Abstract
The pathogenic mechanisms underlying cancer development are complex and heterogeneous, involving multiple cellular signaling transduction pathways that usually function redundantly. In addition, crosstalk between these pathways generates a complicated and robust signaling network that is regulated by compensatory mechanisms. Given the complexity of cancer pathogenesis and progression, many of the currently used antitumor agents, which typically target a single intracellular pathway, might not always be effective on complex tumor systems. Moreover, long-term use of these agents often generates drug resistance and toxicity against normal cells. Therefore, the development of novel anticancer chemotherapies is urgently needed. Scutellaria barbata D Don (SB) is a medicinal herb that has long been used in China to treat various types of cancer. We previously reported that the ethanol extract of SB (EESB) is able to induce colon cancer cell apoptosis, inhibit cell proliferation and tumor angiogenesis via modulation of several pathways, including Hedgehog, Akt, and p53. To further elucidate the precise mechanisms of SB’s antitumor activity, using a colorectal cancer (CRC) mouse xenograft model in the present study, we evaluated the therapeutic efficacy and molecular mechanisms of EESB against tumor growth. We found that EESB reduced tumor volume and tumor weight but had no effect on body weight gain in CRC mice, demonstrating that EESB could inhibit colon cancer growth in vivo without apparent adverse effect. In addition, EESB treatment could significantly suppress the activation of several CRC-related pathways, including STAT3, Erk, and p38 signalings in tumor tissues, and alter the expression of multiple critical target genes such as Bcl-2, Bax, Cyclin D1, CDK4, and p21. These molecular effects lead to the induction of cancer cell apoptosis and inhibition of cell proliferation. Our findings demonstrate that SB possesses a broad range of antitumor activities because of its ability to affect multiple intracellular targets.
Introduction
Colorectal cancer (CRC) is the third most common human malignancy with more than 1 million new cases and more than a half million deaths each year. 1 Chemotherapy is one of the major therapeutic approaches for patients with advanced CRC. 2 However, both drug resistance and toxicity against normal tissues limit the effectiveness of current CRC chemotherapies, thus increasing the necessity for the development of novel antitumor agents. 3 The development of CRC is strongly correlated with the aberrant activation of multiple intracellular signaling transduction cascades, including STAT3, Erk, and p38 pathways. Signal transducer and activator of transcription 3 (STAT3), a transcription factor critical for the processes of cell survival and proliferation, is strongly associated with tumorigenesis.4-7 Constitutive activation of STAT3 is found in many types of human cancer and commonly suggests poor prognosis.8,9 STAT3 activation is mediated by phosphorylation at tyrosine 705 by receptor and nonreceptor protein tyrosine kinases; and the phosphorylated STAT3 proteins in the cytoplasm dimerize and translocate to the nucleus where they regulate the expression of critical genes involved in cell apoptosis and proliferation.7,10,11 The mitogen-activated protein kinase (MAPK) cascade is a highly conserved module that is involved in various cellular functions, such as cell proliferation, differentiation, and apoptosis. There are at least 3 distinct MAPK signaling modules which mediate extracellular signals into the nucleus to turn on the responsive genes in mammalian cells, including Erk, JNK, and p38 kinase. Activation of all MAPK pathways is regulated by a central 3-tiered kinase core consisting of MAPK kinase kinase (MAP3K), MAPK kinase (MAP2K), and MAPK; wherein MAP3K phosphorylates MAP2K and activated MAP2K in turn phosphorylates and activates MAPK. 12 MAPK signaling is important in intestinal epithelial differentiation 13 ; however, aberrant activation of MAPK pathway is involved in colon carcinogenesis. 14 , 15
It is noteworthy that intracellular signaling transduction pathways usually function redundantly. Moreover, cross-talks exist between these pathways forming a complicated and robust cellular signal transduction network that is regulated by compensatory mechanisms. Therefore, inhibitors that target only a single pathway might not be effective initially and their long-term use might generate drug resistance. Natural products have received recent interest as therapeutic agents for CRC as they have relatively few side effects and have been used for thousands of years as alternative remedies for a variety of diseases, including cancer. 16 Numerous plants and their constituents have been shown to possess beneficial therapeutic effects for cancers; and their effect is believed to be the result of targeting multiple molecular pathways.17,18 Therefore, discovering naturally occurring agents is a promising approach for cancer chemotherapy. Scutellaria barbata D Don (SB) is a medicinal herb widely distributed in northeast Asia. As a well-known traditional Chinese folk-medicine, it has long been used to clinically treat various kinds of cancer. 19 We previously reported that the ethanol extract of SB (EESB) is able to induce colon cancer cell apoptosis, and inhibit cell proliferation and tumor angiogenesis via modulation of several pathways, including Hedgehog, Akt, and p53.20-23 To further elucidate the precise mechanisms of SB’s antitumor activity, using a CRC mouse xenograft model in the present study we evaluated the therapeutic efficacy of EESB against tumor growth and investigated the underlying molecular mechanisms.
