Abstract
BACKGROUND:
Inflammation-based prognostic scores have been increasingly used for prognosis prediction in malignant tumors. However, no existing study has comprehensively evaluated these scores in combined hepatocellular-cholangiocarcinoma (cHCC-CCA).
OBJECTIVE:
This study aimed to identify a robust inflammation-based prognostic predictor for cHCC-CCA.
METHODS:
We retrospectively analyzed 220 patients pathologically confirmed as Allen type C cHCC-CCA. The univariate and multivariate analyses were used to explore the associations between clinical variables and prognosis of cHCC-CCA. The propensity score-matching (PSM) was performed to reduce the effects of potential cofounders and selection bias. Finally, the predictive values of different inflammation-based indices were compared by using time-dependent receiver operating characteristic (ROC) curves.
RESULTS:
The systemic immune-inflammation index (SII) and aspartate aminotransferase to platelet ratio index (APRI) were identified as independent prognostic predictors in multivariate analysis. After PSM, the survival differences were still significant between SII-high group and SII-low group (
CONCLUSIONS:
The SII may serve as a preferable predictor of both recurrence-free survival (RFS) and overall survival (OS) in patients with cHCC-CCA.
Keywords
Introduction
Combined hepatocellular-cholangiocarcinoma (cHCC-CCA) is a distinct subtype of primary liver cancer (PLC) defined by both hepatocellular and biliary differentiations within the same tumor nodule [1]. cHCC-CC is a rare liver malignancy, only accounting for about 2%–5% of PLCs [2]. A host of evidences have suggested that the demographic and clinical features of cHCC-CCA are intermediate between hepatocellular carcinoma (HCC) and intrahepatic cholangiocarcinoma (iCCA) [3]. Hepatic resection (HR) is the mainstay curative treatment for cHCC-CCA patients [4]. Unfortunately, recent researches showed that cHCC-CCA exhibited significantly higher early recurrence potential and worse prognosis than other primary liver cancers, even after curative resection [5, 6]. Nevertheless, due to the rarity of cHCC-CCA, the prognostic characteristics of this malignancy remain poorly understood. To date, no effective prognostic predictor for cHCC-CCA has been identified.
Inflammation is a hallmark of cancer, which plays a crucial role in tumorigenesis, tumor progression and metastasis [7]. The severity of underlying tumor-associated inflammation can be measured by a number of clinical markers, including peripheral blood counts (neutrophil, lymphocyte and platelet counts) and acute-phase protein (albumin (ALB) and C-reactive protein) [8]. Recently, several inflammation-based prognostic scores have been constructed for prognosis prediction in cancer patients, such as the neutrophil to lymphocyte ratio (NLR) [9], platelet to lymphocyte ratio (PLR) [10], lymphocyte to monocyte ratio (LMR) [11], prognostic nutritional index (PNI) [12], systemic immune-inflammation index (SII) [13] and aspartate aminotransferase to platelet ratio index (APRI) [14]. These scores have showed significant prognostic values in various malignancies including HCC and iCCA [15, 16, 17, 18]. However, no existing study has comprehensively evaluated the prognostic values of inflammation-based indices in cHCC-CCA patients.
In this study, we aimed to evaluate and compare the prognostic values of inflammation-based prognostic scores in a cohort of 220 patients with Allen type C cHCC-CCA following curative resection.
Materials and methods
Patients selection
A total of 307 patients with cHCC-CCA who underwent hepatic resection at liver cancer institute, Zhongshan Hospital between 1993 and 2015 were retrospectively screened. Given that the 2019 World Health Organization (WHO) classification simplifies the subcategorization of cHCC-CCA and defines cHCC-CCA as the subtype with unequivocal components of both HCC and CC within the same mass [19], we only enrolled patients with Allen type C cHCC-CCA in this research [20]. The detailed inclusion criteria were as follows: 1) received liver resection and pathologically confirmed as Allen type C cHCC-CCA; 2) Eastern Cooperative Oncology Group (ECOG) performance-status score of 0 or 1; 3) Child-Pugh grade A or B; 4) with complete preoperative laboratory results and survival data. The exclusion criteria were as follows: 1) received previous anti-tumor therapy before surgery; 2) concomitance with other malignancies in addition to cHCC-CCA; 3) presence of serious medical comorbidities such as heart or renal dysfunction. Finally, 220 consecutive cHCC-CCA patients were enrolled for further analyses.
