Abstract
Colorectal cancer is a major public health problem and fourth guiding cause of cancer-induced mortality worldwide. The five-year survival rate for patients with colorectal cancer remains poor, and almost half of colorectal cancer patients present recurrence and die within five years. The increasing studies showed that long non-coding RNA (lncRNA) was involved in colorectal cancer. Therefore, this study was used to explore molecular mechanisms of nuclear paraspeckle assembly transcript 1 (NEAT1) in colorectal cancer. The real-time quantitative polymerase chain reaction (RT-qPCR) was employed to estimate the expression levels of NEAT1, Nuclear receptor 4 A1 (NR4A1), and miR-486-5p in colorectal cancer tissues and cells. Kaplan-Meier curve was conducted to analyze relationship between survival time of colorectal cancer patients and level of NEAT1. The protein levels of NR4A1,
Introduction
Colorectal cancer is commonly diagnosed cancers worldwide, and its incidence and mortality rates are increasing per year, especial in developed regions and countries [1, 2]. In the past few decades, chemotherapy was a mainstream treatment method to suppress invasion and metastatic of tumor cells for colorectal cancer patients at advanced stages, which resulted in huge burden of economy and body for colorectal cancer patients. Besides, formation of drug resistance is a major obstacle for increasing the overall survival time of colorectal cancer patients [3]. Therefore, enormous efforts are needed to clarify the pathogenesis for colorectal cancer.
Recently, long non-coding RNAs (lncRNAs), more than 200 nucleotides (nt) in length and without coding-protein ability [4], have been widely acknowledged as pivot roles in multiple biological activities, including cell differentiation [5], maintains active chromatin [6], and carcinomas development [7]. Besides, previous studies have shown that nuclear paraspeckle assembly transcript 1 (NEAT1) was dysregulation in various cancers, such as gastric cancer [8], ovarian cancer [9], and breast cancer [10]. Importantly, recent studies have highlighted that NEAT1 was highly expressed and acted as a biomarker for survival prognosis in colorectal cancer [11]. Therefore, functional effects of NEAT1 was investigated in colorectal cancer.
Additionally, microRNAs (miRNAs) are a group of non-coding RNA molecules with 18–25 nucleotides [12]. Although miRNAs are no ability to encode protein, it is closely associated with the occurrence and development of human malignant tumors [13]. For instance, Liu et al. indicated that miR-19a was involved in occurrence and progression of colorectal cancer via mediating proliferation and migration of colorectal tumor cells [14]. As for miR-486-5p, a previous research revealed that miR-486-5p was decreased and functioned as an inhibitor in the development of colorectal cancer [15]. Zhang et al. also confirmed that transfection with miR-486-5p mimic into colorectal cancer cells could inhibit migratory and invasive ability of tumor cells [16].
Nuclear receptor 4 A1 (NR4A1), a transcription factor belong to the member of the steroid/thyroid hormone receptor family, is regulated by multiple factors, including serum, inflammatory factors, and stress [17, 18]. In addition, it has been suggested that frequent dysregulation of NR4A1 was closely associated with the development of most cancers by acting oncogene or tumor inhibitor [19, 20]. This adverse function of NR4A1 possible because of the different environments and regulatory patterns of tumors. In summary, NR4A1 could be served as biomarkers and targets for cancer diagnosis and treatment. Therefore, this study aimed to explore molecule mechanism of NEAT1, along with association relationship among NEAT1, miR-486-5p, and NR4A1 in colorectal cancer.
Materials and methods
Patient specimens
All colorectal cancer tissues (
Cells culture
Human NCM460 cells (INCELL, San Antonio, TX, USA) were maintained in Dulbecco’s Modified Eagle Medium (GIBCO BRL, Grand Island, NY, USA) supplemented with 10% (v/v) fetal bovine serum (FBS; PAN, Aidenbach, Bavaria, Germany). SW620 and HT29 cells were purchased from Shanghai Cell Bank at the Chinese Academy of Sciences (Shanghai, China) and cultured in 1640 medium (GIBCO BRL) containing 10% FBS (PAN). The above cell lines were cultured at 37
RNA isolation and real-time quantitative polymerase chain reaction (RT-qPCR)
Total RNA was extracted with TRIzol reagent (Solarbio, Beijing, China) from colorectal cancer tissues or cells. Afterward, qualified RNA was reverse transcribed to complementary DNA by Prime Script RT Reagent kit (Takara, Dalian, China). The transcription levels of NEAT1, NR4A1, and miR-486-5p was estimated by SYBR-Green Reagent kit (Qiagen, Hilden, Germany) under ABI Prism 7900 (Applied Biosystems, Foster City, CA). The expression level of RNA was normalized to glyceraldehyde-3-phosphate dehydrogenase (GAPDH) or endogenous small nuclear RNA U6 by using 2
The sequences of primers were listed:
NEAT1 (Forward, 5’-GUCUGUGUGGAAGGAGGAATT-3’; Reverse, 5’-UUCCUCCUUCCACACAGACTT-3’); NR4A1 (Forward, 5’-AGGGCTGCAAGGGCTTCT-3’; Reverse, 5’-GGCAGATGTACTTGGCGTTTTT-3’); miR-486-5p (Forward, 5’-CAGTCCTGTACTGAGCTGC-3’; Reverse, 5’-GTGCAGGGTCCGAGGT-3’); GAPDH (Forward, 5’-TCCCATCACCATCTTCCAGG-3’; Reverse, 5’-GATGACCCTTTTGGCTCCC-3’); U6 (Forward, 5’-AACGCTTCACGAATTTGCGT-3’; Reverse, 5’-CTCGCTTCGGCAGCACA-3’).
