Abstract
BACKGROUND:
Secreted protein acidic and rich in cysteine (SPARC) is implicated in cancer progression, but its role and associated molecular mechanism in the sorafenib sensitivity of hepatocellular carcinoma cells (HCC) remains elusive.
METHODS:
Human HCC cell lines Hep3B and HepG2 were treated with sorafenib alone or combined with activator or inhibitor of ferroptosis. Cell viability assay, reactive oxygen species (ROS) assay, lactate dehydrogenase (LDH) assay and western blot were used to study the regulatory mechanism of SPARC on HCC cells.
RESULTS:
Overexpression of SPARC enhanced the cytotoxic effect of sorafenib in Hep3B and HepG2 cells compared with parental cells. Depletion of SPARC decreased the cytotoxic effect of sorafenib in Hep3B and HepG2 cells compared with parental cells. Moreover, overexpression of SPARC significantly induced LDH release, whereas depletion of SPARC suppressed the release of LDH in Hep3B and HepG2 cells. Inhibition of ferroptosis exerted a clear inhibitory role against LDH release, whereas activation of ferroptosis promoted the release of LDH in HCC cells, as accompanied with deregulated expression of ferroptosis-related proteins. Furthermore, overexpression of SPARC induced oxidative stress, whereas depletion of SPARC suppressed the production of ROS. Deferoxamine (DFX)-induced inhibition of ferroptosis suppressed the production of ROS, while activation of ferroptosis promoted the contents of ROS in HCC cells exposed to sorafenib.
CONCLUSION:
Our findings give a better understanding of ferroptosis and its molecular mechanism in HCC cells that is regulated by SPARC in response to sorafenib.
Introduction
Hepatocellular carcinoma (HCC) is the most frequent malignancy of primary liver cancer, and causes great health and economic concerns around the world [1]. Sorafenib, a multi-kinase inhibitor, is currently approved for advanced HCC with a proven efficacy in terms of overall survival [2]. However, long-term therapeutic efficacy of sorafenib usually cannot be achieved in patients with HCC. Although HCC presents great heterogeneity in individual sensitivity to sorafenib [3, 4], there are currently no biological assays that can predict its efficacy in individual patients. It is critical to identify the biological alterations that are associated with an optimal efficacy of sorafenib in HCC [5].
Secreted protein acidic and rich in cysteine (SPARC), a calcium-binding matricellular glycoprotein, regulates cell adhesion, migration, tissue repair and remodeling. Growing evidence have shown that SPARC is implicated in the progression of many cancers. SPARC is considered as an oncogene and up-regulated levels of SPARC expression have been reported in prostate cancer, colorectal cancer, breast cancer, and hepatocellular carcinoma [6, 7, 8]. However, an opposite correlation regarding to the SPARC expression levels in tumors has also been demonstrated in hepatocellular carcinoma, prostate cancer, and ovarian cancer [9, 10, 11]. Additionally, the correlation between SPARC and anti-cancer drug sensitivity has been reported. For example, SPARC has been considered as a putative resistance-reversal gene and overexpression of SPARC in tumor xenografts restored their sensitivity to 5-fluorouracil (5-FU) in colorectal cancer and HCC [12, 13]. Knockdown of SPARC promoted the 5-FU chemoresistance and increased tumor growth in gastric cancer [14]. Moreover, SPARC has been reported to act as a regulator of imatinib sensitivity in Chronic myelogenous leukemia (CML) cells [15]. However, it remains unknown whether SPARC could regulate the sensitivity of sorafenib in human liver cancer cells.
Induction of apoptosis has been remarkably elevated in HCC cells exposed to sorafenib
In the present study, we reported a novel function of SPARC on the regulation of ferroptosis in HCC. Up-regulation of SPARC sensitized sorafenib and promoted sorafenib-induced ferroptosis as elevated LDH release and ROS production in HCC cells. These results proposed a novel function of SPARC in the regulation of ferroptosis and provided additional treatment option for the treatments of patients.
