Abstract
BACKGROUND:
p16 has often been found to be overexpressed in patients oral squamous cell carcinoma (OSCC), but its prognostic value between anatomic subsites is still unclear.
OBJECTIVE:
The aim of this study was to investigate the diagnostic and prognostic values of p16 in OSCC originating from tongue, gingiva or buccal mucosa.
METHODS:
A total of 147 OSCC patients with tumors arising from the tongue, gingiva or buccal mucosa were enrolled in this study. p16 expression was detected using immunohistochemistry (IHC), and the presence of HPV16 was determined by real-time PCR in p16 positive patients. The correlation of p16 expression with the clinical parameters was evaluated.
RESULTS:
Only one p16 positive patient with a cut off value of 25% and 75% was HPV16 positive. Although overall survival (OS), recurrence free survival (RFS) and metastasis free survival (MFS) had no significant differences between the p16 positive and negative patients, p16 negative patients (cut off value 25%) had more RFS in the buccal mucosa cancer (
CONCLUSIONS:
The prevalence of HPV16 in Chinese OSCC patients was low. p16 overexpression decoupled from HPV infection was not a prognostic marker for OSCC patients except for patients with the buccal mucosa cancer.
Introduction
Oral squamous cell carcinoma (OSCC) is one of the most common cancers affecting the oral cavity, with a recent increase in prevalence [1, 2]. Traditionally, oral cancers are broadly defined as carcinomas originating in the oral and oropharyngeal subsites. However, the oral cavity comprises different subsites, including the lips, gingiva and retromolar trigone, and cancers originating from different subsites behave differently. For example, tongue cancers appear to grow faster than cancers at other sites of the oral cavity and more frequently metastasize to the cervical lymph nodes, with a poorer prognosis [3]. As proteins and miRNAs are diversely expressed in different anatomic subsites in OSCC, biomarkers and treatments should vary at different subsites, which highlights the importance of a precise anatomic subsite classification in OSCC diagnosis [3, 4, 5, 6, 7].
OSCC development is influenced by lifestyle and exogenous factors, including smoking, alcohol consumption and high-risk human papillomavirus (HR-HPV) infection. Among them, HR-HPV has been reported to be a risky etiologic agent in some types of head and neck cancer patients, especially in oropharyngeal squamous cell carcinoma (OPSCC) patients [8]. Mechanisms of how HPV causes cancer have been investigated [9], with E6 and E7 proteins of HPV reported to be the main contributors to cancer [10]. The E6 protein stimulates the degradation of the tumor suppressor p53 protein, which interferes with cell cycle regulation and leads to tumor cell growth. In addition, the binding between the E7 protein and pRb leads to the transcriptional activator E2F constitutionally active, and a release of p16 from negative feedback of free pRb. Overexpression of p16 ensues. Thus, the immunohistochemical detection of p16 is often used as a surrogate marker for HPV oncogenic activity, especially in OPSCC [11]. However, overexpression of p16 has been reported to not always be associated with the presence of HPV in none-OPSCC, and the independent prognostic role of p16 expression decoupled from HPV is still unclear, especially in OSCC patients [12, 13, 14]. Additionally, although it is widely accepted that over 70% or more positive staining of p16 in tumors has predictive value for identifying HPV-related cancer in OPSCC, no unambiguous conclusions have been drawn for other head and neck sites outside the oropharynx [15]. It has been reported that a 25% cut off value for p16 positivity is used in the different HNC subsites [16]. Therefore, the discordant definition of p16 also makes the diagnostic and prognostic value of p16 confusing.
Therefore, in this study we investigated the expression and prognostic value of p16 with two reported cut off values in 147 OSCC patients with tumors originating from different anatomic subsites [17, 18]. In addition, we tested HPV16 DNA in p16 positive samples to determine the correlation of p16 expression and the presence of HPV16 in OSCC patients.
