Abstract
Hepatocellular carcinoma (HCC) is one of the most severe diseases worldwide. For the different stages of HCC, there are different clinical treatment strategies, such as surgical therapy for the early stage, and transarterial chemoembolization (TACE) and selective internal radiation therapy (SIRT) for intermediate-stage disease. Systemic treatment, which uses mainly targeted drugs, is the standard therapy against advanced HCC. Sorafenib is an important first-line therapy for advanced HCC. As a classically effective drug, sorafenib can increase overall survival markedly. However, it still has room for improvement because of the heterogeneity of HCC and acquired resistance. Scientists have reported the acquired sorafenib resistance is associated with the anomalous expression of certain genes, most of which are also related with HCC onset and development. Combining sorafenib with inhibitors targeting these genes may be an effective treatment. Combined treatment may not only overcome drug resistance, but also inhibit the expression of carcinoma-related genes. This review focuses on the current status of sorafenib in advanced HCC, summarizes the inhibitors that can combine with sorafenib in the treatment against HCC, and provides the rationale for clinical trials of sorafenib in combination with other inhibitors in HCC. The era of sorafenib in the treatment of HCC is far from over, as long as we find better methods of medication.
Background
Humans have not yet overcome cancer, which is the second most common disease causing death worldwide. Liver cancer is the sixth most common malignancy among all types of cancer. Liver cancer with a 5-year survival of 18% is the second most lethal tumor after pancreatic cancer. In 2018, there were 841,080 new liver cancer cases and 781,631 deaths. WHO estimates that more than 1 million patients will die from liver cancer in 2030. The most common type of liver cancer is hepatocellular carcinoma (HCC). In 2012, 782,000 patients were diagnosed with HCC and 746,000 patients died from it.1–4
For patients with early stage HCC [Barcelona Clinic Liver Cancer (BCLC) stage 0/A], surgical therapy is the main choice. Usually, resection of tumors can result in more than 60% survival at 5 years. Some patients who are not candidates for resection because of tumor location or other reasons can also choose liver transplantation if they have a limited tumor burden. Ablation is another treatment for patients with BCLC stage 0/A disease. For patients with intermediate-stage (BCLC stage B), transarterial chemoembolization (TACE) and selective internal radiation therapy (SIRT) are the main treatment methods. For the patients who have advanced HCC (BCLC stage C), whose tumors cannot be resected, systemic therapies are recommended. Systemic therapies are based on targeted drugs.1,4,5
Sorafenib is the first United Stated Food and Drug Administration (US FDA)-approved first-line systemic therapy, and is also the standard therapeutic agent for advanced HCC. In fact, until lenvatinib was approved as a frontline therapy in 2018, sorafenib was the only first-line therapy in the last 10 years. It is a multi-kinase inhibitor targeting vascular endothelial growth factor receptor (VEGFR) and platelet-derived growth factor receptor (PDGFR), and is also an activator of AMP-activated protein kinase (AMPK), which can suppress tumors.
6
Sorafenib brings an obvious survival benefit to patients with HCC. According to two clinical trials, sorafenib could improve overall survival (OS) significantly compared with placebo (10.7 months
However, there are several factors that prevent more patients benefitting from sorafenib (Figure 1). Only about 40% of patients with HCC can benefit from sorafenib because of the genetic heterogeneity of HCC and other reasons. Some studies showed that sorafenib had more benefits for specific groups of patients. The two clinical trials above involved strictly selected patients, that is, patients with well-preserved liver function, and these patients were named SHARP eligible patients (named after the original SHARP clinical trial). A retrospective study showed sorafenib could only benefit SHARP eligible patients.
8
Besides, the benefit from sorafenib is higher in patients with hepatitis C patients than in others.
