Abstract
Introduction
Modern clinical genomic practices are now being applied for discovering targeted therapeutics against the “culprit key ring leader genes,” also called driver gene(s), 1 leading to dramatic enhancement of personalized medicine approach against specific cancers. However, the perceived success of targeted therapies still adds merely a few months of life for patients with late-stage cancers, particularly against aggressive metastatic tumors. The prospects of survival for patients with metastasis seem even murkier as adjuvant therapies developed to reduce metastasis have hit a plateau and new drugs are hard to come by. It seems imperative to translate the tumor biology knowledge that has been elucidated from the Cancer Genome Anatomy Project (CGAT) 2 and leverage this knowhow against metastasizing cells in designing improved treatment strategies. 3 Proper utilization of technological advances in understanding tumor biology could help expedite the process of targeted new drug discoveries that are safe and more effective against metastatic tumors.
The bottleneck in tackling metastasis, a complex biological process that involves dissemination of tumor initiating cells evading immune response to distant organs, displays remarkable tumor heterogeneity among patients. Key clinical genomic signatures observed also vary depending on the type and stage of cancer, disseminating distant organ site(s) and period of dormancy seen before clinical diagnosis of metastasis. In one such study 4 that included 41 different cancer types, it was found that breast cancer remains one of the most aggressive metastasizing malignancies and is usually recalcitrant to current therapies. 5 Detecting metastasis correlates with the size of primary tumor, 6 as large tumors tend to shed more cells into circulation, and the feat of metastasis quite often depends on the late-stage tumor’s growth kinetics. Cells having a knack for proliferation are more able to do so. 7 This could be due to genetic heterogeneity that tumors acquire during the course of their development, from benign to highly aggressive over a long period of time, which makes the targeted new drug discovery a challenge against the metastatic process.
Breast cancer is highly prevalent among women worldwide, particularly in developed countries and its high probability of metastasizing to distant organs 4 makes it one of the deadliest and most hard-to-treat malignancies. Metastasis in breast cancer patients remains a leading cause of death 8 and accounts for 15% of cancer related mortalities. 9 Because of lifestyle changes, the incidence of breast cancer is steadily increasing in the developing countries. 10
Our understanding of cellular signal transduction pathways that are differentially regulated in cancer has been of tremendous help in devising therapeutic strategies to target tumors. Nevertheless, cancer-related mortalities remain high, requiring urgency in the development of additional means of controlling cancer progression. Understanding the molecular biology of chemoprevention could help identify new and novel chemical entities that aid current therapeutic agents and might become standalone therapies as well. In this regard, traditional medicinal plants used for treating various ailments for centuries could be of immense therapeutic importance. Identification and purification of individual plant compounds as new chemical entities (NCEs) that inhibit signaling pathways of cancer progression and metastasis would certainly benefit and expedite the process of developing efficient treatments.11-15 Polyphenolic compound like epigallocatechin-3-gallate (EGCG), 15 fisetin, delphinidin, and lupeol derived from dietary plants have shown promising results as therapeutic agents.14,16-18 However, these compounds affect numerous signaling pathways 15 and the identification and characterization of key driver genes and the resultant signal transduction pathways affected by these compounds is necessary. 19
Rhazya stricta Decne (Apocynaceae) (RS), an excellent source of antioxidants, is used for treating numerous diseases in Asia.20-22 Although many studies in the 1980s and 1990s identified and characterized alkaloids (> 100), flavonoids, triterpenes, and volatile substances from this plant. These studies did not link the therapeutic effect of individual compounds to specific biochemical, physiological and toxicological parameters in experimental animal models.20,21 Alkaloids present in herbs exhibited antiproliferative and anti-metastatic properties in both in vitro and in vivo studies. 23 In recent decades, alkaloids such as camptothecin 24 and Vinca alkaloids from Catharanthus roseus (L.) G.Don (Apocynaceae) 25 were used as anticancer agents. Similarly, alkaloids from R. stricta exhibit antimicrobial26,27 and anticancer activities. 20 Total alkaloidal extracts from RS downregulated the proliferation of the lung cancer cell line A549, 28 partly by inducing the expression of NQO1, a chemopreventive enzyme. 29 Elucidating key gene(s) involved in the signal transduction pathways in normal and cancerous cell lines and how their inhibition in breast cancer affects metastasis is one of the possible ways to design safe and effective breast cancer therapeutics. In this study, we have isolated and characterized individual compounds and total extracts from RS by nuclear magnetic resonance (NMR) structure, attenuated total reflectance–Fourier transform infrared spectroscopy (ATR-FTIR) spectra chemical studies. Then we examined one of the newly characterized active compounds, rhazyaminine (R.A), for its physiological, cellular, and molecular effects on the breast cancer cell line-MCF7, using the scratch assay, immunostaining, RNA-seq, and quantitative real-time polymerase chain reaction (qRT-PCR) to delineate its gene expression signatures and work out its specific mechanism of action.