Data collection
Clinical variables were retrospectively retrieved from the electronic medical record, including patient characteristics (age, sex, etiology, liver cirrhosis, diabetes mellitus, ascites, Child-Pugh grade and TNM stage), hepatitis B virus (HBV) status (HBV-DNA load and HbeAg positivity), tumor-related factors (tumor size, tumor number, tumor capsule, microvascular/macrovascular invasion, hilar lymph node involvement and tumor differentiation), laboratory results (ALB (g/L), total bilirubin (TB, umol/L), alanine aminotransferase (ALT, U/L), aspartate aminotransferase (AST, U/L), alkaline phosphatase (AKP, U/L),
Inflammation-based prognostic scores
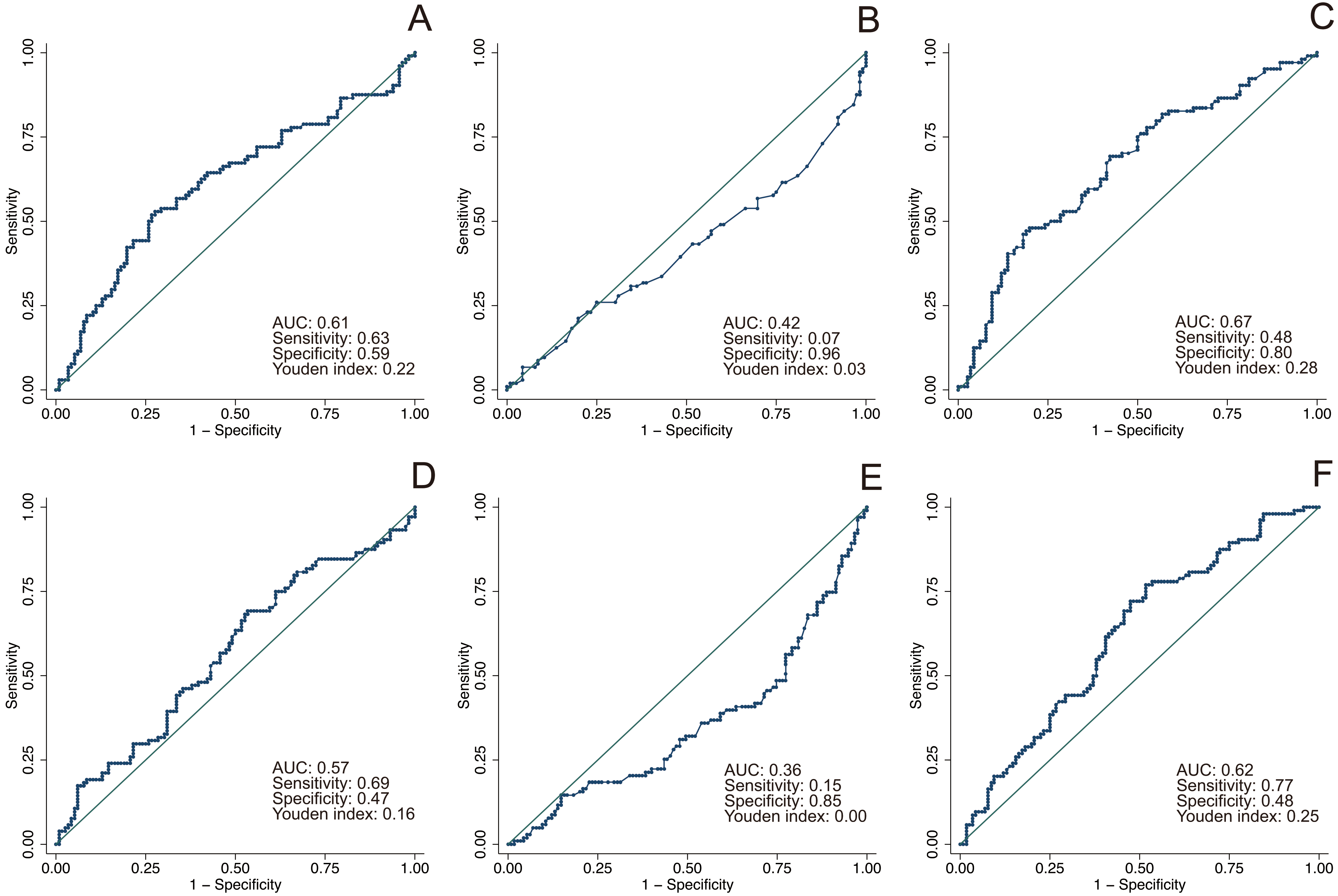
All the blood samples were acquired within 5 days before surgery for measurement of ALB, AST, neutrophil, lymphocyte, monocyte and platelet counts. The inflammation-based prognostic scores including the SII, PNI, NLR, PLR, LMR and APRI were calculated and described in Supplementary Table 1. The optimal threshold value for each score was determined by performing receiver operating characteristic (ROC) analysis regarding overall survival (OS). The value with the maximum Youden index was chosen as the optimal cut-off value(Supplementary Fig. 1).
Treatment and follow-up
The standard technique for hepatic resection has been described in previous studies [21]. The adjuvant TACE was performed under the standard protocol of our department as described by Ren and Yin et al. [22, 23]. Briefly, a 5F or 4F catheter (Cordis
Statistical analysis
Continuous variables were presented as median with interquartile range (IQR), whereas categorical variables were summarized as counts with percentages. Binary variables were compared by using either the chi-square test or Fisher’s exact test, whereas ordinal categorical variables were compared by using the Kruskal-Wallis test. Survival curves were estimated using the Kaplan-Meier method and compared through log-rank test. The univariate analysis was employed to identify significant factors associated with RFS and OS. Then, all variables that had a
All statistical analyses were performed using the Stata version 15.1 (StataCorp, College station, TX). Differences were considered statistically significant when two-tailed
Ethics approval statement
This study was approved by Medical Ethics Committee of Zhongshan Hospital affiliated to Fudan University (No: B2019-169) and complied with the standards of the Declaration of Helsinki. Informed consent was obtained from each patient for using their data for research.