Materials and Methods
Materials and Reagents
Dulbecco’s modified Eagle’s medium (DMEM), fetal bovine serum (FBS), penicillin–streptomycin, trypsin–EDTA, Trizol reagent were purchased from Invitrogen (Carlsbad, CA). Matrigel was provided by Becton Dickinson (San Jose, CA, USA). SuperScript II reverse transcriptase was provided by Promega (Madison, WI). Phospho-STAT3 (p-STAT3), total STAT3, Bcl-2, Bax, p21, cyclinD1, CDK4 antibodies, horseradish peroxidase (HRP)–conjugated secondary antibodies were obtained from Cell Signaling (Beverly, MA). Proliferating cell nuclear antigen (PCNA) and terminal deoxynucleotidyl transferase (TdT)–mediated dUTP nick end labeling (TUNEL) assay kit (TumorTACS in situ) were purchased from R&D Systems (Minneapolis, MN). ABC kit (Rabbit IgG,) was provided by VECTOR Laboratories (Burlingame, CA). BCA Protein Assay Kit was purchased from Tiangen Biotech (Beijing) Co., Ltd. Erk1/2 and p38 Bio-Plex assay kits were obtained from Bio-Rad (Hercules, CA). All the other chemicals used, unless otherwise stated, were obtained from Sigma Chemicals (St Louis, MO).
Preparation of Ethanol Extract From Scutellaria barbata D Don (EESB)
Authentic plant material was purchased from Guo Yi Tang Chinese Herbal Medicine store, Fujian, China. The original herb was identified as Scutellaria Barbata D Don by Dr Wei Xu at Department of Pharmacology, Fujian University of Traditional Chinese Medicine (TCM), China. EESB was prepared as described before. 20 Scutellaria Barbata D Don (500 g) were extracted with 5000 mL of 85% ethanol using refluxing method and filtered. Stock solutions of EESB were prepared by dissolving the EESB powder in PBS to a concentration of 2 g/mL, and stored at −20°C. The working concentrations of EESB were made by diluting the stock solution in saline to a concentration of 0.2 g/mL.
High-Performance Liquid Chromatography Analysis
Ethanol extract of SB was analyzed on an Agilent 1200 HPLC system (Agilent, Santa Clara, CA) using a C-18 column. The absorbance was measured at 355 nm (Figure 1). The mobile phase consisted of methanol: 0.2% acetic acid at 85:15 at a flow rate of 1 mL/min with an injection volume of 10 µL. A sample containing Scutellarin and Apigenin was used as control.

High-performance liquid chromatography profiles of (A) ethanol extract of Scutellaria barbata D Don (EESB) and (B) control sample. The mobile phase consisted of methanol:0.2% acetic acid at 85:15. The control sample comprised scutellarin (peak R1) and Apigenin (peak R2).
Cell Culture
Human colon carcinoma HT-29 cell line was obtained from American Type Culture Collection (ATCC,
Animals
Twenty male athymic BALB/C nude mice (with an initial body weight of 20-22 g) were obtained from Shanghai Si-Lai-Ke Experimental Animal Ltd (Shanghai, China). The mice were housed 5 per cage in specific pathogen-free room in an environment with controlled temperature (22°C), humidity, and a 12-hour light/dark cycles with free access to water and standard laboratory food. All animal treatments were strictly in accordance with international ethical guidelines and the National Institutes of Health Guide concerning the Care and Use of Laboratory Animals, and the experiments were approved by the Institutional Animal Care and Use Committee of Fujian University of Traditional Chinese Medicine.
