Abstract
Objective:
Emerging studies have identified that long non-coding RNAs (lncRNAs) play critical roles in cancer development. This study aims to explore the mechanism of NF-KappaB (NF-κB) interacting lncRNA (NKILA) in the pathological process of oral squamous cell carcinoma (OSCC).
Methods:
NKILA expression in OSCC tissues, paracancerous tissues, and normal human oral keratinocytes and OSCC cell lines was detected using RT-qPCR. KB cells were selected for the follow-up experiments. The role of NKILA in cell proliferation, migration, invasion, and NF-κB signaling pathway was identified using the gain- and loss-of function of NKILA in OSCC cells. Additionally, the role of NKILA
Results:
NKILA was poorly expressed in OSCC tissues and cells. Cell proliferation, invasion and migration, tumor volume and weight were significantly suppressed in cells with overexpressed NKILA, while silencing NKILA led to opposite trends. Moreover, the protein levels of p-IκBα and nuclear-p65 were markedly decreased, while the levels of IκBα and cytoplasm-p65 were enhanced in cells with overexpressed NKILA.
Conclusion:
This study provided evidence that NKILA could reduce proliferation, invasion and migration of OSCC cells through inhibiting the NF-κB signaling pathway. The findings may offer new insights for OSCC prevention and treatment.
Keywords
Introduction
Oral squamous cell carcinoma (OSCC) is a widely known malignancy which accounts for over 90% of all oral cancer cases. 1 OSCC could result from multiple factors such as tobacco, betel, alcohol, viruses, diet habits, oral health, and host defences. 2 The incidence of OSCC is quite high in Asia, which is owing to the specific lifestyle factors such as chewing of betel quid, drinking, smoking, human papillomavirus infection and poor oral hygiene. 3 Early detection is crucial for survival; however, most of the OSCC in early stage are difficult to be observed since they hardly cause visible changes. 4 Despite advances in current therapeutic strategies for OSCC including surgical incision, radiotherapy and chemotherapy, the survival rate of OSCC patients has not notably increased yet in recent years, with the overall 5-year survival rate only improved from 63% to 65% during the past 8 years. 5,6 This unfavorable prognosis is caused by frequent recurrence resulted from aggressive local cell invasion and metastasis, the inadequate early detection, and a lack of advanced lesions in response to chemotherapy. 7 Therefore, a more intensive understanding of the underlying biological mechanisms of OSCC progression is promising to offer more useful diagnostic markers and targets for OSCC treatment.
Long non-coding RNAs (lncRNAs) are defined as the molecules of transcribed RNAs with no less than 200 nucleotides, which are an important category of genes participating in diverse biological functions. 8 LncRNAs are deregulated in multiple cancers, showing as both cancer contributor and tumor suppressor in different cancers, thus their abnormal expression might be an important promoter in cancer progression. 9 Recently, a NF-KappaB (NF-κB) interacting lncRNA (NKILA), which is demonstrated to suppress the metastasis of breast cancer via the blockage of phosphorylation site of IκB and the inactivation of NF-κB pathway, has aroused wide concerns. 10 Currently, it has been suggested that NF-κB family of transcription factors have a key role in inflammation and are increasingly recognized as crucial factors in the initiation and progression of cancers. 11 NKILA has recently been revealed to inhibit the malignant progression and serves as a key mediator of epithelial mesenchymal transition in several diseases. 12 -14 However, the biological mechanisms of NKILA in OSCC are not entirely clear. Taken together, this study was carried out to investigate the mechanism of NKILA in OSCC progression through the NF-κB signaling pathway.
Materials and Methods
Sample Collection
Thirty-five pairs of surgically resected OSCC tissues and paracancerous tissues (no less than 2 cm away from tissues in surgical sites of the same patient) of patients were collected. The clinicopathological features of 35 OSCC patients are shown in supplementary Table 1. Patients were included if: (1) they accorded with the diagnostic criteria of OSCC and were identified with pathological examination; (2) they were aged over 40 years; (3) they and their family members were informed; (4) they didn’t have other diseases including congenital heart disease, endocrine diseases; coagulopathy or other potential interferences. The tissue samples were preserved at −80°C.
