Abstract
Overall survival of non-small cell lung cancer (NSCLC) patients remains disappointingly low. The estrogen receptor (ER) was considered a promising therapeutic target for NSCLC. Numerous studies have linked expression of ERβ to lung cancer outcome. However, results are conflicting regarding the association of ERβ with surviving lung cancer. The aim of this meta-analysis was to evaluate the prognostic aspect of ERβ expression on survival among NSCLC patients. We performed a final analysis of prognostic value of overexpression ERβ on 3500 patients from 18 evaluable studies (from January 1, 2000 to May 1, 2021). The reference category is specified as low ERβ expression levels. Summarized hazard ratios were calculated. Our study showed that the pooled hazard ratios of ERβ overexpression for overall survival in NSCLC was 0.81 (95% confidence interval (CI): 0.64–1.02, P = 0.07) by univariate analysis and 1.06 (95% CI: 0.83–1.36, P = 0.63) by multivariate analysis. Pooled hazard ratio by univariate analysis in Asian studies was 0.73 (95%CI: 0.59–0.89, P = 0.002). Pooled hazard ratio by univariate analysis was 0.75 (95% CI: 0.61–0.93, P = 0.009) from seven studies reported for nuclear ERβ. No significant results were found in subgroups by multivariate analysis. No significant results were found in studies outside Asia or in studies reported for cytoplasmic ERβ. Our results suggested that expression of ERβ might not be a direct prognostic factor for NSCLC patients. More detailed prospective studies are needed to identify direct prognostic factors in these patients.
Introduction
Lung cancer is the leading cause of cancer-related deaths among males and the second leading cause among females worldwide.1,2 Modern advances in genomics allow development of therapies that target oncogenic driver mutations or translocations in genes such as EGFR and ALK. 3 More recently, immune checkpoint inhibitors have shown efficacy in the metastatic and locally advanced settings. 4 Small molecule tyrosine kinase inhibitors and immunotherapy have brought very good survival benefits in some patients. However, the overall survival (OS) rate of non-small cell lung cancer (NSCLC) is still very low, especially in metastatic NSCLC. 5 The estrogen receptor (ER) is another promising target. 6 Many studies have demonstrated that estrogens and estrogen signaling play significant roles not only in normal lung development but also in lung cancer pathophysiology.7,8 Cell lines derived from lung tumors of both men and women express ERs and respond to estrogens. Estrogen can be synthesized in the lung by the enzyme aromatase (CYP19A1). Aromatase present in NSCLC cells and tumor tissues is functional. Estrogens are known to stimulate NSCLC cell proliferation, whereas the antiestrogen fulvestrant inhibits this effect. 9 Cellular responses to estrogen and its analogues are mediated by two distinct receptors, ERα and ERβ. In lung cancer cells, ERβ is sufficient to induce the full-range of estrogenic responses when no detectable full-length ERα protein is present. 10 In addition, ERβ appears to be the predominant form in lung cancer in the literature.11,13
Consequently, numerous studies have linked ERβ expression to lung cancer outcome.9,10, Some studies suggested that high expression of ERβ was unfavorable to the prognosis of lung cancer patients.19,20,24 However, some studies found that high expression of ERβ was beneficial for lung cancer survival.21,23 There were even two meta-analyses of these studies with opposite conclusions: Luo et al. 26 suggested high expression of ERβ was significantly associated with improved OS in NSCLC patients based on univariate analysis, while Ma et al. 27 concluded that ERβ overexpression was not associated with NSCLC prognosis based on univariate analysis. Based on the discordant results, we conducted this meta-analysis in order to evaluate the prognostic impact of ERβ expression on survival among NSCLC patients.
Luo et al. 26 published their meta-analysis in 2015, which included 13 studies, including 3 Chinese studies. Ma et al. 27 published the meta-analysis in 2016, which included 11 studies. Until now, at least 4 new studies have been published.25,28–30 Therefore, we considered our systematic review to be an extension and update of the meta-analyses by Luo et al. 26 and Ma et al. 27
Materials and methods
We conducted the systematic review of cohort studies on the effect of ERβ expression on survival among NSCLC patients based on the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) and the Meta-analysis of Observational Studies in Epidemiology (MOOSE) statements.31,32
Literature search
We performed extensive literature searches in 15 databases: PubMed, Web of Science, EMBASE, Cochrane Library, Cochrane Central Register of Controlled Trials, Cochrane Database of Systematic Reviews, CINAHL, Open-Grey, metaRegister of Controlled Trials, Clinical-Trials.gov, WHO Clinical Trials Database, WanFangData, CQVIP, COMPENDEX, and China National Knowledge Infrastructure. We searched the following keywords in the above databases: (“estrogen receptor” OR “ER” OR “ERβ” OR “estrogen receptor beta”) AND (“non-small cell lung cancer” OR “NSCLC” OR “lung cancer” OR “lung carcinoma” OR “carcinoma of lung”) AND (outcome OR survival OR prognosis OR OS). We included articles published from January 1, 2000 to May 1, 2021. We searched the citations and references of the identified literature for more potentially relevant studies to ensure literature saturation. Since this meta-analysis is an extension and update of Luo et al. 26 and Ma et al., 27 we also searched for articles considered by them, to avoid missing any existing literature.