Materials and methods
Bioinformatics Analysis
Detailed bioinformatics analyses were performed as discussed previously. 30 Briefly, Fastq files obtained from European Nucleotide Archive (ENA) were mapped to human reference genome (UCSC hg38) using HISAT2.31,32 The resulting files were sorted and converted into BAM files using SAMtools, 33 followed by annotation and abundance estimation using Stringtie. 34 Differential gene expression analyses and visualization were performed using the Ballgown package in R-studio.35-37
Extraction and Isolation of Plant Alkaloids
RS plants collected in September 2015 from the Bahrah region, located toward the east of Jeddah, Saudi Arabia, were identified by our departmental taxonomist (Dr. Khalid Hakeem) and submitted to our herbarium in the Department of Biology, King Abdulaziz University, Jeddah, Saudi Arabia. These plants are also available from Prof. Robert K. Jensen, Department of Integrative Biology, University of Texas at Austin, Austin, TX, USA.
Alcoholic Extract and Purification of RA From R stricta
Leaves of RS were dried in the absence of light. Powder obtained (1 kg), was crushed and extracted thrice by chloroform:ethanol (1:2) at a room temperature. The extract was further concentrated under low pressure obtaining dark brown residue (102 g). This was later dissolved in water containing 2% HCl and reextracted by chloroform removing nonbasic compounds. Ammonium hydroxide was used to neutralize aqueous layer and further extracted by chloroform. The chloroform extract was dried over anhydrous sodium sulfate, filtered, and evaporated using vacuum rotatory evaporator, obtaining 20.88 g of crude alkaloidal extract (TERS). Purification of individual alkaloids was performed using aluminum oxide column (800 g, 80 × 2.5 cm) and equilibrated with chloroform. Thin layer chromatography (TLC) and preparative thin layer silica gel chromatography (PTCL) were performed to obtain the purified individual indole-alkaloid, R.A.
UV-VIS, ATR-FTIR, and NMR analyses
Total extract obtained was analyzed using ultraviolet-visible (UV-VIS) and FTIR spectrophotometers for proximal analysis. Sample was diluted 1:10 in dimethyl sulfoxide (DMSO) and scanned at 200 to 700 nm for UV-VIS analysis (Evolution 300, Thermo Fisher Scientific). Afterward, to decipher peaks and their corresponding functional groups, FTIR analysis was performed on the same samples using ATR technique (Thermoscientific ATR-FTIR instrument). 1 H NMR: (400 MHz) and 13 CNMR 100 MHz) were further used to characterize the sample for the detailed structural analysis. The sample was first dried under vacuum and the powder obtained was dissolved in deuterated DMSO and used for further analysis.
Cell Line and Culture Conditions
MCF7, a breast cancer cell line, was obtained from King Fahd Medical Research Center, King Abdualaziz University, Jeddah, Saudi Arabia. Cells were cultured and maintained in Dulbecco’s modified Eagle medium (DMEM-high), containing 10% fetal bovine serum (FBS) and 1% PS (penicillin and streptomycin) in a humidified incubator at 37°C with 5% CO2.
Cell Viability Assay
MCF7 cells plated in 96-well plates at a density of 5 × 103 cells/well were incubated overnight, at 37°C with 5% CO2. These cells were treated with 0, 25, and 50 μg/mL of TERS and R.A, respectively, for 0, 24, and 48 hours. Afterward, 100 μL of fresh media were added to each well. The methyl-thiazole tetrazolium (MTT) assay was performed, as per the instructions provided by the manufacturer. In brief, cells were incubated with 10 μL of MTT solution for 4 hours. Later, 50 μL of DMSO were added to 25 μL of media in each well, followed by homogenization thoroughly and by pipetting vigorously. Subsequently, plates were incubated at 37°C for 10 minutes. The sample in each well was mixed again and absorbance was checked at 540 nm with microplate reader (SYNERGY|HTX multimode reader, BioTek).