Cell Culture
Normal human oral keratinocytes (NHOK) and OSCC cell lines SCC-4, Cal-27, TSCCA, KB (purchased from Cell Bank of Experimental Medical Center of Beijing Union Medical College, Beijing, China) were incubated with Dulbecco’s modified Eagle’s medium (DMEM) containing 10% fetal bovine serum (FBS, Sigma-Aldrich Chemical Company, St Louis MO, USA), 100 U/mL penicillin (Invitrogen Inc., Carlsbad, CA, USA) and 50 μg/mL streptomycin in a 37°C incubator with 5% CO2. When the cell confluence reached 80%-90%, the cells were detached with trypsin and passaged, and then the cells in the logarithmic growth phase were collected for the following experiments.
Reverse Transcription Quantitative Polymerase Chain Reaction (RT-qPCR)
Total RNA of cells and OSCC tissues was extracted using TRIzol (Invitrogen, Carlsbad, CA, USA) and the RNA quality was measured via formaldehyde denaturing gel electrophoresis. The cDNA was produced by RNA reverse transcriptase M-MLV. SYBR Green method was applied to conduct qPCR, and the PCR primers were designed and synthesized by Shanghai Sangon Biotech Co., Ltd (Shanghai, China) (Table 1), with β-actin set as an internal reference. The PCR system was 20 μL, and the PCR condition was as follows: pre-denaturation at 95°C for 5 min, followed by 32 cycles of denaturation at 94°C for 40 s, annealing at 55°C for 40 s and extension at 70°C for 1 min, and a final extension at 72°C for 20 min. The production was identified using agarose gel electrophoresis. Data was analyzed using 2−ΔΔCt method in which −ΔΔCt refers to the ratio of the target gene expression of experimental group to control group. The formula was as follow: ΔΔCt = [Ct (target gene) − Ct (control gene)] experimental group − [Ct (target gene) − Ct (control gene)] control group.
Primer Sequences of qRT-PCR.
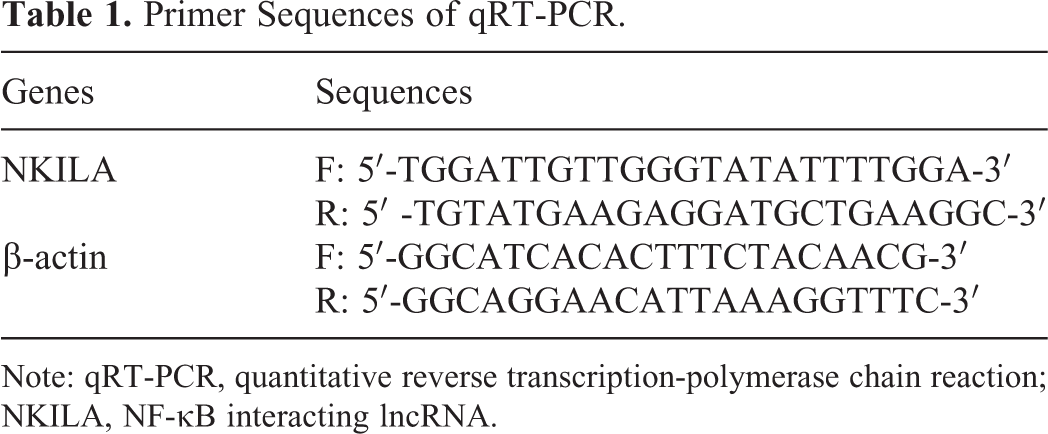
Note: qRT-PCR, quantitative reverse transcription-polymerase chain reaction; NKILA, NF-κB interacting lncRNA.
Cell Transfection and Grouping
KB and Cal-27 cell lines were selected for the following experiments. KB cells were assigned into negative control group (NC group, cells were transfected with 10 µg empty vector), NKILA group (cells were transfected with 10 µg pLVX-NKILA-IRES-ZsGreen vector) and NKILA + PMA group (cells were cultured with 50 ng/mL PMA (NF-κB activator, Sigma) for 24 h and then transfected with 10 µg pLVX-NKILA-IRES-ZsGreen vector).
Meanwhile, the Cal-27 cells were allocated into 3 groups: Sh-NC group (cells were transfected with 10 µg empty vector); ShNKILA-1 group (cells were transfected with 10 µg pLVX–ShNKILA-1-IRES-ZsGreen1 vector) and ShNKILA-2 group (cells were transfected with 10 µg pLVX–ShNKILA-2-IRES-ZsGreen1 vector). All transfections were performed in strict accordance with the instructions of the LipofectamineTM 3000 (Invitrogen Inc., Carlsbad, CA, USA). Cells were used for following experiments 48 h after transfection.
