Abstract
BACKGROUND:
The prognosis of lung cancer patients is poor without useful prognostic and diagnostic biomarker. To search for novel prognostic and diagnostic markers, we previously found homeobox-A13 (HOXA13) as a promising candidate in lung cancer.
OBJECTIVE:
To determine the precisely clinical feature, prognostic and diagnostic value, possible role and mechanism of HOXA13.
METHODS:
Gene-expression was explored by real-time quantitative-PCR, western-blot and tissue-microarray. The associations were analyzed by Chi-square test, Kaplan-Meier and Cox-regression. The roles and mechanisms were evaluated by MTS, EdU, transwell, xenograft tumor and luciferase-reporter assays.
RESULTS:
HOXA13 expression is increased in tumors, and correlated with age of patients. HOXA13 expression is associated with unfavorable overall survival and relapse-free survival of patients in four cohorts. Interestingly, HOXA13 has different prognostic significance in adenocarcinoma (ADC) and squamous-cell carcinoma (SCC), and is a sex- and smoke-related prognostic factor only in ADC. Importantly, HOXA13 can serve as a diagnostic biomarker for lung cancer, especially for SCC. HOXA13 can promote cancer-cell proliferation, migration and invasion in vitro, and facilitate tumorigenicity and tumor metastasis in vivo. HOXA13 acts the oncogenic roles on tumor growth and metastasis by regulating P53 and Wnt/
CONCLUSIONS:
HOXA13 is a new prognostic and diagnostic biomarker associated with P53 and Wnt/
Keywords
Abbreviations
Introduction
Lung cancer is the most frequent malignant cancer in the worldwide [1]. Non-small cell lung cancer (NSCLC) is the common type of lung cancer, accounting for more than 80% of total lung cancer. The main types of NSCLC are lung squamous cell carcinoma (SCC) and lung adenocarcinoma (ADC) [2, 3, 4]. The prognosis of patients with NSCLC is very poor and the rate of 5-year survival is less than 15% [5, 6]. This poor survival of NSCLC patients is mainly due to lack of useful prognostic and early diagnostic marker. Therefore, identification of new, useful prognostic and diagnostic biomarker is crucial for early treatment and prolonging the survival of NSCLC patients.
Homeobox (HOX) family genes, as the nuclear transcription factors, play important roles in embryogenesis and cellular differentiation [7]. HOX family was divided into four HOX gene subgroups, which are HOXA, HOXB, HOXC and HOXD clusters. Previous study has revealed that the position of HOX genes on chromosome is closely associated with site and time of their expression, the genes at 3’ end of the subgroups (for example HOXD1 and HOXA1) are usually expressed early in proximal and anterior regions, however the genes at 5’ end (for example HOXD13 and HOXA13) are generally expressed later in more distal and posterior regions [8]. Increasing evidences have indicated that the expression of HOX genes is usually abnormal in many tumors [9, 10, 11, 12, 13, 14].
HOXA13 (homeobox-A13), as the most distal member of HOXA cluster in HOX family, acts key roles in distal limb and genitourinary development [15]. HOXA13 has been found expressed highly in the hindgut region, but extremely low or absent in anterior areas in human adult [16]. HOXA13 may involve in the carcinogenesis of esophageal carcinoma and hepatocellular carcinoma [17, 18, 19]. In the recent studies, HOXA13 is associated with cancer progression and may be a predictive factor for outcome in hepatocellular cancer, gastric cancer, prostate cancer, cervical cancer, ovarian cancer and glioma [20, 21, 22, 23, 24, 25]. However, the possible role, potential mechanism, prognostic and diagnostic significance of HOXA13 are largely unexplored in human cancers, including lung cancer.
In our present study, HOXA13 was identified as a preferential high expression molecule in tumor tissues by RT-PCR, qRT-PCR and immunohistochemistry in lung cancer. Then the clinical relevance, prognostic significance, diagnostic value, functional role and possible mechanism of HOXA13 were further explored. Our study reveals that HOXA13 is an important prognostic and diagnostic biomarker, and acts as an important oncogene by transcriptional regulating P53 and Wnt/
Materials and methods
Patient samples and cell lines
A total of 130 lung cancer patients (who undergone surgical resection from 2008 to 2010) were acquired from Southwest Hospital in Chongqing, China. The clinico-pathologic data including gender, age, tumor size, histological grade, lymph node status, tumor location, overall survival (OS) and clinical stage was obtained. The present study was approved by the ethics committee from Southwest Hospital which affiliated to Army Medical University. The experiments were performed in accordance with approved guidelines of Southwest Hospital and Army Medical University. The informed consents were all signed by the patients.
The cell lines LTEP-a-2 (LTEP) and SPC-A-1 obtained from the Chinese Academy of Sciences Cell Bank of Type Culture Collection (Shanghai, China) were cultured in RPMI-1640 medium (HyClone, USA) which was supplemented with 10% FBS (fetal bovine serum, HyClone).
RNA isolation
The total RNA was extracted by Trizol reagent (Invitrogen, Carlsbad, CA) from the frozen tissues according to the manufacturer’s instruction. The RNA (5.0
RT-PCR and qRT-PCR
The expression of HOXA13 was evaluated by RT-PCR and quantitative real-time PCR (qRT-PCR). A series of different PCR cycles were performed to determine the appropriate cycles. The products of PCR were subjected to electrophoresis in 3.0% agarose gel. An endogenous control of
Tissue microarray (TMA)
In order to construct lung cancer TMA slides, a core was taken from tumor and adjacent non-tumor tissue within a distance of approximately 30 mm, respectively. The adjacent noncancerous tissues were stained with H&E (hematoxylin-eosin) and evaluated by comparing with the normal tissues. Then, the TMA slides containing lung cancer were constructed as described previously [26].
Immunohistochemical and quantified analyses of HOXA13
Immunohistochemical (IHC) analysis was performed to evaluate HOXA13 expression in patient samples using HOXA13 antibody with 1:200 (Abcam, ab106503), P53 antibody with 1:100 (Santa Cruz, sc-71820), P21 antibody with 1:100 (Santa Cruz, sc-6246), BAX antibody with 1:100 (Santa Cruz, sc-20067),
Construction of HOXA13 expression vector
Full-length of HOXA13 gene was amplified using genomic DNA extracted from human normal blood by PCR, verified by sequencing and then cloned to pIRES2-EGFP vector (stored by Invitrogen, USA). The vector was transfected into cancer cells using the transfection reagent of lipofectamine 2000 (Invitrogen, USA).
Construction of stable transfected cells
The transfection was performed and the stable transfected cells were selected as described in previous study [28].
HOXA13 siRNA assay
The HOXA13 siRNA of this study was purchased from Santa Cruz Biotechnology (sc-45666), and was transfected into cancer cells according to the manufacturer’s instruction.