Materials and methods
Cell culture
Hep3B and HepG2 human hepatocellular carcinoma cells were purchased from American Type Culture Condition (ATCC, Rockville, MD, USA) and were grown in Dulbecco’s Modified Eagles Medium (DMEM, Gibco, Grand Island, NY, USA) supplemented with 10% v/v fetal bovine serum (FBS), 100 U/mL penicillin and 100
Cell viability assay
Cell Counting Kit-8 (CCK-8) assay was performed for determining cell viability, using the CCK-8 assay kit (Sigma, St. Louis, MO, USA) according to the manufacturer’s protocol. In brief, cells were seeded into 96-well plates at a density of 0.5
LDH activity assay
The total LDH activity in cell lysates was examined according to the manufacturer’s instructions of the LDH cytotoxicity assay kit (BioVision, Tucson, AZ, USA). The assay is conducted based on the enzymatic coupling reaction in which LDH reduces NAD to NADH, which then reacts with a specific probe to generate a color. Briefly, 2
ROS production
Cells were incubated with chloro-methyl-dichlorofl- uorescein diacetate (CM-DCFDA) (Molecular Probes, Eugene, OR, USA). Samples were then centrifuged at 1000 g for 3 min, and the pellets were resuspended in 500
Western blot analysis
Whole cells were lysed in lysing buffer containing protease inhibitor (Roche, Mannheim, Germany). Protein samples (20
Statistical analysis