Immunofluorescence Staining to Observe Apoptosis
Cells were cultured in 6-well plates and treated with 0 and 50 μg/mL of TERS and R.A, respectively, for 24 hours. The latter cells were washed with cold PBS (1×), 3 times and incubated with 10% formaldehyde for 20 to 30 minutes at 37°C, and again washed with PBS thrice and incubated with 0.1% Triton X 100 for 3 to 5 minutes at room temperature. Cells were washed 3 times with PBS followed by staining with Hoechst 33258 stain for 15 to 30 minutes at 37°C. Stain was removed and cells were washed with PBS 3 times and observed under inverted immunofluorescence microscope using DAPI filter (EX-350-410 nm, DM460 nm BA460-480 nm).
Scratch/Wound Healing Assay
Cells cultured in 6-well plates at a density of 5 × 105 cells/well were allowed to form a confluent monolayer for 24 hours. A scratch was made using 10-μL pipette tip in each well and washed with 1× PBS to remove cell debris. Fresh media added to each well, followed by the addition of 50 μg/mL TERS and R.A. Pictures were taken at 0, 24, and 48 hours to observe cell migration/wound closure in the presence and absence of TERS and R.A using inverted microscope.
RNA-seq Analysis
MCF7 cells (1.5 × 105 cells) were exposed to sublethal dose of purified R.A, that is, 50 μg/mL, in triplicates, for 24 hours at 37°C. Total RNA was isolated from drug-treated cells using TRIzol reagent and RNA-seq experiment was carried out using Illumina HiSeq. RNA was ribo-depleted (Ribo-Zero, Epicentre) before preparing cDNA library (ScriptSeq epicentre). Each cDNA was sequenced in multiplex generating >40 million 100 bp reads per sample. The Illumina reads were mapped to the assembled reference transcriptome sequence using the software TopHat and Cufflinks. 38 Differentially expressed genes were identified and functionally annotated. Clusters of differentially expressed transcripts were annotated to identify GO/KEGG pathways. 39 Genes were considered significantly upregulated in alkaloid-treated MCF7 cells in comparison with DMSO-treated control cells if log2 fold change >1; P value <.05 and genes were considered significantly downregulated if log2 fold change <−1; P value <.05 in alkaloid-treated MCF7 cells as compared with DMSO control.
RNA Extraction and qPCR Analysis
Cells were grown in 6-well plates at a density of 5 × 105 and were treated with control and 50 μg/mL of TERS and R.A for 24 hours. mRNA was extracted using Trizol reagent (Invitrogen). The pellet was dissolved in 50 μL RNase-free water and quantified using NanoDrop. A total of 1 µg/µL of RNA was used to prepare cDNA. qPCR was performed (Bio-Rad, CFX Connect, Real-Time System) with SYBR green master mix to analyze the expression of Bcl-2 gene. The conditions used were as following. Initial activation at 98°C for 30 seconds followed by 39 cycles of 98°C for 30 seconds and 60°C for 30 seconds. The sequences of primers used are described in Supplementary Table 1 (available in the online version of the article).
Statistical Analysis
All the data are represented as mean ± standard error. Significance was analyzed using one-way analysis of variance test. P < .05 was considered significant.
Results
Differential Expression Comparison Between MCF10A and MCF7
To evaluate the differential expression of genes between MCF10A and MCF7 cell lines, RNA-seq data was downloaded from ENA and analyzed, as described earlier. 30 A total of 935 genes with at least 2-fold differential expression were identified (Supplementary Table 2, available in the online version of the article). A scatterplot showing the distribution of differentially expressed genes (log2) depicts the significant genes as blue dots (Supplementary Figure 1a, available in the online version of the article). Multidimensional scaling (MDS) shows the relationship between samples. Samples closely correlated are grouped together. MCF10A samples, colored in red, and MCF7 samples, colored in blue on the MDS plot, are seen clustering in close proximity, respectively (Supplementary Figure 1b). In addition, we observed the distribution of differentially expressed genes; the histogram between dashed lines represents genes that are 4-fold differentially expressed (Supplementary Figure 1c). A heatmap of log2 (FC > 2) genes between the 2 datasets shows the gene expression differences between epithelial cell line (MCF10A) and MCF7 (Supplementary Figure 1d).
Isolation and Structural Determination of Individual Alkaloid
Total extract and individual alkaloids were isolated, and fractionated as discussed, under the Materials and Methods section. The structure of individual alkaloid-R.A was determined, and further validated by the NMR and FTIR spectroscopic studies, as shown in Figure 1.