Results
Patient characteristics
A total of 220 patients with Allen type C cHCC-CCA were enrolled in this study. The entire cohort consists of 162 (73.6%) males and 58 (26.4%) females, with a median age of 54 (IQR: 47, 61) years. Among these patients, 168 (76.4%) were positive for HBV surface antigen (HBsAg), 43 (19.6%) were positive for HBV e antigen (HBeAg), and 2 (0.9%) were positive for hepatitis C virus (HCV) antibody. Liver cirrhosis was observed in 126 (57.3%) patients and a number of 41 (18.6%) patients exhibited Child-Pugh grade B. With regard to tumor characteristics, the median tumor size was 4.7 (IQR: 3, 7.5) cm and 63 (28.6%) patients presented with multiple tumors. Microvascular invasion, macrovascular invasion and hilar lymph node involvement were detected in 40 (18.2%) patients, 16 (7.3%) patients and 21 (9.6%) patients, respectively. The numbers of patients with AJCC TNM-8 stage I, II, III and IV were 107 (48.6%), 44 (20.0%), 38 (17.3%) and 31 (14.1%), respectively. Other clinical characteristics including the 6 inflammation-based scores (SII, PNI, NLR, PLR, LMR and APRI) were presented in Table 1.
Clinical characteristics of enrolled patients (
220)
Clinical characteristics of enrolled patients (
Abbreviations: IQR: interquartile range; HBV: hepatitis B virus; HCV: hepatitis C virus; HBeAg: hepatitis B virus e antigen; AJCC TNM-8: the 8
Univariate and multivariate analyses of prognostic factors in patients with combined hepatocellular-cholangiocarcinoma (