3-(4, 5-dimethylthiazol-2-yl)-2, 5-Diphenyltetrazolium Bromide (MTT) Assay
Cells in each group were detached with trypsin and then dispersed into 1 × 104 cells/mL single cell suspension with Roswell Park Memorial Institute (RPMI) 1640 medium containing 10% FBS. Then the suspension was added to 96-well plates with 200 µL per well and incubated at 37°C with 5% CO2. To each well, 20 µL of 5 mg/mL MTT solution was added at 0 h, 24 h, 48 h and 72h, after which the cells were further cultured for 4 h and then the supernatant of each well was discarded. Next, each well was added with 200 µL dimethyl sulfoxide and vibrated for 10 min to fully dissolve the crystal. The optical density (OD) at 490 nm of each well was detected.
Colony Formation Assay
Well-grown cells of passage 3 in each group were collected, detached with trypsin, and seeded into 6-well plates at the density of 1000 cells/well. After culturing in a 37°C incubator with 5% CO2 for 2 weeks, the cells were treated with 75% methanol for 30 min and stained by crystal violet (Beijing Solarbio Science & Technology Co., Ltd., Beijing, China) for 15 min. Then the number of cell clones was observed and calculated under an optical microscope (IX51, Olympus Optical Co., Ltd, Tokyo, Japan). Each experiment was repeated 3 times.
Transwell Assay
Cells starved for 24 hours after serum removal. The cells were then detached, washed twice with serum-free medium, and then resuspended with serum-free medium. The bottom membrane surface of each chamber was coated with 40 µL Matrigel (biosciences, Le Pont de Claix, France) diluted by serum-free medium (the cell migration assay didn’t apply this procedure). After that, the apical chamber (8 μmin diameter) was added with 100 µL cell suspension and the basolateral chamber was added with 600 µL DMEM containing 10% FBS, followed by 48 h of incubation at 37°C with 5% CO2. Then the remaining liquid in the apical chamber was discarded and the cells didn’t pass through the membrane were wiped out with a wet cotton swab, and the remaining cells were stained with 0.1% crystal violet for 20 min. The cells passing through the membrane were calculated based on 5 randomly selected views under a microscope. The experiment was repeated 3 times.
Scratch Test
Cells were seeded into 6-well plates at 5 × 105 cells per well and incubated at 37°C with 5% CO2 for 24 h. Subsequently, a 1 mL pipette tip was used to produce a scratch on single-layer cells, and then the cells were washed 3 times with PBS and added with serum-free medium. The scratch width was photographed and recorded under a microscope at 0 h, 24 h and 48 h, with 5 randomly selected fields observed in each group. The outcome of scratch width at 0 h subtracted that at 24 h or 48 h was regarded as the migration distance. Each experiment was repeated 3 times.
Western Blot Analysis
Total proteins of cells were extracted, and the protein concentration was detected in accordance with the instructions of the bicinchoninic acid (BCA) kit (Qiagen, Wiessberg, Deutch). Then the extracted proteins were run on sodium dodecyl sulfate polyacrylamide gel electrophoresis and transferred onto polyvinylidene fluoride membranes. Next, the membranes were blocked with 5% bovine serum albumin (BSA) at room temperature for 1 h, and then incubated with following primary antibodies at 4°C overnight: IκBα (1:1000, ab32518,), p-IκBα (1:10000, ab133462), p65(1:1000, ab32536), p-P65 (1:5000, ab86299) (all purchased from Abcam, Cambridge, MA, USA). Next, the membranes were incubated with secondary antibody goat anti-rabbit immunoglobulin G (IgG, 1:2000, ab6721, Abcam) at room temperature for 1 h. Glyceraldehyde-3-phosphate dehydrogenase (1:10000, ab181602) was set as an internal reference. The protein bands were developed using chemiluminescence reagents. The gray value analysis for target bands was conducted by the Image J software (National Institutes of Health. Bethesda, Maryland, USA).