All data were shown as mean
Results
SPARC regulates the cytotoxic effect of sorafenib in HCC cells
As shown in Fig. 1A and B, expression of SPARC was significantly increased by about three folds in Hep3B and HepG2 cells transfected with vectors encoding SPARC. Meanwhile, we successfully depleted SPARC in Hep3B cells transfected with siRNA-SPARC (Fig. 1A and B). Following treatment with sorafenib, the viability of parental Hep3B and HepG2 cells gradually decreased in a dose dependent manner (Fig. 2A and B). In order to investigate the role of SPARC on chemosensitivity, SPARC-overexpressed or silenced-Hep3B and HepG2 cells were exposed to sorafenib at 16
Manipulation of SPARC expression in HCC cells. Hep3B and HepG2 cells were transfected with vectors encoding SPARC or with siRNA-SPARC. Forty-eight hours later, the cells were harvested and the expression of SPARC in Hep3B (A) and HepG2 (B) cells was determined by western blot. The results are based on the analysis of three independent experiments. 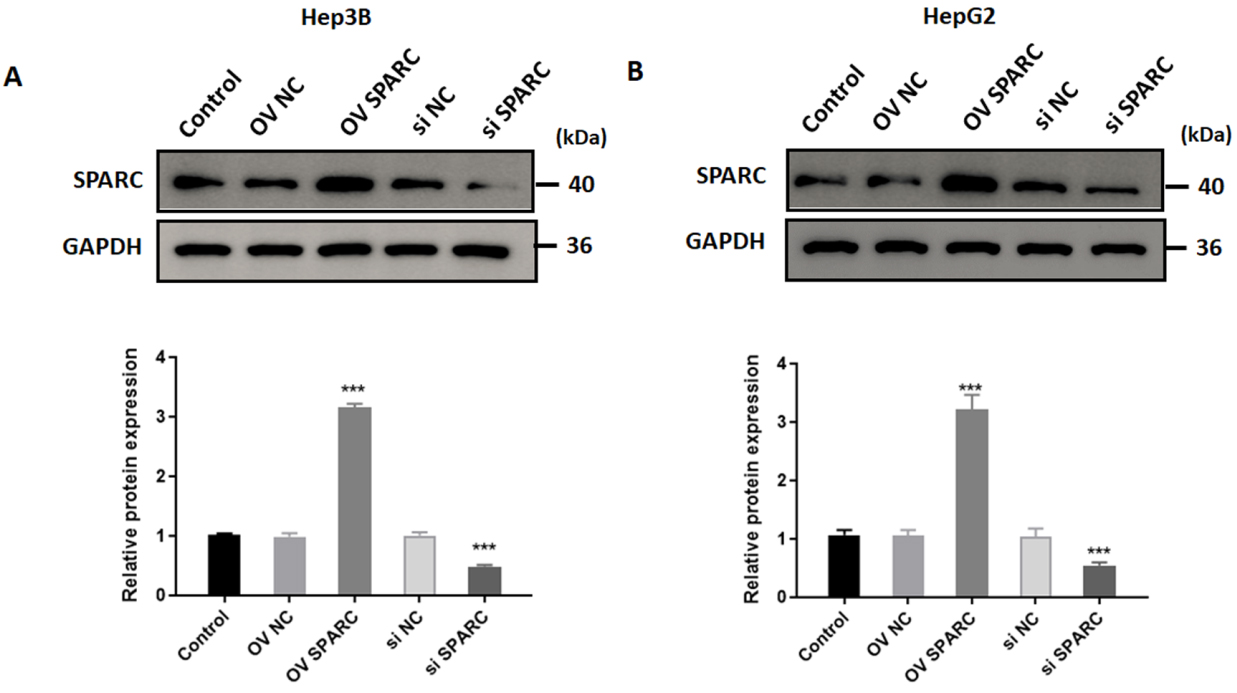
Effects of SPARC on cell viability in HCC cells in response to sorafenib. Hep3B and HepG2 cells were treated with sorafenib at different doses (0, 8, 16, and 32 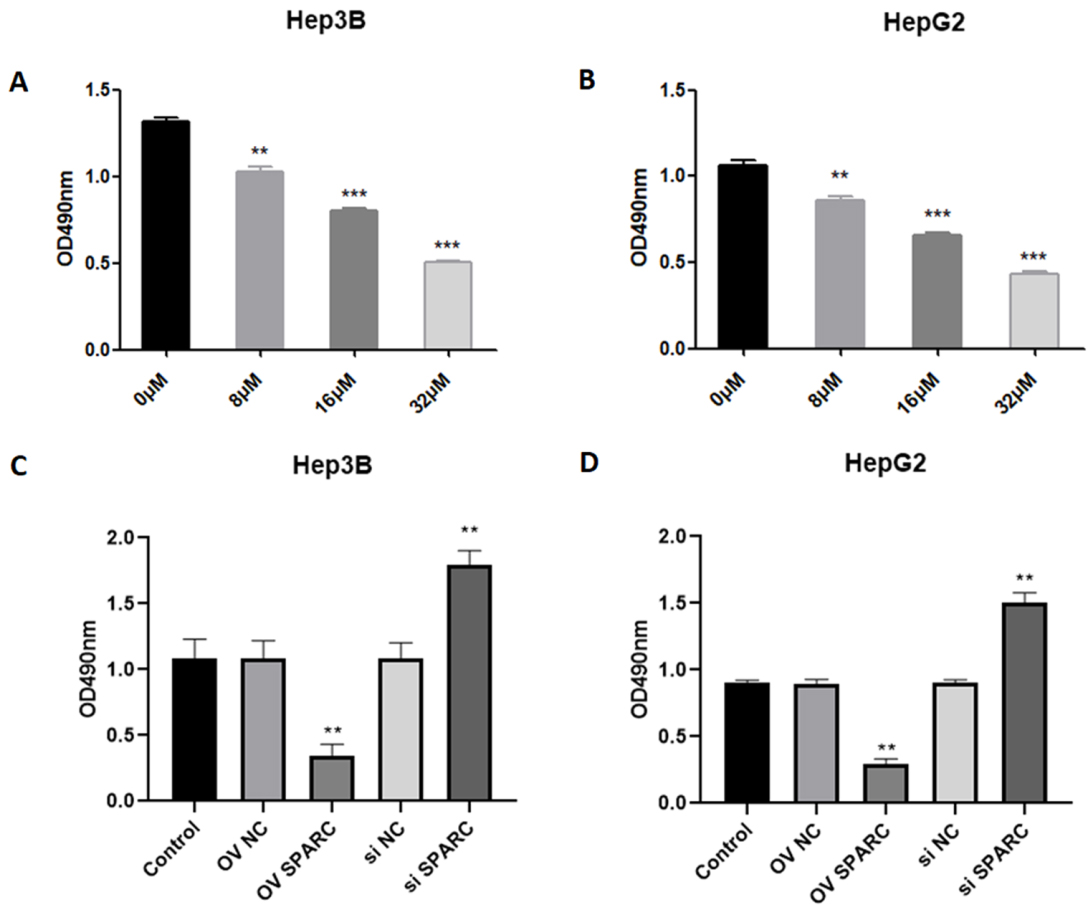