Nuclear magnetic resonance (NMR) structure, attenuated total reflectance–Fourier transform infrared (ATR-FTIR) spectra of rhazyaminine/dimethyl sulfoxide (R.A/DMSO) solution and blank DMSO: (a) Structure of isolated compound. (b) Structure of DMSO used to dissolve the extracted compound. (c). The FTIR spectra of the samples were obtained for the compound’s identification. ATR-FTIR spectra of R.A (red) in DMSO and blank DMSO (blue). In the FTIR spectrum of DMSO, the two small peaks located at 2910 cm−1 were due to the C-H asymmetric stretching vibrations. Other peaks at 1300, 1200, and 1000 cm−1 represent –CH3 bending and S-O stretching vibrations. In addition to the presence of these peaks, two new peaks appeared at the 3400 and 1600 cm−1 in the FTIR spectrum of R.A/DMSO solution. The broad peak at 3400 cm−1 is due to the N-H asymmetric stretching and at 1629 cm−1 is due to the C=O ester group. The aromatic C-H wagging and twisting vibrations are confirmed by the peak locks located below 1000 cm−1. (d) Ultraviolet-visible (UV-VIS) spectral data of R.A/DMSO with different dilutions. The visual appearance of R.A/DMSO solution was a colorless and nearly transparent liquid. In all the 2 UV-VIS spectra of the samples, a single peak at 200 nm was observed which is due to the electronic transition from the n to π*. Moreover, the peak’s intensity increased with increasing the R.A amount in the R.A/DMSO obeying the Beer-Lambert’s law. No other peak in the visible region of the spectra was identified, which represents that the solution was colorless and transparent in nature.
Effect of Crude Extract and Isolated Compound on Cell Viability
The effectiveness of crude extract and individual alkaloid from RS was investigated at different concentrations on MCF7 cell line. Cytotoxicity of crude extract has already been reported by our departmental colleague. 28 In this study, evaluation of cytotoxicity of R.A in comparison to TERS was performed as outlined below. After investigating the optimum dosage for MTT assay (data not shown here), two doses (25 and 50 µg) were selected for TERS and R.A, respectively. MTT assay was performed to assess the cellular cytotoxicity. The assay indirectly measures cell growth by observing the amount of tetrazolium compound converted into colored formazan whose absorbance was measured at the end of the assay. Both TERS and R.A showed cellular cytotoxicity toward MCF7 cell line in a dose and time dependent manner, as shown in Figure 2.