Xenograft Tumors in Nude Mice
BALB/C nude mice (male, 16-18 g and 1-6 weeks old) purchased from Shanghai Cancer Institute (Shanghai, China) were raised in a specific-pathogen-free grade cell center (License: SYXK (Shanghai) 2017-0011). Animal grouping was the same as the cell grouping, with the well-transfected cells from each group injected into the nude mice. The tumor volume (V) was recorded every 5 days according to the length (L) and width (W), the tumor volume (mm3) was calculated as follows: V = L × W2 × 0.5, and a growth curve was drawn with 30-day recorded data. The nude mice were killed on ice via cervical dislocation at day 30, and the tumors were extracted and weighted. Meanwhile, the lung tissues in xenograft tumors were collected for the following experiments.
Hematoxylin and Eosin (HE) Staining
The collected lung tissues from nude mice were fixed in 4% paraformaldehyde, embedded with paraffin and sectioned. The sections in each group were dewaxed, dehydrated, and stained with hematoxylin at room temperature for 10 min, and further stained with 0.5% eosin (Boster Biological Technology Co., Ltd, Wuhan, Hubei, China) at room temperature for 1 h. Next, the sections were dehydrated with increasing concentrations of ethanol and xylene, sealed with neutral balsam, and observed under an optical microscope (Olympus Optical Co., Ltd, Tokyo, Japan) for pathological change analysis.
Statistical Analysis
The SPSS 21.0 (IBM Corp. Armonk, NY, USA) was applied for data analysis. Measurement data were in normal distribution according to Kolmogorov-Smirnov test and expressed as mean ± standard deviation. Differences between 2 groups were evaluated using the
Results
NKILA Is Poorly Expressed in OSCC Tissues and Cells
NKILA expression in human OSCC tissues and paracancerous tissues was detected using RT-qPCR, and the results showed that NKILA expression was lower in OSCC tissues than that in paracancerous tissues (

NKILA is poorly expressed in OSCC tissues and cells. (A) NKILA expression in OSCC tissues and normal tissues measured using RT-qPCR (the paracancerous normal tissue did not include epithelial dysplasia), n = 35; (B) NKILA expression in NHOK and OSCC cell lines (SCC-4, Cal-27, TSCCA, and KB) measured using RT-qPCR, Repetition = 3;*,
RT-qPCR results for NKILA expression in NHOK and OSCC cell lines (SCC-4, Cal-27, TSCCA and KB) suggested that NKILA was poorly expressed in OSCC cell lines compared to NHOK cells, and among all OSCC cell lines, NKILA expression was significantly low in KB cells, while compared to the other OSCC cell lines, Cal-27 cells showed relative high expression (