Abbreviations: HR: hazard ratio; CI: confidence interval; HBV: hepatitis B virus; HCV: hepatitis C virus; HBeAg: hepatitis B virus e antigen; AJCC TNM-8: the 8
Kaplan-Meier analysis of the recurrence-free survival of patients with combined hepatocellular-cholangiocarcinoma. (A) systemic immune-inflammation index (SII); (B) prognostic nutritional index (PNI); (C) neutrophil to lymphocyte ratio (NLR); (D) platelet to lymphocyte ratio (PLR); (E) lymphocyte to monocyte ratio (LMR); (F) aspartate aminotransferase to platelet ratio index (APRI). 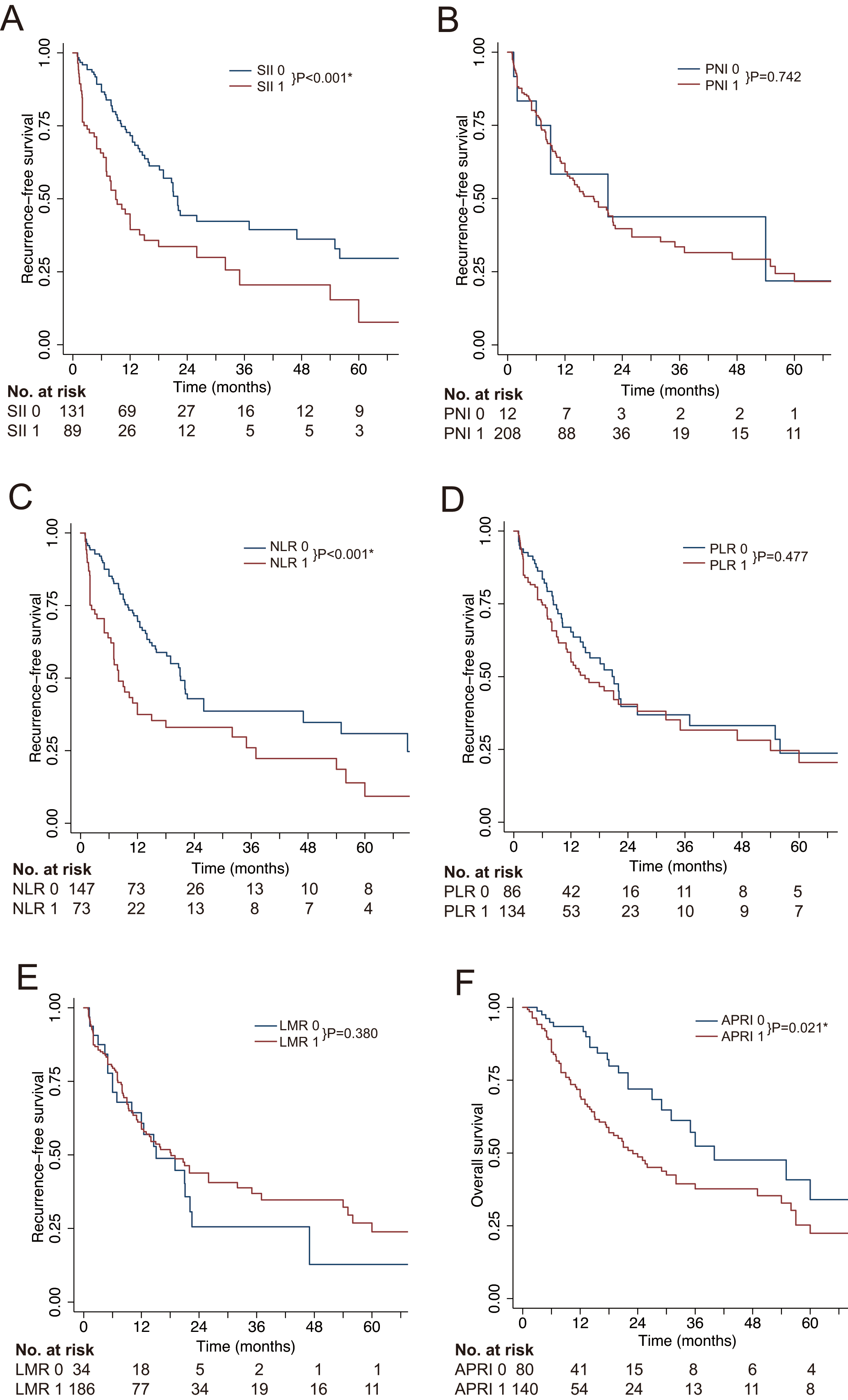
Kaplan-Meier analysis of the overall survival of patients with combined hepatocellular-cholangiocarcinoma. (A) systemic immune-inflammation index (SII); (B) prognostic nutritional index (PNI); (C) neutrophil to lymphocyte ratio (NLR); (D) platelet to lymphocyte ratio (PLR); (E) lymphocyte to monocyte ratio (LMR); (F) aspartate aminotransferase to platelet ratio index (APRI). 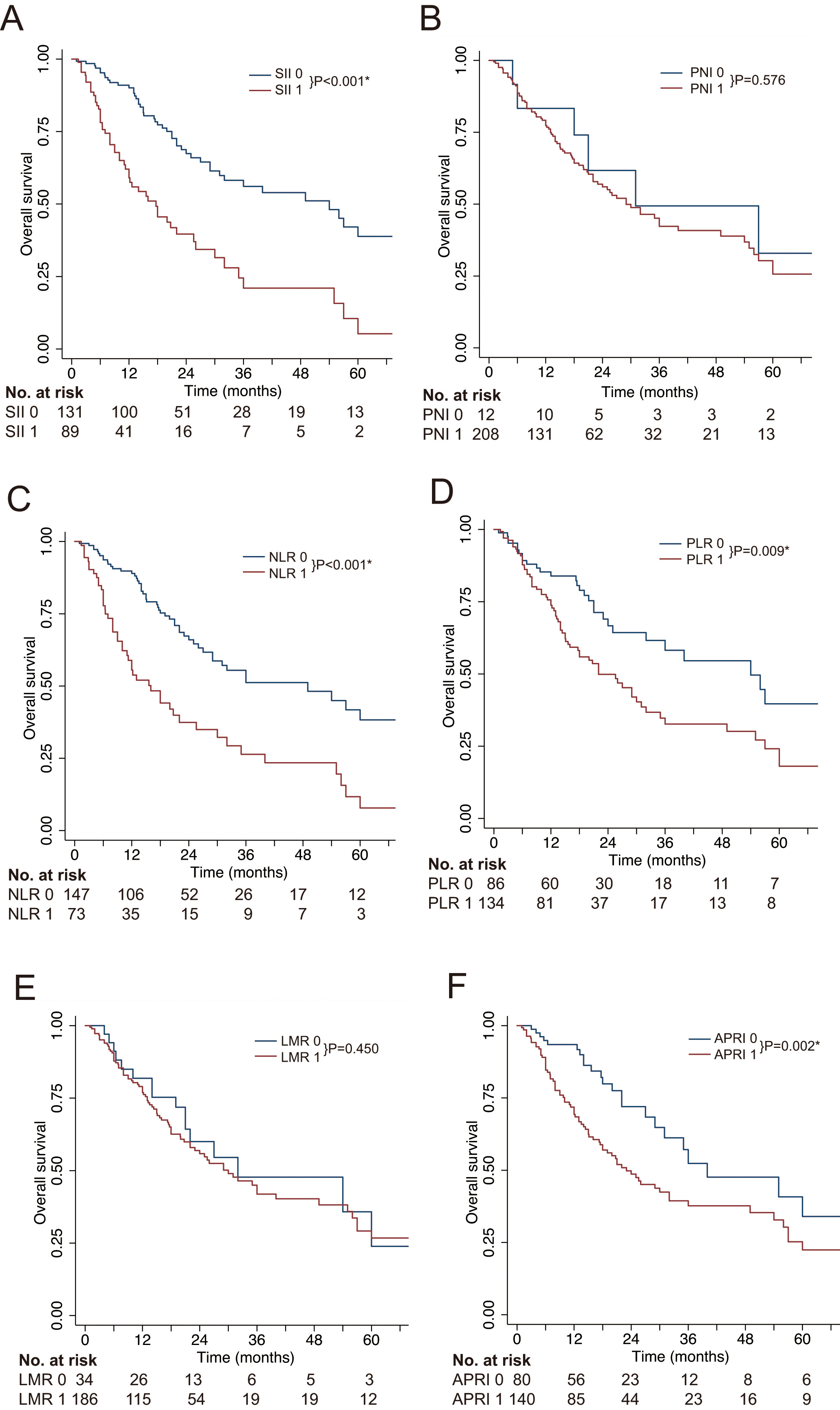
The mean duration of follow-up was 22.1 (Range: 1–118) months. A total of 104 (47.3%) patients died and 110 (50.0%) patients suffered tumor recurrence at the end of the follow-up. The median RFS was 18.2 (95% CI: 13, 22.1) months and the 1-, 3- and 5-year RFS rates were 59.8%, 34.5% and 21.8%, respectively. The median OS was 30 (95% CI: 23, 49) months and the 1-, 3- and 5-year OS rates were 77.8%, 42.8% and 26.3%, respectively.