Western blot analyses
Proteins of transfected cancer cells were run on 12% SDS-PAGE which were transferred into the PVDF membrane (Millipore, USA), and then incubated in the primary antibody (HOXA13, 1:1000, Abcam, ab106503;
MTS assay
The cancer cells transfected with HOXA13 expressing vector were plated at 3, 500 cells per well in 96-well plate. Cell viability was determined via MTS (Promega, USA) at 1, 2, 3, 4 and 5 days posttransfection as described previously [28]. These experiments were done in triplicate.
5-ethynyl-2’-deoxyuridine (EdU) assay
EdU assay was carried out as described previously [28]. Briefly, cancer cells were cultured in 96-well plates at 4,000 cells per well and transfected with pIRES2-EGFP-HOXA13 or empty vectors. After 46 h of transfection, cancer cells were incubated in EdU for about 2 h, and fixed with 4% formaldehyde for 0.5 h. The fixed cells were incubated with glycine for 5 min, and then treated for 10 min with the Triton X-100. The cancer cells were thereafter incubated for 30 min in the Apollo reaction cocktail after phosphate-buffered saline washing, and then treated with the Triton X-100 twice. DNA was stained in Hoechst 33342 stain for 30 min and was observed with fluorescence microscopy.
Migration and invasion assay
Transwell assays were carried out in 24-well plates without or with the matrigel (8
Clinical microarray database analyses
The Kaplan-Meier Plotter (
ROC (receiver operating characteristic) curve analyses
The diagnostic values of HOXA13 expression were evaluated in NSCLC, ADC and SCC patients using the expression data of tumor and normal tissues from TCGA database. The x and y axes in the ROC curve are the specificity and the sensitivity, respectively. The area under curve (AUC) in the ROC curve was calculated to evaluate the ability of HOXA13 expression in predicting the diagnostic outcome of the patients.
Tumorigenicity assay in nude mice
The LTEP stably cells with control and HOXA13 vector were suspended in PBS and were subcutaneously injected into the flank of male Balb/c nude mice (6-week-old) using 6
Experimental metastasis assay in nude mice
The LTEP stably cells with control and HOXA13 vector were suspended in PBS and were injected into tail vein of male Balb/c nude mice (6-week-old) using 2
P53 and Wnt/
-catenin signaling activity and luciferase reporter assays
LTEP and SPC-A-1 cells were co-transfected with TOP/FOP-Flash plasmids or pp53-TA-Luc plasmid with pIRES2-EGFP-HOXA13 and empty vectors, respectively. The plasmids of TOP/FOP-Flash were previously purchased from the Addgene (No. 12456 and 12457, Cambridge, USA), and the plasmids of pp53-TA-Luc was purchased from Beyotime Biotechnology (D2223, Shanghai, China). The Varioskan-LUX (a fluorescence microplate reader measurement system, Thermo Fisher, MA, USA) and the Dual-luciferase reporter kit (Promega) were used to analyze the luciferase reporter assays as described previously [28]. The activities of luciferase were measured at 32 h post transfection, and the experiment was performed thrice.
Statistical analyses
Statistical analyses were carried out via SPSS 13.0 software (SPSS, Chicago, IL). The differences of categorical variables were analyzed via Chi-square test. The OS was calculated and evaluated using Kaplan-Meier with log-rank test. Multivariate analysis of prognostic predictors was performed using Cox-regression. The high and low expression groups were categorized base on the average score. When
HOXA13 expression is increased in tumor tissue of lung cancer. (A) HOXA13 mRNA expression was determined by RT-PCR and qRT-PCR analyses in normal and tumor tissues. The “N” represents normal lung tissue. The “M” represents DNA marker. The “T” represents tumor tissue. 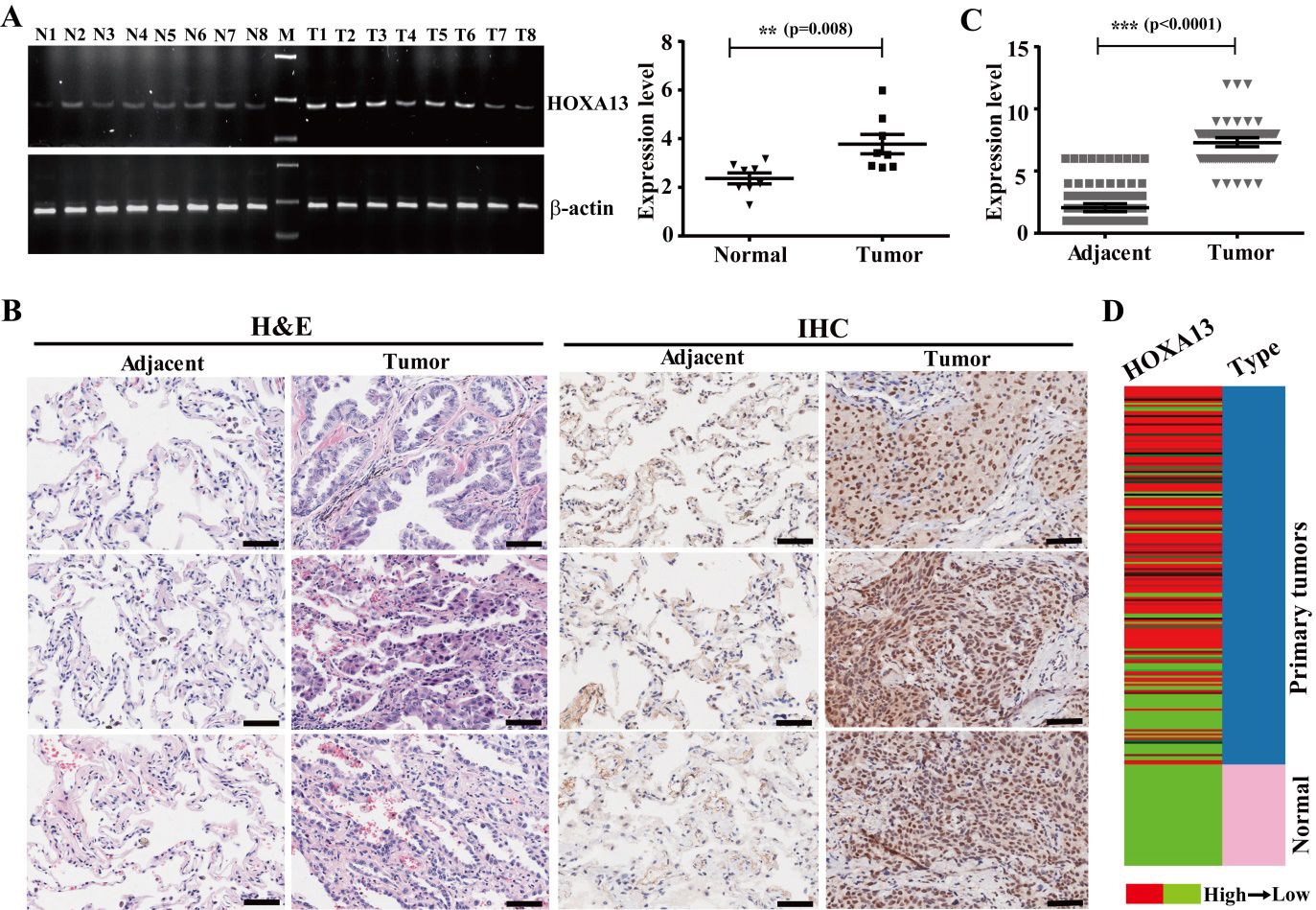
HOXA13 expression is sharply increased in NSCLC tissues
To investigate HOXA13 expression in tissues of NSCLC, HOXA13 expression was tested in normal lung and NSCLC tissues by RT-PCR and qRT-PCR. The expression of HOXA13 was elevated in NSCLC tissues (3.86
HOXA13 expression is associated with age of NSCLC patients
Two groups of the patients were classified according to the quantified expression of HOXA13, the high group (score greater than average expression) and the low group (score equal to and less than average expression) groups. Then the associations between HOXA13 expression and the clinico-pathologic features of the NSCLC patients were investigated, and the expression of HOXA13 was found to be closely correlated with age (
Correlations of HOXA13 expression with clinicopathologic features in human patients (
123)
Correlations of HOXA13 expression with clinicopathologic features in human patients (
The age of two patients and the clinical stage of seventeen patients are missing.