Over-Expressed NKILA Suppresses KB Cell Proliferation
To further investigate the underlying mechanism of NKILA in OSCC cells, we transfected NKILA overexpression vector into KB cells, and transfected ShNKILA-1 and ShNKILA-2 vectors into Cal-27 cells. RT-qPCR results demonstrated that the transfections were successfully performed (all
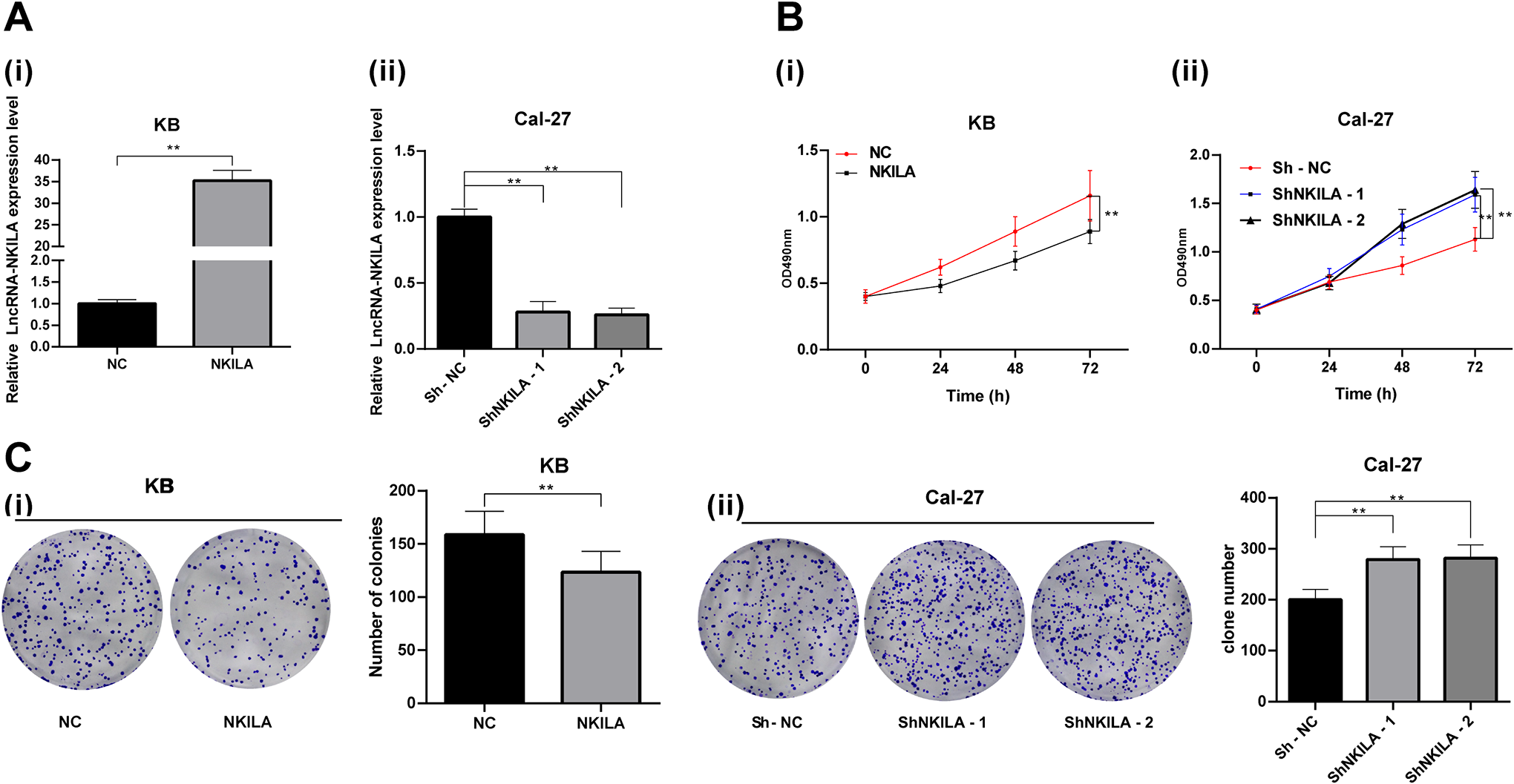
Overexpressed NKILA inhibits OSCC cell proliferation. (A) NKILA expression in KB and Cal-27 cells after different transfections measured using RT-qPCR; (B) Cell proliferation at 0 h, 24 h, 48 h, and 72 h detected using MTT assay; (C) number of cell clones of each group measured using colony formation assay; Repetition = 3, in panels A (i) and C (i), data were analyzed using the
MTT and colony formation assays suggested that overexpressed NKILA reduced KB cell proliferation, while Cal-27 cells transfected with ShNKILA-1 and ShNKILA-2 showed elevated proliferation ability (all
Overexpressed NKILA Inhibits the Invasion and Migration of OSCC Cells
Transwell assay and scratch test was applied to investigate the role NKILA in invasion and migration of OSCC cells. The results demonstrated that overexpression of NKILA significantly reduced invasion and migration of KB cells, while downregulation of NKILA promoted cell invasion and migration in Cal-27 cells (all

Overexpressed NKILA inhibits OSCC cell invasion and migration. (A) Invasion and migration of OSCC cells in each group detected with Transwell assay; (B) migration of OSCC cells in each group detected using scratch test, with the migration distance at 0 h, 24 h, and 48 h detected; Repetition = 3; in panel A (i)/(ii), data were analyzed using the
Overexpressed NKILA Inactivated the NF-κB Signaling Pathway
The protein levels of p-IκBα, IκBα, p-p65 and p65 in each group were measured using Western blot analysis. The result showed that the KB cells with overexpression of NKILA presented significantly reduced protein levels of p-IκBα/IκBα and p-p65/p65, but opposite trends were shown in Cal-27 cells which transfected with ShNKILA-1 and ShNKILA-2 vectors (all
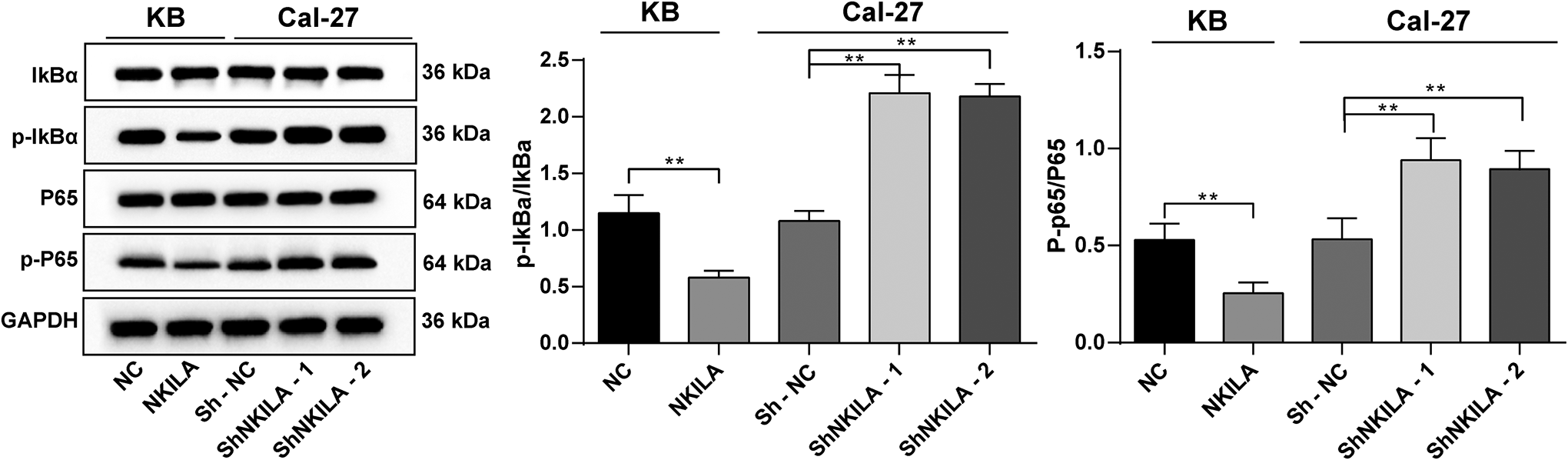
Overexpressed NKILA inactivates the NF-κB signaling pathway. Relative levels of NF-κB signaling pathway-related proteins (p-IκBα, IκBα, p-p65 and p65) in cells measured using Western blot analysis; Repetition = 3; data were analyzed using one-way ANOVA, and Tukey’s multiple comparisons test was used for the pairwise comparison after ANOVA analysis; **,