9
However, other groups are resistant to sorafenib from the beginning. This phenomenon is also named primary resistance, whose mechanism is still unknown. However, some studies have revealed possible reasons. Gene polymorphism may be a key factor that can influence the function of sorafenib. Scientists have showed that polymorphisms of ATP binding cassette (ABC) subfamily B member 1 (

Factors preventing more patients benefitting from sorafenib. To date, about six factors have been identified to interfere with the effect of sorafenib. Economic burden, acquired resistance, genetic heterogeneity, and adverse reaction are widely accepted factors. The liver is the main metabolic site of sorafenib; therefore, the status of the liver can also influence the effect of sorafenib. Sorafenib cannot kill cancer stem cells effectively; therefore, the existence of cancer stemness is another important factor.
Apart from the low response rate, another common problem of sorafenib is the acquired resistance of HCC cells, and patients who are sensitive to sorafenib at the beginning usually develop resistance within 6 months.6,15 These shortcomings, plus the emergence of new drugs, have made scientists propose that the era of sorafenib is over. These weaknesses of sorafenib have prompted many researchers to find novel and effective methods to treat HCC using sorafenib. One important solution is to identify the genetic changes before and after sorafenib resistance, and then use drugs targeting these molecules. Scientists have shown that several pathways, such as glycolysis and autophagy, are related to resistance to sorafenib.16,17 Meanwhile, most targets that are related to resistance are also associated with HCC development. Thus, combinations of sorafenib and other drugs might play synergistic roles, which represent a novel strategy against HCC. This review focuses on the combinations of sorafenib with other inhibitors to treat HCC
Glycolysis-related HCC development and sorafenib resistance
Glycolysis is the main source of energy for cancer cells. In normal cells, the energy source is glucose oxidative phosphorylation (OXPHOS). 18 OXPHOS involves slower ATP production compared with glycolysis. Therefore, glycolysis can support the faster growth of more tumor cells. 19 This phenomenon, named the Warburg effect, was reported to be related closely to cell proliferation and drug resistance. 16 Sorafenib can inhibit angiogenesis, which will induce hypoxia and glycolysis. Therefore, combinations of sorafenib and glycolytic inhibitors could significantly reduce sorafenib resistance, suppress cell reproduction, and improve the effect of killing HCC cells. This part mainly summarizes studies of the combinations of sorafenib and glycolytic inhibitors to treat HCC.
Glycolysis inhibitors that function by activating AMPK
As the main source of energy in cancer cells, glycolysis produces ATP and promotes the growth and reproduction of tumor cells. Suppressing this metabolic progress can slow down the growth rate of tumor cells. Besides, a central metabolic switch, AMPK, is also activated by an increased AMP/ATP ratio, which can be induced by the absorption of glycolytic inhibitors. AMPK can promote catabolic pathways and inhibit cell proliferation. Meanwhile, AMPK can also inhibit the function of mammalian target of rapamycin (mTOR), which is also closely related to drug resistance. 20
Combining sorafenib with the glycolytic inhibitor 2-deoxyglucose (2DG) could drastically inhibit the viability of HCC cells, including sorafenib-sensitive and -resistant cells. The mechanism is that the combination of the two drugs inhibits ATP production, and then activates AMPK, which inhibits mTOR, and finally suppresses the cell cycle.
21
Tomizawa
Glycolysis inhibitors that function by inhibiting HIFs
Hypoxia-inducible factors (HIFs) including HIF-1α and HIF-2α are important transcription factors that regulate the induction of several enzymes involved in glycolysis. In cancer cells, increased HIFs and glycolysis activities are observed, which help tumors grow.24,25 As important regulators of glycolysis, HIFs can be ideal targets in the glycolysis-related treatment against HCC. Meanwhile, apart from glycolysis, HIFs are also involved in sorafenib resistance through multiple other downstream factors. Some autophagy processes in HCC cells depend on the stabilization of HIFs. Autophagy was widely reported as being able to promote tumor growth and drug resistance.