Study selection
We restricted publications to be in either English or Chinese. We searched for observational studies (study design criteria) conducted in NSCLC patients (setting criteria) and studies reporting the OS (outcome criteria) in at least two different ERβ expression levels using comparable methods (design criteria). In order to assess the quality of individual studies and methodological differences between studies, we included only studies that reported ERβ testing and assessment methods. We included primary NSCLC patients and excluded those with other cancers, severe organ dysfunction, or severe infections. Two reviewers independently identified potentially eligible articles by screening all titles and abstracts; in cases of disagreement, a third researcher was involved.
Data extraction and quality assessment
The following data were collected from included studies using a predesigned abstraction form: name of first author, year of publication, country, sample size, average age, sex ratio, pathological type, laboratory methodology, follow-up period, number of people with high ERβ expression, number of smoking people, study year, and univariate analysis hazard ratio (HR) and 95% confidence interval (CI), multivariate analysis HR and 95% CI. Data from each study were collected independently and in duplicate by two reviewers; in cases of disagreement, a third researcher was involved. Two reviewers independently scored the quality of included studies using the Newcastle–Ottawa Scale (NOS). 33 In cases of disagreement, a third researcher was involved.
Statistical methods
The intensity of the relationship between the expression levels of ERβ and OS was described as HR. The reference category was specified as low ERβ expression levels, and an HR greater than 1 implied a greater rate of mortality of high ERβ compared with low ERβ, and vice versa. HR and 95% CI were obtained from published papers or calculated by Kaplan–Meier survival curves using the software Engauge Digitizer Version 4.1 (http://digitizer.sourceforge.net/) and the method previously presented by Parmar et al and used by Zhang et al.34,35 The pooled HR and corresponding 95% CI were used to assess the prognostic value of ERβ in NSCLC patients. According to the Cochrane Handbook, we supposed a moderate level of heterogeneity between studies for I2 values ranging from 30% to 60%. 