77/85 cases (90%). Loss of HOXA5 expression (score 0) was found in the stromal/endothelial cells (45%) more than in tumor cells (21%). Our results are in line with those previously reported in breast cancer in which loss of HOXA5 expression was observed in more than 60% of breast cancer cell lines [29]. Comparing to normal breast tissues, nearly 70% of all breast carcinomas showed decreased HOXA5 protein levels [30].
In our study the expression of HOXA5 in adjacent non-tumor tissues of all samples included (54 samples) was similar to that in tumor cells, these results are in agreement with the study of Watanabe et al. in which they did not identify a difference between HOXA5 expression in tumor tissue and in their corresponding non-tumor tissue by RT-qPCR [31]. These adjacent non tumoral areas couldn’t be considered as normal tissues.
Our immunohistochemical staining showed a cytoplasmic location of HOXA5 expression in both normal and tumor cells. Ordonez-Moran et al. reported both cytoplasmic and nuclear HOXA5 expression, in primary colorectal adenocarcinoma [12], suggesting that HOXA5 could play besides its function as transcription factor, other molecular functions [32]. IHC is a gold standard for evaluating the expression level of proteins on FFPE tissues and it is used as a diagnostic tool for many diseases. This technique is very sensitive when we compare normal to pathologic tissues but lack reproducibility when the pre-analytical steps are not standardized. We didn’t found nuclear staining on our samples; this could be related to the use of a different antibody or differences in the pre-analytical steps.
Previous studies of HOXA5 mRNA expression levels demonstrated a down-regulation in colorectal cancer [12, 28, 33]. HOXA5 mRNA was expressed in quiescent endothelial cells, but it was absent in activated angiogenic endothelial cells [34, 35]. This loss could be explained by several molecular alterations, including HOXA5 promoter hyper-methylation. Several studies demonstrated an inverse correlation between HOXA5 expression and methylation level in colorectal cancer compared to normal tissues [28]. Furthermore, HOXA5 high methylation levels, was associated with a slightly worse survival rate [28]. A significant progressive down-regulation of HOXA5 mRNA expression was shown from normal colon tissue to adenoma and to carcinoma [12]. This down-regulation has been found in several other malignant tumors such as breast, lung, cervix, oral cavity and stomach. Moreover, the levels of HOXA5 expression are significantly down-regulated in cervical cancer cell lines and [11] as well as in oral squamous cell carcinoma and gastric cancer tissues and cell lines [36, 37].
HOXA5 down-regulation might be due to microRNAs (miRNAs) overexpression which promotes tumor proliferation and metastasis. The miRNAs are a class of small, non-coding single-stranded, endogenous RNAs that negatively regulate expression targeted gene at the post-transcriptional level by base pairing complementarity of miRNA with the targeted messenger RNA [38, 39, 40]. These miRNAs have an important role in the pathogenesis of various diseases, including cancer [41]. In CRC cells, it was reported that HOXA5 expression was down-regulated by miR196B [42]. In addition, it has been demonstrated that miR-429 mediates tumor growth and metastasis by targeting HOXA5 [33]. Moreover, reported results in non-small cell lung cancer, showed that HOXA5 expression could be down-regulated by miRNA-196a which promote cell proliferation, invasion and migration [22] and by miR-130a which dynamically regulate HOXA5 gene expression [43].
In our study, the HOXA5 abnormal expression in stromal/endothelial cells was associated with age specifically with middle-aged adult male group (
HOXA5 is known as an inhibitor of angiogenesis, invasion and metastasis. In our series, among the 85 patients with CRC, lymph node metastasis was reported 54 cases. Loss or low expression of HOXA5 was significantly associated with lymph node metastatic status in 77 cases in stromal/endothelial (
Ordonez-Moran et al., reported that the high-level expression of HOXA5 gene was a marker of a good prognosis in patients with colon cancer [12]. In addition, it was shown that the high expression level of HOXA5 was associated to an improved probability of relapse-free survival in colorectal cancer [27]. HOXA5 interacts with VEGF and EGF signaling pathways implicated respectively in angiogenesis and proliferation. The over-expression of HOXA5 down-regulates the expression of proangiogenic factors such as VEGFR2 and promotes anti-angiogenic factors [35]. In contrast, the under-expression of HOXA5 was associated to tumor progression and could be a poor prognosis marker in patients with gastric cancer [17], oral squamous cell carcinoma [36] as well as non-small cell lung cancer [44]. The loss of HOXA5 expression was inversely correlated to EGFR expression in breast cancer [30].
The abnormal HOXA5 expression alters its interaction with key molecular pathways involved in proliferation, apoptosis, angiogenesis and metastasis. The loss of HOXA5 expression in malignant breast cells results in the acquisition of more aggressive phenotypes by loss of epithelial cell characteristics and an increase in stemness and cellular plasticity [45]. In the other hand, HOXA5 is suppressed by Wnt Signaling pathway which promotes tumor progression and metastasis [12, 46]. In gastric cancer, it was demonstrated that HOXA5 increased the expression of p21 and decreased c-Myc and Ki67, which leads to the inhibition of cancer cell proliferation [17]. In other malignant tumors such as cervical cancer, HOXA5 overexpression stimulates apoptosis by inducing caspase 3 activity and abolished cell viability, proliferation and invasion by regulating protein kinase B (AKT)/p27 [11]. Moreover, it was demonstrated that the high expression of HOXA5 in osteosarcoma cells inhibited their survival and proliferation, and promoted apoptosis through activation of p53-p38
No association was found between RAS status and HOXA5 expression (
Given the association between HOXA5 expression and LNM and known its role as an inhibitor factor of cell proliferation and angiogenesis, our findings suggest that loss or under-expression of HOXA5 could enhance the tumor aggressiveness in colorectal cancer and the resistance to anti-EGFR therapy. These results could be improved by increasing the number of patients and controls to better assess the associations between HOXA5 expression and prognosis.
Conclusion
In summary, our study demonstrates that HOXA5 abnormal expression (loss or low expression) was associated to lymph node metastasis, suggesting hence its pivotal role as prognostic biomarker for patient with colorectal cancer. A better understanding of the interactions between HOXA5 and the key molecular pathways could bring new insights to the use of HOXA5 as new wide range potential biomarker.
Footnotes
Conflict of interest
None.