NF-κB Activation Reverses the Inhibition of NKILA on KB Cell Proliferation and Invasion
To further validate the correlation between NKILA and the NF-κB signaling pathway, another group of KB cells were pre-treated with PMA for 24 h and then transfected with NKILA vector. When PMA was added, NF-κB signaling pathway was activated. Compared with the overexpression of NKILA alone, the proliferation, invasion and migration of KB cells were significantly enhanced by the combination of overexpression of NKILA and PMA, the activator of NF-κB signaling pathway (all

NF-κB activation reverses the inhibition of NKILA on KB cell proliferation and invasion. A, Western blot was used to detect the expression of NF-κB signaling pathway related proteins (p-IKBα, IKBα, p-p65 and p65) in OSCC cells of each group; B, KB cell proliferation at 0 h, 24 h, 48 h and 72 h detected using MTT assay; C, number of cell clones in each group measured using colony formation assay; D, number of invaded and migrated KB cells measured using Transwell assay; E, migration of KB cells detected using scratch test, with the migration distance at 0 h, 24 h and 48 h detected; Repetition = 3; in panels A and D, data were analyzed using 2-way ANOVA, and Tukey’s multiple comparisons test was used for the pairwise comparison after ANOVA analysis; in panels B and C, data were analyzed using the
NKILA Inhibited OSCC Tumor Formation In Vivo
Well-transfected KB cells and Cal-27 cells were subcutaneously injected into nude mice. Then the tumor volume was observed and measured. The result showed that the tumor volume was obviously decreased in the mice in the NKILA group while increased in the ShNKILA-1- and ShNKILA-2 groups (all

NKILA suppresses tumor formation
Mice were sacrificed 30 d after xenograft tumor transplantation. The tumors were weighed and it was shown that the tumor weight was notably decreased in the mice in the NKILA group while elevated in the ShNKILA-1 and ShNKILA-2 groups (all
Discussion
OSCC is a common cancer type all over the world and also the most frequent malignant tumor in human oral and maxillofacial areas. 15 Despite an abundance of basic and clinical studies and reports, the overall 5-year survival rate for patients with OSCC is still unfavorable. 16 It has been revealed that lncRNAs play crucial roles in many cancer processes including cell apoptosis and proliferation. 17 In the current study, we explored the biological mechanism of NKILA in OSCC, and it was suggested that NKILA could suppress the malignant behaviors of OSCC cells via suppression of the NF-κB signaling pathway.