Univariable and multivariate analyses
The associations between patient characteristics and clinical outcomes were examined by univariable and multivariate analyses (Table 2 and Figs 1 and 2). With regard to RFS, our univariable analysis indicated that sex, HBeAg positivity, liver cirrhosis, TNM stage, tumor size, tumor number, tumor capsule, macrovascular invasion, lymph node involvement, adjuvant TACE, AKP, SII, NLR and APRI were significant factors. And sex (
Propensity score-matching analysis
Clinical characteristics were compared between groups divided by the SII and APRI (Tables 3 and 4). Before PSM, clinical parameters of SII 0 group and SII 1 group statistically differed in TNM stage (
Baseline characteristics of the patients classified by the SII before and after propensity score-matching
Baseline characteristics of the patients classified by the SII before and after propensity score-matching
Abbreviations: SII: systemic immune-inflammation index; HBV: hepatitis B virus; HCV: hepatitis C virus; HBeAg: hepatitis B virus e antigen; AJCC TNM-8: the 8
Baseline characteristics of the patients classified by the APRI before and after propensity score-matching
Abbreviations: APRI: aspartate aminotransferase to platelet ratio index; HBV: hepatitis B virus; HCV: hepatitis C virus; HBeAg: hepatitis B virus e antigen; AJCC TNM-8: the 8
Kaplan-Meier analysis of patients with combined hepatocellular-cholangiocarcinoma after propensity-score matching. (A) recurrence-free survival by the SII; (B) overall survival by the SII; (C) recurrence-free survival by the APRI; (D) overall survival by the APRI. 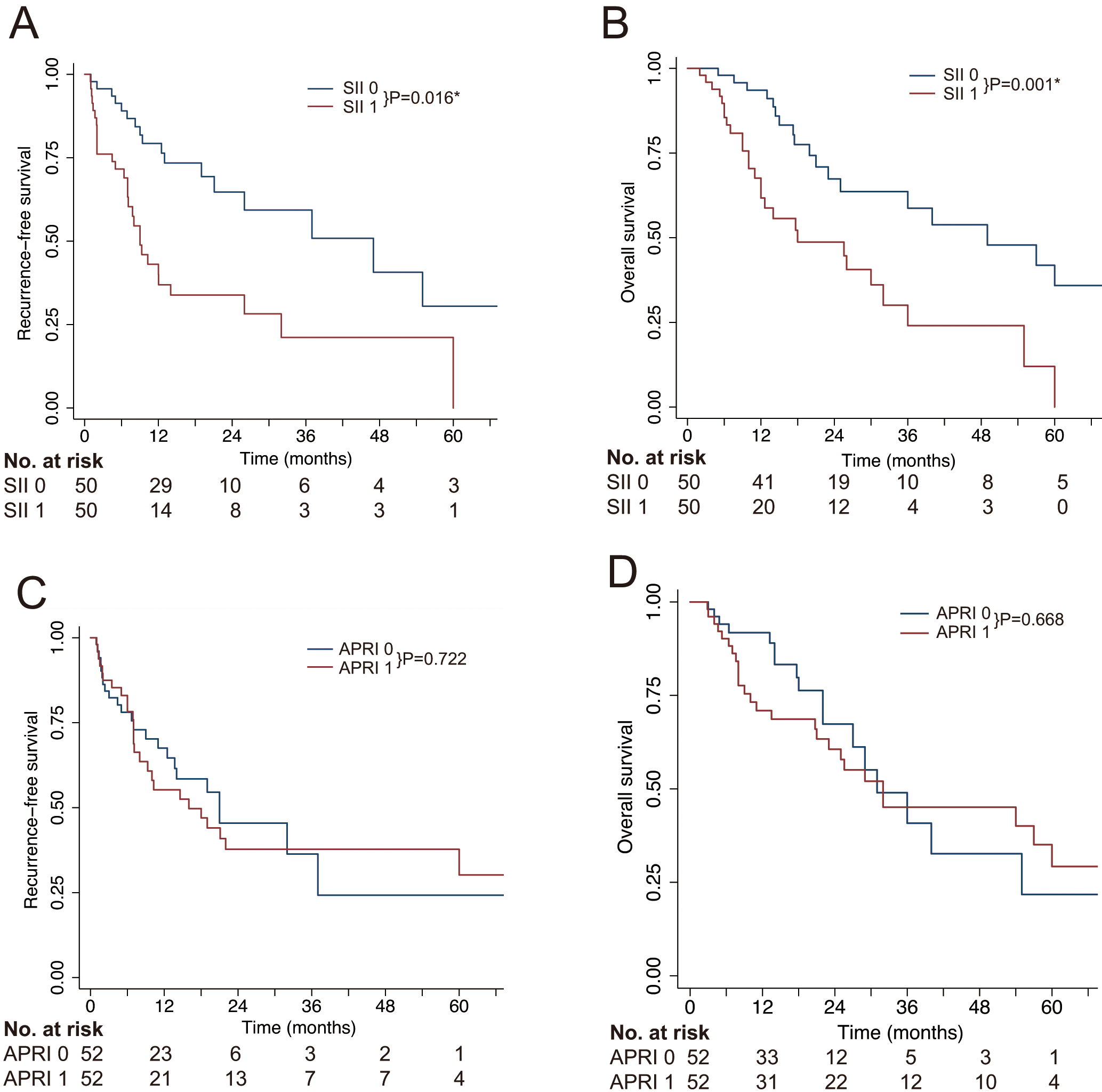
