Abstract
BACKGROUND:
Hepatoblastoma (HB) is an embryonic solid tumor and the most common primary malignant liver tumor in children. HB usually occurs in infants and children. Although treatment diversity is increasing, some patients still have very poor prognosis. Many studies have investigated USP7 inhibitors for tumors. Using database information, we found that USP7 is highly expressed in HB.
METHODS:
Lentivirus-mediated USP7 knockdown and overexpression was performed in HB cell lines HepG2 and Huh6. CCK8 and transwell assays were used to determine cell viability and metastasis. Flow cytometry was used to study cell cycle and apoptosis. Levels of proteins were detected using western blots.
RESULTS:
Downregulation of USP7 resulted in significant decrease in cell proliferation, clonal formation, and cell migration and invasion. With overexpression of USP7, cellular malignant behavior increased. Cell cycle assays showed that USP7 knockdown inhibited G1 to S phase transition in the cell cycle. Upregulation of USP7 promoted the transition. Animal experiments showed USP7 facilitated tumor growth in vivo. Western blots indicated that USP7 may affect HB tumorigenesis through the PI3K/AKT signaling pathway. Furthermore, USP7 inhibitor P5091 inhibited HB development and PI3K/AKT pathway.
CONCLUSION:
USP7 upregulation contributed to HB genesis and development through the PI3K/AKT signaling pathway. USP7 could be a potential target for future HB treatment.
Introduction
Hepatoblastoma (HB) is highly prevalent in children and has a high degree of malignancy [1, 2]. HB is the most common hepatic malignancy in children [3, 4] and is more common in under age five [5]. It may be closely related to chromosomal abnormalities, adverse external factors of maternal pregnancy, overactivation of oncogenes or inactivation of tumor suppressor genes [1, 6, 7]. If HB is found early, surgical treatment can have therapeutic effects. However, some children have late-stage disease and overall poor prognosis [8, 9, 10, 11]. Therefore, effective diagnosis and treatment targets are urgently needed.
Ubiquitination is the process by which ubiquitin specifically modifies a target protein under catalysis of a series of enzymes [12, 13]. Ubiquitination is an important pathway for post-translational modification of proteins [14, 15, 16]. Deubiquitinating enzymes specifically cleave the isopeptide bond between the C-terminus of ubiquitin and the target protein, detaching ubiquitin from the target protein so it is not degraded, but relocated or activated [17, 18]. Ubiquitin-specific protease 7 (USP7) is one of the deubiquitinating enzymes [19, 20, 21, 22]. The substrate range of USP7 is very broad. Most substrates such as p53, PTEN and FOXO4 are involved in tumor suppression, DNA repair, or immune response [23, 24, 25, 26]. USP7 degrades the E3 ligase MDM2, which is oncogenic because it causes the tumor suppressor gene p53 to be inactivated in a variety of cancers [27, 28]. Monoubiquitination regulates the localization of PTEN protein. Monoubiquitinated PTEN transfers from the cytosol to the nucleus and USP7 removes it from the nucleus by deubiquitinating PTEN [25, 29]. Studies show that USP7 protein is highly expressed in a variety of malignant tumors such as multiple myeloma [30], colon cancer [31], lung cancer [32] and prostate cancer [33]. High expression positively correlates with the occurrence and development of tumors, suggesting involvement of oncogenes.
This evidence strongly suggests that USP7 is a promising target for disease treatment. In studies on small molecule inhibitors of USP7, some identified small molecules such as P5091 show antiproliferative activity against cancer cells [23]. Thus, USP7 inhibitors are a promising new anticancer approach. Currently, ubiquitin-like protease inhibitors are studied in the field of cancer therapy [34].
Materials and methods
Human tissues and HB cell lines
Human HB and adjacent nontumor liver tissues were collected from patients treated at Children’s Hospital of Fudan University (Shanghai, China). Tumor tissues were diagnosed as HB by the Department of Pathology. The stage of the disease adopted the COG staging system. HepG2 and Huh6 cells were from the Cell Bank of the Chinese Academy of Science (Shanghai, China) and cultured in Dulbecco’s modified Eagle’s medium with 10% fetal bovine serum (FBS) in a humidified incubator with 5% CO2 at 37
Construction of stably transfected cell lines
USP7 shRNA plasmids were designed and constructed with the pLko1 vector. The shRNA target was 5’-CCTGGATTTGTGGTTACGTTA-3’. USP7 overexpression was achieved using the PCDH vector. Lentivirus packaging was in 293T cells with l-pei. Following viral infection and puromycin screening, stable HepG2 and Huh6 cells were acquired.