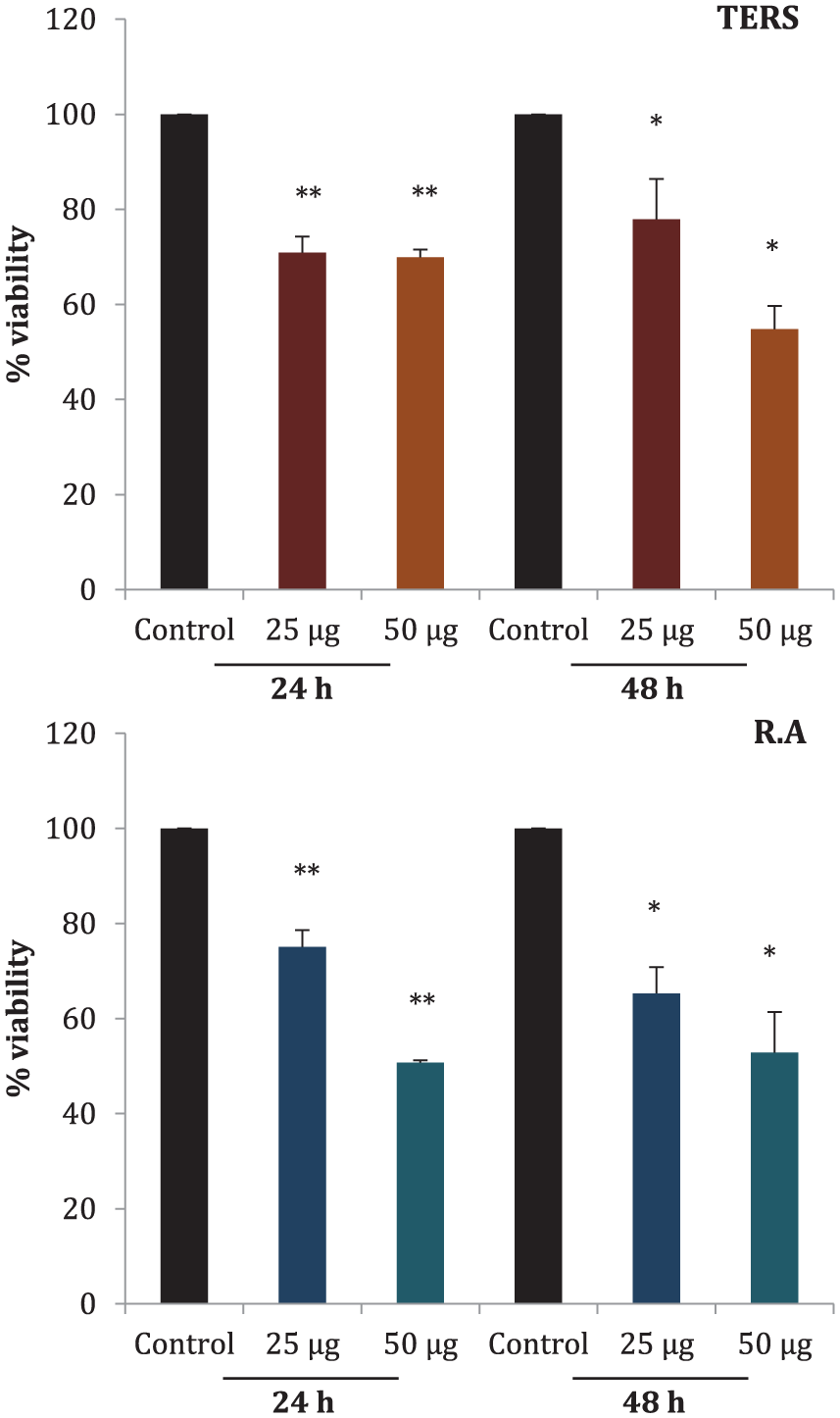
Cytotoxicty of total extract of Rhazya stricta (TERS) and rhazyaminine (R.A). Breast cancer cells, MCF7, were treated with 0, 25, and 50 μg/mL TERS and R.A for 24 and 48 hours. Percentage of relative cell viability was evaluated compared with control (0 μg/mL). All data presented as mean ± SEM. Data were analyzed by one-way analysis of variance where * indicates significance (P < .05) and ** indicates high significance (P < .001).
Induction of Apoptosis in MCF7 Cell Line
Apoptosis in MCF7 cell line by TERS and R.A was observed under the fluorescence microscope. Cells were treated with TERS and R.A for 24 hours, and later stained with the Hoechst 33258 dye. This stain binds to A-T rich regions in DNA and emits blue fluorescence that could be observed under a 4′,6-diamidino-2-phenylindole DAPI filter. Normal cells emit uniform fluorescence while apoptotic cells emit bright fluorescence due to the densely stained condensed genomic DNA. Induction of apoptosis was observed in MCF7 cell lines treated with TERS and R.A. Emission of bright blue fluorescence was observed in both TERS- and R.A-treated cells (Figure 3). Our data indicate the induction of apoptosis in MCF7 cells by both crude extract and individual alkaloid-R.A.

Cells were cultured in 24-well plates and treated with 0 and 50 µg/mL of total extract of Rhazya stricta (TERS) and rhazyaminine (R.A) for 24 hours. Cells were stained with Hoechst 33258 and observed under immunofluorescence microscope (20× magnification, 50 µm scale bars).
Inhibition of Cell Migration
Scratch assay is a technique used to measure cell migration in vitro 40 where a scratch is made in a cell monolayer and images are captured at different time points during cell migration (wound healing). Images were compared to evaluate migration inhibition by TERS and R.A at 24 and 48 hours. Scratch was made in monolayer of confluent MCF7 cells. Cells were exposed to 0 and 50 µg of TERS and R.A for 24 and 48 hours. Both TERS and R.A impeded wound healing (migration) of MFC7 cell line compared with control (Figures 4 and 5).

To evaluate wound closure, a vertical scratch was made with 10-µL pipette tip in confluent monolayer of MCF7 cell line. Cells were exposed to 0 and 50 µg/mL of total extract of Rhazya stricta (TERS) and rhazyaminine (R.A) and wound closure was observed at 24 and 48 hours. Images were taken at 5× magnification.