Initially, our study found that NKILA was poorly expressed both in OSCC tissues and cells. It has been suggested that NKILA expression was notably higher in the paracancerous tissues and normal tongue squamous epithelial cells than in the tongue squamous cell carcinoma (TSCC) tissues and cells. 18 Significantly lower NKILA expression has been documented in several types of cancer tissues and cells than that in the paired normal tissues and cells, including in rectal cancer, 19 non-small cell lung cancer (NSCLC), 14 esophageal squamous cell carcinoma, 20 laryngeal cancer, 21 malignant melanoma 12 and retinoblastoma. 22 In this study, NKILA expression was lowest in KB cells and relative highest in Cal-27 cells than other OSCC cells, thus KB and Cal-27 cells were selected for the following studies. KB cells were transfected with NKILA vector, while Cal-27 cells were transfected with ShNKILA-1 or ShNKILA-2 vectors. Then, our study found that KB cells with overexpressed NKILA showed suppressed cell proliferation, invasion and migration abilities, while Cal-27 cells transfected with ShNKILA-1 and ShNKILA-2 vectors presented opposite trends. Increased expression of NKILA has been proved to inhibit cell invasion, migration and proliferation, and silenced NKILA could reverse the changes and led to enhanced cell viability in NSCLC. 14 NKILA has also been revealed to suppress cell proliferation and invasion of TSCC cells, 18 ESCC cells, 20 laryngeal cancer 21 and retinoblastoma cells. 22
Moreover, in our study, overexpression of NKILA decreased the tumor volume and weight in nude mice. Increased expression of NKILA has been suggested to inhibit cell malignant behaviors and suppress the tumor growth
To sum up, the current study identified that NKILA was poorly expressed in OSCC cells. Upregulation of NKILA could inhibit OSCC cell proliferation, invasion and migration, as well as tumor growth. Most of all, increased expression of NKILA could reduce the malignant development of OSCC through the inactivation of NF-κB signaling pathway. Hopefully, these findings could provide a novel theoretical target for OSCC control and treatment, and hope more studies in this field would be conduct to validate our findings and develop more ways to overcome OSCC and other severe diseases.
Supplemental Material
Supplemental_Table_1 - Long Non-Coding RNA NKILA Reduces Oral Squamous Cell Carcinoma Development Through the NF-KappaB Signaling Pathway
Supplemental_Table_1 for Long Non-Coding RNA NKILA Reduces Oral Squamous Cell Carcinoma Development Through the NF-KappaB Signaling Pathway by Daoyong Hu, Tian Zhong and Qun Dai in Technology in Cancer Research & Treatment
Supplemental Material
Supplementary_material - Long Non-Coding RNA NKILA Reduces Oral Squamous Cell Carcinoma Development Through the NF-KappaB Signaling Pathway
Supplementary_material for Long Non-Coding RNA NKILA Reduces Oral Squamous Cell Carcinoma Development Through the NF-KappaB Signaling Pathway by Daoyong Hu, Tian Zhong and Qun Dai in Technology in Cancer Research & Treatment
Footnotes
Authors’ Note
The data that support the findings of this study are available from the corresponding author upon reasonable request. The present study was approved by the Committee of The Affiliated Stomatological Hospital of Nanchang University (SYXK 2015-0003). This study has gained the approval of the Ethics Committee of the Affiliated Stomatological Hospital of Nanchang University. All individuals signed informed written consent documents. All the procedures conducted in animals in this experiment were approved by the Animal Ethics Committee.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