Effects of SPARC on ferroptosis in HCC cells in response to sorafenib. Hep3B and HepG2 cells were transfected with vectors encoding SPARC or with siRNA-SPARC. Forty-eight hours later, the cells were harvested and the LDH release was determined in Hep3B (A) and HepG2 (B) cells. Moreover, Hep3B and HepG2 cells were treated with sorafenib alone, or in combination with DFX or Erastin. Forty-eight hours later, the LDH release was determined in Hep3B (C) and HepG2 (D). Moreover, western blot was used to detect the expression of ferroptosis-related proteins in Hep3B (E) and HepG2 (F), including long-chain acyl-CoA synthetase 4 (ACST4), prostaglandin-endoperoxide synthase 2 (PTGS2), NADPH oxidase 1 (NOX1), and glutathione peroxidase 4 (GPX4). The results are based on the analysis of three independent experiments. 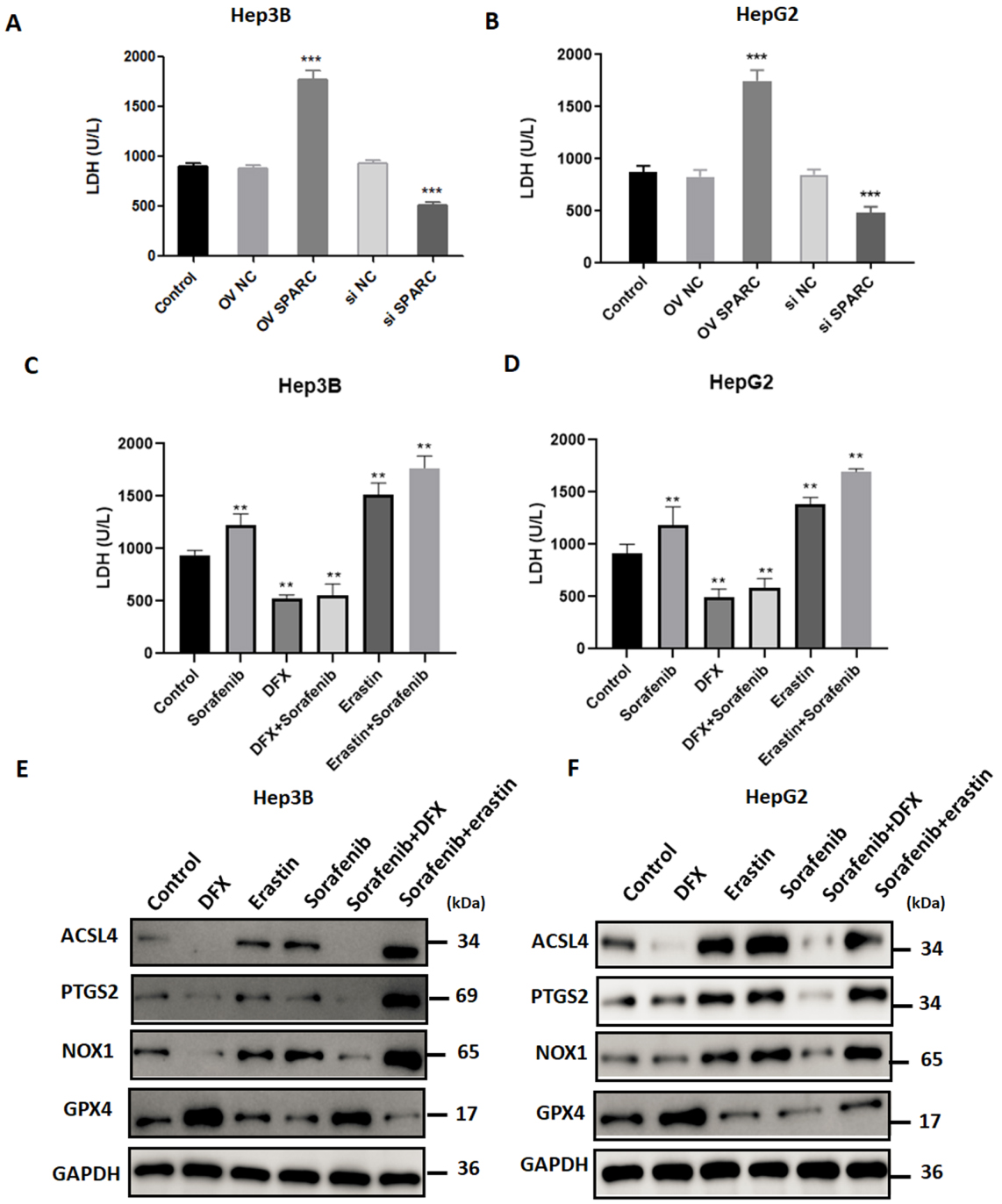
To explore the role of SPARC on ferroptosis, we measured cell membrane permeability by the lactate dehydrogenase (LDH) release assay. Interestingly, we found that overexpression of SPARC significantly induced LDH release in Hep3B (1772.2
Effects of SPARC on oxidative stress in HCC cells exposed to sorafenib. SPARC-overexpressed or silenced-Hep3B and HepG2 cells were treated with sorafenib at 16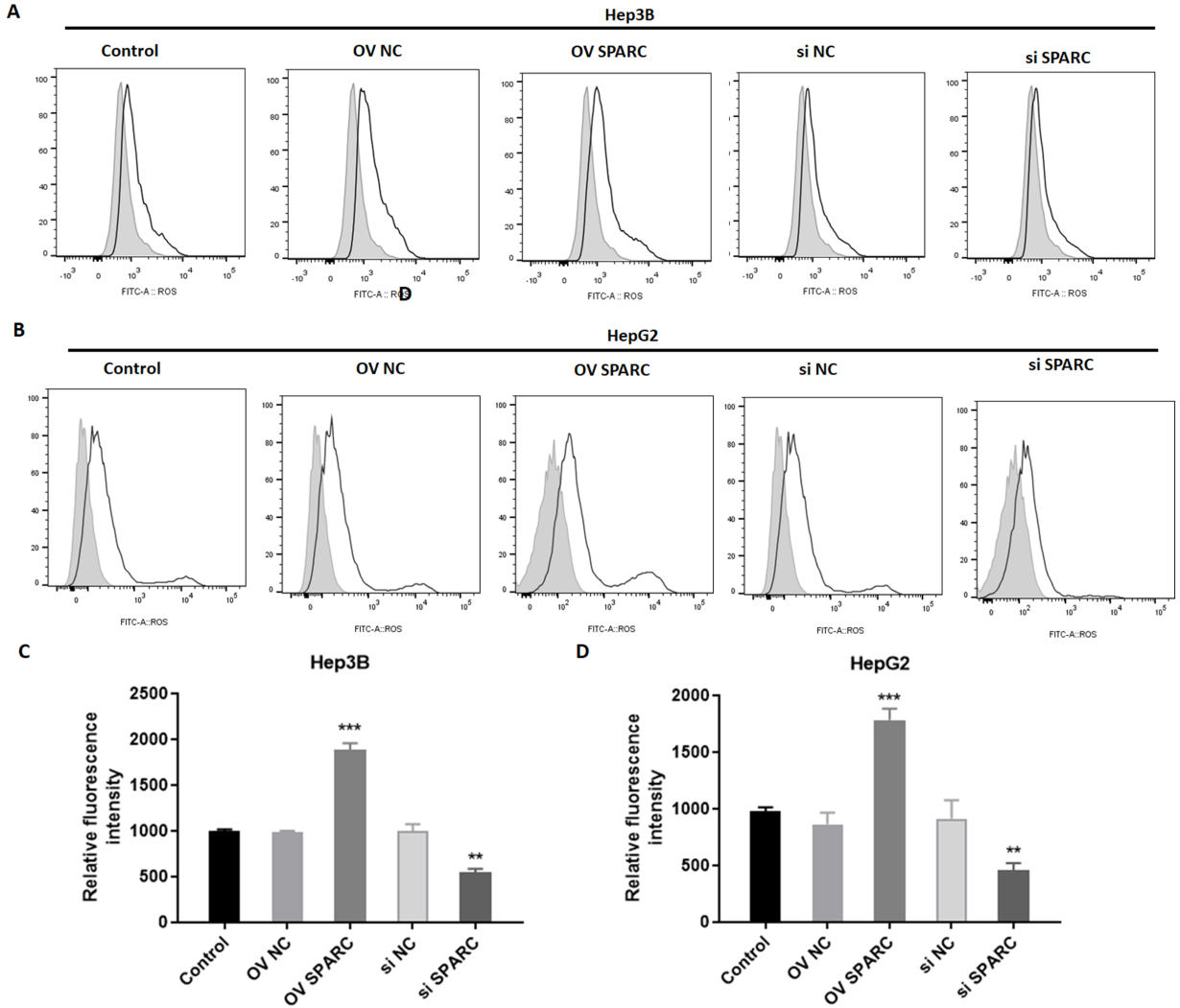