26
HIFs can also stabilize multidrug resistance protein 1 (MDR1), glucose transporter 1 (GLUT-1), galectin-1, and other drug-resistance-associated proteins and induce sorafenib resistance.27–30 Besides, some upstream factors of HIFs can regulate the expression of HIFs and induce sorafenib resistance. Qiu
Li
OXPHOS activators
Notably, most current research has focused on inhibitors targeting glycolysis rather than activators targeting OXPHOS. Shen
Other glycolysis inhibitors
There are also some inhibitors can target glycolysis directly. Sorafenib combined with the β-catenin inhibitor FH535 could simultaneously suppress mitochondrial respiration and glycolytic flux, which plays an important role in cell proliferation and apoptosis. 40 Using resveratrol can reduce the expression of hexokinase 2 (HK2), which plays a key role in glycolysis. Thus, combining resveratrol with sorafenib could potentially target sorafenib-resistant HCC cells. 41
Summary and discussion
Changes to the metabolic pathways in tumor cells have been recognized as a feature of malignancies, and the Warburg effect is representative of these changes. Glycolysis, which can be activated using sorafenib, is closely related to hepatocarcinogenesis and drug resistance. Most research attempting to inhibit glycolysis showed the feasibility of combining sorafenib and glycolysis inhibitors. However, little research has investigated restoring OXPHOS in tumor cells. Theoretically, targeting OXPHOS is another strategy to solve the problems with sorafenib. OXPHOS depends on mitochondrial function, and activation of OXPHOS also switches on the mitochondria-mediated apoptosis pathway, whereas mitochondrial inhibition induces cell proliferation and cyclin D1 expression.
42
Therefore, targeting both OXPHOS and glycolysis is expected to be a reliable way to treat HCC and ameliorate sorafenib resistance. In addition to DCA, Sun
PI3K/AKT/mTOR-related HCC development and sorafenib resistance
The phosphatidylinositol 3-kinase (PI3K)/protein kinase B (AKT)/mTOR pathway is an important metabolic pathway in cells. It is related closely to cell proliferation, cell growth, protein translation, and other important life processes in organisms. This pathway is overactivated in 30–50% of patients with HCC, and is associated with the occurrence and development of HCC.44–46 Inhibiting the PI3K/AKT/mTOR pathway is a vital anti-cancer therapy, and inhibitors targeting the pathway have been confirmed as an effective way to treat HCC. 47
Sorafenib has been reported to suppress cell proliferation and cell growth by inhibiting the PI3K/AKT/mTOR pathway; however, the inhibition of this pathway is not dominant in sorafenib therapy.48,49 Interestingly, research has shown that this pathway is reactivated in sorafenib-resistant cells.50–52 Simultaneously, sorafenib can downregulate mTOR, and then induce autophagy. Autophagy is also a potential cause of resistance to sorafenib. 17 Thus, the PI3K/AKT/mTOR pathway is instrumental in HCC and sorafenib resistance. Combinations of sorafenib with inhibitors targeting this pathway are potential treatments for HCC (Figure 2).

Theoretical research and clinical application of PI3K/AKT/mTOR inhibitors and sorafenib. The PI3K/AKT/mTOR signal pathway plays a key role in the occurrence and development of HCC, and resistance to sorafenib. Combinations of PI3K/AKT/mTOR inhibitors and sorafenib showed satisfactory results in the treatment of HCC. The two inhibitors above plus 3-MA (an autophagy inhibitor) demonstrated a better treatment effect. As a type of immune inhibitor, mTOR inhibitors combined with sorafenib may also prevent post-transplant HCC.
PI3K/AKT/mTOR pathway inhibitors
Cotreatment with C2-ceramide and sorafenib could significantly enhance ROS levels and mitochondrial depolarization, which cause caspase-dependent cell apoptosis. An intensive study found that the phenomenon is induced by targeting the PI3K/AKT/mTOR pathway and the extracellular regulated protein kinase (ERK) signaling pathway. This combination therapy could markedly suppress cell growth, cell migration, and cell proliferation in HCC.