To determine the relationships between HOXA13 expression and survival prognosis of NSCLC patients, we analyzed the patients’ survival data. The results of Kaplan-Meier analyses revealed that the patients with high HOXA13 expression owned a poor OS compared to the patients with low HOXA13 expression (
Multivariate analysis of different prognostic factors in human lung cancer patients (
123)
Multivariate analysis of different prognostic factors in human lung cancer patients (
Abbreviations: HR, hazard ratio; CI, confidence interval.
High expression of HOXA13 is associated with adverse OS of the patients with lung cancer. (A) The high expression and low expression groups of HOXA13 was shown in human NSCLC tissues. Scale bars represent 50 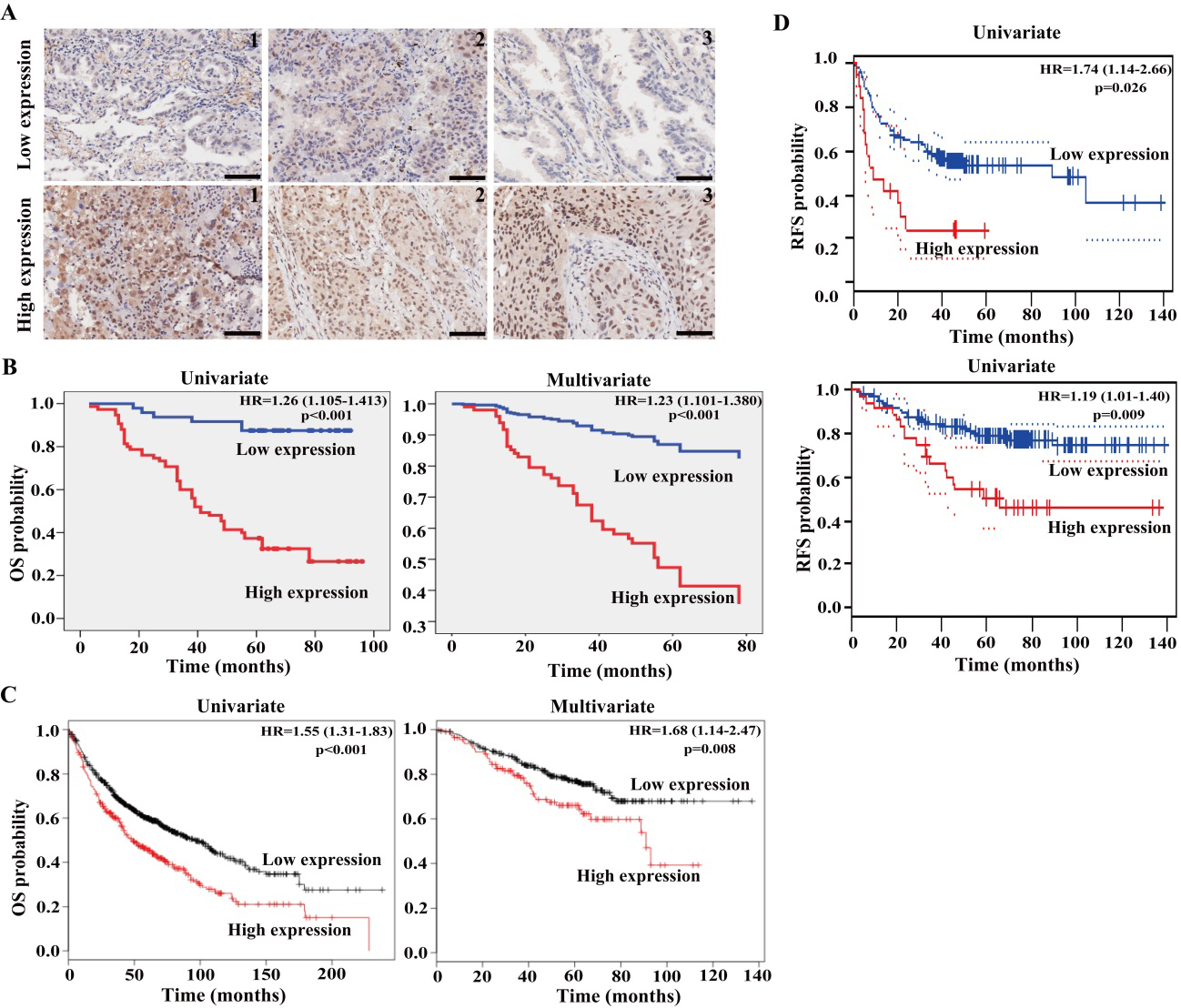
The correlations were then investigated between HOXA13 expression and the survival in the patients with ADC and SCC, respectively. The ADC patients with high HOXA13 expression owned a shortened OS when compared to the patients with low HOXA13 expression (HR
HOXA13 is a sex- and smoke-related prognostic factor and potential diagnostic biomarker. (A) High expression of HOXA13 is associated with unfavorable OS in the patients with ADC, but with better OS in the patients with SCC. Kaplan-Meier survival analysis was performed between HOXA13 expression and the OS of 673 ADC patients and 271 SCC patients from database of Kaplan-Meier Plotter. Auto best cutoff was chosen. (B) HOXA13 expression is an unfavorable prognostic biomarker of the male ADC patients, but a favorable prognostic biomarker of the