Cell proliferation and viability assays
Cell proliferation was detected using CCK-8 kits (Yeasen, Shanghai, China) in accordance with the manufacturer’s instructions. Cells were cultured in 96-well plates at 1,000 cells in 100
Cell apoptosis detection
After digestion with trypsin without EDTA, cells were collected and washed twice with pre-cooled PBS. Suitable cells were transferred into 5-mL flow tubes and 5
USP7 expression from database and patients. A, USP7 expression in children with HB (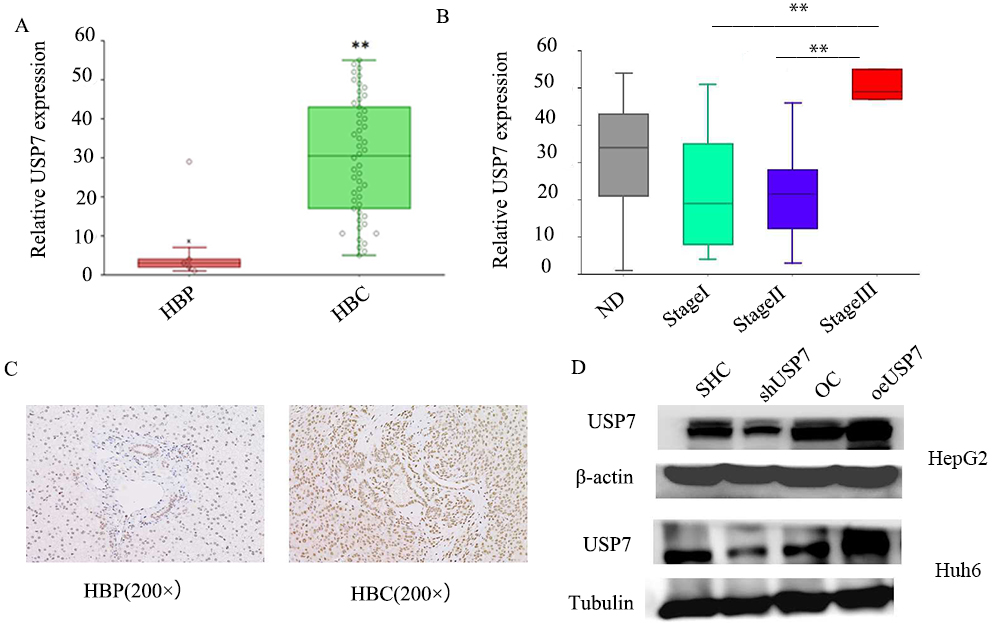
Cell migration and invasion assays used 24-well culture plates with 8-
Western blots
Cell proteins were extracted with Protein Lysis Buffer (Beyotime Institute of Biotechnology). After blocking with 8% skim milk, membranes were incubated with primary antibodies: USP7 (4833T, CST), PI3K (4249T, CST), AKT (9272S, CST), p-AKT (4060T, CST) at 4
Immunohistochemical analysis