53
Badawi
In addition to the above-mentioned combinations, there are also many inhibitors that play a synergistic effect with sorafenib by inhibiting the PI3K/AKT/mTOR pathway, including emodin, 56 SB-3CT, 57 20(S)-Ginsenoside Rg3, 58 bufalin, 59 capsaicin, 60 silibinin,61,62 amentoflavone, 63 MLN8237, 64 KU-55933, 65 melatonin, 35 PT-2385, 38 Valproic acid, 66 PF-03084014, 67 and ARQ 092. 68
Multiple inhibitors targeting more targets to improve the curative effect
Notably, mTOR inhibitors combined with sorafenib might induce high level of autophagy, and autophagy could cause resistance. Suppressing autophagy on the basis of the combinations of sorafenib and mTOR inhibitors might be a solution. Ling
Clinical intervention
Inhibitors of mTOR combined with sorafenib have been tested in some clinical trials. Everolimus is a potent mTOR inhibitor, and Koeberle
As classical immune inhibitors, mTOR inhibitors are used widely after liver transplantation. One of the most severe problems of liver transplantation is the recurrence of HCC. Several cases using sorafenib and mTOR inhibitors together showed surprising results in the treatment against post-transplant HCC, and provided a novel method in the post-transplant drug delivery strategy. Wang
Summary and discussion
The PI3K/AKT/mTOR pathway plays different roles at diverse stages in HCC cells. At the beginning, when HCC cells are still sensitive to sorafenib, sorafenib can downregulate the PI3K/AKT/mTOR pathway; however, this action also induces the activation of autophagy. The role of autophagy in drug resistance remains controversial; however, most evidence shows that autophagy can induce drug resistance. Briefly, the use of sorafenib might actuate HCC cells from sorafenib-sensitive to sorafenib-resistant
Autophagy-related HCC development and sorafenib resistance
Autophagy is a mechanism for the degradation of unwanted or damaged organelles in cells. Autophagy is important to maintain cellular homeostasis. Various stimulations can induce autophagy, such as drug treatment and nutrient deprivation. 77 Based on current research, the roles of autophagy in the progression of cancer and the reaction to drugs remain controversial. Research has shown that sorafenib can induce autophagy in HCC.78,79 Most studies have demonstrated that autophagy contributes to the resistance of HCC cells to sorafenib, whereas a few scientists hold the opinion that autophagy can enhance the toxicity of sorafenib.17,80 Based on these theories, the combinations of drugs are also divided into two groups; those that activate autophagy and those that inhibit autophagy (Figure 3).

Roles and solutions of autophagy in HCC development and sorafenib resistance. The role of autophagy in HCC development and sorafenib resistance remains controversial. Most studies showed that inhibiting autophagy could enhance the effect of sorafenib through multiple pathways. A few research studies have reported that autophagy can induce cell apoptosis and plays a synergistic role with sorafenib.
Most research concluded that autophagy leads to drug resistance. Rong
Almost all studies showed that autophagy has a negative effect on treatment of HCC with sorafenib; however, one study by Tai
Autophagy plays roles in both cell survival and cell death. Similarly, autophagy also plays a dual role in drug resistance. The function of autophagy depends on the tumor type and the treatment characteristics.
86
There are only a few reports in various tumors of the anti-tumor effect of excessive autophagy induced by targeted drugs. For HCC, only one study mentioned the positive role of autophagy in treatment against HCC. Lee
Almost all studies stand by the opinion that autophagy induces drug resistance. Apart from the combinations of sorafenib and autophagy inhibitors in HCC mentioned above, there are also many reports in other kinds of tumors, for example, gemcitabine and 3-MA in lung cancer. 89 These studies combined two or more inhibitors together and showed ideal results, and are expected to contribute to the treatment of cancer in the future.