Western blot assay
Total protein was extracted from tissues or transfected cells by cell lysis buffer (Thermo Fisher Scientific, Waltham, MA, USA). The 60
RNA transfection and vector construction
The Specific small interfering RNA (siRNA) for NEAT1 (si-NEAT1), siRNA for NR4A1 (si-NR4A1) and its negative control si-NC, overexpressed plasmid of NR4A1 (NR4A1) and overexpressed plasmid of NEAT1 (NEAT1) and its negative control pcDNA, miR-486-5p mimic, and its negative control miR-NC were obtained from GenePharma (Shanghai, China). The oligonucleotides and vectors were transfected into SW620 and HT29 cells utilizing Lipofectamine 3000 (Thermo Fisher Scientific) referring to manufacturer’s instructions. Transfected cells were incubated in a humidified incubator at 37
3-(4, 5-dimethylthiazol-2-yl)-2, 5-diphenyl- 2H-tetrazol-3-ium bromide (MTT) assay
The transfected SW620 and HT29 were seeded into 96-well culture plates at the density 4
Flow cytometry
Flow cytometry was conducted for cell apoptosis assay. In brief, SW620 and HT29 cells were harvested and washed with cold phosphate buffer saline. Apoptotic cells were stained with 5
Transwell assay
The migration experiment was conducted with transwell plates that had 8-
The expression levels of NEAT1 and NR4A1 in colorectal cancer tissues and cell lines. (A and B) The relative expression level of NEAT1 was determined by RT-qPCR assay in colorectal cancer tissues and adjacent non-tumor tissues, as well as in colorectal cancer cell lines (HT29 and SW620) and normal colonic epithelial cells (NCM460). (C) Kaplan-Meier curve for survival time of colorectal cancer patients was displayed with log-rank test. (D, E, F and G) RT-qPCR and western blot assays were performed to assess the expression level of NR4A1 in colorectal cancer tissues and cell lines (HT29 and SW620), along with matched controls. (H) The correlation between NEAT1 and NR4A1 expression was analyzed in colorectal cancer tissues. 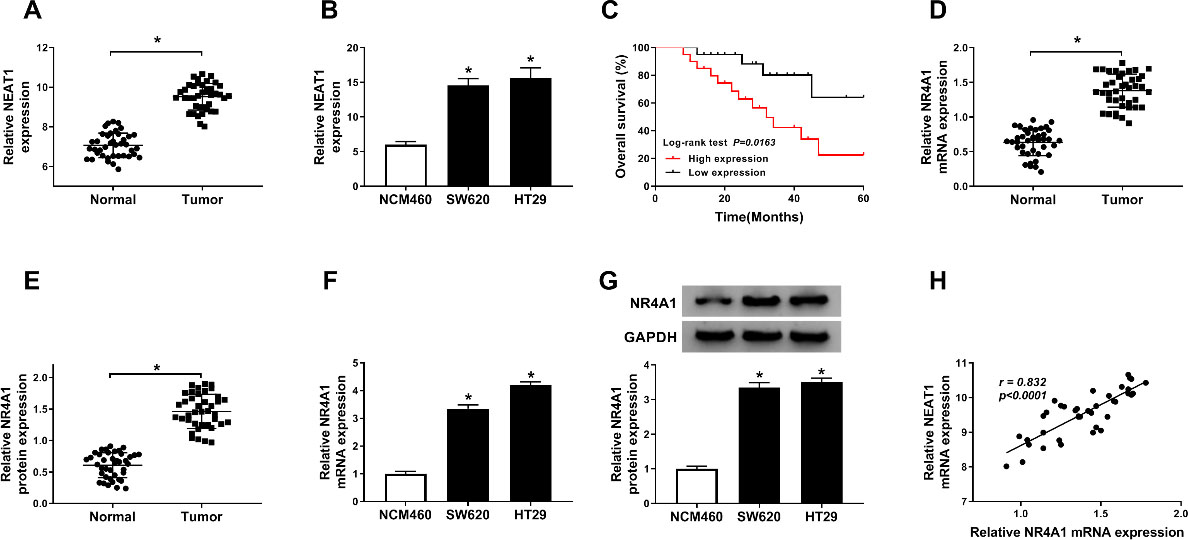