Immunohistochemical analysis of HB tissue samples was by a standard procedure. Tissue sections were dewaxed, rehydrated and antigen-repaired under high temperature and high pressure. Myeloperoxidase was inactivated with 3% H2O2 and sections were blocked with 3% BSA. USP7 (4833T, CST) was used for immunohistochemical in 4
Tumor xenografts
HepG2 cells were cultured for animal experiments. Cells were collected by trypsin digestion, washed with precooled PBS, and converted to single-cell suspensions in PBS. Inoculation was subcutaneous with 2
Statistical analysis
All assays were repeated three times. All results are presented as mean
USP7 affects tumor cell proliferation. A and B, CCK8 assays for HepG2 and Huh6 cells for four days. C, Clone formation assays for HepG2 and Huh6 cells. D, Statistics for clone numbers of HepG2 cells. E, Statistics for clone numbers of Huh6 cells. 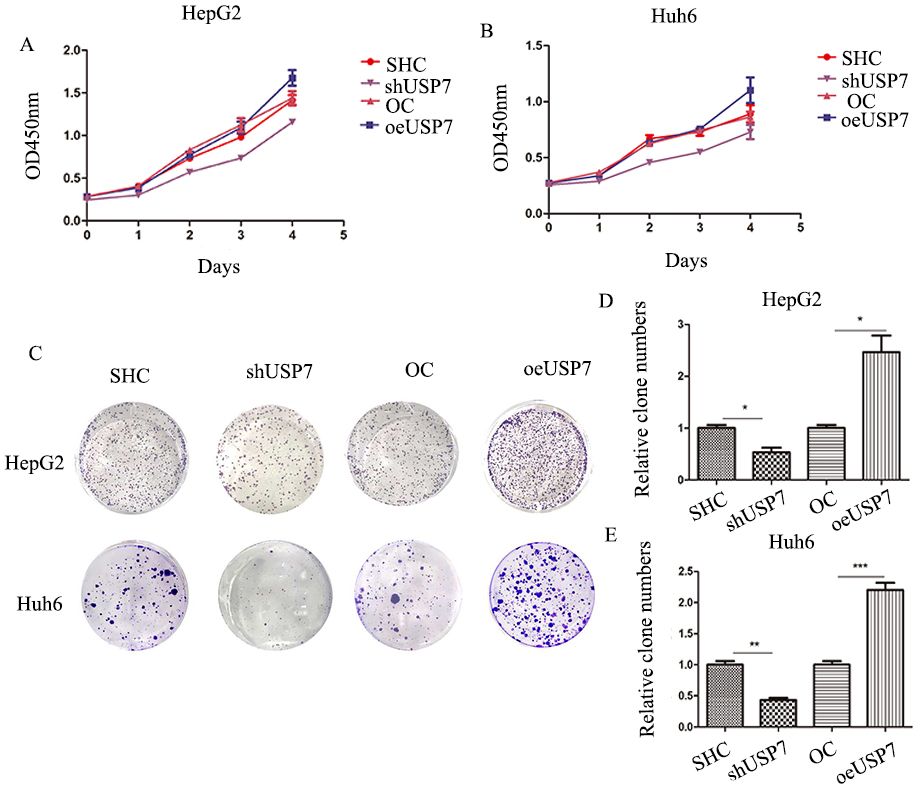