Changes of oxidative stress in HCC cells exposed to sorafenib. Hep3B and HepG2 cells were treated with sorafenib alone, or in combination with DFX or Erastin. Forty-eight hours later, the ROS production was determined in Hep3B (A and B) and HepG2 (C and D) cells. The results are based on the analysis of three independent experiments. 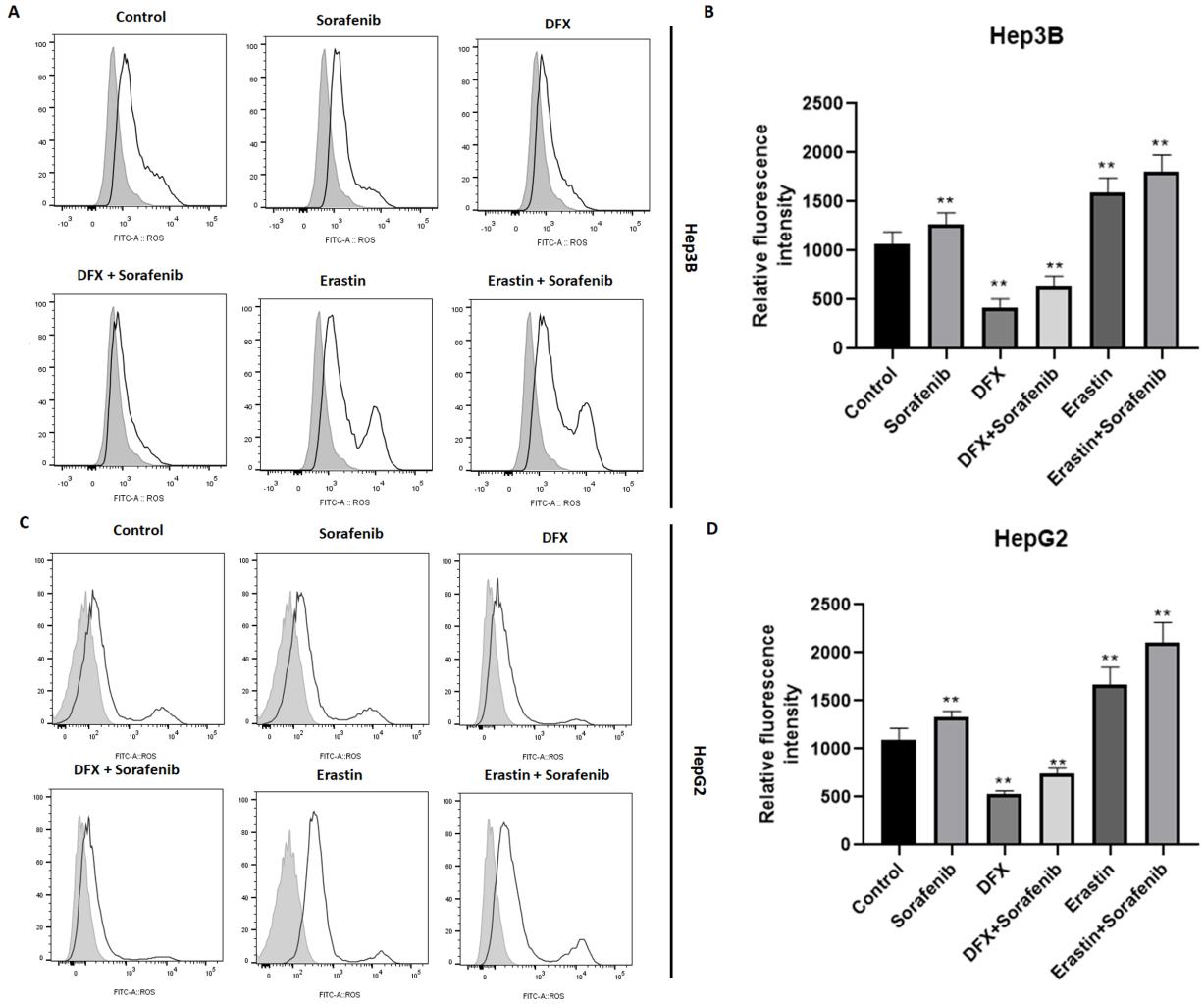
It is known that ferroptosis is characterized by the occurrence of intense oxidative stress. In order to detect the role of SPARC on oxidative stress, the probe CM-DCFDA was used, whose fluorescence intensity reflects the production of reactive oxygen species (ROS). As a result, overexpression of SPARC induced oxidative stress in Hep3B (1887.3
Discussion
Although SPARC has been widely studied in different human disease, the specific role and molecular mechanism of SPARC in HCC is unclear [7]. Previous investigations demonstrate the association of SPARC with aggressive tumor phenotype (i.e., gliomas, melanoma, breast, gastric cancers) or with anti-tumor activity (i.e., ovarian, colorectal, liver, neuroblastoma) [6, 7, 8, 9, 10, 11]. Our previous study found that SPARC appears to function as a tumor suppressor in HCC: SPARC was down regulated in 5-FU resistant cells and overexpression of SPARC facilitated the re-sensitization of 5-FU resistance [8]. In the present study, we examined the impact of the SPARC of HCC cells on their response to sorafenib. We found that the overexpression of SPARC remarkably increased the induction of ferroptosis, a form of necrosis recently reported to be induced by sorafenib.