The online bioinformatics prediction software Starbase (
Statistical analysis
The function effects of NEAT1 silencing on proliferation, apoptosis, migration, and invasion of colorectal cancer cells. SW620 and HT29 cells were transfected with si-NEAT1 or si-NC. (A) The relative expression level of NEAT1 was assessed by RT-qPCR assay in SW620 and HT29 cells. (B and C) MTT assay was carried out to evaluate cell viability in SW620 and HT29 cells post-transfection. (D) Apoptosis of transfected SW620 and HT29 cells was measured by flow cytometry. (E and F) Transwell assay was employed to measure cell abilities of migration and invasion of SW620 and HT29 cells. 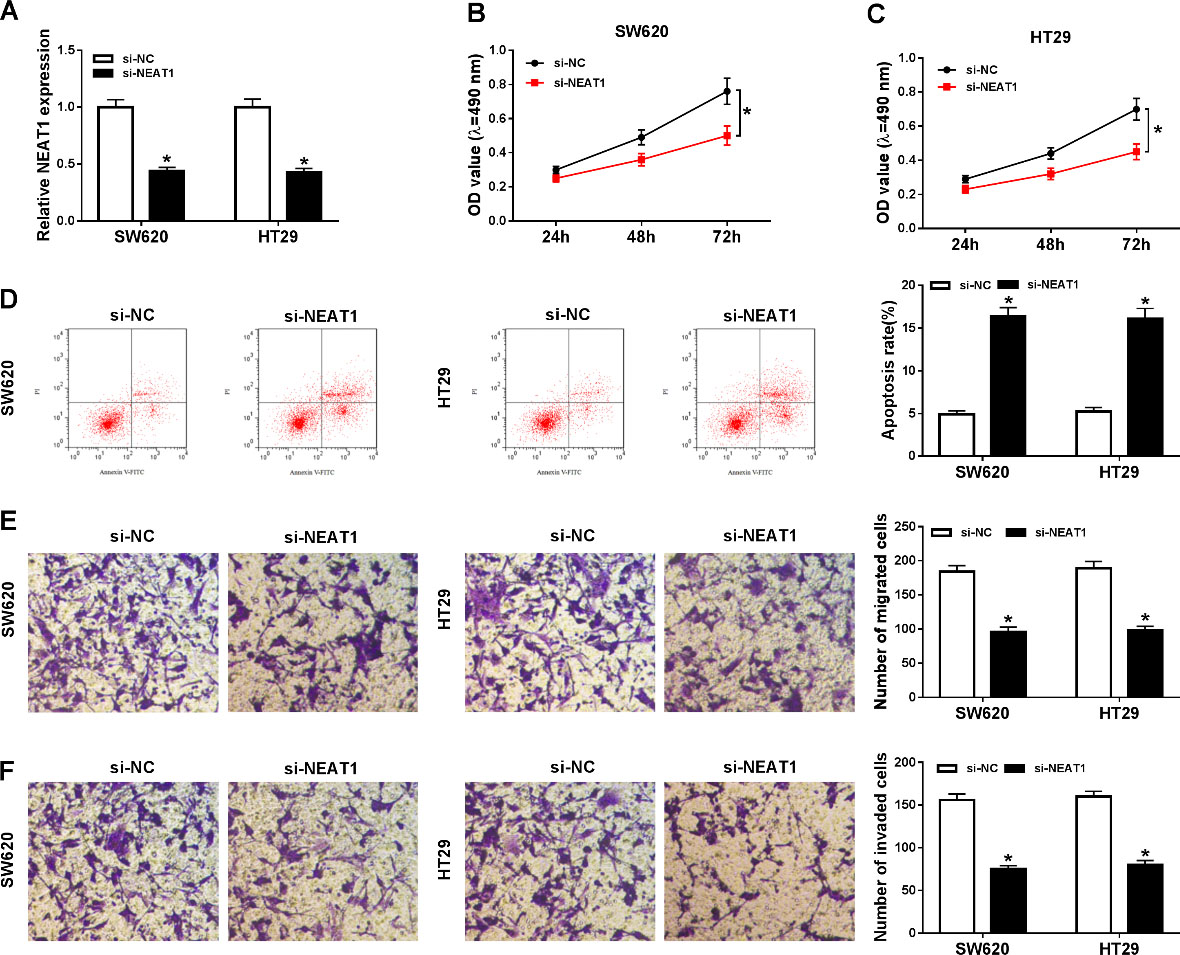
NEAT1 and NR4A1 were highly expressed in colorectal cancer tissues and cells
To begin with, we first examined NEAT1 level in colorectal cancer tissues and adjacent non-tumor tissues by RT-qPCR assay. As presented in Fig. 1A, when compared with the normal tissues, the expression level of NEAT1 was significantly upregulated in colorectal cancer tissues. The same conclusion was confirmed in Fig. 1B, RT-qPCR results indicated that NEAT1 was higher expression in SW620 and HT29 cells than that in normal colonic epithelial cells NCM460. In addition, Kaplan-Meier analysis results implied that patients with low NEAT1 expression had a longer overall survival time than those with high NEAT1 expression (Fig. 1C). We also discovered that NR4A1 also was overexpressed in colorectal cancer tissues when compared with normal tissues. No matter mRNA and protein (Fig. 1D–E). Moreover, our data also revealed that NR4A1 was upregulated in colorectal cancer cell lines in comparison with NCM460 cells (Fig. 1F–G). A positive correlation between NR4A1 and NEAT1 expression was found in colorectal cancer tissues (Fig. 1H). Above data suggested that overexpression of NEAT1 and NR4A1 was correlated with the development of colorectal cancer.