USP7 promotes cell cycle G1 to S phase transition. A, Curve fitting was by Modfit software for HepG2 and Huh6 cells. B, Statistics for cell proportions of HepG2 cells. C, Statistics for cell proportion of Huh6 cells.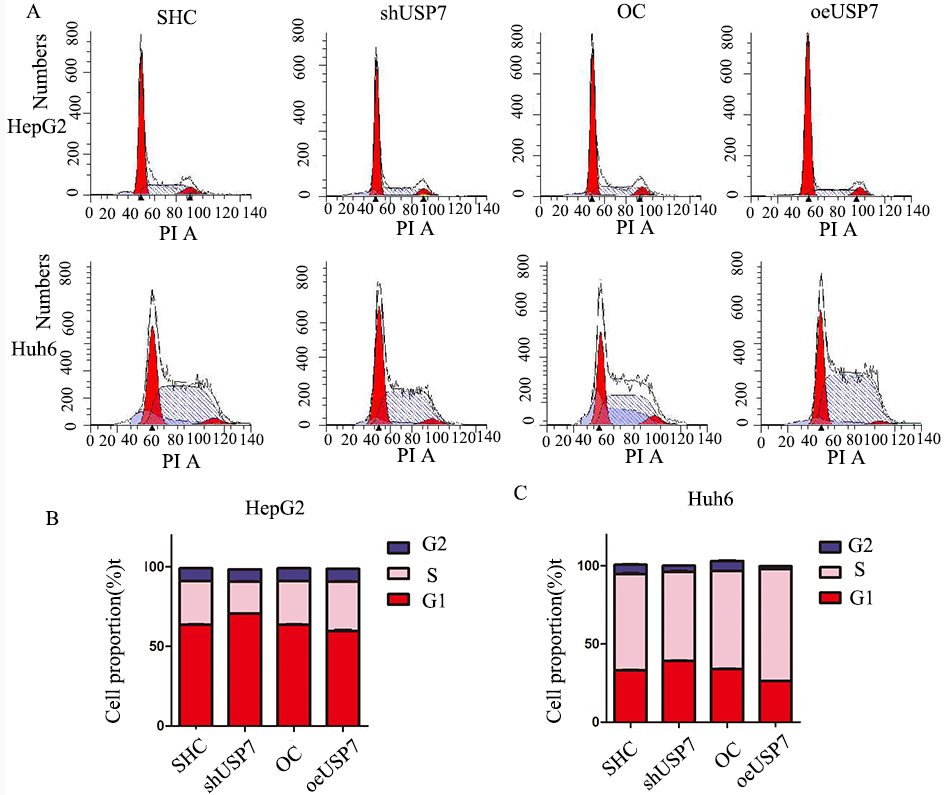
USP7 is an apoptosis suppressor. A, Flow cytometry to detect proportion of apoptotic HepG2 and Huh6 cells. Dyes were annexinV APC and PI. HepG2 cells were induced into apoptosis with 50 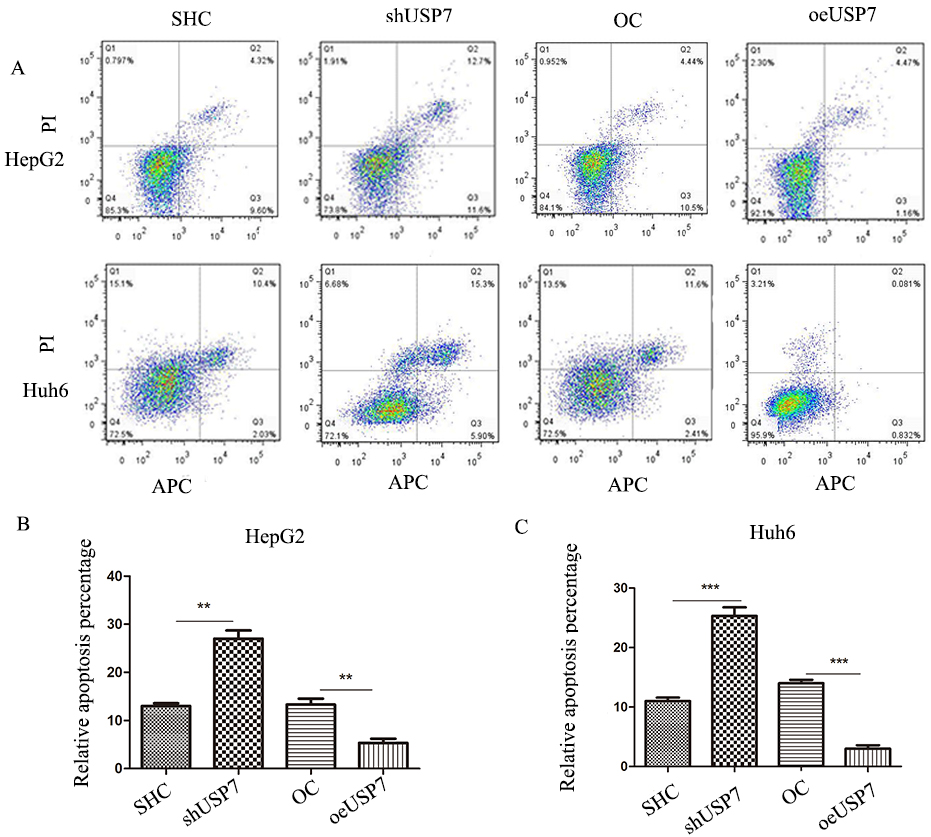
USP7 participates HB metastasis in vitro. A, Transwell assays for migration and invasion of HepG2 and Huh6 cells. B and C, HepG2 cell migration and invasion. D and E, Huh6 cell migration and invasion. 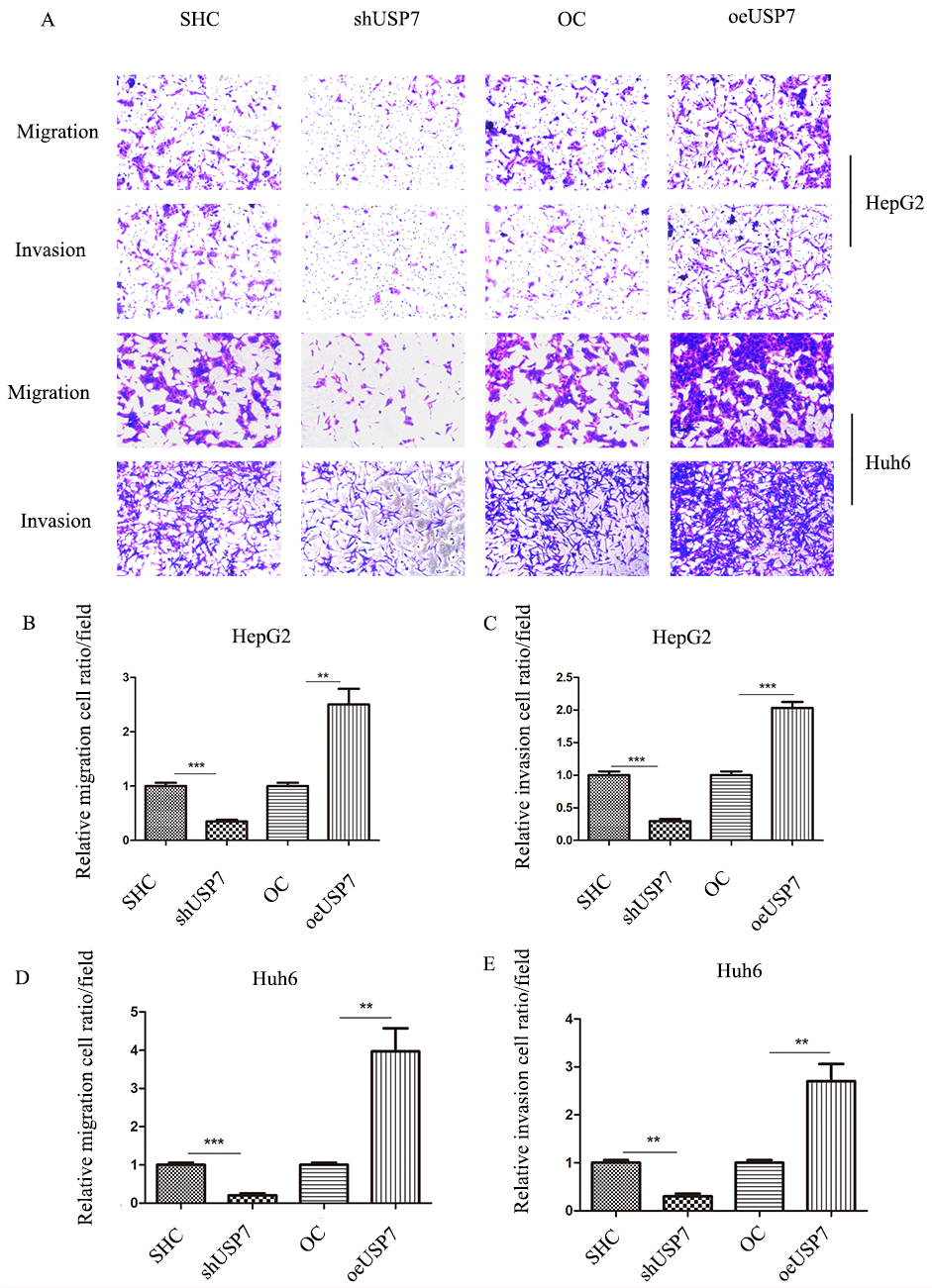
USP7 has an influence on HB growth in vivo. A, Tumor formation was observed and nude mice were randomly divided into groups by USP7 downregulation, upregulation, or control (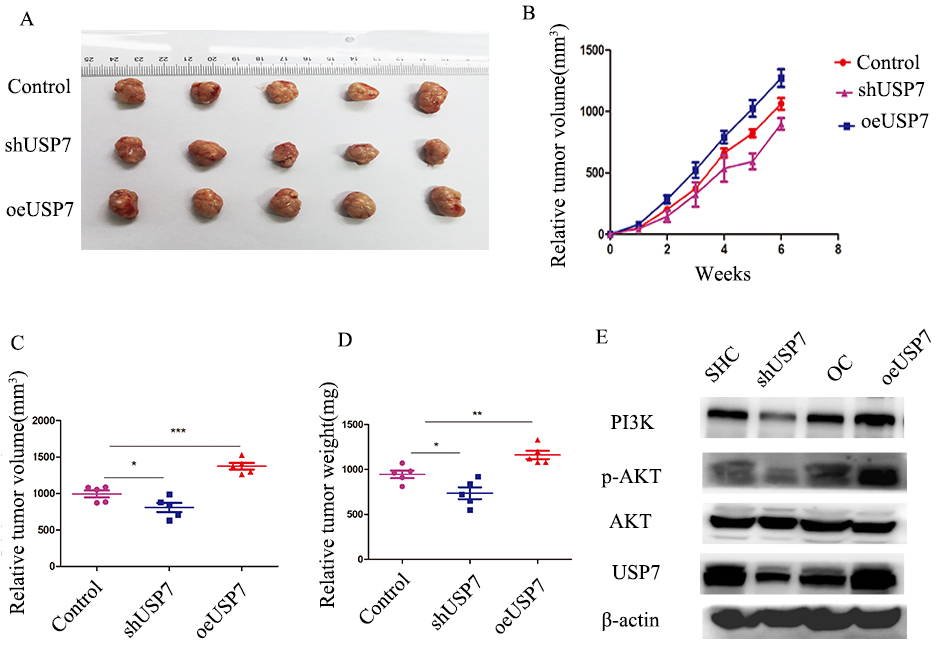
