Abstract
OBJECTIVE:
To assess the expression levels of IFITM1 in human tissue samples and laryngeal squamous cell carcinoma (LSCC) cells, and to explore the potential mechanisms of IFITM1 in LSCC progression.
METHODS:
Quantitative PCR and immunohistochemical (IHC) assays were performed to detect IFITM1 expression in 62 LSCC tissues and corresponding normal tissues. We further detected the effects of IFITM1 on the proliferation, migration and invasion of LSCC cells and NF-
RESULTS:
We demonstrated the possible involvement of IFITM1 in the progression of LSCC. We found the upregulated expression of IFITM1 in human LSCC tissues and cells, and analyzed the correlations between IFITM1 expression and osteopontin. Our data further confirmed that IFITM1 affected cell proliferation, migration, and invasion of LSCC cells via the regulation of NF-
CONCLUSIONS:
We investigated the potential involvement of IFITM1 in the progression of LSCC, and therefore confirmed that IFITM1 was a potential therapeutic target for LSCC.
Keywords
Introduction
Laryngeal squamous cell carcinoma (LSCC) is one of the most common tumor types with aggressive clinical characteristics [1, 2, 3]. Patients with advanced LSCC have poor prognosis and high mortality [4, 5]. LSCC patients usually lack obvious early symptoms, and are often accompanied by cervical lymph node metastases and carotid sinus syndrome at advanced stages [6, 7]. The treatment of LSCC usually adopts surgical excision, chemoradiotherapy and combination therapy [8]. Recently, targeted therapy for LSCC has shown broad prospect [9]. However, there is still an urgent need to explore more promising therapeutic targets for treatment of LSCC.
IFN-induced transmembrane protein 1 (IFITM1), a17-kDa membrane protein, is known as a cell surface protein and is involved in the inhibition of virus entry into host cell cytoplasm [10, 11]. In lymphocytes, IFITM1 is also a part of membrane complexes that transduces homotypic adhesion signals [12]. For decades, IFITM1 has been widely known as a regulator in multiple cellular processes such as cell proliferation [13]. The essential role of IFITM1 in immunity, osteogenesis and angiogenesis has also been reported in previous studies [14, 15].
In the past decade, the key role of IFITM1 in the progression of multiple types of cancers has been documented [16, 17]. IFITM1 shows abnormally high expression in several cancers, such as colorectal cancer, breast cancer and lung cancer [16, 18, 19]. A previous study indicated that depletion of IFITM1 suppressed the proliferation, migration, and invasion of lung cancer cells in vitro [19]. In addition, the combination of IFITM1 depletion and radiotherapy suppresses the progression of oral cancer [20]. Similarly, IFITM1 also affects the growth of cervical squamous cell carcinoma [21]. So far, IFITM1 has been shown to be involved in the progression and metastasis of multiple types of cancers However, the possible role of IFITM1 in the progression and development of LSCC is still unclear.
In this study, we demonstrated the abnormally high expression of IFITM1 in human LSCC tissues and cell lines, and further found that IFITM1 could affect the proliferation, migration, and invasion of LSCC cells via p65-I
Materials and methods
Antibodies, plasmids, and primers
Rabbit anti-IFITM1 (1:200 dilution for IHC; 1:2000 dilution for Immunoblot, ab11259, abcam, Cambridge, UK), mouse anti-GAPDH (1:2000 dilution, ab8227, abcam, Cambridge, UK), rabbit anti-cyclin A (1:2000 dilution, ab16667, abcam, Cambridge, UK), rabbit anti-cyclin D1 (1:1000 dilution, ab37150, abcam, Cambridge, UK), rabbit anti-CDK2 (1:2000 dilution, ab38898, abcam, Cambridge, UK), rabbit anti-OPN antibody (1:2000 dilution, ab184276, abcam, USA), rabbit anti-p-I
The quantitative PCR primer sequences for the analysis of IFITM1 were as follows: forward, 5’-ATAGTT GCCGTTCGCATG-3’ and reverse, 5’-CTGACCGACT GATATCAGAG-3’; The quantitative PCR primer sequences for the analysis of GAPDH are as follows: 5’-CGACCACTTCGTCAAGCTCA-3’ and reverse, 5’-GGTGGAGCACAGGGTACCTTATT-3’. shRNA plasmids (Ready-to-package AAV) for the knockdown of IFITM1 were bought from the Addgene.