Knockdown of NEAT1 repressed proliferation, migration, and invasion while induced apoptosis of colorectal cancer cells
To identify potential effects of NEAT1 silencing on proliferation, apoptosis, migration, and invasion in colorectal cancer cells, SW620 and HT29 cells were transfected with si-NEAT1 or si-NC. Transfection efficiency was measured by RT-qPCR assay, and the results implied that knockdown of NEAT1 led to an obvious reduction of NEAT1 expression (Fig. 2A). Additionally, MTT analysis revealed that NEAT1 knockdown effectively impeded the proliferation of SW620 and HT29 cells (Fig. 2B–C). Conversely, SW620 and HT29 cells transfected si-NEAT1 showed higher apoptosis rate when compared with si-NC-transfected cells (Fig. 2D). Furthermore, the results of transwell assay indicated that the migration ability of SW620 and HT29 cells was greatly wakened following NEAT1 knockdown (Fig. 2E). Similar results were observed in invasion assay (Fig. 2F). These results demonstrated that NEAT1 knockdown obviously facilitated apoptosis and constrained proliferation and motility of colorectal cancer cells.
Knockdown of NR4A1 regulated proliferation, apoptosis, migration, and invasion of colorectal cancer cells. SW620 and HT29 cells were transfected with si-NR4A1 or si-NC. (A and B) The mRNA and protein levels of NR4A1 were measured by RT-qPCR and western blot assays in SW620 and HT29 cells, respectively. (C and D) The effect of si-NR4A1 on cellular viability was assessed by MTT assay. (E) Flow cytometry assay was conducted to test apoptosis of transfected SW620 and HT29 cells. (F and G) Cell migration and invasion capabilities were evaluated by transwell assay in transfected SW620 and HT29 cells. 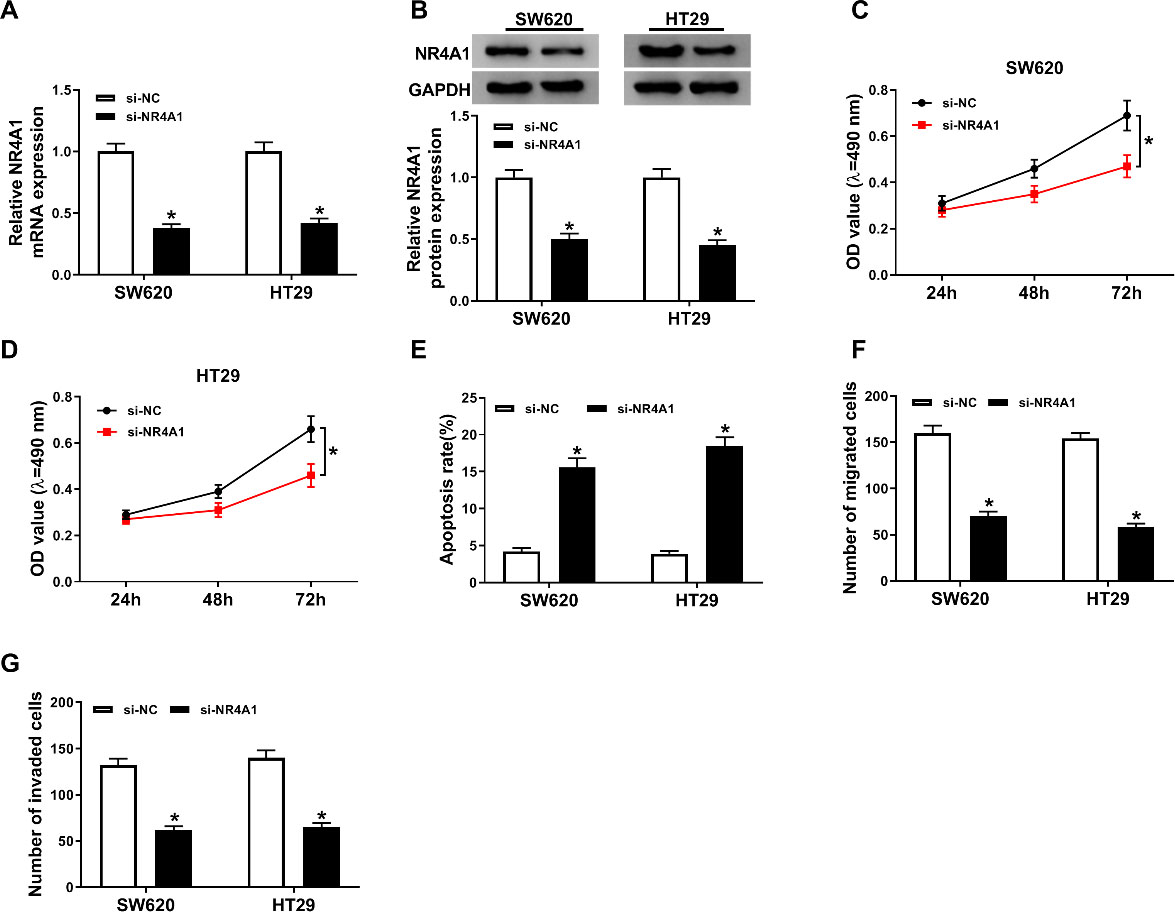
As NR4A1 was upregulated in colorectal cancer tissues and cells, we knocked down the expression of NR4A1 in SW620 and HT29 cells by performing loss-functional experiment. As shown in Fig. 3A, compared with si-NC-transfected SW620 and HT29 cells, the expression level of NR4A1 was decreased in SW620 and HT29 cells transfected with si-NR4A1. Moreover, knockdown of NR4A1 evidently repressed protein expression level of NR4A1 in SW620 and HT29 cells by western blot assay (Fig. 3B). Analogously, NR4A1 silencing impeded proliferation in in