USP7 inhibitor P5091 suppresses HB malignant behavior. A, CCK8 assays for HepG2 cells treated with DMSO or P5091. B, Clone formation assays for P5091 treatment and control groups of HepG2 cells. C and D, Cell cycle experiments by flow cytometry and statistics in HepG2 cells. E and F, Flow cytometry to detect the proportion of apoptotic HepG2 cells treated with DMSO and P5091. G–I, Transwell assays for migration and invasion of HepG2 cells treated with DMSO and P5091. J, Western blot for PI3K/AKT signaling pathway proteins in HepG2 cells treated with DMSO and P5091. Cells were incubated for 48 hours with 10 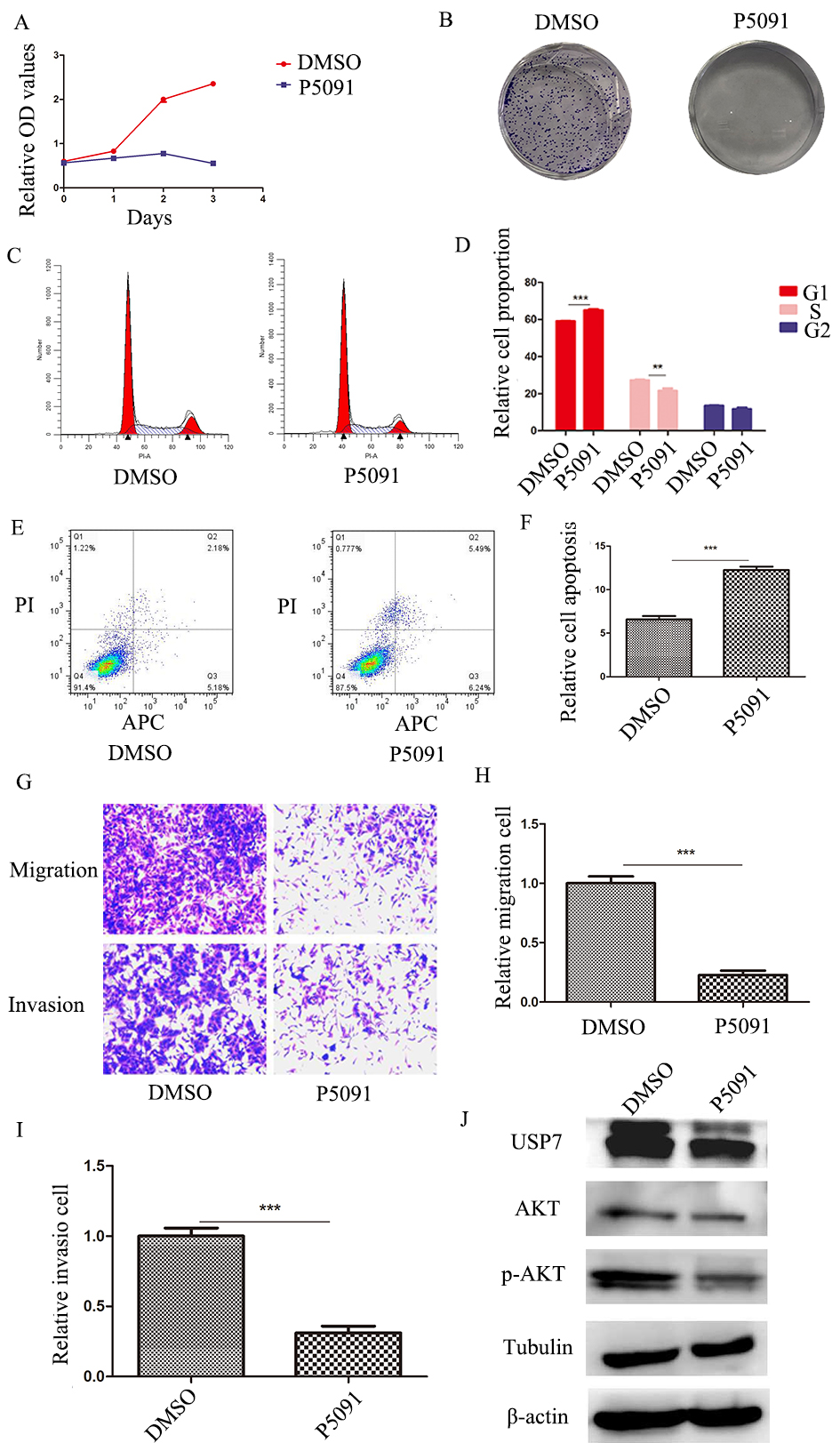
USP7 was significantly upregulated in HB tissues and cell lines
To explore the relationship between USP7 expression and clinical relevance to patients with HB, the GEO dataset GSE75271 (
USP7 promoted tumor growth and clonal formation