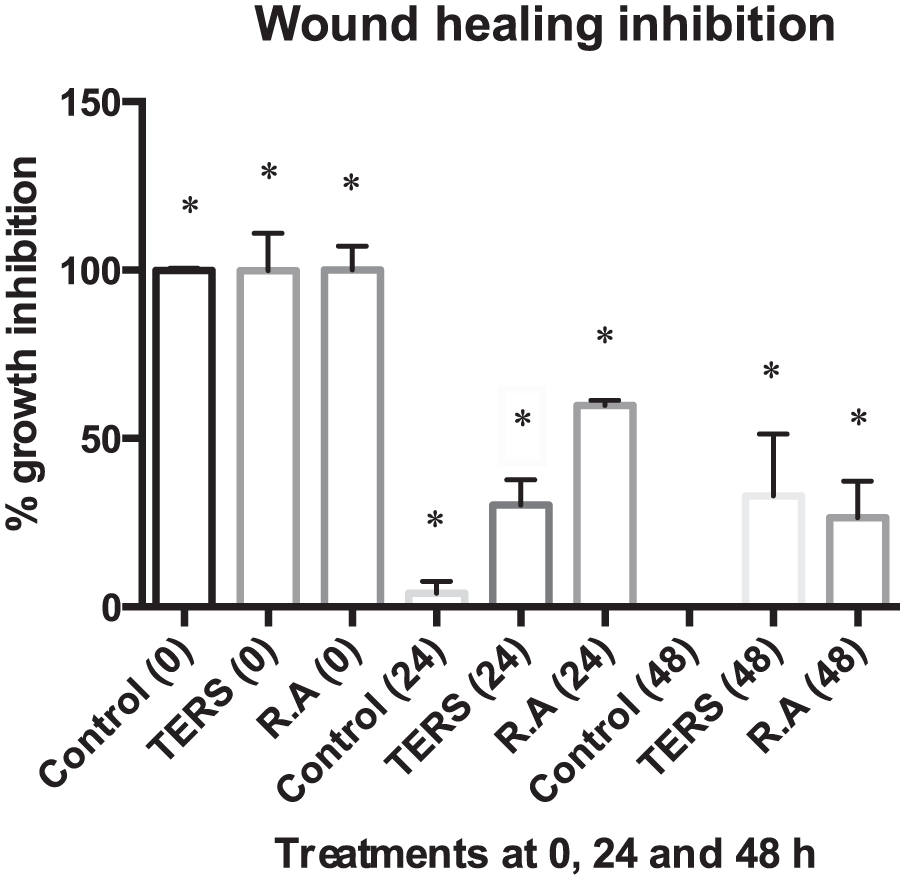
MCF7 cell lines were treated with 50 µg/mL total extract of Rhazya stricta (TERS) and rhazyaminine (R.A) for 24 and 48 hours and percentage of migration inhibition was calculated using ImageJ software. Both TERS and R.A successfully managed to inhibit MCF7 movement even after 48 hours of treatment.
Regulation of the Bcl-2 Gene
Real-time PCR was performed to evaluate the quantitative expression of Bcl-2 gene in MCF7 cell line after treatment with TERS and R.A for 24 hours. Both TERS and R.A reduced the expression of Bcl-2 gene in MCF7 cell line (Figure 6).

MCF7 cell line was treated with 50 µg/mL total extract of Rhazya stricta (TERS) and rhazyaminine (R.A) for 24 hours and quantitative polymerase chain reaction (qPCR) was performed. Analysis showing fold change in expression of bcl-2 gene after treatment with TERS and R.A. All results were determined by one-way analysis of variance (mean ± SEM, n = 3). * indicates P < .05.
Differential Expression of Metastasis-Related Genes
MCF7 cell line was treated with 50 µg/mL of R.A for 24 hours and total RNA was extracted. RNA-seq was performed and R.A-treated MCF7 samples were compared with control samples. A total of 43 genes were log2 differentially expressed between control and R.A-treated cells (Supplementary Table 3, available in the online version of the article). We further analyzed the role of differentially expressed genes to elucidate their specific role in the regulation of metastasis. We searched the role of all the 43 genes against articles present in Google Scholar and PubMed library. Thirteen of the genes downregulated by R.A treatment had a direct or indirect role in metastasis (Table 1). A schematic representation of these genes and their role in metastasis is shown in Figure 7.
List of Differentially Expressed Genes: Differential Expression of Metastasis-Related Genes in Untreated Control and R.A-Treated MCF7 Cell Line.