The PSM analysis generated 50 pairs of SII-matched patients and 52 pairs of APRI-matched patients, respectively. As shown in Fig. 3, after PSM, APRI 0 and APRI 1 groups showed no significantly survival differences (
The time-dependent ROC curves at 1, 3 and 5 years were employed to compare the predictive values of the 6 inflammation-based scores (Table 5 and Fig. 4). The SII had 1-, 3- and 5-year area under the curve (AUC) values of 0.66 (95% CI: 0.59–0.72), 0.63 (95% CI: 0.56–0.71) and 0.65 (95% CI: 0.55–0.74), respectively, which significantly outperformed other scores. Moreover, the positive predictive value (PPV) and negative predictive value (NPV) of various inflammation-based scores were presented in Table 6. These results supported our finding that the SII was superior to other inflammation-based scores in predicting prognosis in cHCC-CCA.
Comparison of the predictive performances between the inflammation-based scores
Comparison of the predictive performances between the inflammation-based scores
Abbreviations: AUC: area under the curve; CI: confidence interval; SII: systemic immune-inflammation index; PNI: prognostic nutritional index; NLR: neutrophil to lymphocyte ratio; PLR: platelet to lymphocyte ratio; LMR: lymphocyte to monocyte ratio; APRI: aspartate aminotransferase to platelet ratio index.
The positive predictive value and negative predictive value of various scores
Abbreviations: PPV: positive predictive value; NPV: negative predictive value; SII: systemic immune-inflammation index; PNI: prognostic nutritional index; NLR: neutrophil to lymphocyte ratio; PLR: platelet to lymphocyte ratio; LMR: lymphocyte to monocyte ratio; APRI: aspartate aminotransferase to platelet ratio index.