Currently, most studies have focused on apoptosis of cancer cells induced by anticancer agents [5, 19, 20]. Because the anti-apoptotic mechanisms usually render cancer cells resistant to therapeutic drugs, the induction of new forms of cell death is necessary for development of novel therapies against solid tumors [21, 22]. Since the concept of ferroptosis was first reported in 2012, accumulating evidence suggest that this form of regulated cell death is closely associated with human malignancies [23, 24]. For instance, sorafenib, the first drug used for advanced HCC, is able to induce ferroptosis in HCC cells [16]. Our study suggests that SPARC could enhance the cytotoxic effect of sorafenib in HCC cells. Moreover, SPARC significantly induced LDH release and promoted ferroptosis accompanied with deregulated expression of ferroptosis-related proteins, such as elevated expression of COX2. These finding suggest that SPARC is a parameter that regulates the susceptibility of tumor cells to ferroptosis induced by sorafenib.
How sorafenib promotes oxidative stress and ferroptosis is not fully elucidated [25]. We found that overexpression of SPARC induced oxidative stress, whereas depletion of SPARC suppressed the production of ROS. This role of mitochondrial production of ROS is reported by two previous studies, suggesting the mitochondrial respiratory chain as a potential vulnerability in HCC cells exposed to sorafenib [26, 27]. Our findings showed that DFX-induced inhibition of ferroptosis suppressed the production of ROS, while activation of ferroptosis promoted the contents of ROS in HCC cells exposed to sorafenib.
In conclusion, the current study provides the first evidence that SPARC plays a critical role in sorafenib-induced ferroptosis. Suppression of SPARC restored sorafenib sensitivity and diminished ROS levels. Future studies on the role of SPARC in ferroptosis may provide novel treatment options that is based on ferroptosis-mediated therapeutic strategies for human cancer.
Footnotes
Conflict of interest
The authors in this study declare no conflicts of interest.
Funding
This paper was supported by: The Fundamental Research Fund of Xinhua Hospital in 2017 (XH1926), The Medical Interdisciplinary Research Fund of Shanghai Jiaotong University in 2017 (YG2017QN64); The Project of Shanghai Municipal Health and Family Planning Commission in 2018 (201840131); Projects of Shanghai Municipal Health and Family Planning Commission in 2019 (201940403). Training plan of leading medical talents in Shanghai in 2019 (2019LJ28).