In Vivo Nude Mice Xenograft Study
HT-29 cells were grown in culture and then detached by trypsinization, washed, and resuspended in serum-free DMEM. Cells (1.5 × 106) mixed with Matrigel (1:1) were subcutaneously injected in the right flank area of athymic nude mice to initiate tumor growth. After 5 days of xenograft implantation, mice were randomized into 2 groups (n = 10) and given intragastric administration with 2 g/kg of EESB or saline daily, 6 days a week for 16 days. Body weight and tumor size were measured. Tumor size was determined by measuring the major (L) and minor (W) diameter with a caliper. The tumor volume was calculated according to the following formula: tumor volume = π/6 × L × W2. At the end of the experiment, the animals were anaesthetized with diethyl ether, and tumors were excised and weighed.
Immunohistochemical Staining for PCNA
Tumor samples were fixed with 10% buffered formalin for 12 hours. Samples were then processed conventionally for paraffin-embedded tumor sections (5 µm thick). The tumor sections were then deparaffinized and rehydrated via treatment with a series of xylenes and graded alcohols. Endogenous peroxidase activity of the sections was quenched by incubating in 0.3% H2O2 in water for 30 minutes. Immunohistochemical staining was performed using the VECTASTAIN elite ABC kit according to the manufacturer’s instructions. After blocking nonspecific proteins with normal serum in PBS (0.1% Tween 20), the sections were treated with PCNA antibody (at a dilution of 1:200) for 1 hour. Then sections were incubated with a biotinylated anti-rabbit IgG antibody for 30 minutes and then treated with the ABC reagent for 30 minutes. They were finally treated with 3,3′-diaminobenzidine (DAB) for 10 minutes. After the sections were washed and air-dried, cover slips were applied to the sections using Permount slide mounting medium. PCNA was quantified by counting respective positive cells and total number of cells at 5 high-power fields (400×) were randomly selected in each slide, and the average proportion of positive cells in each field were counted using the true color multi-functional cell image analysis management system (Image-Pro Plus, Media Cybernetics, Rockville, MD).
In Situ Apoptosis Detection by TUNEL Staining
Formalin-fixed (12 hours) and paraffin-embedded sections of tumors (5 µm thick) were analyzed by TUNEL staining using apoptag peroxidase In situ apoptosis detection kit (R&D Systems) as per manufacturer’s information. Apoptotic cells were counted as DAB-positive cells (brown stained) at five arbitrarily selected microscopic fields at a magnification of 400×. TUNEL-positive cells were counted as a percentage of the total cells.
Bio-Plex Phosphoprotein Assay
The phosphorylation of Erk1/2 andp38 was detected by Bio-Plex Phosphoprotein Assay kit (Bio-Rad Laboratories, Hercules, CA) and Phosphoprotein Testing Reagent kit (Bio-Rad) according to the manufacturer’s protocols as described previously. 24 Briefly, 8 tumors were selected randomly from control or EESB group, homogenized in. a commercially available lysis kit (Bio-Rad) and centrifuged at 15 000 × g for 15 minutes. Protein concentrations of the clarified supernatants were determined by BCA protein assay. Twenty-five microliters of protein extract and 25 µL of testing assay buffer were transferred into 96-well filter plate coated with antibodies against phospho-Erk and phospho-p38 and then incubated overnight on a platform shaker at room temperature. After a series of washes to remove the unbound proteins, a mixture of biotinylated detection antibodies, each specific for a different epitope, was added to the reaction. Streptavidin–phycoerythrin was then added to bind to the biotinylated detection antibodies. Data were acquired and analyzed using the Bio-Plex 200 suspension array system (Bio-Rad). The total proteins for Erk1/2 and p38 were tested using the Bio-Plex total protein assay kit (Bio-Rad). The phosphorylation level was expressed as the ratio of phosphoprotein to total protein.
Preparation of Tumor Homogenate and Western Immunoblotting
Three tumors were selected randomly from control and EESB groups, homogenized in nondenaturing lysis buffer using Polytron, and centrifuged at 15 000 × g for 15 minutes followed by protein concentration determination in supernatants. Equal protein per lysate was resolved on Tris-glycine gel, transferred onto PVDF (polyvinylidine fluoride) membrane, and blocked for 2 hours with 5% nonfat dry milk. Membranes were incubated with desired primary antibody p-STAT3, total-STAT3 (STAT3), Bcl-2, Bax, p21, Cyclin D1, CDK4, and β-actin (at a dilution of 1:1000) overnight at 4°C and then with appropriate HRP-conjugated secondary antibody followed by enhanced chemiluminescence detection.