Material and methods
Study population and design
Samples and the follow-up data of 147 eligible histopathologically confirmed OSCC patients were obtained from the head and neck tumor bio-bank of Nanjing Stomatological Hospital, Medical school of Nanjing University. All the enrolled patients underwent surgeries in the department of Oral and Maxillofacial Surgery, Nanjing Stomatological hospital between February 1, 2013 and November 13, 2014. Specifically, the OSCC primary tumors were removed with adequate surgical margins, which were sent for histopathology analysis. If margins were not deemed to be tumor free, further resection was performed. A total of 136/147 (92.5%) of patients received follow-up examinations every two months after surgery until death. Follow-up visits continued until February 9, 2018, and only 7.5% (11/147) of patients did not attend the follow-up appointment because of an incorrect phone number or address.
This study was conducted according to the ethical principles of the Helsinki Declaration and approved by Nanjing Stomatological Hospital ethics committee (date of approval, 26th February 2018; approval number 2018NL-022 (KS)).
p16 immunohistochemistry
Paraffin sections (4-
DNA extraction from FFPE tissues
Samples that met the criteria for inclusion were then re-cut into 10-
Real-time PCR
All samples were run in triplicate in a 10-
Sequence of HPV16 E6/E7 and
-globin primers used for real-time PCR
Sequence of HPV16 E6/E7 and
Baseline characteristics of OSCC patients (
Clinical characteristics of 147 cases of oral squamous cell carcinoma according to p16 expression
Representative immunohistochemical micrographs of p16 expression. (A, F) absence of p16 expression; (B, G) p16 positive cells less than 5%; (C, H) p16 positive cells between 5% and 25%; (D, I) p16 positive cells more than 25%; (E, J) p16 positive cells more than 75%; Original magnification: 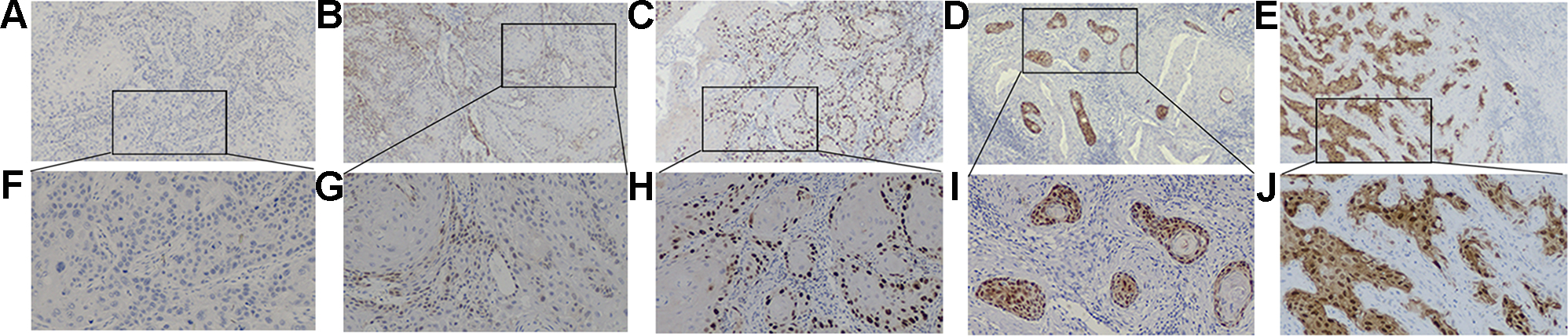
Correlation of p16 expression to the clinical parameters was evaluated with the chi-square test. OS was defined as the time from the date of surgery to the date of death or the date of censorship. RFS was defined as the time of surgery to the date of recurrence and relapse or the date of censorship. MFS was defined as the time of surgery to the date of metastasis or the date of censorship. The OS, RFS and MFS were estimated with Kaplan-Meier methods for all patients and subpopulations with different anatomic subsites. Log rank tests were performed to test the differences in survival between p16-positive and p16-negative patients with 25% and 75% cut off values, which were previously used to determine p16 positivity. Univariate and multivariate analyses of all patients using Cox proportional hazards regression were performed using the following variables: age, primary tumor location, TNM, differentiation, metastasis, recurrence and p16 positivity. In all statistical tests, a