Immunohistochemistry
Tumor tissues were obtained from Affiliated Hospital of Nantong University. The experiments was the approved by the Ethics Committee at/of the Affiliated Hospital of Nantong University (Approval no. 2019-K047). To explore the expression levels of IFITM1 in human LSCC tissues, we performed immunohistochemistry (IHC) assays. Briefly, sections were fixed with 4% paraformaldehyde (PFA) for 30 minutes at room temperature and subsequently blocked with 2% BSA for 20 minutes. Slides were subsequently incubated with IFITM1 or GAPDH antibodies at room temperature for 2 hours. Then the sections were incubated with biotinylated secondary antibody for 1.5 hours, and diaminobenzidine was used as a chromogen substrate.
Cell culture and transfection
Three types of human LSCC cell lines, including AMC-HN-8 (BNCC33877, Beijing beina technology co. LTD, China), TU177 (YS578C, Shanghai Yaji Biology, China) and TU686 (BNCC100479, Beijing beina technology co. LTD, China), which were bought from the indicated company, were maintained in DMEM culture medium supplemented with 10% of fetal bovine serum (FBS), at 37
The shRNA plasmids were transfected into LSCC cells using lipofectamine 2000 (11668019, Invitrogen, USA). In a 6-well plate, we diluted 1
Quantitative PCR assay
Trizol (15596026, Invitrogen, USA) was used to extract total mRNA from human LSCC cells. Then the mRNAs from each type of cells were reverse-transcribed to cDNA by reverse transcriptase (M1701, Promega, Wisconsin, USA). Quantitative PCR was performed using SYBR Ex Taq kit (638319, Takara, Japan), and the expression levels of IFITM1 were normalized to the expression of GAPDH.
Immunoblot assays
Cells were lysed by RIPA lysis buffer (Beyotime, China) containing of protease inhibitors (Beyotime, China). Then the protein concentration was measured by BCA kit (Beyotime, China) and total proteins were were separated through SDS-PAGE (8% gels). After transfered onto polyvinylidene fluoride (PVDF) membranes, the membranes were blocked with 5% skim milk in TBST and incubated with primary antibodies for the detection of IFITM1, cyclin A, cyclin D1, CDK2, OPN (Osteopontin), p-I
MTT assays
Approximately 500 LSCC cells were seeded into 96-well plates, transfected with control or shIFITM1 plasmids and maintained for 48 hours. LSCC cells were then washed with PBS twice and incubated with MTT agent for 4 hours. Then dissolved in 150