In conclusion, it could be speculated that autophagy participates mainly in sorafenib resistance, and, in only very few cases, excessive autophagy can induce cell death. This is consistent with the duality of autophagy in cell survival. Some reports showed there are different markers in the stages of autophagy and subsequent apoptosis in sorafenib-treated HCC cells. Autophagy has been generally acknowledged to suppress tumor by inhibiting inflammation. So, changes in some inflammation-related factors can be markers of the roles of autophagy in suppressing tumor. HIFs and reactive oxygen species (ROS) have been reported related with tumor-promoting role of autophagy.26,90 Another study found the dynamic features of specific markers of autophagy after treating sorafenib, and revealed important roles of endoplasmic reticulum stress in the transformation of autophagy to excessive autophagy. 91 So, finding specific markers may clarify specific roles of autophagy in sorafenib resistance and provide better treatment strategies.
Defining the role of autophagy in drug resistance and cancer treatment, and determining the conditions leading to the appearance of excessive autophagy are challenges in future research.
Immune checkpoint-related HCC development
Inhibiting immune checkpoints is a novel and promising way to treat HCC and other cancers. Two inhibitors of immune checkpoint, nivolumab, and pembrolizumab, have been approved as second-line treatments for HCC by the US FDA. They showed satisfactory response rate according to phase I/II clinical trials.92,93 In following phase III trials, though the primary endpoints did not achieve statistical significances both in CheckMate 459 (nivolumab
Though immune checkpoint inhibitors showed meaningful results in some clinical trials, the effects of them might be influenced by immunosuppression. 96 Scientists have proven that the VEGF/VEGFR pathway leads to immunosuppression, such as inhibition of the maturation of dendritic cells (DCs) and accumulation of immunosuppressive inflammatory cells.97,98 Inhibiting the VEGF/VEGFR pathway using sorafenib could modulate the immune microenvironment and enhance the activity of inhibitors of immune checkpoints. Theoretically, combinations of sorafenib and immune checkpoint inhibitors could have synergistic roles.
Currently, there are only a few reports on this aspect. Chen
There has also been a clinical case of the use of sorafenib and immune checkpoint inhibitors. Adcock
Sorafenib is an inhibitor targeting VEGF/VEGFR pathway. Recently, a phase III clinical trial showed exciting progress in the combination therapy for HCC. The phase III IMbrave150 study showed the combination of atezolizumab (targeting programmed cell death ligand 1, PD-L1) and bevacizumab (targeting VEGF) can improve OS significantly compared with sorafenib [not evaluable (NE)
Another phase Ib trial of the combination of lenvatinib (targeting VEGFR) and pembrolizumab (targeting PD-1) also showed promising antitumor activity in HCC; ORR was 44.8% compared with using lenvatinib alone (24.1%) in another study. An acceptable safety profile was also observed.103,104 A phase III trial of this combination is also ongoing (NCT03713593). These results show the great potential of the combinations of VEGF/VEGFR inhibitors and immune checkpoint inhibitors. In addition, there are also several ongoing phase III clinical trials of combinations of VEGF/VEGFR inhibitors and immune checkpoint inhibitors. These combinations include: apatinib and SHR-1210 (NCT03764293); IBI305 and sintilimab (NCT03794440); bevacizumab and durvalumab (NCT03847428); and cabozantinib and atezolizumab (NCT03755791).
Therapy using combinations of VEGF/VEGFR inhibitors with immune checkpoint inhibitors has showed significant effects in several kinds of tumors, including HCC. Sorafenib is a representative drug targeting VEGFR; however, there have been few studies about sorafenib plus immune checkpoint inhibitors to treat HCC. We speculate that using immune checkpoint inhibitors could be an important method to allow more patients with HCC to benefit from sorafenib. In summary, targeting immune checkpoints and VEGF/VEGFR is expected to become a treatment with great potential against HCC. These molecular pathways have complex connections; therefore, targeting multiple targets together might produce better treatment results.
Other pathways-related HCC development and sorafenib resistance
The targets above are important factors in the development of HCC. They also play important roles in the resistance of HCC cells to sorafenib. In addition, there are other pathways related to HCC and sorafenib resistance.
Wnt/β-catenin
The Wnt/β-catenin pathway is a de-regulation pathway in HCC and is one of the pathways that is difficult to treat.