36 If I2 was equal or less than 60%, the fixed effects model was used to estimate the pooled HR; if I2 exceeded 60% for the pooled analysis, we explored sources of heterogeneity in subgroups of studies and used a random effects model. We used a funnel plot and Egger's test 37 to evaluate reports for publication bias. A P-value less than 0.05 was considered statistically significant. Review Manager 5.3 38 and STATA 15 39 were used to perform statistical analysis.
Results
Study selection and characteristics
A total of 208 articles were retrieved from the 15 electronic databases according to our defined search terms. Then, 87 duplicates were removed. Through careful reading the abstracts, 49 studies that focused on the association between ERβ expression and NSCLC survival were included in our full-text review process. After reading the full-text studies, 31 papers had to be excluded because the data were not extractable or could not provide enough information about OS. As a result, 18 eligible studies comprising 3500 patients were included in this meta-analysis. 13, 19,25,29,40–53 The process of literature screening is shown in the PRISMA Flow Diagram. (Figure 1) Among all the included studies, the individual characteristics and HRs are summarized in Table 1. The quality of studies with a NOS score of 7 or more was considered high. All of the included studies had NOS scores of 8–9 (as shown in Table 2 in the supplementary file).
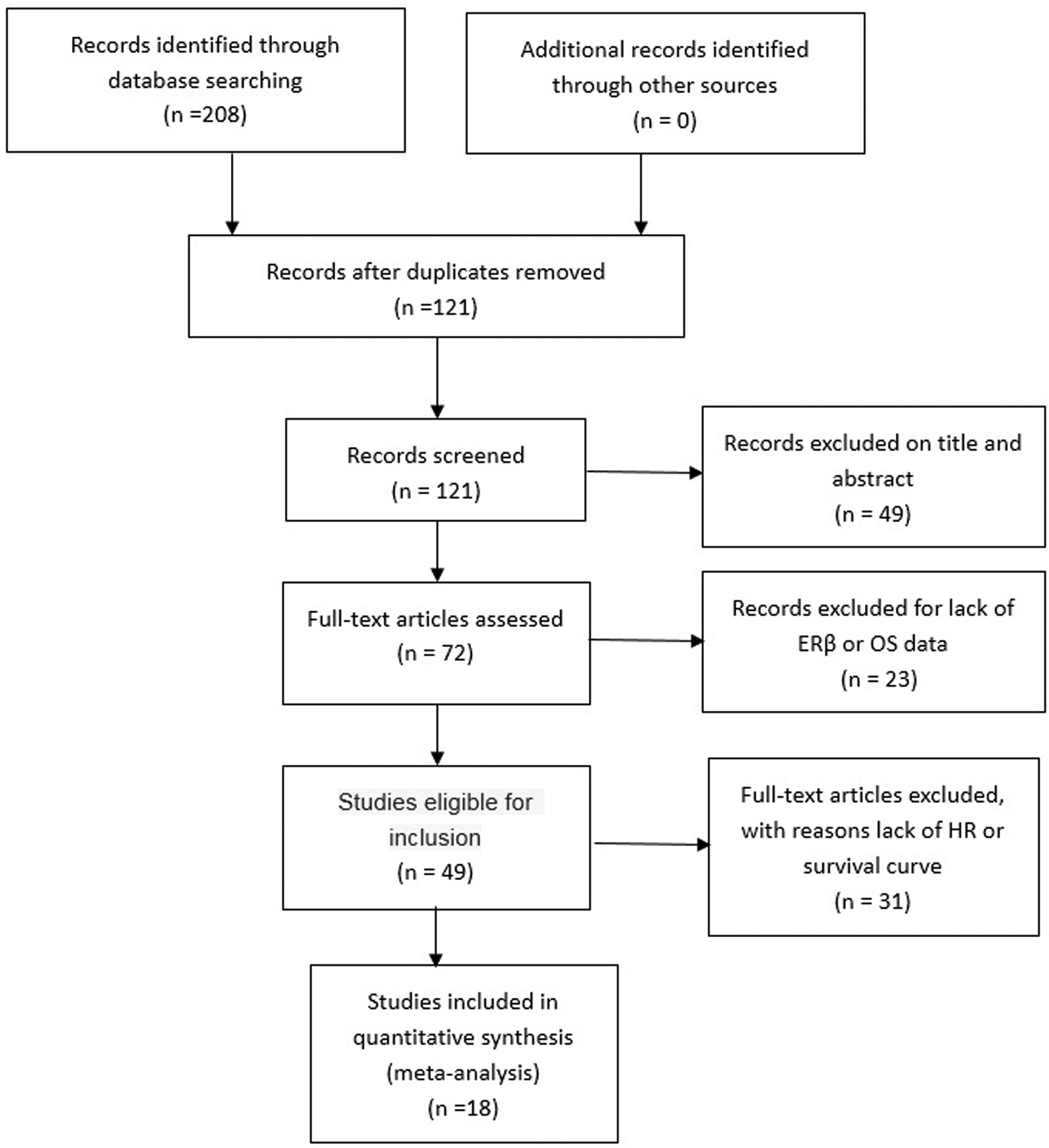
PRISMA flow diagram.
Characteristics of the included studies.
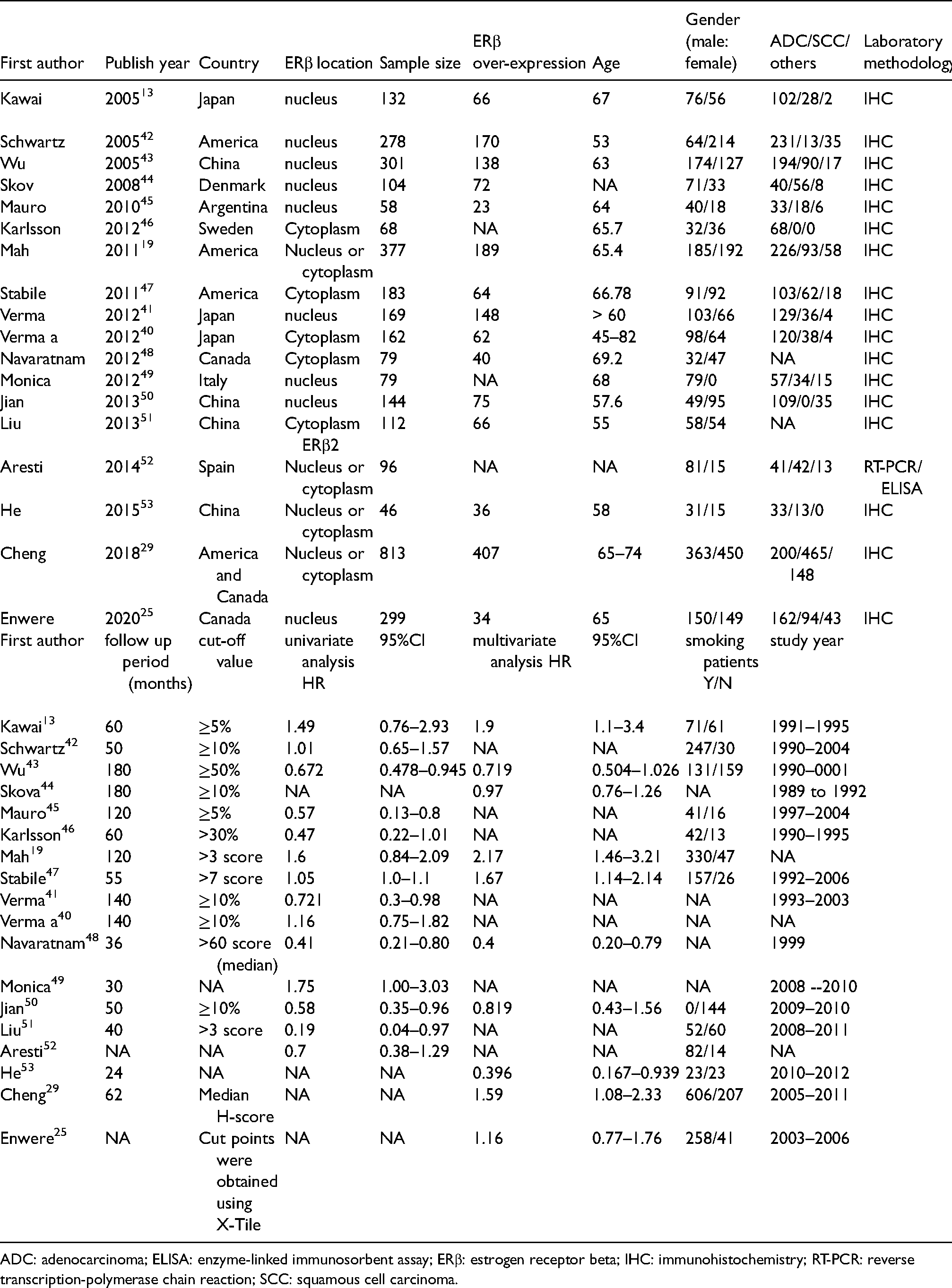
ADC: adenocarcinoma; ELISA: enzyme-linked immunosorbent assay; ERβ: estrogen receptor beta; IHC: immunohistochemistry; RT-PCR: reverse transcription-polymerase chain reaction; SCC: squamous cell carcinoma.