Colony formation assay
One thousand cells were seeded into a 6-well culture plate and transfected with shRNA plasmids and maintained in a 37
Wound healing assay
LSCC cells were transfected with shRNA plasmids, maintained in serum-free culture medium for 48 hours and the wound was made mechanically with a 20-
Transwell assay
LSCC cells were transfected with control or shIFITM1 plasmids for 48 hours and then re-suspended in serum-free RPMI1640 culture medium. The upper chambers of filters (8.0
Reporter gene assay
LSCC cells were co-transfected with 0.8
Statistical analysis
GraphPad 6.0 was used for statistical analysis in this study. All data were represented as mean
IFITM1 was upregulared in human laryngeal squamous cell carcinoma (LSCC) tissues and cells. Quantitative PCR and immunohistochemical assays were performed to analyze the expression of IFITM1 in LSCC tissues and cells. A. Quantitative PCR assay showed an upregulation ofin IFITM1 in 62 human LSCC tissues samples. B. Immunohistochemical analysis of IFITM1 expression levels in LSCC tissues and the corresponding normal tissues (100 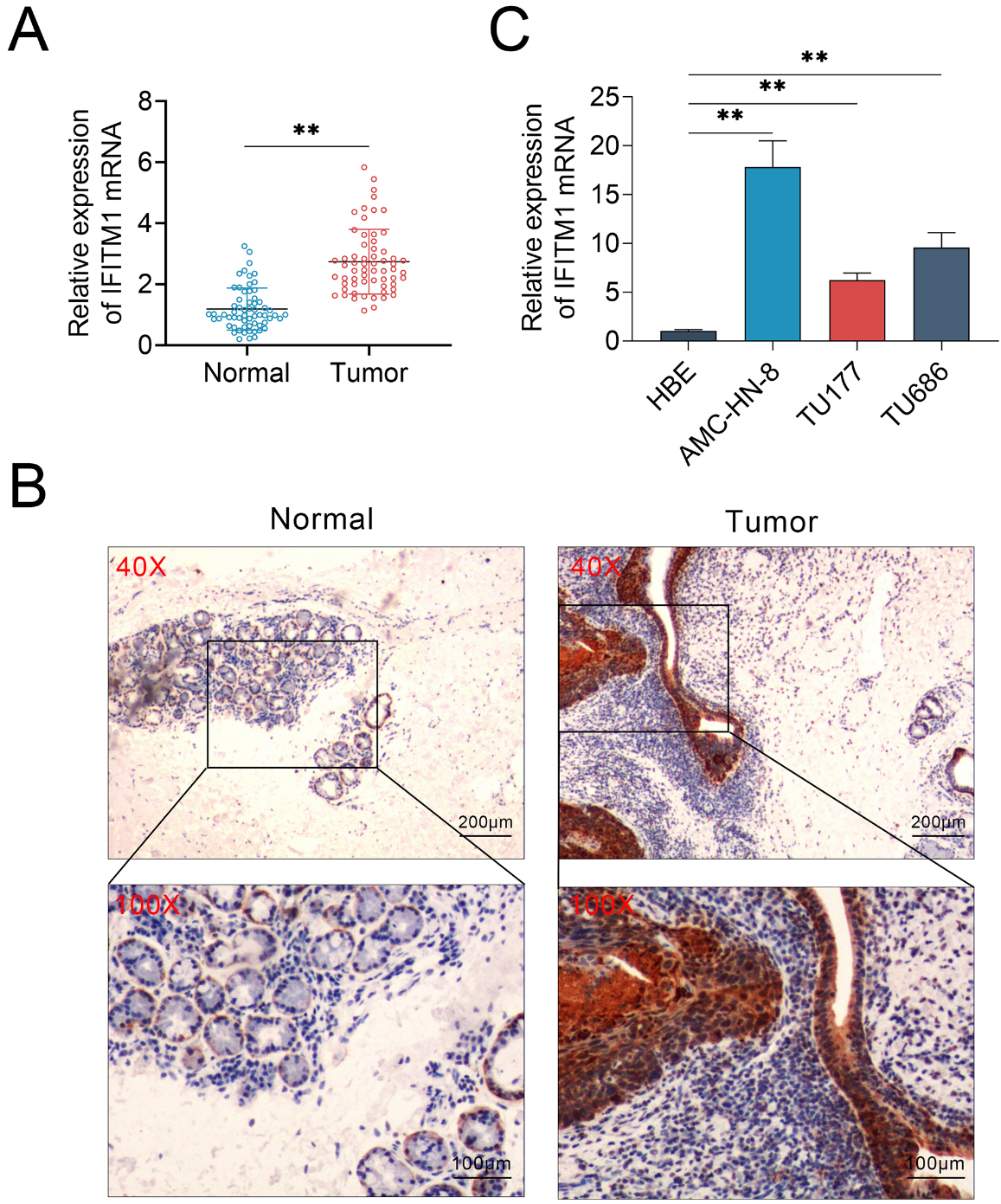
IFITM1 is upregulated in human laryngeal squamous cell carcinoma tissues (or IFITM1 over-expressed in human LSCC)
To explore the potential role of IFITM1 in the progression of laryngeal squamous cell carcinoma (LSCC), we compared its expression levels between tumor tissues and the adjacent normal tissues. A total of 62 LSCC tumor tissue were used in this study, and the mRNA levels or expression levels of IFITM1 in tumor tissues or corresponding normal tissues were detected through quantitative PCR assay and immunohistochemistry (IHC) assay, respectively. Interestingly, our results showed that the expression levels of IFITM1 mRNA was significantly upregulated in tumor tissues, compared to adjacent normal tissues (Fig. 1A). To further confirm the expression difference of IFITM1 in tumor and normal tissues, we performed IHC assays. In line with the quantitative PCR assays, the expression of IFITM1 in human LSCC tissues was profoundly increased, whereas the expression levels of IFITM1 in normal tissues were lower than in tumor tissues (Fig. 1B).