RNA Extraction and RT-PCR analysis
Total RNA was isolated from fresh tumor with Trizol reagent. Oligo(dT)-primed RNA (1 µg) was reverse transcribed with SuperScript II reverse transcriptase according to the manufacturer’s instructions. The obtained cDNA was used to determine the mRNA amount of Bcl-2, Bax, p21, Cyclin D1 and CDK4 by PCR with Taq DNA polymerase (Fermentas).GAPDH was used as an internal control. The primers used for amplification of Bcl-2, Bax, p21, Cyclin D1, CDK4, and GAPDH transcripts are as follows: Bcl-2 forward 5′-CAG CTG CAC CTG ACG CCCTT-3 and reverse 5′-GCC TCC GTT ATC CTG GAT CC-3′ (Tm = 55°C, 198 bp); Bax forward 5′-TGC TTC AGG GTT TCA TCC AGG-3′ and reverse 5′-TGG CAA AGT AGA AAA GGG CGA-3′ (Tm = 55°C, 276 bp); p21 forward 5′-GCG ACT GTG ATG CGC TAA TGG-3′ and reverse 5′-TAG AAA TCT GTC ATG CTG GTC TGC-3′ (Tm = 55°C, 328 bp); Cyclin D1 forward 5′-TGG ATG CTG GAG GTC TGC GAG GAA-3′ and reverse 5′-GGC TTC GAT CTG CTC CTG GCA GGC-3′ (Tm = 57°C, 573 bp); CDK4 forward 5′-CAT GTA GAC CAG GAC CTA AGC-3′ and reverse 5′-AAC TGG CGC ATC AGA TCC TAG-3′ (Tm = 58°C, 206 bp); GAPDH forward 5′-GT CAT CCA TGA CAA CTT TGG-3′ and reverse 5′-GA GCT TGA CAA AGT GGT CGT-3′ (Tm = 58°C, 240 bp). Samples were analyzed by gel electrophoresis (1.5% agarose). The DNA bands were examined using a Gel Documentation System (Model Gel Doc 2000, Bio-Rad, Hercules, CA).
Statistical Analysis
Data are presented as mean ± standard deviation. The data were analyzed using the SPSS package for Windows (Version 17.0). The statistical analysis between groups was carried out using the Student’s t test and P < .05 was considered to be statistically significant.
Results
EESB Inhibits HT-29 Tumor Xenograft Growth in Nude Mice
To study EESB efficacy against tumor growth, we compared the tumor weight and volume in treated and control CRC xenograft mice, while adverse effect of this agent was determined in the same mice by measuring the body weight change. As shown in Figure 2A and B, tumor volume and tumor weight per mouse was 0.27 ± 0.09 cm3 and 0.32 ± 0.11 g, respectively, in EESB-treated group; whereas that in control group was 0.72 ± 0.18 cm3 and 0.55 ± 0.14 g, respectively (P < .01). However, administration of EESB had no effect on the change of body weight during the course of the study (Figure 2C). Taken together, it is suggested that EESB is effective in suppressing colorectal tumor growth in vivo, without noticeable toxicity.

Effect of ethanol extract of Scutellaria barbata D Don (EESB) feeding on HT-29 colorectal cancer (CRC) xenograft growth and body gain in mice. After tumor development, the mice were given intragastric administration of 2 g/kg/d dose of EESB or saline daily, 6 days a week for 16 days. Tumor volume (cm3) per mouse was measured at different times (A), tumor weight (g) per mouse was measured at the end of study (B) and body weight (g) per mouse was measured at various times (C). Each value represents mean ± standard deviation (error bars) of 10 individual mice in each group. *P < .05, versus controls.
EESB Inhibits Tumor Cell Proliferation in CRC Xenograft Mice via Modulation of Cyclin D1, CDK4, and p21 Expression
The effect of EESB on cell proliferation in CRC tumors was determined via immunohistochemical staining (IHS) for PCNA, a proliferation marker specifically expressed in proliferating cell nuclei. As shown in Figure 3A, the percentage of PCNA-positive cells in CRC tumors from control and EESB-treated group was 37.83% ± 4.31% and 23.17% ± 4.79%, respectively (P < .01), suggesting that EESB inhibits tumor cell proliferation.