NOS score of included studies.
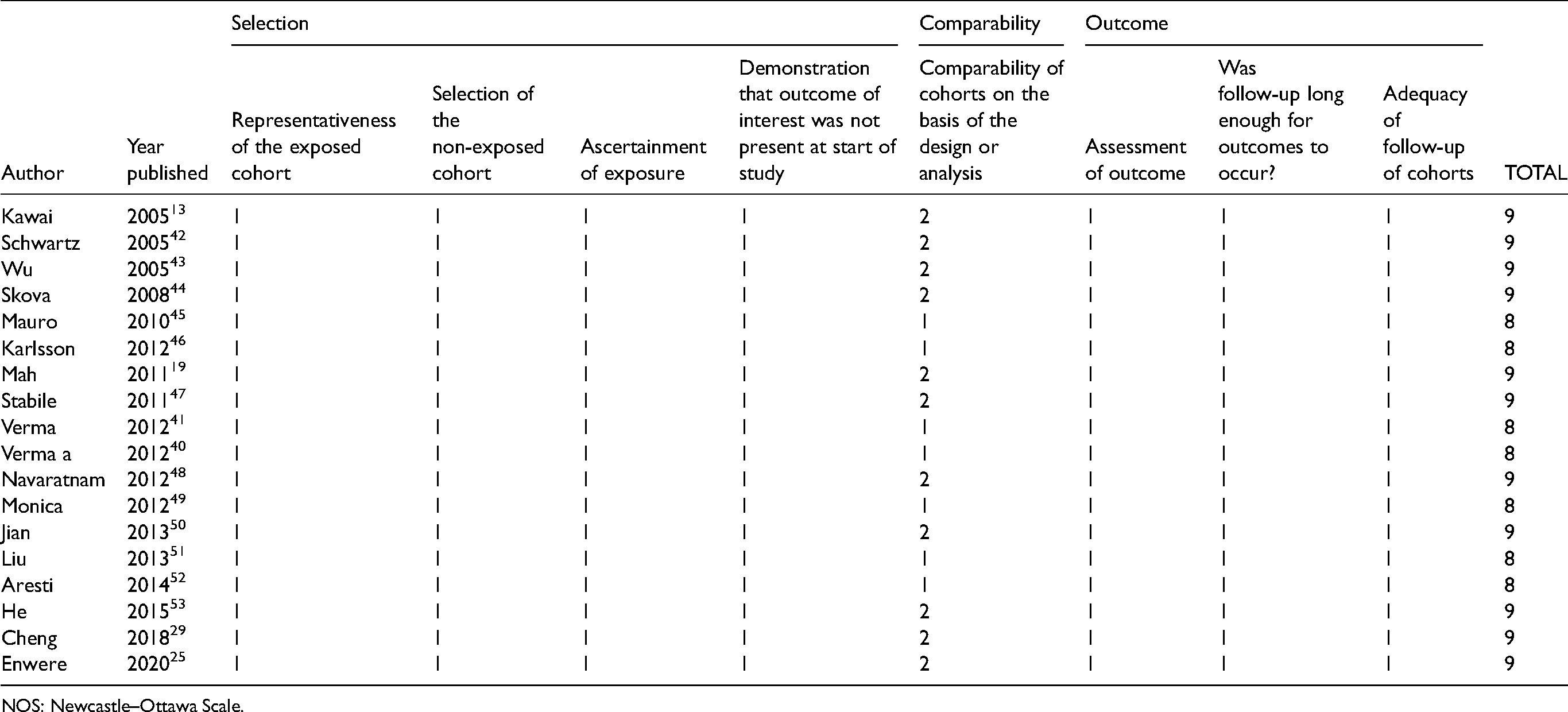
NOS: Newcastle–Ottawa Scale.