After USP7 knockdown and overexpression stable transfected cells were constructed (Fig. 1D), CCK-8 assays were performed to study the effect of USP7 on cell proliferation. Silencing USP7 inhibited HB cell proliferation and overexpression of USP7 promoted cell proliferation compared to a negative control group for both HepG2 and Huh6 cells (Fig. 2A and B). Clonal formation experiments showed that USP7 knockdown inhibited colony formation, while overexpression facilitated colony formation (Fig. 2C–E).
USP7 participated in HB cell cycle transition
Cell cycle assays were performed on HepG2 and Huh6 cells. Overexpression of USP7 promoted cell cycle transition from G1 to S phase (Fig. 3). Knockdown of USP7 inhibited cell cycle transition from G1 to S phase (Fig. 3).
USP7 was an apoptosis repressor in HB
Cells were stained with annexinV APC/PI and detected by flow cytometry. The percentage of apoptotic cells significantly increased in the USP7 knockdown cell population and decreased in the population of cells overexpressing USP7. These results were seen for both HepG2 cells and Huh6 cells compared to the scramble groups (Fig. 4).
USP7 was involved in migration and invasion of tumor cells
Transwell assays were performed to determine the effect of USP7 on HB cell migration and invasion. Knockdown of USP7 inhibited HB cell migration and invasion compared to a control group. Upregulation of USP7 significantly increased cell migration and invasion in both HepG2 and Huh6 cells (Fig. 5).
USP7 affected tumor growth in vivo
Tumor formation of nude mice showed that tumor volume and weight were significantly lower than a control group after USP7 silencing. Upregulation of USP7 increased tumor growth in vivo (Fig. 6A–D). Western blots indicated that USP7 knockdown decreased the level of PI3K and phosphorylation of AKT and overexpression increased PI3K and phosphorylation of AKT (Fig. 6E).