105
This pathway is also reported to be closely linked to sorafenib resistance in HCC.106,107 Scientists combined sorafenib with inhibitors targeting the Wnt/β-catenin pathway and observed positive therapeutic effects. Galuppo
c-Met/HGF
c-Met is a receptor tyrosine kinase that is involved in epithelial–mesenchymal transition. It is regulated by hepatocyte growth factor (HGF). Overexpression of c-Met increases cell proliferation, survival, mobilization, and invasiveness. Aberrant c-Met/HGF activity is associated with the development of HCC and resistance to sorafenib.70,111 Thus, the c-Met/HGF pathway is a potential target to treat HCC, and combining sorafenib with c-Met/HGF inhibitors may be an effective strategy against HCC. Sorafenib and vitamin K can have a synergistic effect on the suppression of HCC cell migration and metastasis by inhibiting the c-Met/HGF pathway, and could suppress HCC cell growth. These conclusions provided a theoretical basis for subsequent clinical trials.112,113 Jiang
Tivantinib, an inhibitor targeting c-Met, has been proved to play a synergistic role with sorafenib in treating HCC in a phase I trial. This therapy strategy showed preliminary evidence of anticancer activity in 10% of HCC patients, including those refractory to sorafenib. Though there was no phase II/III trial of the combination of tivantinib and sorafenib in HCC, this result still indicated the feasibility of the combination strategy and provided impetus for further research. 115
HDAC
HDAC is a novel target in the treatment of cancer. High HDAC activity correlates with higher incidence of HCC. HDAC inhibitors have been proven as a novel treatment against cancer. HDAC inhibitors can induce apoptosis, autophagy, differentiation, and inhibition of tumor vascularization
Resminostat is an HDAC inhibitor that has promising anti-tumor activity in HCC. A SHELTER study invested the role of resminostat in sorafenib resistance. The results showed that the OS of patients treated with the combination of sorafenib and resminostat was 8 months, compared with 4.1 months after sorafenib treatment alone. 122 This trial provided evidence that using sorafenib with HDAC inhibitors can treat HCC more effectively. Further testing of this strategy appears necessary.
Clinical trials performed to date
Leveraging basic research, scientists have conducted clinical trials to test the effects of drug combinations. Most of these clinical trials are in phase I or phase II trials. Though some trials produced a positive conclusion, they still need more research because of their small sample size. 124 Some trials did not set a group of sorafenib alone, and some did not calculate the OS, and these factors might lead to inaccuracy. Some drugs can inhibit HCC markedly when combined with sorafenib; however, the trials finally failed because of their poor safety. Some drugs in the trials are targeted drugs and have specific treatment mechanisms; however, for most of these drugs, the anti-tumor mechanisms have not been determined. In conclusion, in the future, more clinical trials should be conducted to find better combinations, and the mechanisms of some effective combinations should be further clarified (Table 1).
Recent clinical trials investigating the combinations of drugs and sorafenib in HCC.

S-1 is an anticancer drug comprising three components: Tegafur, 5-chloride-2,4-dihydroxypyridine, and oteracil potassium (molar concentration ratio = 1:0.4:1).
HCC, hepatocellular carcinoma; HDAC, histone deacetylase; MAPK, mitogen-activated protein kinase; mTOR, mammalian target of rapamycin; OS, overall survival; VEGF, vascular endothelial growth factor.