female ADC patients. Kaplan-Meier analysis was performed between HOXA13 expression and the OS of 328 male and 287 female patients with ADC in the database of Kaplan-Meier Plotter. Auto best cutoff was used. (C) HOXA13 is a smoke-related prognostic biomarker of the ADC patients. Kaplan-Meier analysis was performed between HOXA13 expression and OS of 140 non-smoked and 231 smoked ADC patients in the database of Kaplan-Meier Plotter. Auto best cutoff was used. (D) ROC curves and AUC analysis were performed to evaluate the diagnostic values of HOXA13 expression in NSCLC, ADC and SCC patients. The gray and red lines represent the random performance and the ROC curve, respectively.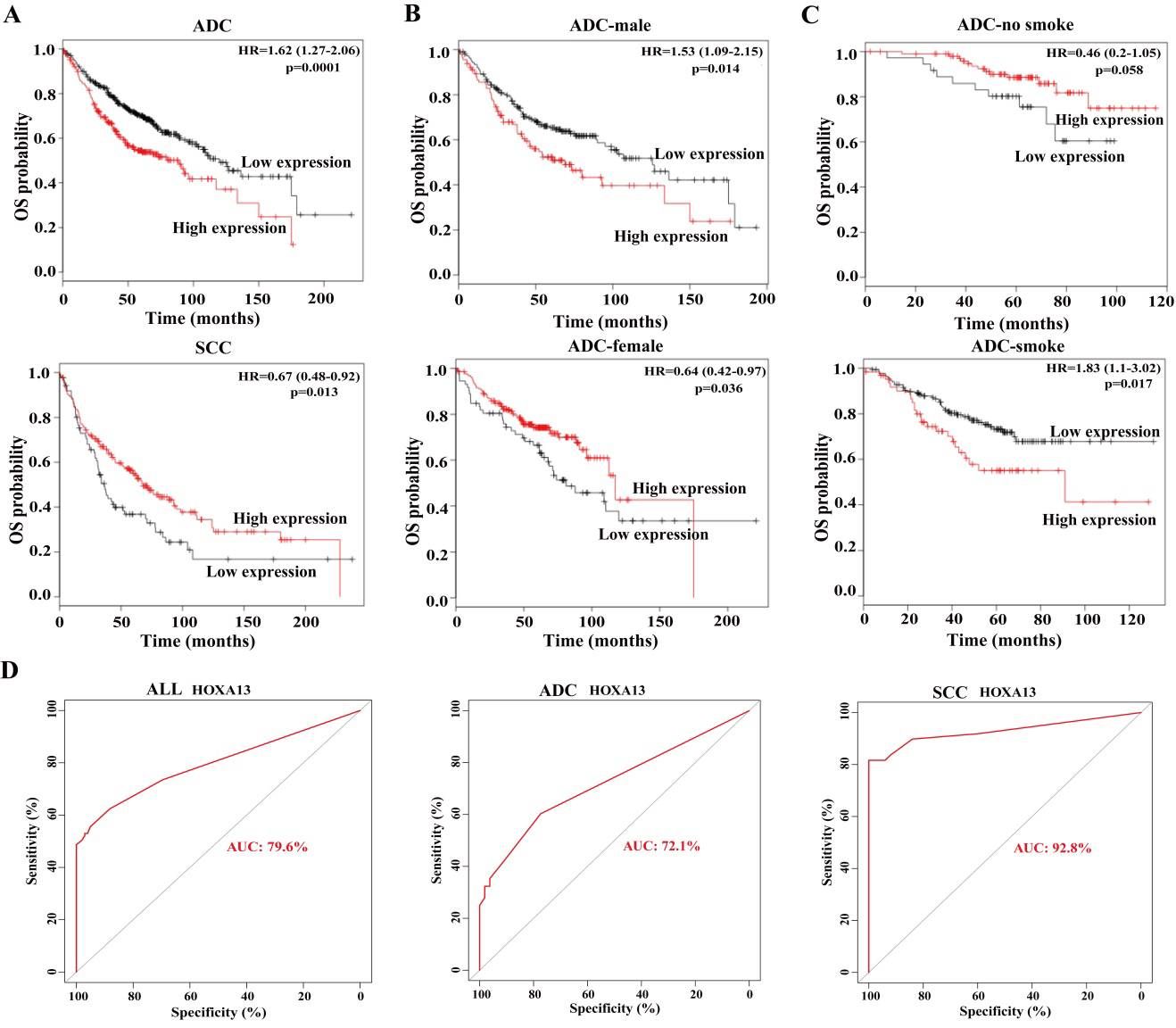
HOXA13 accelerates cancer cell growth and proliferation. (A) Cancer cell transfectants of HOXA13 expressing vector and empty vector (mock) control were identified in SPC-A-1 and LTEP cells by western blot. Strong expression of HOXA13 was detected after transfection of HOXA13, whereas not detected after transfection of empty vector. (B) MTS assay was carried out to examine the role of HOXA13 on cancer cell growth and proliferation in SPC-A-1 and LTEP cells. Cell viability was determined by the CellTiter 96 AQueous One Solution Cell-Proliferation-Assay (Promega) in triplicate. The “*” represents 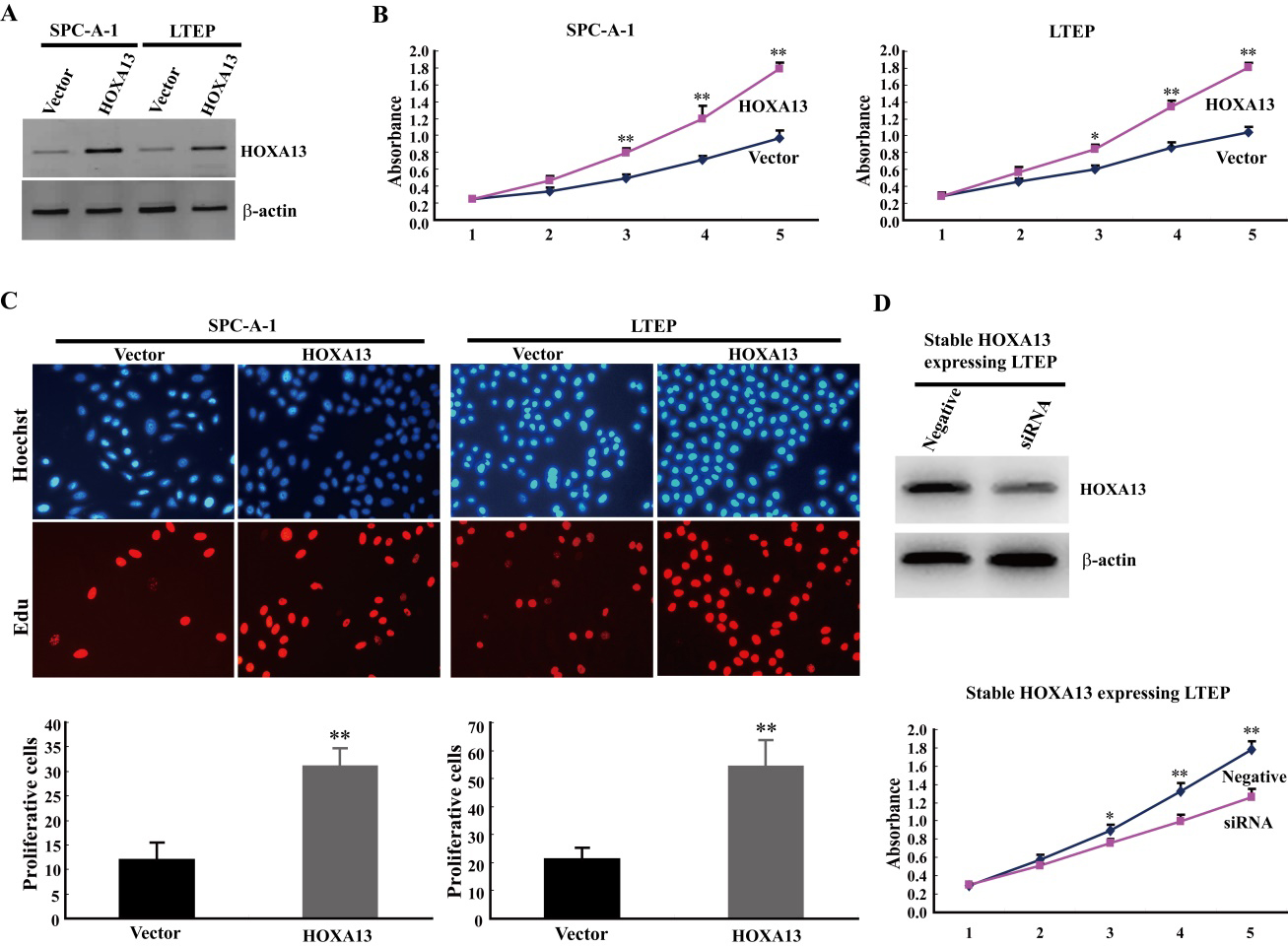