Real-time PCR for HPV DNA loads. (A) The standard curves for HPV 16 E6 and E6/E7 genes using 10-fold diluted DNA of Caski cells. Data are mean 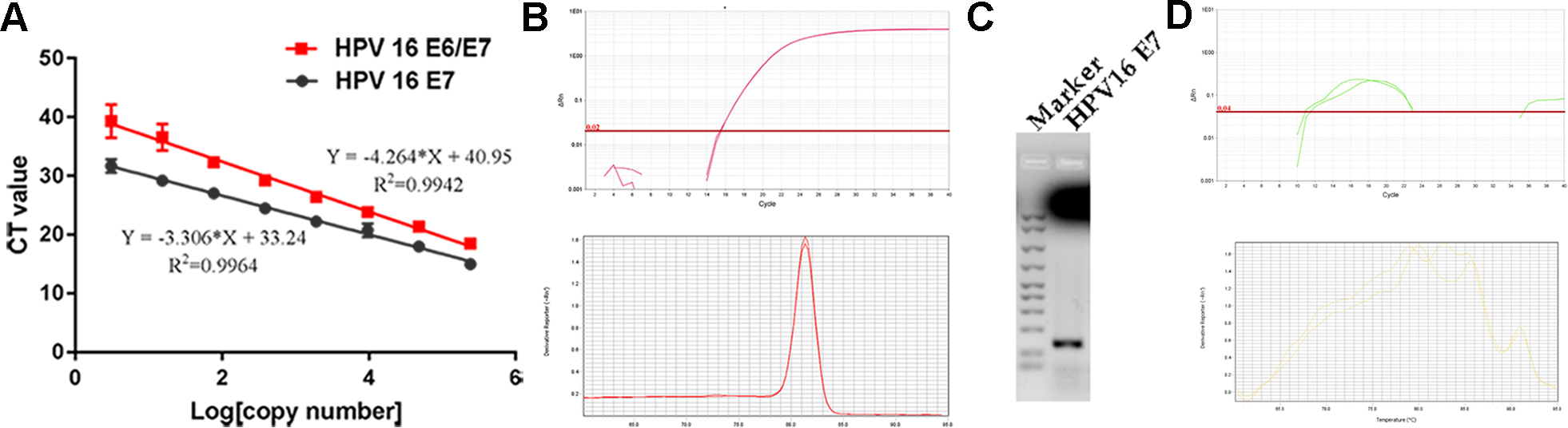
Clinicopathological characteristics of patients with OSCC
147 OSCC patients, including primary sites in the tongue (
Prevalence of HPV16 was rare in OSCC patients
Although p16 expression has been regarded as a surrogate marker for HR-HPV in OPSCC, its diagnostic value in OSCC is still confusing because of the diverse anatomical tumor subsites and discordant definition of p16 positivity. Therefore, real-time PCR was performed to detect HPV16 E6/E7 and E7 DNA using the two kinds of primers in p16 positive OSCC patients. Moreover, in order to confirm the copy number of HPV16 per cell, the standard curves of HPV16 E6/E7 and E7 using a 5-fold dilution of CaSki gDNA samples with known copy numbers were set up.
Only 1 out of 21 p16 positive patients (cut off value 25% and 75%) showed a standard amplifying curve and melt curve for HPV16 E7 (Fig. 2B). The result of real-time PCR was confirmed by agarose gel electrophoresis, demonstrating the presence of HPV16 in the sample (Fig. 2C). The copy number of HPV E7 DNA was 0.312 copies/genome. Moreover, the matched adjacent group without tumors did not show a standard amplifying and melt curve for HPV16 E6/E7, showing that normal epithelial cells were not infected by HPV16 (Fig. 2D).
The information of the patient was as follows, age: 69, sex: female, smoking history: no smoking, primary tumor site: Buccal mucosa, Grade: poor differentiation, T stage: 2, N stage: negative, live status: live.
The expression of p16 had no prognostic value for OSCC patients
In order to evaluate the prognostic values of p16 in OSCC, we investigated the associations between p16 expression and patients’ clinical outcomes with 25% and 75% cut off values. Whether the cut off value of p16 expression was set at 25% or 75%, no significant differences in OS, DFS and RFS between patients with p16-positivity and p16-negativity was observed (Fig. 3).
The Cox regression model was then used to determine whether the known prognostic values and p16 expression status were associated with OS. Multivariate analysis showed that TNM, metastasis and recurrence were associated with worse OS. p16 status was not statistically significant in univariate and multivariate analysis. These results showed that p16 expression had no prognostic values in OSCC patients.