Perspectives
Systemic treatment is the standard therapy against advanced HCC. This treatment depends mainly on targeted drugs. However, a key problem is that there are too few of these drugs and their effects are unsatisfactory. Sorafenib was approved as the first frontline drug against advanced HCC, and was the only one until 2018. Sorafenib has two problems; one is primary resistance, whose mechanism remains unclear, which leads to a low response rate at the start of sorafenib therapy. The other is acquired resistance, which means that, as HCC progresses, the potency of sorafenib becomes lower and lower until the cancer is completely resistant. Patients encountering these problems will be offered other treatments. Lenvatinib, which was approved as the second first-line treatment against advanced HCC, was used in a randomized phase III non-inferiority trial, which showed that it was non-inferior to sorafenib for OS in untreated advanced HCC (13.6 months with lenvatinib and 12.3 months with sorafenib). And lenvatinib showed statistically significant improvements in progression-free survival, time to progression, and ORR compared with sorafenib. 103 In general, both drugs have reasonable safety profiles. For lenvatinib, more hypertension, proteinuria, dysphonia, and hypothyroidism occurred; and more palmar-plantar erythrodysaesthesia, diarrhoea, and alopecia were observed in the patients who received sorafenib. These differences can provide reference for the choices of first-line treatment and subsequent second-line treatment against HCC.
If patients cannot benefit from first-line treatments, they have to turn to second-line drugs. Up to now, there are five second-line treatments (Table 2).
137
Regorafenib is a second-line drug approved by the FDA for use by patients showing progression on sorafenib therapy. It is also a multi-kinase inhibitor targeting STAT3, nuclear factor-κB (NF-κB) and other HCC-related factors.138–140 The RESORCE trial showed that regorafenib could improve OS significantly compared with placebo (10.6 months
US FDA-approved targeted drugs for HCC.
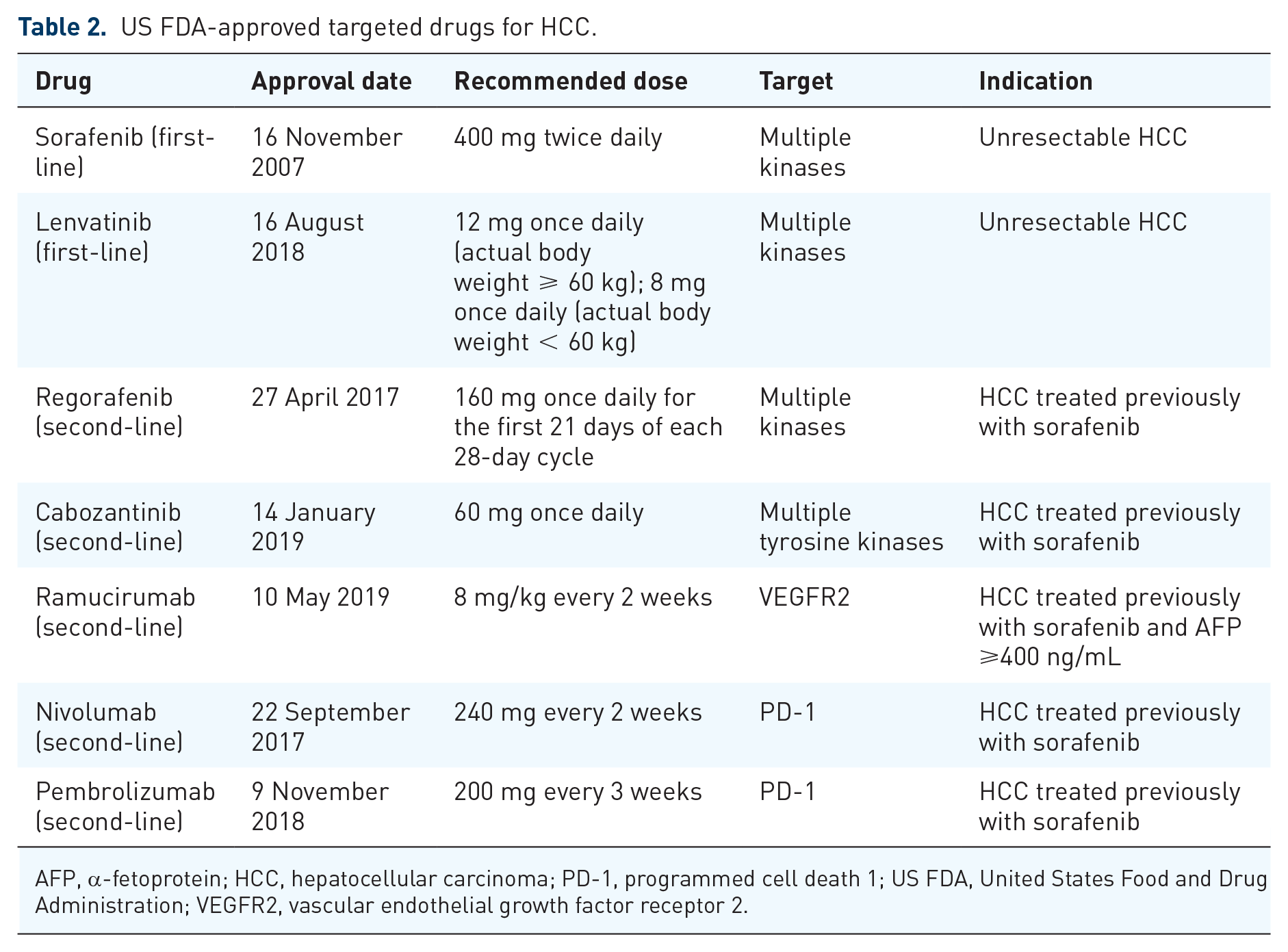
AFP, α-fetoprotein; HCC, hepatocellular carcinoma; PD-1, programmed cell death 1; US FDA, United States Food and Drug Administration; VEGFR2, vascular endothelial growth factor receptor 2.
There are many reports about the combinations of sorafenib and other drugs to treat HCC (Figure 4). To date, most research has investigated small molecule targeted drugs, and several clinical trials concerning their combination with sorafenib are in progress. Inhibiting immune checkpoints has been proven as a promising method to treat HCC and other types of cancer. Theoretically, sorafenib could enhance the effects of immune checkpoint inhibitors. However, research on combining sorafenib and immune checkpoint inhibitors is still at an early stage. Notably, the combination of sorafenib and one inhibitor cannot produce satisfactory results because of complex connections between pathways; therefore, it is critical to determine their molecular mechanisms and test more combinations comprising several drugs.