Meta-analysis
Of the 18 studies, 14 (including 2238 patients)13,19,40–4345–52 were included in a univariate meta-analysis and 10 (including 2478 patients)13,19,25,29,43,44,47,48,50,53 were included in a multivariate meta-analysis. Among the 10 studies that conducted the multivariate analysis, age, sex, and stage were taken as covariables in 9 studies,13,19,25,29,43,44,47,48,53 and smoking status and pathological type were taken as covariables in 8 studies.13,19,25,29,43,,47,53 Univariate and multivariate analyses showed no significant relationship between ERβ expression and NSCLC OS, with a pooled HR of 0.81 (95% CI: 0.64–1.02, P = 0.07, I2 = 69%, random-effect) by univariate analysis and a pooled HR of 1.06 (95% CI: 0.83–1.36, P = 0.63, I2 = 77%, random-effect) by multivariate analysis. (Figure 2(a) and (b))
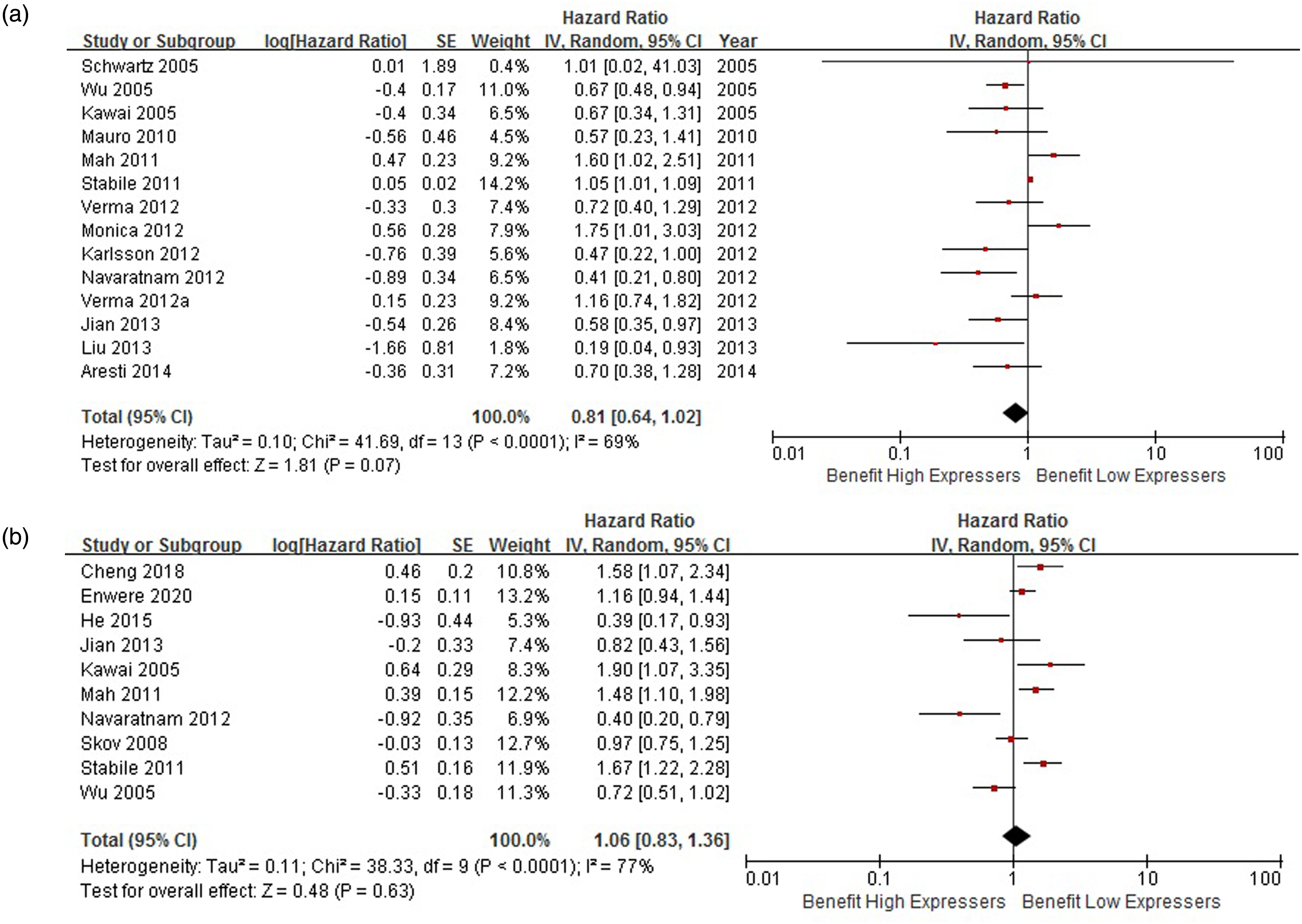
(a) Univariate meta-analysis of 14 studies (2238 patients). (b) Multivariate meta-analysis of 10 studies (2478 patients).
Subgroup analysis
After reading all the included literature, we found that the effect of ERβ on lung cancer prognosis was different among different races or different ERβ distribution locations, so we performed a subgroup analysis.
Asian patients
To assess the factor of race, we conducted a subgroup analysis for Asian patients. Six Asian studies (including 1020 patients) reported univariate analysis results—three from China and three from Japan—and the pooled HR was 0.73 (95% CI: 0.59–0.89, P = 0.002, I2 = 37%, fixed-effect) (Figure 3(a)).