SW620 and HT29 cells when compared with the control group (Fig. 3C-D). We further probed the effect of NR4A1 knockdown on the apoptosis in SW620 and HT29 cells, and results implied that apoptosis of SW620 and HT29 cells was dramatically increased after inhibition of NR4A1 (Fig. 3E). Additionally, the migration and invasion results suggested that SW620 and HT29 cells transfected with si-NR4A1 remarkably inhibited the cell capabilities of migration and invasion in comparison with si-NC group (Fig. 3F-G). The above data indicated knockdown of NR4A1 might act as a tumor suppressor in colorectal cancer.
Overexpression of NR4A1 abolished the effects of NEAT1 silencing on proliferation, motility, and apoptosis of colorectal cancer cells. (A and B) The expression level of NR4A1 was measured by RT-qPCR and western blot assays in SW620 and HT29 cells transfected with si-NEAT1 or si-NC. SW620 and HT29 cells were transfected with si-NC, si-NEAT1, si-NEAT1+pcDNA, or si-NEAT1+NR4A1. (C and D) Cell viability of SW620 and HT29 cells was assessed by MTT assay. (E) Flow cytometry showed apoptosis rate of SW620 and HT29 cells after transfection. (F and G) Transwell analysis was used to show the migration and invasion abilities of SW620 and HT29 cells. 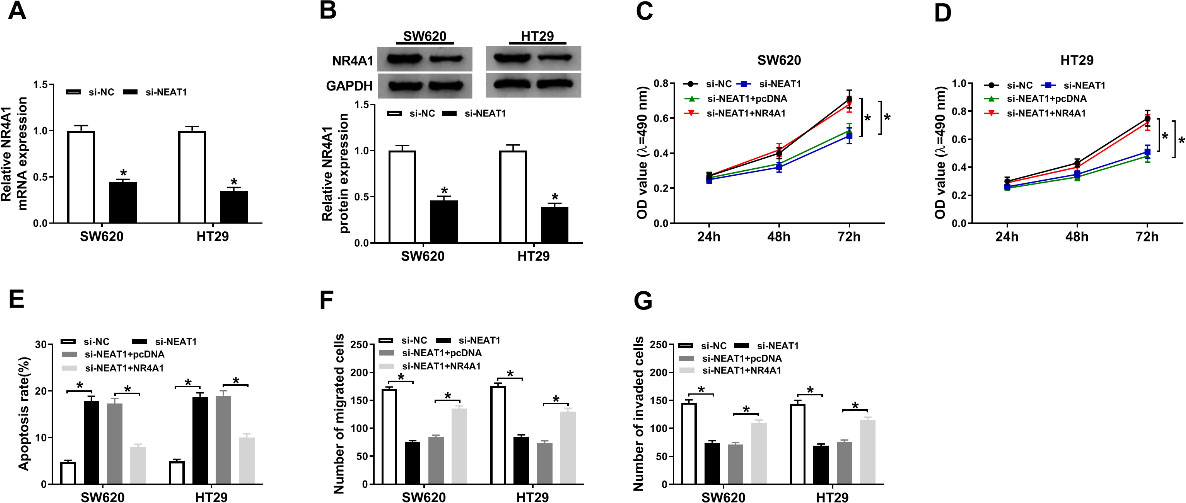
It reported that NEAT1 and NR4A1 were involved in proliferation, motility, and apoptosis of colorectal cancer cells in above results. As presented in Fig. 4A–B, RT-qPCR and western blot assays implied that the expression level of NR4A1 was inhibited in SW620 and HT29 cells after transfection with si-NEAT1 (Fig. 4A–B). In addition, overexpression of NR4A1 could significantly reduce cell viability loss in SW620 and HT29 cells caused by silencing NR4A1 (Fig. 4C–D). Flow cytometry results showed apoptosis rate was notably decreased in SW620 and HT29 cells transfected with si-NEAT1+NR4A1 when compared with cells only transfected with si-NEAT1 (Fig. 4E). Finally, transwell assay indicated that overexpression of NR4A1 effectively prevented knockdown of NEAT1-induced inhibitory effects on migration and invasion (Fig. 4F–G). Collectively, these data suggested that NEAT1 silencing repressed proliferation, motility, and facilitated apoptosis in colorectal cancer cells by regulating NR4A1 expression.