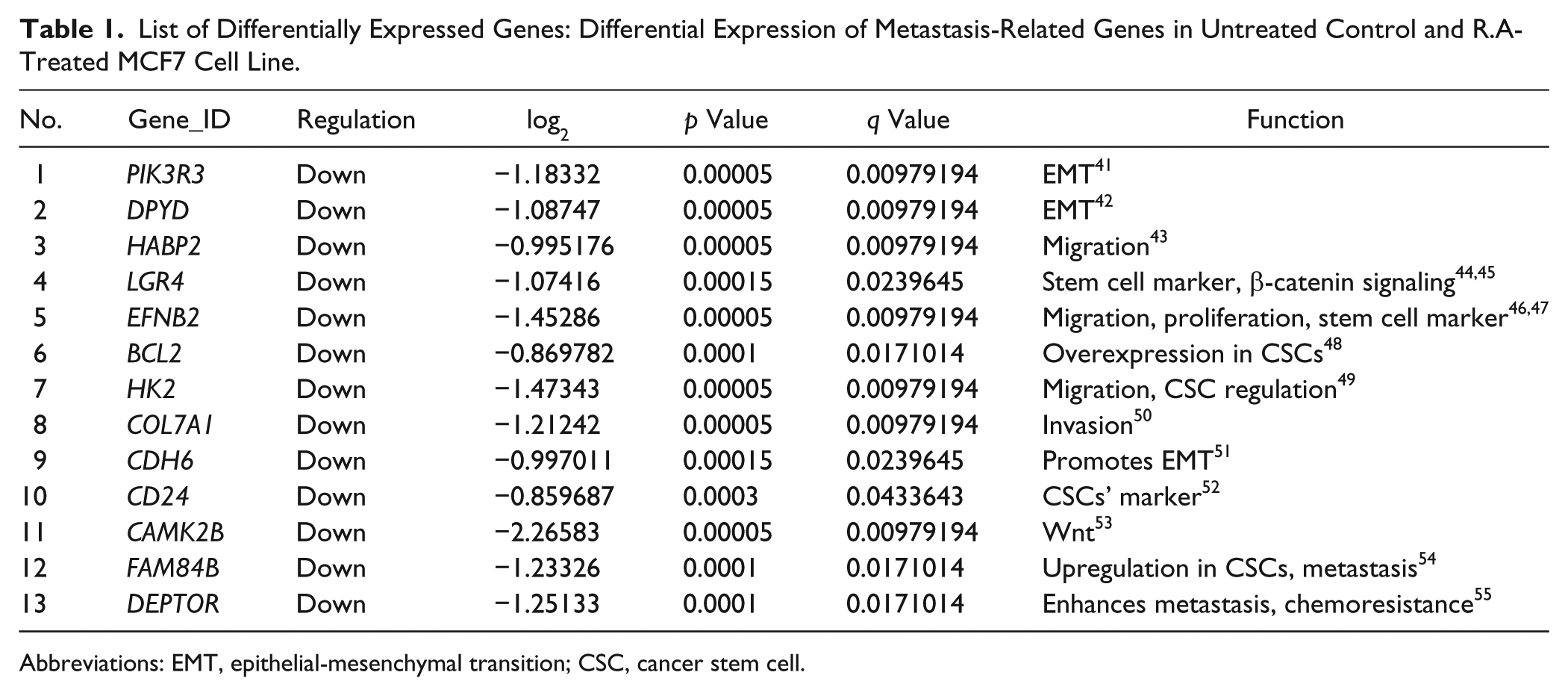
Abbreviations: EMT, epithelial-mesenchymal transition; CSC, cancer stem cell.

A schematic representation of genes inhibited and their role in abetting metastasis: Rhazyaminine (R.A) inhibited a number of genes that have been previously documented to play role in epithelial-mesenchymal transition (EMT), migration of cancer cells to secondary sites, invasiveness and proliferation of cancer stem cells (CSCs).
Discussion
Alkaloids derived from different plant sources have proven efficacy in suppressing various genes involved in the development of oncogenesis. This is because of their multifaceted role in modulating signal transduction pathways of cell proliferation, apoptosis and metastasis. 56 The widely used drug paclitaxel was derived from the bark of Taxus brevifolia Nutt (Taxaceae) and is currently employed in the treatment of various malignancies, such as breast, lung, and ovarian cancer.
Our study has shown the effectiveness of TERS and R.A on cellular cytotoxicity. Both TERS and R.A proved cytotoxic (Figure 2) and were effective in inducing apoptosis as DNA condensation was observed in Hoechst-stained cells after 24 hours of treatment (Figure 3). Further investigations into the antimetastatic potential of both TERS and R.A in a scratch assay on MCF7 cells after 24 and 48 hours of treatment showed clear reduction in the wound-healing ability (Figure 4). Comparative bioinformatics analyses, between MCF10A and MCF7, helped us elucidate and establish differential gene expression patterns, which are the key determinants of cancer progression. In RNA-seq data analyses, we found numerous genes deregulated by R.A (Supplementary Table 3, available in the online version of the article), which could be of interest to us for targeted drug discovery, as the regulation of these genes could have significant influence in abetting metastasis (Table 1). Another major finding was the downregulation of Deptor (DEP domain-containing mTOR-interacting protein) having role in metastasis and chemoresistance 55 by R.A (Table 1), which is one of the 935 genes upregulated in MCF7 cell line compared with MCF10A (Supplementary Table 2). We found 13 genes associated with metastasis or metastatic cancer stem cells (mCSCs). Epithelial-mesenchymal transition (EMT), a process during which cells in the epithelial state undergo transition to the mesenchymal (fibroblast-like) state, is crucial for cancer metastasis. 57 We discovered that PIK3R3, DPYD, and CDH6, key genes involved in EMT, were downregulated by R.A. HABP2, EFNB2, COL7A1, CD24, FAM84B, and HK2 involved in migration/invasion and proliferation of CSCs were also downregulated.