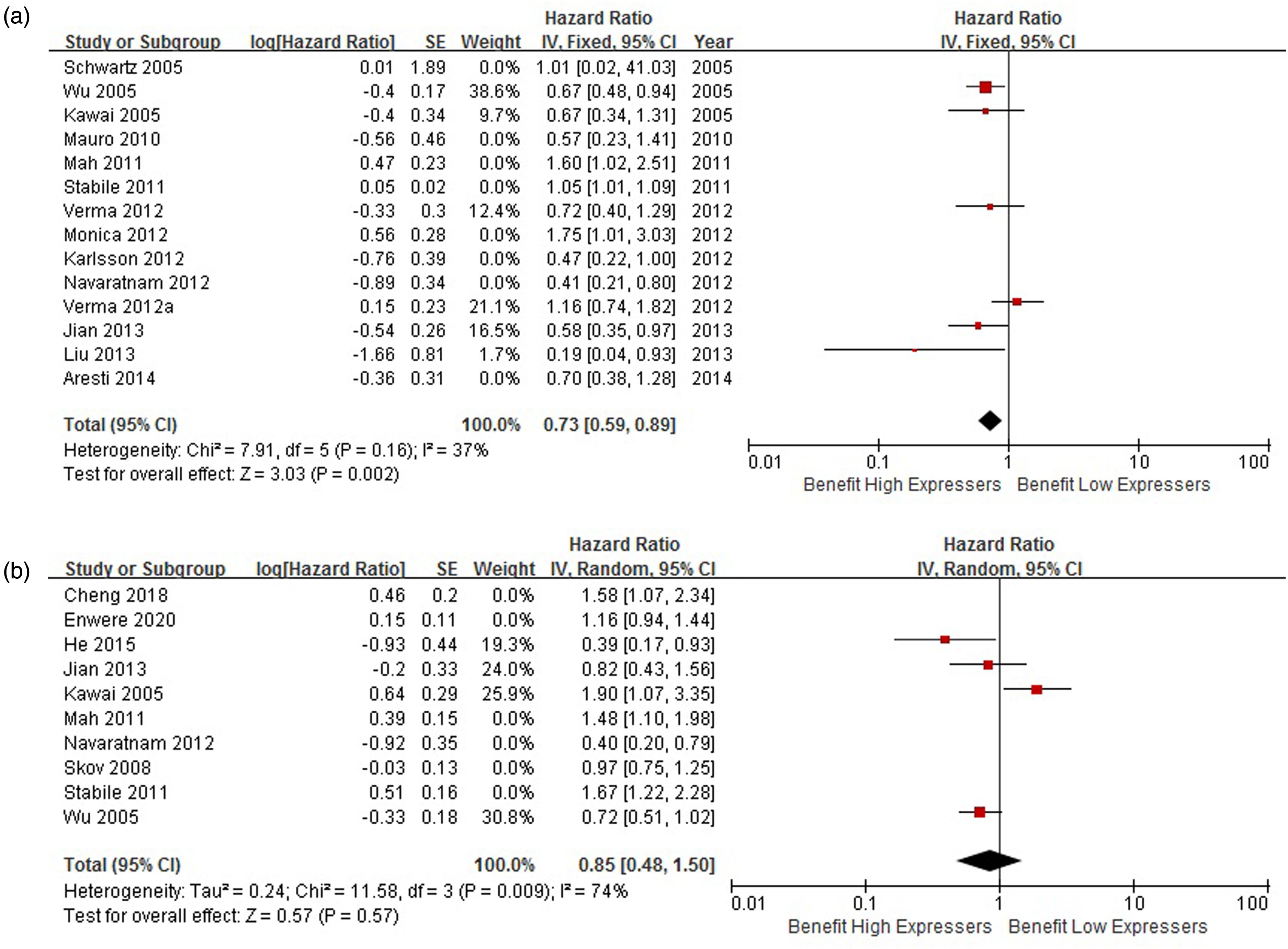
(a) Subgroup analysis for Asian patients (six studies including 1020 patients for univariate analysis). (b) Subgroup analysis for Asian patients (four studies including 623 patients for multivariate analysis).
Four Asian studies (including 623 patients) reported multivariate analysis results—three from China and one from Japan—and the pooled HR was 0.85 (95% CI: 0.48–1.50, P = 0.57, I2 = 74%, random-effect) (Figure 3(b)). There was no significant result for studies out of Asia.
Nuclear ERβ
To assess the factor of ERβ location, we conducted a subgroup analysis. 7 studies (including 1161 patients) conducted a univariate analysis for the effect of nuclear ERβ on lung cancer prognosis, and the pooled HR was 0.75 (95% CI: 0.61–0.93, P = 0.009, I2 = 46%, fixed-effect) (Figure 4(a)). Five studies (including 980 patients) conducted a multivariate analysis for the effect of nuclear ERβ on lung cancer prognosis, and the pooled HR was 1.03 (95%CI: 0.80—1.32, P = 0.84, I2 = 61%, random-effect) (Figure 4(b)). There were no significant results for cytoplasmic ERβ.