The time-dependent receiver operating characteristic (ROC) curves of inflammation-based prognostic scores for predicting overall survival. (A) ROC curves for predicting 1-year overall survival; (B) ROC curves for predicting 3-year overall survival; (C) ROC curves for predicting 5-year overall survival.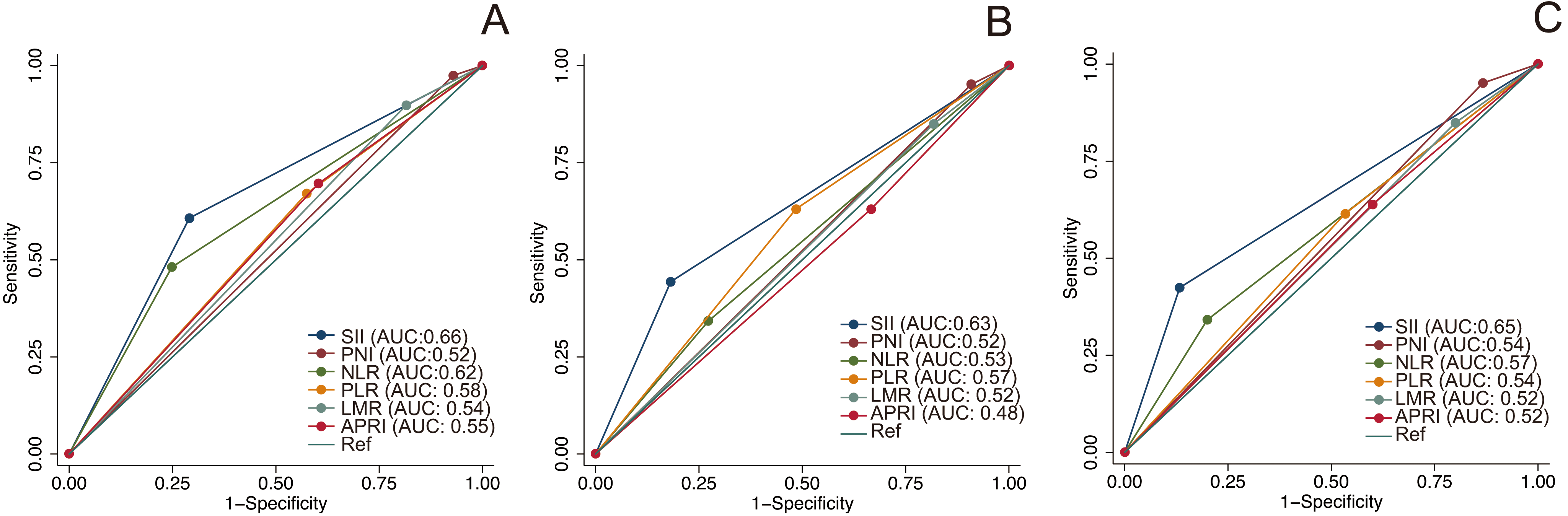
The discrimination efficacy of the SII in different subgroups based on age, sex, serum AFP level and TNM stage. (A) recurrence-free survival by the SII in subgroups based on age, sex, serum AFP level and TNM stage. (B) overall survival by the SII in subgroups based on age, sex, serum AFP level and TNM stage. 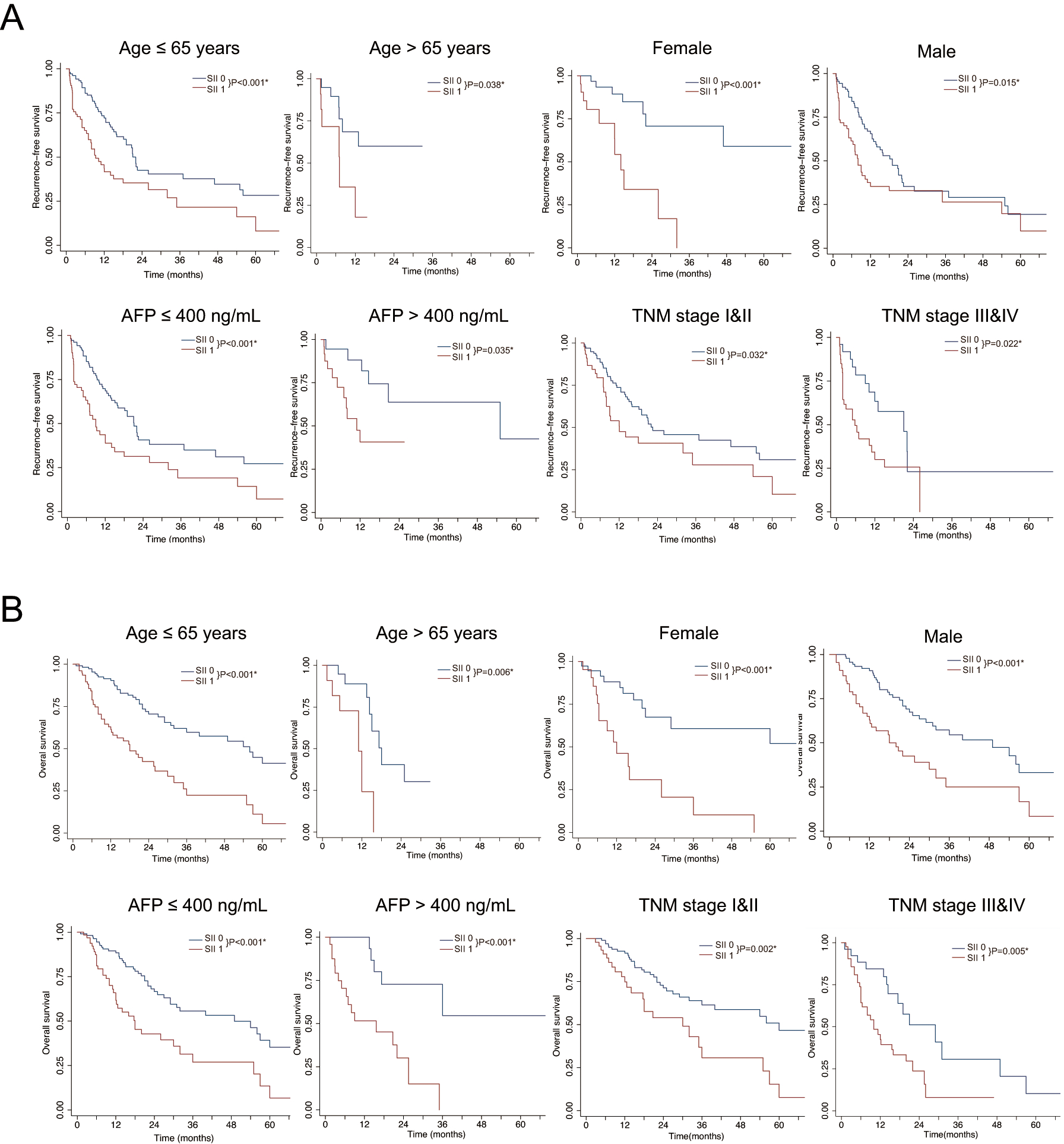
To further verify the predictive value of the SII in patients with different clinical characteristics, we divided the entire cohort into various subgroups based on age, sex, serum AFP level and TNM stage. Our results showed that the SII could significantly discriminate both RFS (Fig. 5A) and OS (Fig. 5B) in all these subgroups (
Discussion
cHCC-CCA is a rare subtype of primary liver cancer, which tends to appear more aggressive behaviors and poorer outcome than HCC [5, 24]. However, the prognostic characteristics of cHCC-CCA still remains undetermined. Due to the rarity of cHCC-CCA, most of the previous clinical researches were derived from small patient cohort, which were statistically inadequate for identifying definitive prognostic predictors for cHCC-CCA [24, 25, 26]. To bridge this gap, in present study, we for the first time compared various inflammation-based prognostic scores in a large cohort of 220 patients with Allen type C cHCC-CCA. Our results indicated that the SII could independently predict RFS and OS in cHCC-CCA, with superior predictive performance to other inflammation-based indices.