USP7 inhibitor P5091 decreased HB cell malignant behavior
To further study USP7 function, we used the USP7 inhibitor P5091 for cell function assays. CCK8 and clonal formation assays showed that P5091 inhibited cell proliferation (Fig. 7A and B). P5091 inhibited the cell cycle transition from G1 to S phase (Fig. 7C and D). Cell apoptosis significantly increased in a dosing group measured by flow cytometry (Fig. 7E and F). Transwell experiments demonstrated that P5091 decreased HB cell migration and invasion (Fig. 7G–I). Western blots indicated that P5091 inactivated the PI3K/AKT signaling pathway (Fig. 7J).
Discussion
Despite a variety of treatments, some high-risk HB prognoses are still unsatisfactory [3]. Understanding the mechanism of HB development is especially important. Abnormal activation of oncogenes is known to be an important factor in HB development. Studies indicate that USP7, an important deubiquitinating enzyme, has a range of substrates and is important in the progression of variety of tumors [24, 27].
In a study of neuroblastoma (NB) in children, high expression of USP7 was used to predict the poor prognosis of patients. The small molecule inhibitor P22077 efficiently promotes NB through the USP7-HDM2-p53 axis in the wildtype p53. Apoptosis of NB cells reverses the resistance of NB cell line LA-N-6, and in vivo experiments show that P22077 significantly inhibits the growth of transplanted tumors [35]. An osteosarcoma (OS) study showed that USP7 is highly expressed in OS and associated with TNM stages and tumor metastasis [36]. Knockdown of USP7 affects epithelial-mesenchymal transition through a Wnt/
In our study, we first found that USP7 knockdown significantly inhibits HB cell proliferation. Therefore, we analyzed 50 HB cases in the GEO database. In line with our hypothesis, USP7 was significantly increased in HB tissues compared with normal liver tissues. Moreover, high expression of USP7 was associated with later stage. However, the database used COG stage system which is an outdated staging system and this is a limitation to the data. Immunohistochemistry showed that USP7 was markedly higher in HB than normal liver tissue adjacent to the tumors. In vitro functional studies showed that after knocking down USP7, cell proliferation, clonal formation ability, and cell migration and invasion were significantly reduced. Conversely, overexpression of USP7 increased cellular malignant behavior. Cell cycle assays demonstrated that overexpression of USP7 facilitated cell cycle G1 to S phase transition and knockdown of USP7 suppressed the transition. We hypothesized the influence of USP7 on the cell cycle affected cell proliferation. Downregulation of USP7 promoted apoptosis and upregulation of USP7 inhibited apoptosis, indicating that USP7 may function as an apoptosis inhibitor in HB. Animal experiments demonstrated that USP7 was involved in HB growth in vivo. Some studies report that USP7 participates in various tumor biological processes through WNT, Hippo and NF-
In conclusion, our study demonstrated the USP7 was highly expressed in tumors from HB patients. Furthermore, USP7 influenced tumor malignant behavior such as proliferation, apoptosis, cell cycle and metastasis. Changes in cell phenotype may be caused by the PI3K/AKT signaling pathway. This study had some limitations. First, we did not detect USP7 expression in many patients because HB is an extremely rare disease. Second, we did not find the substrate of USP7 as it is a ubiquitin-regulated enzyme. Because of these shortcomings, we will carry out more in-depth research on these issues. With increasing studies on USP7, we believe it could be a future potential target for HB treatment.
Footnotes
Acknowledgments
This study received financial support from Shanghai Key Disciplines (no. 2017ZZ02022), National Natural Science Foundation of China (no. 81771633 and no. 81572324), The Science Foundation of Shanghai Excellent Youth Scholars (no. 2017YQ042) and Science Foundation of Shanghai (no. 17411960600 and no. 15ZR1404200). This study also received financial support from Clinical Research Plan of SHDC (no. SHDC2020CR2009A), Shanghai Municipal Key Clinical Specialty (no. shslczdzk05703).
Conflict of interest
The authors declare that they have no competing interests.
Ethic approval
Written informed consent has been obtained from each patient. The study methodologies have been approved by Ethics Committee of Children’s Hospital of Fudan University. And all experiments do conform to the Declaration of Helsinki.