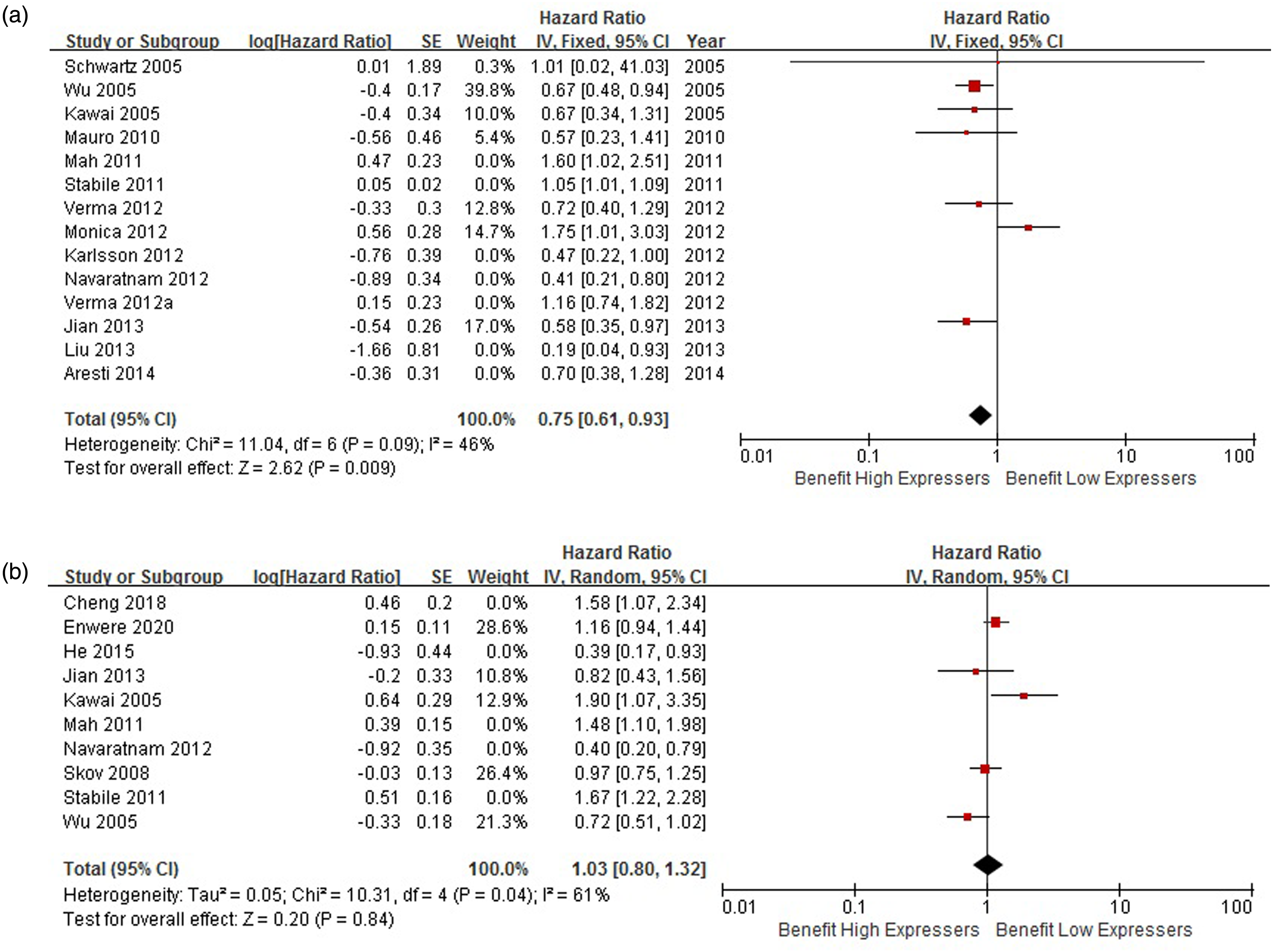
(a) Subgroup analysis for nuclear ERβ (seven studies including 1161 patients for univariate analysis). (b) Subgroup analysis for nuclear ERβ (five studies including 980 patients for multivariate analysis).
Publication bias
The 14 studies for the univariate meta-analysis yielded the funnel plot in Figure 5(a) and an Egger's test score of P = 0.138, while the 10 studies for the multivariate meta-analysis yielded the funnel plot in Figure 5(b) and an Egger's test score of P = 0.202. Publication bias was within acceptable limits.
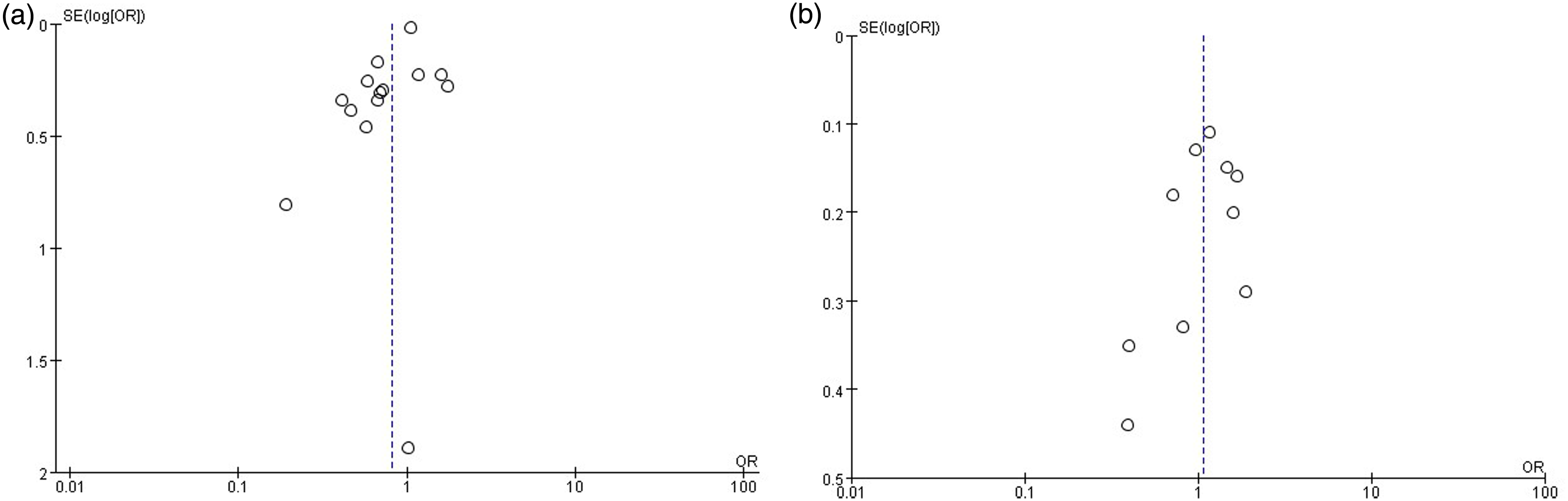
(a) Funnel plot of 14 studies included in univariate analysis. (b) Funnel plot of 10 studies included in multivariate analysis.