Effect of ethanol extract of Scutellaria barbata D Don (EESB) on cell proliferation and the expression of Cyclin D1, CDK4, and p21 in colorectal cancer (CRC) xenograft mice. (A) At the end of the study, tumor tissues were processed for immunohistochemical (IHC) staining for proliferating cell nuclear antigen (PCNA). The photographs are representative images taken at a magnification of 400×. Quantification of IHC assay was represented as percentage of positively stained cells. Data shown are mean ± standard deviation (error bars) of 6 individual mice in each group. *P < .05, versus controls. (B, C) The mRNA or protein expression levels of Cyclin D1, CDK4, and p21 were determined by reverse transcriptase–polymerase chain reaction (RT-PCR) and Western blot analyses. GAPDH and β-actin were used as the internal controls for the RT-PCR and Western blotting, respectively. Images are representatives of 3 individual mice in each group. (D, E) Quantitation of mRNA and protein expression was assayed by densitometric analysis. The data were normalized to the mean mRNA or protein expression of untreated control (100%). *P < .01, versus controls.
Eukaryotic cell proliferation is regulated by the cell cycle. G1 to S transition is 1 of the 2 main checkpoints for the control of cell cycle progress, which is tightly regulated by Cyclin D1, CDK4, and p21. To investigate the mechanism of EESB’s anti-proliferative action, we examined its effect on the expression of these cell cycle–regulatory factors. As shown in Figure 3B and C, EESB treatment profoundly reduced mRNA level of pro-proliferative Cyclin D1 and CDK4 in CRC tumors, whereas that of anti-proliferative p21 was significantly enhanced by EESB treatment. In addition, the pattern of protein expression of Cyclin D1, CDK4, and p21 was similar to their respective mRNA levels (Figure 3D and E).
EESB Induces Tumor Cell Apoptosis in CRC Xenograft Mice by Increasing the Pro-Apoptotic Bax/Bcl-2 Ratio
Apoptosis in tumors from CRC xenograft mice was examined via TUNEL assay. As shown in Figure 4A, the percentage of TUNEL-positive cells in tumor tissues from control and EESB-treated CRC mice was 22.0% ± 4.47% or 32.0% ± 7.32%, respectively (P < .01), demonstrating its pro-apoptotic activity in vivo. We next examined effect of EESB on the expression of Bcl-2 family proteins that are key regulators of apoptosis. As shown in Figure 4B to E, EESB significantly increased the ratio of pro-apoptotic Bax to anti-apoptotic Bcl-2, at both transcriptional and translational levels.

Effect of ethanol extract of Scutellaria barbata D Don (EESB) on cell apoptosis and the expression of Bcl-2 and Bax in colorectal cancer (CRC) xenograft mice. (A) At the end of the study, tumor tissues were processed for immunohistochemical staining (HIS) for TUNEL. The photographs are representative images taken at a magnification of 400×. Quantification of IHS assay was represented as percentage of positively stained cells. Data shown are averages with standard deviation (error bars) from 6 individual mice in each group (*P < .01, versus controls). (B, C) The mRNA or protein expression levels of Bcl-2 and Bax were determined by reverse transcriptase–polymerase chain reaction (RT-PCR) and Western blot analyses. GAPDH and β-actin were used as the internal controls for the RT-PCR and Western blotting, respectively. Images are representatives of 3 individual mice in each group. (D, E) Quantitation of mRNA and protein expression was assayed by densitometric analysis. The data were normalized to the mean mRNA or protein expression of untreated control (100%). *P < .01, versus controls.
EESB Suppresses Multiple Intracellular Signaling Transduction Pathways in CRC Xenograft Mice
STAT3 activation in CRC tumors was examined by Western Blot analysis using an antibody that recognizes STAT3 phosphorylation at Tyr705; and the activation (phosphoralytion) of Erk1/2 and p38 in tumor tissues was determined by Bio-Plex phosphoprotein assay. As shown in Figure 5, after EESB treatment the phosphorylation levels of STAT3, Erk1/2, and p38 in CRC tumors were decreased as compared with controls (P < .01), suggesting that EESB significantly suppresses the activation of multiple CRC-related signaling pathways.