IFITM1 promotes the proliferation of LSCC cells in vitro. AMC-HN-8 cells were transfected with shIFITM1 plasmid or a control plasmid. A. Quantitative PCR assays showed significantly decreased expression levels of IFITM1 mRNA in IFITM1-shRNA transfected AMC-HN-8 cells. B. Immunoblot assays confirmed the efficient silencing of IFITM1 expression in IFITM1-shRNA-transfected AMC-HN-8 cells. C. The results of MTT assays showed the inhibition of cell proliferation caused by IFITM1 silence. D. Representive images showing the results of colony formation assays of AMC-HN-8 cells transfected with control or IFITM1 shRNA plasmids. The relative proliferation levels were quantified. E. Immunoblot assays indicated the expression level of cyclin A, cyclin D1, CDK2 and GAPDH in AMC-HN-8 cells transfected with the indicated plasmids. The experiments were repeated for at least three times. Results are presented as mean 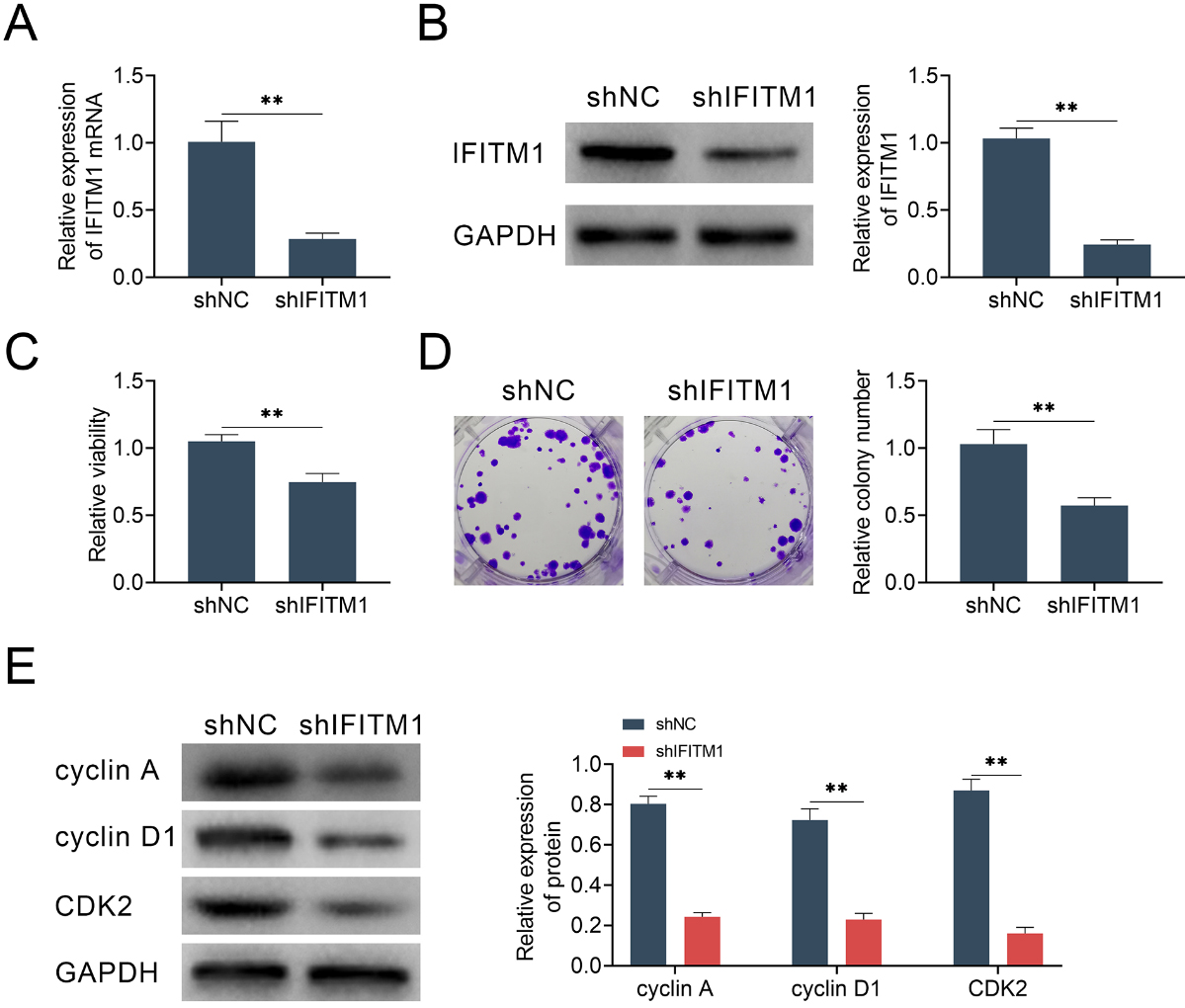