Another key aspect of metastasis is the upregulation of Wnt/β-catenin pathway, 58 and its inhibition could block metastatic process by influencing breast CSCs. 59 We observed that R.A treatment downregulated two genes, LGR4 and CAMK2B, which are key players in the Wnt/β-catenin pathway. Another major finding was the downregulation of the Bcl-2 gene (Figure 6), an antiapoptotic member of the Bcl-2 gene family involved in migration, invasion, and metastasis.60,61 Bcl-2 upregulation is also associated with increased survival in cancer cells. 62 As already evident from various studies, the Bcl-2 gene has diverse roles in tumor initiation, maintenance, progression, invasion, apoptosis and metastasis.60,61,63 Almost 75% of primary breast tumors exhibit elevated levels of Bcl-2 expression.64-66 Clearly, the development of numerous new drugs targeting the BH3 domain of Bcl-2 67 has been central to curbing cancer. 68 In addition, alkaloids such as scutebarbatine A (SBT-A), isolated from Scutellaria barbata D. Don (Lamiaceae), and subditine isolated from Nauclea subdita Steud (Rubiaceae) have also been reported to induce mitochondrial-mediated apoptosis by the downregulating Bcl-2 gene.69,70 The therapeutic potential of the Bcl-2 family, consisting of 25 member proteins, in the regulation of apoptosis and tumorigenesis has been under active investigation; their role in metastasis, however, still remains elusive. siRNA knockdown or upregulation of Mcl-1, Bcl-2 or Bcl-xL in colorectal cancer cell lines (HT29 and SW480) substantially decreased migration and invasion. The effect was increasingly profound for Bcl-2 gene. 60 Furthermore, Bcl-2 overexpression in MCF7 ADR, human breast cancer cell line, not only enhances tumorigenicity but also reduces latency period, leading to lung metastases.71,72 This demonstrates the multifaceted role that Bcl-2 plays and its importance as a plausible small molecule drug target.
Conclusion
A comparative bioinformatics study of MCF7 and MCF10A cell lines led us to elucidate a number of key genes central to breast cancer and metastasis. This gave us an insight on gene expression signatures, which could be exploited for new drug discovery research. Having molecular signature–based knowledge in hand we isolated and characterized RA, a new chemical entity from R stricta. R.A downregulated Bcl-2 gene and modulated several genes crucial to metastasis and cancer stem cells. Further studies in NCI-60 panel of human tumor cell lines for establishing the anticancer and antimetastatic potential of R.A are warranted.
Supplemental Material
Supplementary_material__revised – Supplemental material for Rhazyaminine from Rhazya stricta Inhibits Metastasis and Induces Apoptosis by Downregulating Bcl-2 Gene in MCF7 Cell Line
Supplemental material, Supplementary_material__revised for Rhazyaminine from Rhazya stricta Inhibits Metastasis and Induces Apoptosis by Downregulating Bcl-2 Gene in MCF7 Cell Line by Waqas Iqbal, Saleh Alkarim, Tahseen Kamal, Hani Choudhry, Jamal Sabir, Roop S. Bora and Kulvinder S. Saini in Integrative Cancer Therapies
Footnotes
Acknowledgements
We are grateful to Dr. Ayman I. Elkady, our colleague from the Department of Biology, King Abdulaziz University, Jeddah, for his valuable assistance, guidance, and unflagging support on numerous occasions. We are indebted to the staff in High Performance Computing Center, King Abdulaziz University for allowing us access to the Aziz supercomputer. We also thank Dr. Khalid Hakeem of Plant Sciences Section, Department of Biology for inspecting plant material.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was partially funded by Deanship of Scientific Research (DSR) at King Abdulaziz University (KAU), Jeddah, Saudi Arabia represented by the Unit of Strategic Technologies Research through the Project No. (D 008/431) titled “Identification and Isolation of Salt and Heat-Tolerance Genes of R. stricta and Detection of Metabolites and Their Therapeutic Effects via Cheminformatics.”
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