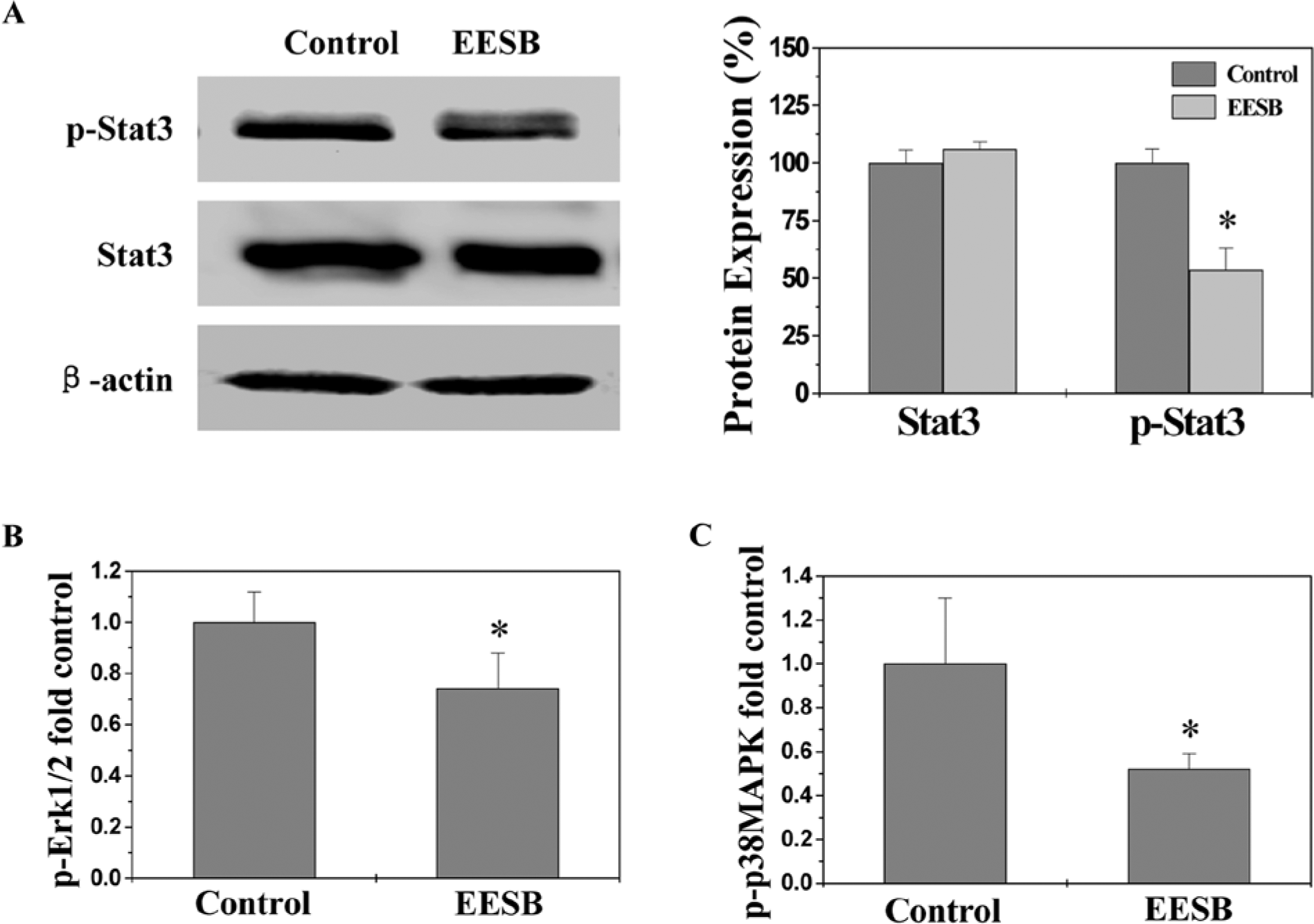
Effect of ethanol extract of Scutellaria barbata D Don (EESB) on phosphorylation of STAT3, Erk1/2, and p38 in colorectal cancer (CRC) xenograft mice. (A) The level of STAT3 phosphorylation in tumor tissues was determined by Western blot using an antibody that recognizes phosphorylated STAT3 at Tyr705 (left). Densitometric analysis was performed using Quantity One Analysis Software (Bio-Rad, Hercules, CA) for Western blot (right). (B, C) The phosphorylation levels of Erk1/2 and p38 in tumor tissues were determined by Bio-Plex phosphoprotein assay. The phosphorylation level was expressed as the ratio of phosphoprotein to total protein. The data were normalized to the phosphorylation level within controls and represented as fold of control. Data are averages with standard deviation (error bars) from 8 individual mice in each group. *P < .01, versus controls.