IFITM1 contributes to the migration and invasion of LSCC cells in vitro. A. Wound healing assays were performed using AMC-HN-8 cells transfected with control or shIFITM1 plasmids, and the relative wound width was detected. B. Transwell assays using control or IFITM1 depletion AMC-HN-8 cells were performed and the extent of transwell migration was quantified by relative cell number. The experiments were repeated for at least 3 times. Results are presented as mean 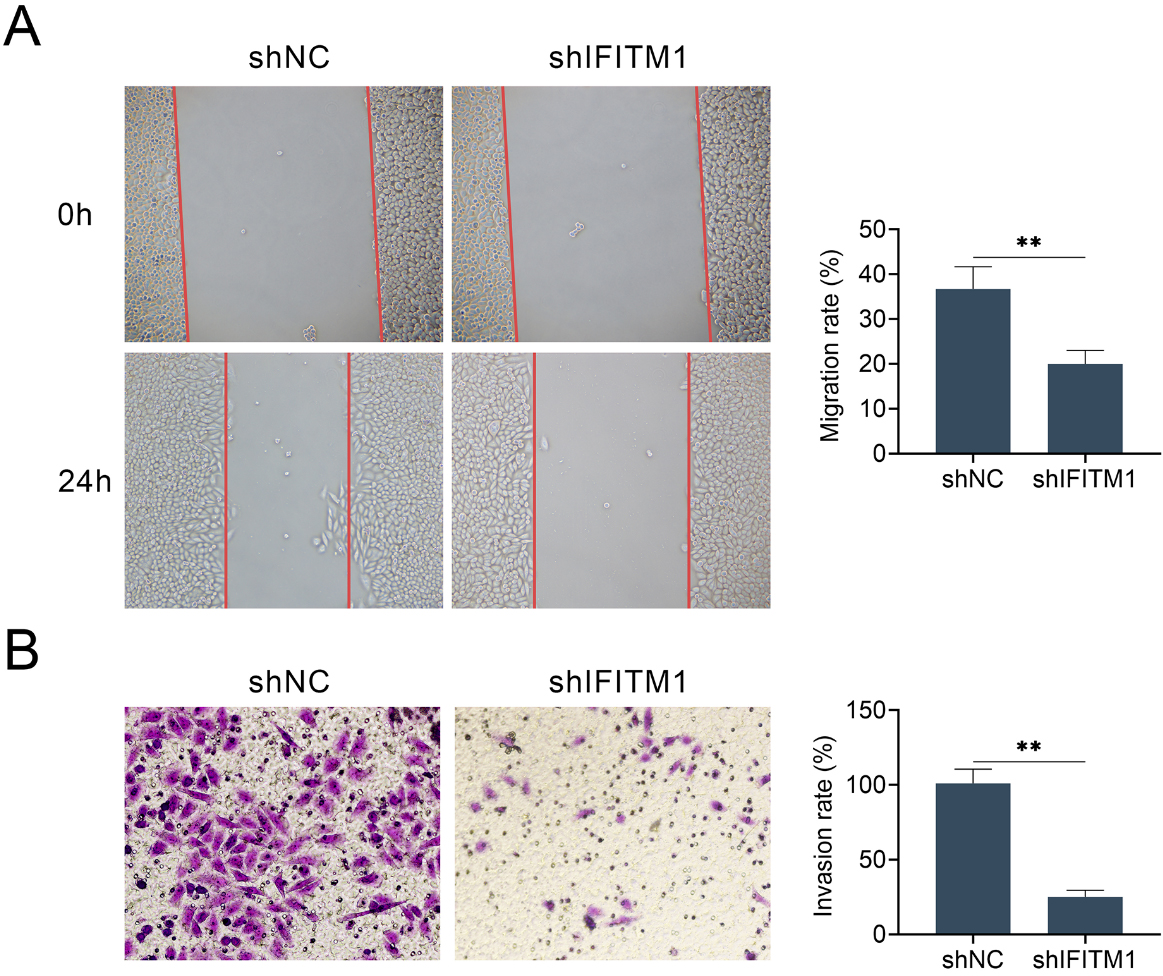
Subsequently, we detected the mRNA levels of IFITM1 in human normal bronchial epithelial cells (HBE) and LSCC cell lines, including AMC-HN-8, TU177 and TU686. Consistent with the previous results, we found that IFITM1 was dramatically up-regulated in LSCC cells, compared with normal bronchial epithelial cells (Fig. 1C). Together, these results suggest that IFITM1 shows abnormally high expression in human LSCC cells and tissues.