In this study, ROC analysis was applied to determine the optimal cut-offs of different inflammation-based prognostic scores (Supplementary Table 1 and Supplementary Fig. 1). The univariate and multivariate analyses demonstrated that the SII and APRI could independently predict both RFS and OS in patients with cHCC-CCA (Table 2). However, after adjusting for cofounding variables, the APRI failed to discriminate survival between low- and high APRI groups (
The SII is an integrated index based on peripheral neutrophil, platelet and lymphocyte counts, which was initially developed for prognosis prediction in HCC patients undergoing curative resection [13]. Recently, its prognostic value has been unveiled in several other cancers including iCCA [15], gastric cancer [27], renal cell cancer [28] and breast cancer [29]. The physiological roles of the three important blood cells may be correlated with tumor malignant behaviors. Experimental studies have showed that neutrophils play a significant promoting-role in tumor development, progression and metastasis [30, 31]. One important mechanism is that neutrophils can promote tumor angiogenesis through several mitogenic and pro-angiogenic molecules including elastase, prokineticin-2 (PROK2) and matrix metalloproteinase-9 (MMP9) [30]. In addition, immunosuppression is another predominant mechanism for neutrophils to facilitate cancer cells proliferation and invasion, achieved by the production of specific cytokines and amino acid depletion [32]. Likewise, activated platelets can bind tumor cells through the adhesion receptor P selectin and promote tumor progression by producing vascular endothelial growth factor, tissue factor, the fibroblast growth factor and platelet-derived growth factor [33, 34]. Moreover, platelet-derived transforming growth factor-
Surgical resection is the most widely used curative approach for cHCC-CCA [4]. However, it’s reported that the recurrence rate and long-term survival of cHCC-CCA patients after resection are poor [4, 6, 37]. Therefore, more effective therapies should be performed for those patients with high risk of recurrence and poor estimated survival. In present study, patients with high-SII levels presented significantly lower 1-, 3- and 5-year RFS rates (41.2%, 20.5% and 7.7% vs. 71.7%, 42.3% and 29.6%,
There are several limitations in this study. First, as a single-center based retrospective study, potential selection bias is inevitable. To reduce selective bias, the PSM was performed to balance baseline variables. Second, the cut-off values for inflammation-based indices in this study was determined by ROC analysis based on survival data, further validation studies determining optimal threshold values for these inflammation-based indices are needed. Third, peripheral neutrophil, platelet and lymphocyte counts may be influenced by other pathological conditions including ischemia, infection, metabolic syndrome and renal dysfunction. Hence, the clinical significance of the SII should be cautiously explained with full consideration of concurrent diseases. Fourth, it’s inadequate to use a single biomarker to guide clinical decision making, therefore, the SII should be regarded as an additional tool in clinical practice.
Conclusion
To sum up, our study identifies the SII as an important prognostic predictor in cHCC-CCA. The low cost, easy access and reproducibility make the SII a promising tool for predicting cHCC-CCA prognosis in future clinical practice. However, large-scale external validation studies are still warranted.
Footnotes
Acknowledgments
This work was supported by the National Natural Science Foundation of China [grant number: 81972889] and Excellent Backbone Foundation of Zhongshan hospital, Fudan University [grant number: 2019ZSGG20].
Conflict of interest
The authors declare that they have no competing interests.
Authors’ contributions
Conception: Feng Zhang, Ying-Hong Shi and Xin Yin.
Interpretation or analysis of data: Feng Zhang, Ke-Shu Hu, Shen-Xin Lu and Miao Li.
Preparation of the manuscript: Feng Zhang, Ke-Shu Hu, Shen-Xin Lu and Miao Li.
Revision for important intellectual content: Rong-Xin Chen, Zheng-Gang Ren and Xin Yin.
Supervision: Rong-Xin Chen, Zheng-Gang ren, Ying-Hong Shi and Xin Yin.
Supplementary data
Inflammation-based prognostic scores
Criteria
Score
Systemic immune-inflammation index (SII)
Platelet count (
0
Platelet count (
1
Prognostic nutritional index (PNI)
Albumin (g/L)
0
Albumin (g/L)
1
Neutrophil to lymphocyte ratio (NLR)
Neutrophil count (
0
Neutrophil count (
1
Platelet to lymphocyte ratio (PLR)
Platelet count (
0
Platelet count (
1
Lymphocyte to monocyte ratio (LMR)
Lymphocyte count (
0
Lymphocyte count (
1
Aspartate aminotransferase to platelet ratio index (APRI)
Aspartate aminotransferase (U/L)/platelet count (
0
Aspartate aminotransferase (U/L)/platelet count (
1
Identification of the optimal cut-off values of different inflammation-base prognostic scores by using receiver operating characteristic (ROC) analysis regarding overall survival. (A) systemic immune-inflammation index (SII); (B) prognostic nutritional index (PNI); (C) neutrophil to lymphocyte ratio (NLR); (D) platelet to lymphocyte ratio (PLR); (E) lymphocyte to monocyte ratio (LMR); (F) aspartate aminotransferase to platelet ratio index (APRI).