HOXA13 stimulates cancer cell migration and invasion. (A) The cancer cell migration was determined by transwell assays without matrigel after transfection of HOXA13 expressing vector or empty vector in SPC-A-1 and LTEP cells. The cancer cells in lower well of chamber were fixed, stained and then quantified. The migration cells were measured with the average count of at least five random microscopic fields. Error bar indicates SEM. The “**” represents 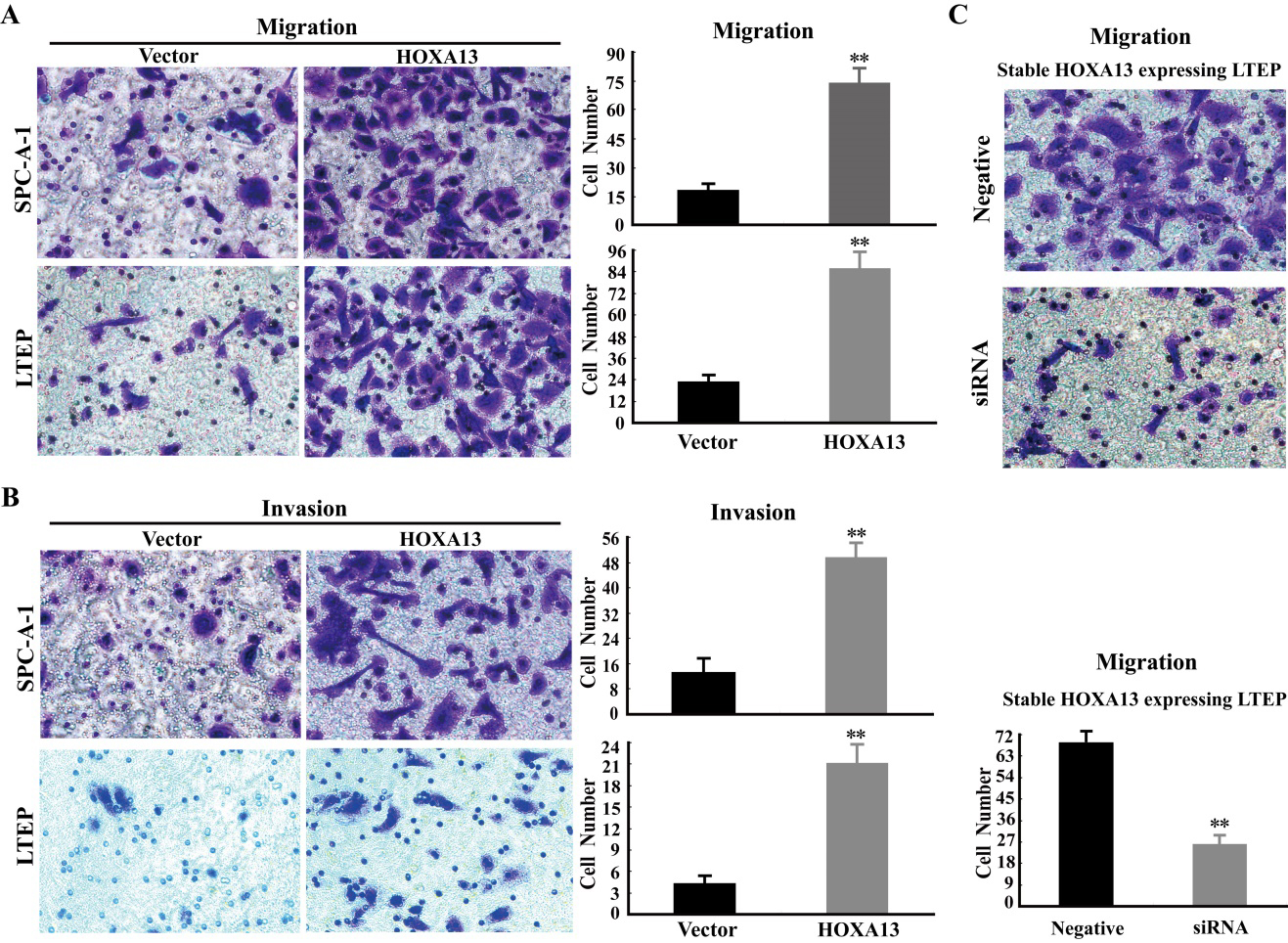
HOXA13 promotes tumor growth and metastasis in vivo. (A) The tumor growth curve and tumor weight were compared between the nude mice with vector control and the nude mice with HOXA13. The result was got from three independent experiments (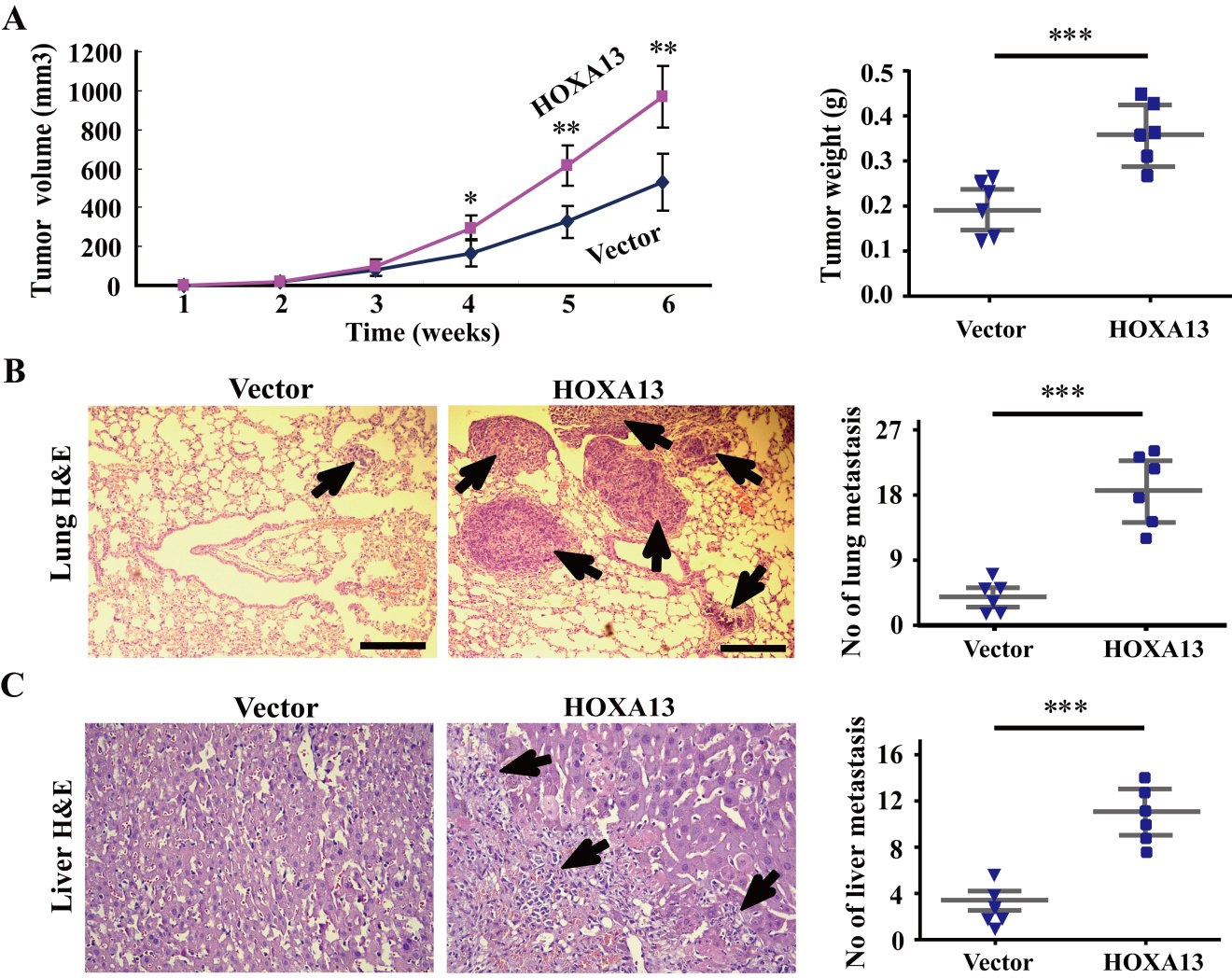
The roles of HOXA13 on proliferation and metastasis are associated with P53 and Wnt/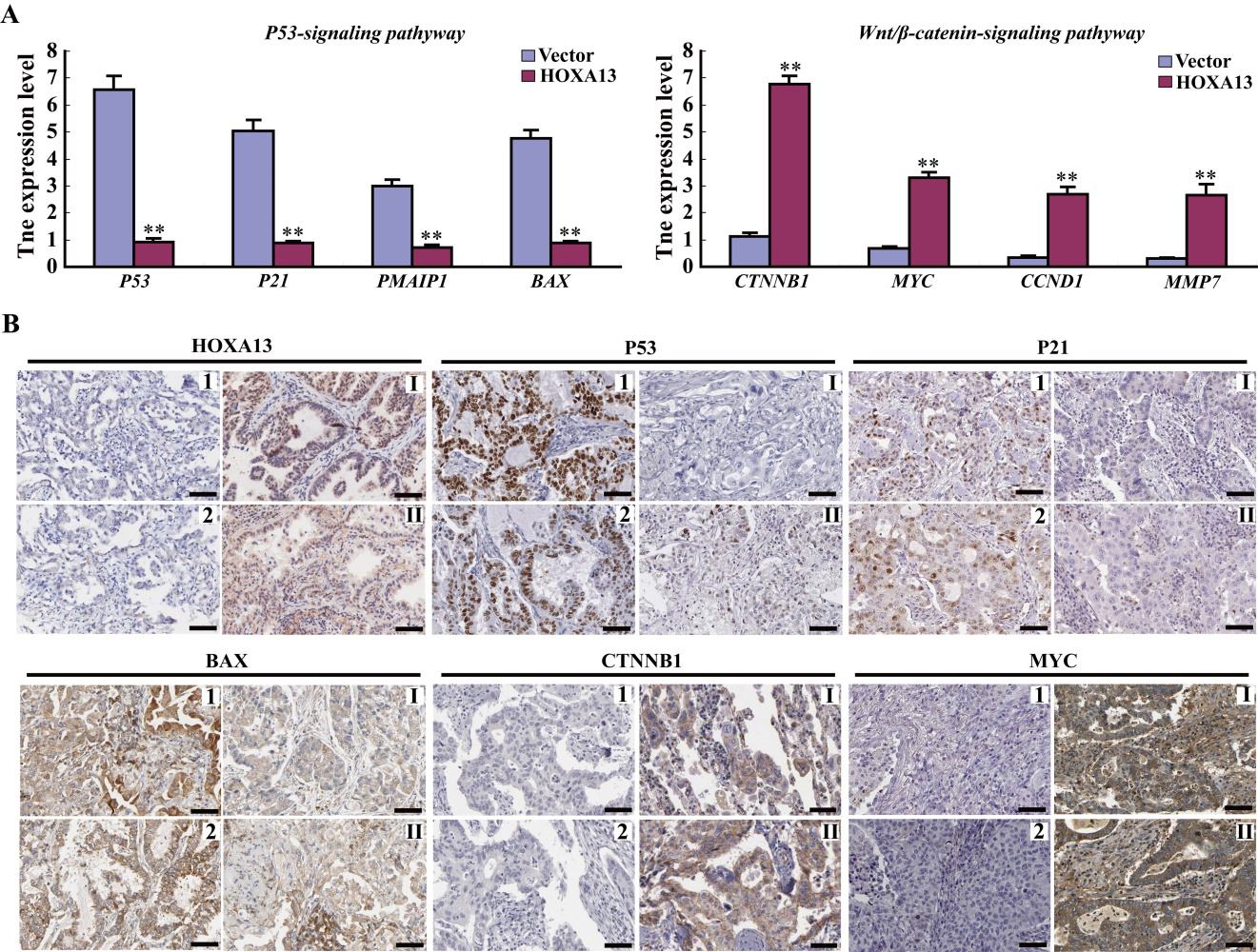
HOXA13 acts as an oncogene by regulating the activities of P53 and Wnt/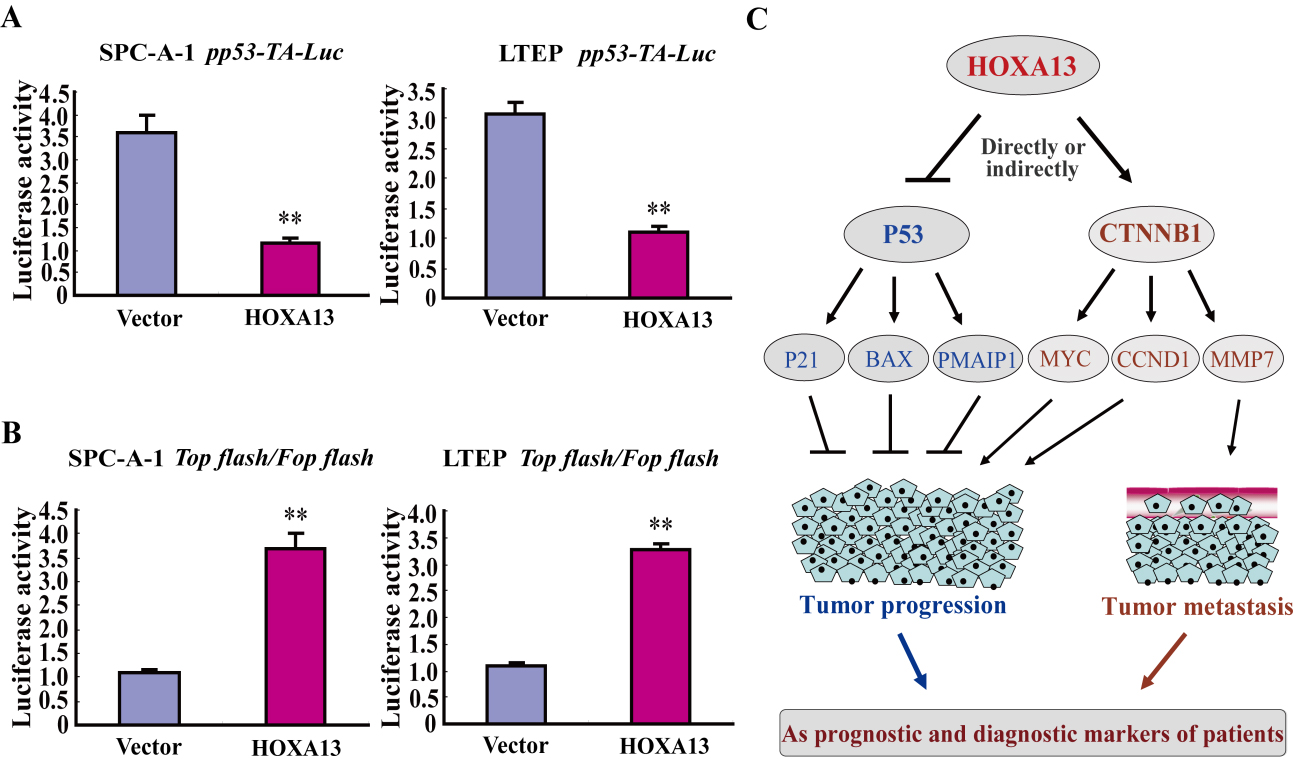
The correlation of HOXA13 expression and the OS of different sex ADC patients was further determined. High HOXA13 expression was prominently associated with poor OS of the male patients with ADC (HR
HOXA13 expression is a smoke-related prognostic factor for ADC patients
Next, we ascertain whether HOXA13 expression predicts unfavorable OS in the ADC patients related to smoking. The analysis results found that high expression of HOXA13 was closely associated with unfavorable OS of the patients with smoking (HR
HOXA13 is a promising diagnostic biomarker for NSCLC patients