To further explore the function of IFITM1, we transfected the shRNA plasmid targeted against IFITM1 into human LSCC cell line AMC-HN-8 (the highest IFITM1 expression level), to suppress the expression level of IFITM1. As excessive cell proliferation can lead to tumorigenesis, therefore, the effect of IFITM1 on LSCC cells proliferation was examined. The results of quantitative PCR assays showed that the transfection of IFITM1 shRNA effectively decreased its expression level in AMC-HN-8 cell (Fig. 2A). Consistent with quantitative PCR results, the immunoblot assays further proved the obvious decrease of IFITM1 expression level in AMC-HN-8 cell transfected with shIFITM1 plasmid (Fig. 2B).
We further detected the effects of IFITM1 on cell proliferation of LSCC by MTT assays and colony formation assay. MTT assays showed that IFITM1 silence significantly decreased cell viability (Fig. 2C). Additionally, colony formation results showed that the colony formation capacity was significantly restrained after knocking-down IFITM1, indicated by the obviously decreased colony numbers (Fig. 2D). Next, we examined the effect of IFITM1 silence on the level of cell cycle control proteins such as cyclin A, cyclin D1 and CDK2. A significantly reduced expression of cyclin A, cyclin D1 and CDK2 in IFITM1 shRNA-transfected group was found, suggesting that the decline in proliferative capacity was mainly caused by cell cycle arrest (Fig. 2E).
Downregulation of IFITM1 blocks cell migration and invasion of LSCC cells in vitro
We next performed wound healing and transwell assays to evaluate the possible effects of IFITM1 on the migration and invasion of LSCC cells. As was expected, our results exhibited that IFITM1 silence remarkably inhibited the extent of wound closure in AMC-HN-8 cell (Fig. 3A). In addition, IFITM1 silence significantly blocked the invasion of AMC-HN-8 cell, with dramatically dropped cell numbers (Fig. 3B), confirmed by transwell assay. Collectively, our results indicated that IFITM1 inhibited cell migration and invasion of LSCC in vitro.