NEAT1 targeted miR-486-5p to regulate NR4A1 expression in colorectal cancer cells
The online bioinformatics tool was used to predict target miRNA of NEAT1. The possible binding sites between miR-486-5p and NEAT1, as well as its mutant sites were shown in Fig. 5A. Moreover, overexpression miR-486-5p conspicuously reduced the relative luciferase activity of WT-NEAT1 group, but not that of the MUT-NEAT1 (Fig. 5B–C). Furthermore, the increased expression of miR-486-5p was observed in SW620 and HT29 cells after silencing NEAT1, while overexpression of NEAT1 significantly repressed miR-486-5p expression in SW620 and HT29 cells (Fig. 5D–E). Besides, we found miR-486-5p could bind to the 3’UTR of NR4A1 by bioinformatics prediction (Fig. 5F). Subsequently, dual-luciferase reporter assay was applied to measure luciferase activity in SW620 and HT29 cells. As indicated in Fig. 5G–H, the luciferase activity in NR4A1 3’UTR-WT group was distinctly decreased by miR-486-5p mimic, while miR-486-5p mimic failed to affect luciferase intensity in NR4A1 3’UTR-WT group. In addition, western blot analysis displayed that transfection of miR-486-5p mimic prominently inhibited protein levels of NR4A1 in SW620 and HT29 cells, while transfection with NR4A1 evidently blocked this reduction (Fig. 5I–J). In summary, NEAT1 mediated NR4A1 expression in colorectal cancer cells by targeting miR-486-5p.
Target assay of NEAT1 and miR-486-5p. (A) The putative binding sites between NEAT1 and miR-486-5p were shown. (B and C) Relative luciferase activity was determined in SW620 and HT29 cells co-transfected with NEAT1 WT/MUT luciferase reporter and miR-486-5p mimic/miR-NC. (D and E) The RT-qPCR assay was conducted to test miR-486-5p level in SW620 and HT29 cells transfected with si-NC, si-NEAT1, pcDNA, or