HOXA13 expression was significantly increased in tumor tissues compared to normal and corresponding tumor-adjacent normal tissues. To explore whether this change of HOXA13 expression can serve as a potential diagnostic biomarker for lung cancer, we performed ROC curves and AUC analyses to evaluate the diagnostic values of HOXA13 expression in NSCLC, ADC and SCC patients. HOXA13 expression exhibited a high sensitivity and specificity with AUC being 79.6%, 72.1% and 92.8% in NSCLC, ADC and SCC patients, respectively (Fig. 3D). Moreover, HOXA13 could exhibit a higher diagnostic efficacy in SCC patients than that in NSCLC and ADC patients (Fig. 3D). These data reveal that HOXA13 can serve as a promising diagnostic biomarker for NSCLC patients, especially for SCC patients.
HOXA13 markedly stimulates cancer cell growth and proliferation in vitro
To determine possible roles of HOXA13 in NSCLC, we performed MTS and EdU assays to determine the cell growth and proliferation in SPC-A-1 and LTEP cell lines (two human NSCLC cell lines lowly expressed HOXA13) following over-expression of HOXA13. Ectopic expression of HOXA13 could significantly promote cancer cell growth and proliferation when compared to the empty vector control in both SPC-A-1 cells and LTEP cells (Fig. 4A–C). Moreover, knockdown of HOXA13 could inhibit cancer cell growth and proliferation when compared to the negative control in stable HOXA13 expressing LTEP cells (Fig. 4D). These results indicate that HOXA13 remarkably activates cancer cell growth and proliferation in NSCLC.
HOXA13 significantly promotes migration and invasion of cancer cells in vitro