IFITM1 mediates NF-
B signaling pathway through the regulation of Osteopontin
LSCC is one of the most common malignancies in head and neck. In order to study the molecular mechanism of IFITM1 affecting the progression of LSCC, we focused on Osteopontin (OPN), a phosphoprotein which functions as a cell attachment protein and cytokine which regulate cancer growth and metastasis through two cell adhesion molecules. Through immunoblot assays, we found that OPN expression was significantly reduced after IFITM1 knockdown (Fig. 4A). It has been known that OPN is necessary for NF-
IFITM1 mediates NF-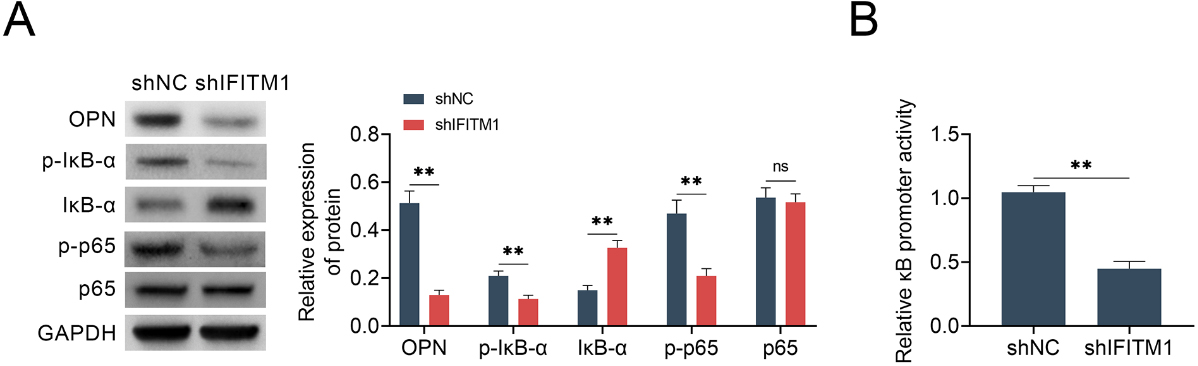
Exogenous OPN rescue cell migration and invasion of LSCC cells blocked by ablation of IFITM1. A. Representive photographs showed the results of colony formation assays of AMC-HN-8 cells transfected with the indicated plasmids. The relative proliferation levels were quantified. B. Wound healing assays were also performed using AMC-HN-8 cells transfected with the indicated plasmids, and the relative wound width was detected. C. Transwell assays using AMC-HN-8 cells transfected with the indicated plasmids were performed and the extent of relative transwell migration was quantified. The experiments were repeated for at least 3 times. Results are presented as mean 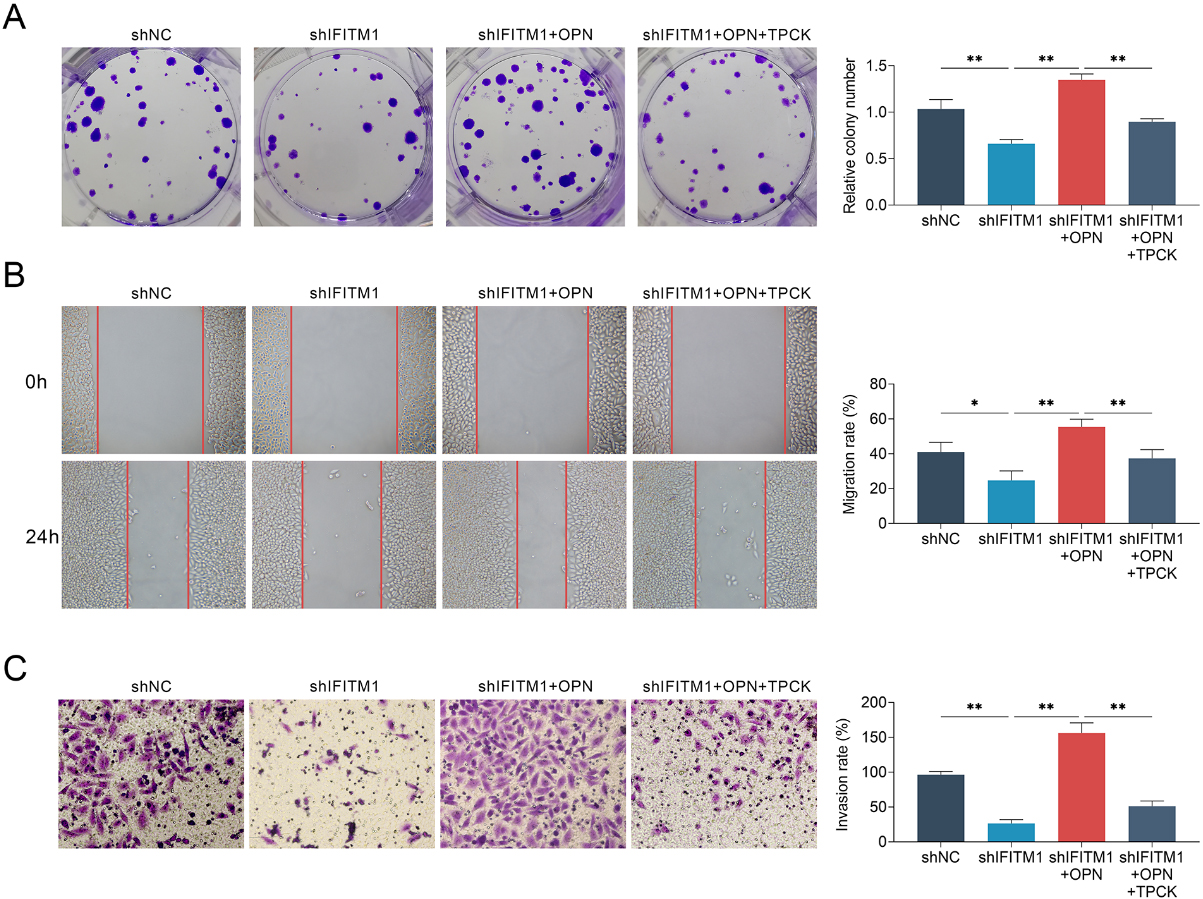
To verify this hypothesis, we evaluated the expression level of I
We next investigated whether adding exogenous OPN or TPCK, a protein inhibitor which inhibiting I
Discussion
Laryngeal squamous cell carcinoma (LSCC) is a common head and neck malignancy with a poor overall 5-year survival rate [22]. Debulking surgery, radiation and chemotherapy have limited efficiency on LSCC treatment due to the high invasiveness [23]. Targeted therapy is a promising treatment for LSCC, however, novel and effective therapeutic targets still need to be developed [9, 24, 25]. Through quantitative PCR and IHC assays, we found the obvious high expression of IFITM1 in human LSCC tissues at mRNA and protein levels, compared to the normal tissues. Similarly, our data also confirmed IFITM1 was highly expressed in LSCC cell lines, including AMC-HN-8, TU177, and TU886 cells. We therefore proposed the possible involvement of IFITM1 in the progression of LSCC. The abnormally high expression of IFITM1 was found in multiple types of tumors, including non-small cell lung cancer, breast cancer, and esophageal adenocarcinoma, and correlated with the prognosis of patients. These findings, together with our data in this study, suggested that IFITM1 has the potential of serving as an independent prognostic factor and therapeutic target for LSCC.