Mechanisms of sorafenib resistance and HCC development, and combination therapies to treat HCC. Current research about the combinations of sorafenib and other inhibitors demonstrates the feasibility of drug combination. Through mechanistic research and clinical trials, scientists can find more treatments against HCC.
Notably, although some reports did not use the identified inhibitors with sorafenib, the results showed that regulating these molecules could play synergistic roles with sorafenib. Therefore, it is worth seeking further inhibitors of these molecules in future research. Lu
Precision therapy is the future direction of cancer treatment. Through profiling cell line cultures’ gene expression and comparing it with that of patients with cancer, machine learning could predict the efficiency of targeted drugs. Sorafenib has been used in this research.
153
Drug combination also depends on the gene expressions of different patients. Using probabilistic modeling, scientists presented an artificial intelligence method to predict different synergistic personalized two-drug combinations for different patients. This system could help more patients benefit from precision therapy.
154
Surprisingly, another group developed an
Previous research and future prospects promise significant progress in the exploration and application of drug combinations. Machine learning predicts targets that might be involved in HCC development and drug resistance, and provides potential drug combinations. Preclinical studies show positive results in cell models and animal models, and clinical trials will confirm their validity and safety further. Finally, based on genetic testing, patients will be treated with specific drug combinations.
In summary, therapy using combinations of sorafenib and other inhibitors is a promising strategy to treat HCC. After further in-depth studies, they are expected to play important roles in fighting HCC in the future. The promising results of combination therapies mean that sorafenib has more possibilities depending on new technologies and new methods.
Footnotes
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Our work is supported by the National Science and Technology Major Project (2017ZX100203205), National Natural Science Funds for Distinguished Young Scholars of China (81625003), Yangtze River Scholar Project, Zhejiang Provincial Natural Science Foundation of China (LQ17H160006).
Conflict of interest statement
The authors declare that there is no conflict of interest.