NEAT1. (F) The pairing regions between miR-486-5p and NR4A1, as well as their mutant regions were presented. (G and H) Dual-luciferase reporter assay was employed to examine luciferase activity in SW620 and HT29 cells. (I and J) The protein expression level of NR4A1 was determined by western blot assay in SW620 and HT29 cells transfected with miR-NC, miR-486-5p, miR-486-5p+pcDNA, or miR-486-5p+NR4A1. 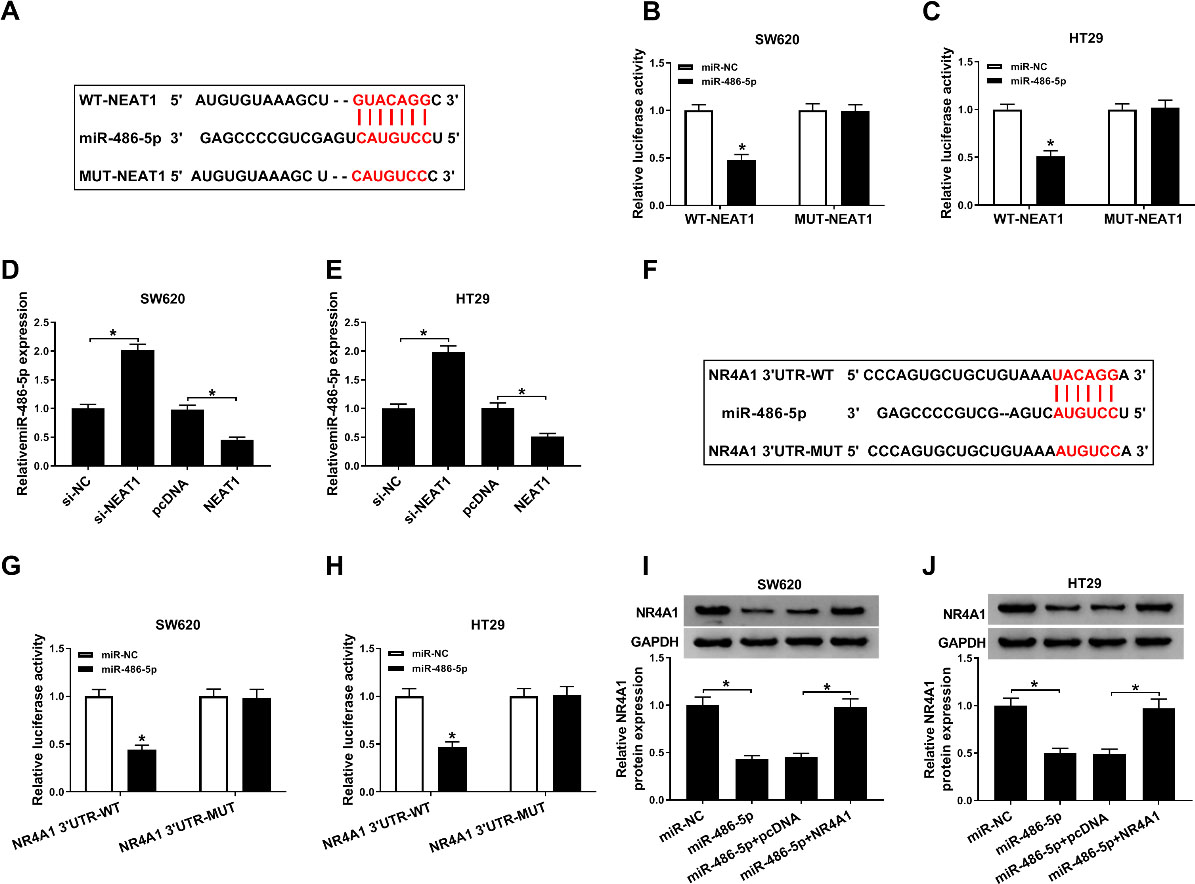
To further analyze the interaction relationship among NEAT1, miR-486-5p, and NR4A1, gain-functional experiment was performed to assess both mRNA and protein levels of NR4A1 by RT-qPCR and western blot analysis. The results elucidated that overexpression of NEAT1 could mitigate suppressive effect on NR4A1 expression in colorectal cancer cells caused by upregulation of miR-486-5p (Fig. 6A–C). Thus NEAT1/miR-486-5p/NR4A1 axis might play key function in colorectal cancer.
NEAT1 regulated NR4A1 expression in colorectal cancer cell lines via affecting miR-486-5p. (A, B and C) The mRNA and protein expression levels of NR4A1 were assessed by RT-qPCR and western blot assays in SW620 and HT29 cells transfected with miR-NC, miR-486-5p, miR-486-5p+pcDNA, or miR-486-5p+NEAT1. 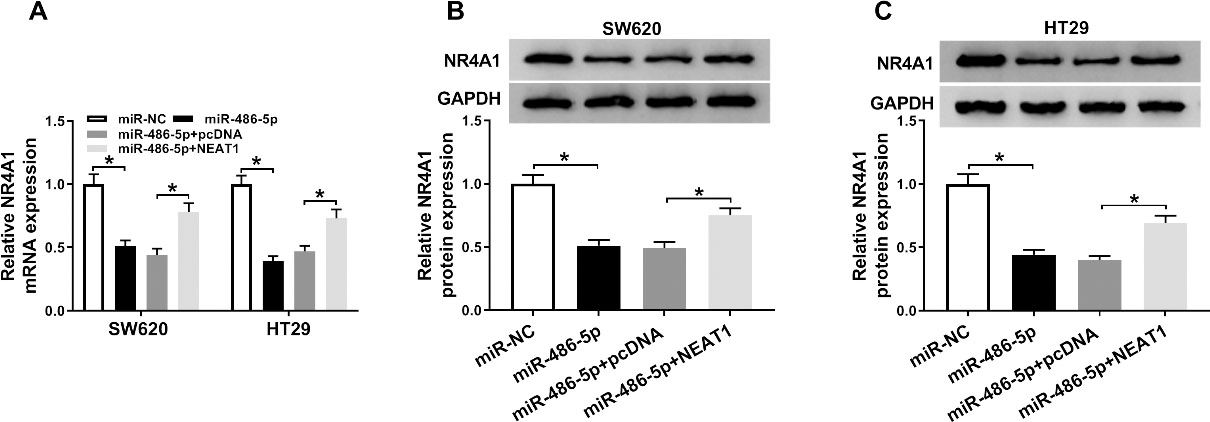
NEAT1 mediated Wnt/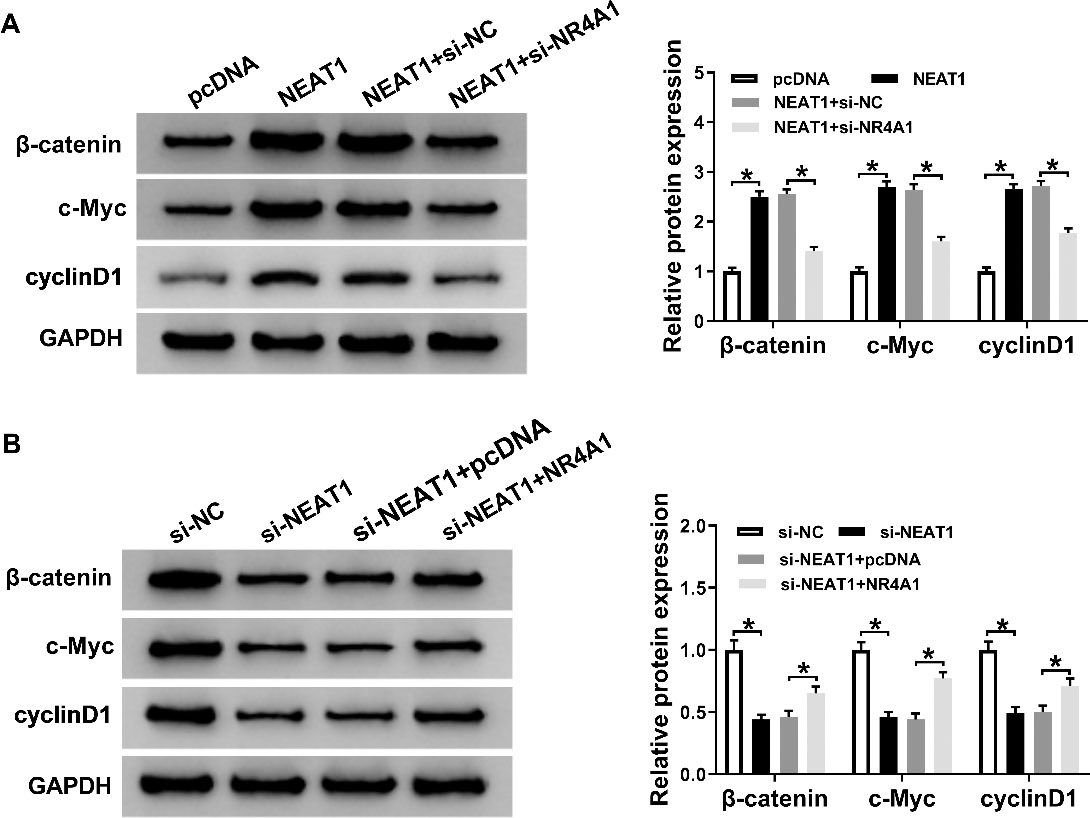
It had reported that Wnt/
Discussion
Currently, our results suggested that NEAT1 was dramatically increased in colorectal cancer tissues and cells compared with controls; at the meanwhile, knockdown of NEAT1 effectively repressed the abilities of proliferation and motility while enhanced apoptosis of colorectal cancer cells by targeting miR-486-5p/NR4A1 axis.