Sensitivity analysis
Sensitivity analysis was performed for the 18 included studies to investigate the effect of every study on the overall meta-analysis by omitting one study each time. The omission of any study made no significant difference, and I2 was greater than 60%.
Discussion
We performed extensive literature searches in 15 databases from January 1, 2000 to May 1, 2021, and included 18 cohort studies incorporating 3500 patients to evaluate the relationship between ERβ expression and NSCLC OS. ERβ expression failed to yield a significant association with NSCLC OS by univariate analysis or multivariate analysis. Then we conducted a subgroup analysis of Asians, and the pooled HR of univariate analysis was 0.73 (P = 0.002). Inspired by Verma et al.,40,41 who obtained different results from his experiment on nuclear ERβ and cytoplasmic ERβ, we further analyzed the subgroup of nuclear ERβ, and the pooled HR of univariate analysis was 0.75 (P = 0.009). However, in multivariate analyses, neither ethnicity nor ERβ position was an important independent factor affecting the OS of NSCLC. Our conclusion was in accordance with Ma et al., 27 who suggested ERβ that overexpression indicated no relationship of prognosis for patients with NSCLC. We included seven more studies than Ma et al. and performed both univariate and multivariate analyses to estimate the HR and 95% CI, while Ma et al. 27 only performed univariate analyses.
Monica et al. 49 suggested ERβ expression was significantly higher in patients with metastatic disease compared with all other disease stages (P = 0.02). Enwere et al. 25 also claimed that higher ERβ expression levels in stage IV disease were apparent in the tumor as well as the surrounding stroma, and in both nuclei and cytoplasmic compartments. However, most studies—such as Schwartz et al. 42 and Skov et al. 44 —reported that there was no statistically significant correlation between ERβ positivity and age, gender, stage, or histology. Researchers have conflicting conclusions on whether tumor stage has an effect on the prognosis of NSCLC, and the literature was insufficient for meta-analysis.
Whether the effect of ERβ expression level on NSCLC prognosis is different for male and female patients remains controversial. Schwartz et al. 42 and Skov et al. 44 found men with ERβ-positive tumors had a significantly reduced mortality compared to men with ERβ-negative tumors, while women with ERβ-negative tumors had a non-significant change in mortality compared with women with Erβ-positive tumors. However, other researchers—such as Enwere et al. 25 —found no differences in ERβ expression between male and female patients. Unfortunately, there were too little data in the literature to draw definitive conclusions.
The expression of ERβ may be closely related to some clinical characteristics, such as gender, stage, race, etc. More observational clinical studies are needed to confirm these possibly-related factors, to provide accurate prognostic prediction for patients with NSCLC, and to then explore corresponding treatment methods.
There were some limitations to our study. First, the presence of heterogeneity could be explained to a great extent by the different population selected, such as sex ratio, age composition, tumor stage, and histological subtypes, which were different in the 18 selected studies. Second, we searched only English and Chinese literature, and thus introduced somewhat of a selection bias. Third, reporting bias existed. For example, Monica et al. 49 reported only significant results for men but not for women, and we didn't have access to the raw data. Another potential source of bias was related to the method used to extrapolate the HR, which was extracted from the data included in the article directly or calculated from the survival curves. Finally, the heterogeneity could be explained by the differences in the laboratory tests of ERβ, such as the different antibodies used for staining and the different criteria for defining stain positivity.
Conclusion
Our results suggested that the expression of ERβ might not be a direct prognostic factor for NSCLC patients. Although high ERβ expression in nuclear and Asian subgroups was significantly associated with improved OS in univariate analysis, it was not in multivariate analysis. More detailed prospective studies are needed to identify direct prognostic factors in NSCLC patients.
Footnotes
Acknowledgments
Not applicable.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Authors’ contributions
Project development: Yuguo Liu and Hui Li. Data collection or management: Haisheng Chen, Jing Shi, and Qing Fan. Data analysis: Hui Li, Zhongxia Zhou, Xiufeng Tang. Manuscript writing/editing: Zhongxia Zhou, Xiufeng Tang, and Yanhong Wang.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China, (grant number 82104051).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