To investigate the roles of HOXA13 on migration and invasion of cancer cells in NSCLC, we performed transwell assays without and with matrigel, respectively. The results of transwell assays without matrigel revealed that there was a significant rise of cancer cell migration in HOXA13-transfected cells when compared to the empty vector control in SPC-A-1 and LTEP cells (Fig. 5A and B). The data of transwell assays with matrigel showed that the invasion of cancer cells was also significantly increased in HOXA13-transfected cells when compared to the empty vector control in SPC-A-1 and LTEP cells. Furthermore, the migration of cancer cells was indeed decreased in HOXA13 knockdown cells when compared to the negative control in stable HOXA13 expressing LTEP cells (Fig. 5C). These results demonstrate that HOXA13 markedly accelerates cancer cell migration and invasion in NSCLC.
HOXA13 strongly accelerates tumor growth and metastasis in nude mice
After determining the roles of HOXA13 in vitro, we want to investigate whether the HOXA13 still plays its mentioned roles in vivo. The xenograft tumor model was used to assess the growth and metastasis of stable tumor cells containing empty and HOXA13 vectors. The data of xenograft tumor model demonstrated that the volume and weight of tumor was much larger in the nude mice receiving stable expressing HOXA13 cells than that of receiving empty vector cells by measuring and weighing (Fig. 6A), and more lung and liver metastatic tumors were found in the nude mice receiving stable expressing HOXA13 cells than that receiving stable empty vector cells by HE staining and quantitative analyses (Fig. 6B and C). These experimental results reveal that HOXA13 strongly accelerates tumor growth and metastasis in vivo.