A series of studies highlighted the importance of lncRNA in colorectal cancer pathogenesis [21]. The regulation mechanism by which lncRNA mediated cellular behaviors was complex, which was involved multiple factors [22]. Typically, increasing studies have reported that lncRNAs functioned as miRNA sponges to regulate multiple cellular behaviors. Jiang et al. discovered that NEAT1 involved in the development of lung cancer by acting as a competing RNA to sponge hsa-mir-98-5p [23]. Similar results were reported by Luo et al. [24], NEAT1 acted as an oncogene in colorectal cancer by sponging miRNA-34a. At the present study, we demonstrated that NEAT1 was a biomarker in clinical outcome of colorectal cancer patients. Mechanistic investigation results suggested that NEAT1 could serve as competing RNA via sponging miR-486-5p to mediated NR4A1 expression in colorectal cancer.
Furthermore, Liu et al. reported that miR-486-5p was notably decreased in colorectal cancer tissues, especial in advanced stage cancer (stage III and IV), while neuropilin-2 was a target of miR-486-5p [15]. Surely, miR-486-5p could target several other pathways, like phosphatidylinositol 3-kinase regulatory subunit 1 (PIK3R1) [16]. In this study, our data revealed that miR-486-5p negatively targeted NR4A1 expression in colorectal cancer cells. Consistent with previous results [25], NR4A1 acted as carcinogenic gene in the development and progression of colorectal cancer. Furthermore, knockdown of NEAT1-induced effects on proliferation, apoptosis, migration, and invasion could be abolished by overexpression of NR4A1. Additionally, Wnt/
In summary, this research reported the cancerogenic roles of NEAT1 in colorectal cancer progression through mediating proliferation, metastasis, and apoptosis of colorectal cancer cells through regulating NR4A1/Wnt/
Footnotes
Conflict of interest
The authors declare that they have no competing interests.