We further evaluated the effects of IFITM1 on LSCC progression in vitro. By performing MTT and colony formation assays, we noticed that downregulation of IFITM1 by siRNA transfection significantly inhibited the LSCC cell proliferation. Our data further confirmed that the expression of cell cycle markers, including cyclin A, cyclin D1, and CDK2, was dramatically decreased after IFITM1 silence. Meanwhile, through wound healing and transwell assays, we further demonstrated that IFITM1 affected the migration and invasion of LSCC cells. Similarly in lung cancer cells, IFITM1 siRNA transfection also resulted in the inhibition of cell proliferation, migration, and invasion [19]. In oral cancer, IFITM1 could affect cell proliferation, DNA damage and cell apoptosis [20]. Additionally, IFITM1 silence had growth inhibitory effects in cervical squamous cell carcinoma [21]. These studies proved that IFITM1 has multiple roles in cancer, and the precise mechanisms of its action still needs to be further studied.
As a cell surface protein, IFITM1 is involved in a variety of physiological and pathological processes through different mechanisms. For example, the depletion of IFITM1 suppressed proliferation and invasion of aromatase inhibitor-resistant breast cancer cells through JAK/STAT signaling pathway [17, 26]. Overexpression of IFITM1 promoted the aggressive phenotype of breast cancer cells via activation of signal transducer and activator of transcription 2 (STAT2)-dependent manner [27]. IFITM1 also contributes to the metastasis of colorectal cancer via the regulation of CAV-1 expression [28]. Here, we showed that IFITM1 regulated the proliferation, migration, and invasion of LSCC cells via the phosphorylation of p65 and I
Footnotes
Acknowledgments
This work was supported by the Project of Nantong Health and Sanitation Council (Grant No. WQ2016070) and Nantong Science and Technology Project (Grant No. MS12017012-1).
Conflict of interest
The authors state that there are no conflicts of interest to disclose.