HOXA13 plays the oncogenic roles by regulating P53 and Wnt/
-catenin signaling-pathways
To gain insights into the mechanism of oncogenic effect of HOXA13, the possible downstream targets were screened by qRT-PCR in LTEP cells transfected with HOXA13, and were confirmed in patient samples by IHC. The expressions of P53 and its targets P21, PMAIP1 and BAX (the P53 signaling pathway) were reduced, whereas the expressions of
To further verify HOXA13 represses P53 signaling activity and activates Wnt/
Discussion
In our present study, we determined the precisely clinical relevance, prognostic and diagnostic significance of HOXA13 expression in patients with NSCLC for the first time. The expression of HOXA13 was much higher in the tumor tissues than that in the non-tumor tissues, and this expression was closely associated with age of the patients. The high HOXA13 expression patients had a short OS and RFS when compared to the low HOXA13 expression patients, and HOXA13 expression was an independent prognostic marker for the patients with NSCLC. Moreover, HOXA13 is a potential and promising diagnostic biomarker for NSCLC patients, especially for SCC patients. These data suggest that the expression of HOXA13 is an important predictor for prognosis and diagnosis of patients in NSCLC.
Previous evidences have indicated that the expression of HOXA13 is higher in tumor tissues than in tumor-adjacent normal tissues and normal tissues in esophageal squamous cell cancer, hepatocellular cancer, prostate cancer, gastric cancer and glioma [17, 19, 20, 22, 25]. In our study, we found that the expression of HOXA13 was strongly increased in tumor tissues of NSCLC, which is consistent with the previous researches [32]. This change of HOXA13 expression suggests that HOXA13 is probably associated with tumorigenesis in NSCLC. HOXA13, as a nuclear transcription factor, is expected to be usually expressed in the nuclei of cells [17, 33]. However, some studies have revealed that HOXA13 is expressed in the cytoplasm of tumor cells [19, 21]. In our study, we showed that HOXA13 was expressed both in the cytoplasm and the nuclei of tumor cells of NSCLC samples (Figs 1B, 2A, 7B), which may be associated with its interaction with a cytoplasmic protein in these samples.
HOXA13 has been reported as a potential biomarker for predicting outcome in esophageal squamous cell cancer, hepatocellular cancer, gastric cancer, prostate cancer and glioma [19, 20, 21, 22, 25]. In the present study, the patients with high HOXA13 expression owned a poor OS compared to the patients with low HOXA13 expression in NSCLC. Further analyses revealed that high expression of HOXA13 is associated with adverse OS in patients with ADC, but with better OS in SCC patients, indicating different prognostic significance of HOXA13 in the ADC and SCC patients, respectively. HOXA13 expression is an adverse prognostic biomarker of the male ADC patients, but is a favorable prognostic biomarker of the female ADC patients, implying a sex-specific prognostic factor in ADC patients. Moreover, high expression of HOXA13 is closely associated with poor OS of the ADC patients with smoking, whereas the expression of HOXA13 is not correlated with OS of ADC patients without smoking, which suggests that HOXA13 is a smoke-related prognostic biomarker in ADC patients. The results suggest that HOXA13 is an important potential and unique biomarker for predicting outcome of NSCLC patients. In addition, HOXA13 expression is a sex- and smoke-related prognostic factor of ADC patients, which suggests that HOXA13 expression may be associated with sex and smoking of the patients. However, the specific correlations and roles between them are still need to explore in the future
Accumulated studies have demonstrated that HOXA13 is involved in tumor progression in esophageal squamous cell cancer, hepatocellular cancer, prostate cancer and glioma [17, 19, 22, 25]. HOXA13 can promote cancer cell growth and proliferation in esophageal cancer, prostate cancer and glioma, and also affects cancer cell cycle, invasion and apoptosis [19, 22, 25]. In the present study, HOXA13 was found to significantly promote cancer cell proliferation, migration and invasion in vitro, and strongly accelerate tumor growth and metastasis in vivo. These data strongly suggest that HOXA13 is a real and key oncogene in lung cancer. However, the roles of HOXA13 on cancer cell cycle and apoptosis are still unknown. Further studies are needed to solve these problems.
For a long time, few studies on the mechanisms of HOXA13 function were performed in human cancers. A recent study shows that HOXA13 stimulates cancer cell proliferation and metastasis partly via activating Erk1/2 in gastric cancer [34]. Another study reveals that HOXA13 confers 5-FU resistance by MRP1 via a p53-dependent-pathway in gastric cancer [35]. However, the clear molecular mechanisms of HOXA13 roles in cancer are still largely undeveloped. In our present study, we found that HOXA13 decreased the expressions of P53 signaling critical and target genes P53, P21, PMAIP1 and BAX, and increased the expressions of Wnt/
In conclusion, HOXA13 is a potential and promising prognostic and diagnostic biomarker, and acts as a new oncogene promoting tumor progression and metastasis through P53 and Wnt/
Author contributions
Conception: FH, JD and JL.
Interpretation or analysis of data: YW, BH, YD, GH, XQ, YL, YY, FH, JD and YR.
Preparation of the manuscript: FH, ZC, XQ and YL.
Revision for important intellectual content: FH, JD, YW and JL.
Supervision: FH, JD and JL.
Footnotes
Acknowledgments
This work was supported by the National Natural Science Foundation of China (81672856; 81502551; 81803028), and the Natural Science Foundation of Chongqing (cstc2015jcyjBX0060).
Conflict of interest
The authors have no conflicts of interest to declare.
Supplemental Table
Sequence of primers used in the present study
Primer
Sequence (5’-3’)
Purpose
HOXA13-F
ACGAACCCTTGGGTCTTCCC
Expression analysis
HOXA13-R
TCGTGGCGTATTCCCGTTCA
Catenin-F
ATGACTCGAGCTCAGAGGGT
Expression analysis
Catenin-R
ATTGCACGTGTGGCAAGTTC
MYC-F
GCGAACACACAACGTCTTGG
MYC-R
TGAGCTTTTGCTCCTCTGCT
CCND1-F
GATGCCAACCTCCTCAACGA
CCND1-R
GGAAGCGGTCCAGGTAGTTC
MMP7-F
CATGATTGGCTTTGCGCGAG
MMP7-R
AGACTGCTACCATCCGTCCA
P53F
GCTGCTCAGATAGCGATGGT
Expression analysis
P53R
CACGCACCTCAAAGCTGTTC
PMAIP1-F
GACTGTTCGTGTTCAGCTCG
PMAIP1-R
TAGCACACTCGACTTCCAGC
P21-F
CACCGAGGCACTCAGAGGAG
P21-R
CATTAGCGCATCACAGTCGC
BAX-F
GTCACTGAAGCGACTGATGT
BAX-R
GGAAAAACACAGTCCAAGGCA
CCACGAAACTACCTTCAACTCC
Internal control
GTGATCTCCTTCTGCATCCTGT