Discussion
Colorectal carcinoma is a serious global health problem with more than 1 million new cases and half a million deaths worldwide each year. The pathogenic mechanisms underlying CRC development are complex and heterogeneous, and involve multiple cellular signaling transduction cascades, including STAT3, Erk, and p38 pathways. These signaling pathways usually function redundantly and form a complicated cross-talking network. Given the complexity of CRC pathogenesis and progression, the currently used anti-CRC agents, which typically target a single intracellular pathway, might not always be effective. Moreover, long-term use of these agents often generates drug resistance and toxicity against normal cells. The problems increase the necessity for development of novel therapeutic agents. Traditional Chinese medicines (TCMs) are composed of many chemical compounds and thus are considered to be multicomponent and multitarget agents exerting their therapeutic function in a more holistic way. In fact, TCMs have been used in China for thousands of years as alternative remedies for a variety of diseases, including cancer.16,25 Moreover, TCMs have relatively few side effects.26,27 Therefore, TCMs have received recent interest as anticancer agents. SB, a well-known traditional Chinese folk medicine, has long been used to clinically treat various kinds of cancer. We previously reported that SB is able to induce cancer cell apoptosis and inhibit cell proliferation and tumor angiogenesis via modulation of several pathways, including Hedgehog, Akt, and p53. However, the precise mechanism of its potential tumoricidal activity remains to be further elucidated.
Cancer cells are characterized by an uncontrolled increase in cell proliferation and/or a reduction in cell apoptosis. Eukaryotic cell proliferation is primarily regulated by cell cycle. G1/S transition is 1 of the 2 main cell cycle checkpoints, which is responsible for initiation and completion of DNA replication. 28 G1/S progression is strongly regulated by Cyclin D1 that exerts its function via forming an active complex with its CDK major catalytic partners (CDK4/6). 29 An unchecked or hyperactivated Cyclin D1/CDK4 complex often leads to uncontrolled cell division and malignancy. 30 As a proliferation inhibitor, p21 protein plays a role in G1 arrest by binding to and inhibiting the activity of Cyclin–CDK complexes; and the decrease of p21 is associated with the promotion of tumor formation and a poor prognosis in many types of cancer. 31 Apoptosis is crucial for animal development and tissue homeostasis. Disturbed regulation of this vital process represents a major causative factor in the pathogenesis of cancers. Bcl-2 family proteins are key regulators of apoptosis, functioning as either suppressors or promoters. The ratio of active anti- and pro-apoptotic Bcl-2 family members determines the fate of cells, and alteration of the ratio by aberrant expression of these proteins impairs the normal apoptotic program contributing to various apoptosis-related diseases. Higher Bcl-2-to-Bax ratios are commonly found in various cancers, which not only confer a survival advantage to the cancer cells but also cause resistance to conventional chemotherapies. Therefore, re-balancing of cell apoptosis and proliferation via regulation of the expression of apoptotic- or cell cycle–related genes is a promising strategy for cancer therapies. Using a CRC mouse xenograft model, we demonstrated that EESB inhibited cancer growth via suppressing cancer cell proliferation and promoting apoptosis in tumor tissues. The pro-apoptotic and anti-proliferative activities of EESB were mediated by its effects on the expression of relevant genes. Administration of EESB profoundly increased the pro-apoptotic Bax/Bcl-2 ratio and the expression of anti-proliferative p21, whereas the expression of pro-proliferative Cyclin D1 and CDK4 was decreased after EESB treatment.
Cancer development is tightly regulated by multiple intracellular signaling pathways, such as STAT3, ERK, and p38 signalings. Aberrant activation of these pathways alters the expression of various critical target genes leading to the imbalance of cell proliferation and apoptosis. In the present study, we demonstrated that EESB significantly suppressed the activation of these cancer-related signaling pathways in tumor tissues.
In summary, here we demonstrate for the first time that SB possesses a broad range of antitumor activities because of its ability to affect multiple intracellular targets. Our findings suggest that SB could be a novel therapeutic agent for the treatment of colorectal and other cancers.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interests with respect to the authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: This study was sponsored by the National Natural Science Foundation of China (81073097) and the Natural Science Foundation of Fujian Province of China (2013J01333).