Cox univariate and multivariate analysis for overall survival of OSCC patients
Cox univariate and multivariate analysis for overall survival of OSCC patients
Kaplan-Meier survival analysis of OSCC using cut off at 25% or 75%. (A) OS, (B) MFS and (C) RFS for OSCC patients using cut off at 25%; (D) OS, (E) MFS and (F) RFS for OSCC patients using cut off at 75%.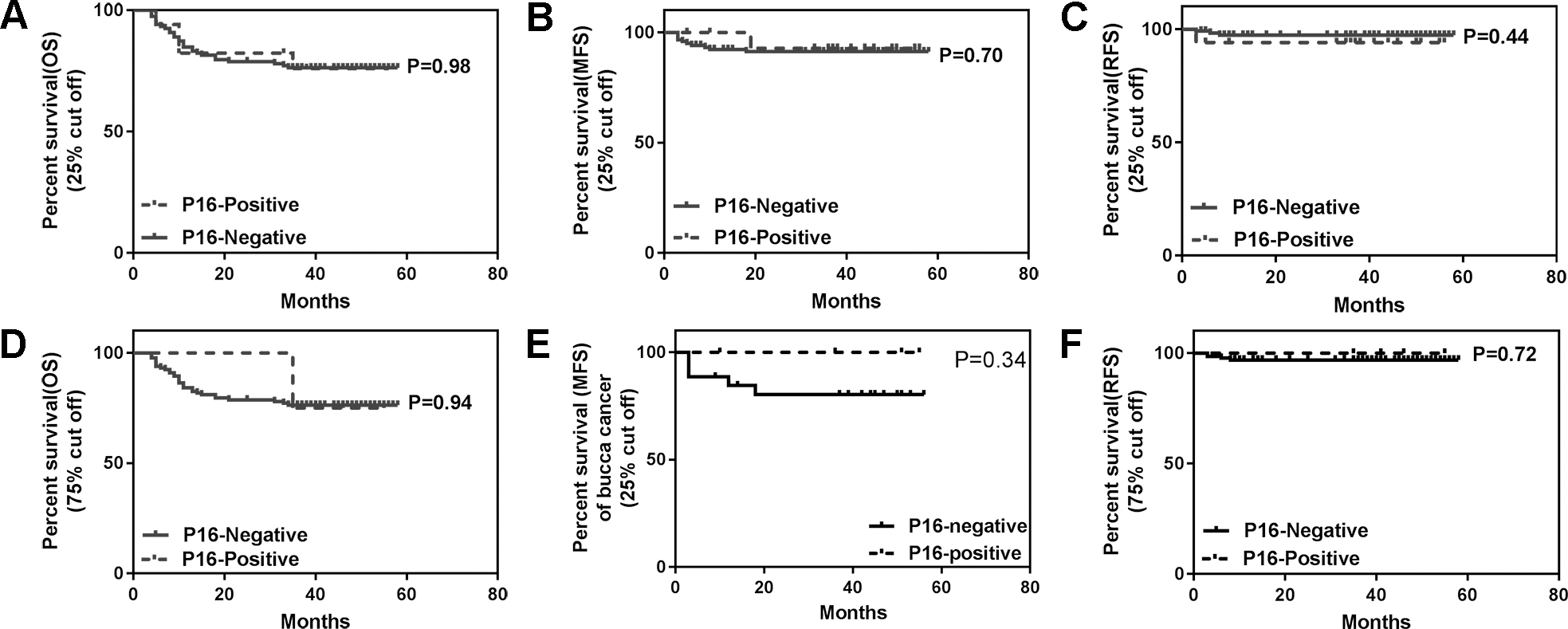
Kaplan-Meier survival analysis of OSCC in different anatomic subsites using cut off at 25% or 75%. (A) OS, (B) MFS and (C) RFS for tongue cancer patients using cut off at 25%; (D)OS, (E) MFS and (F) RFS for buccal mucosa cancer patients using cut off at 25%; (G) OS, (H) MFS and (I) RFS for gingiva cancer patients using cut off at 25%; (J) OS, (K) MFS and (L) RFS for tongue cancer patients using cut off at 75%.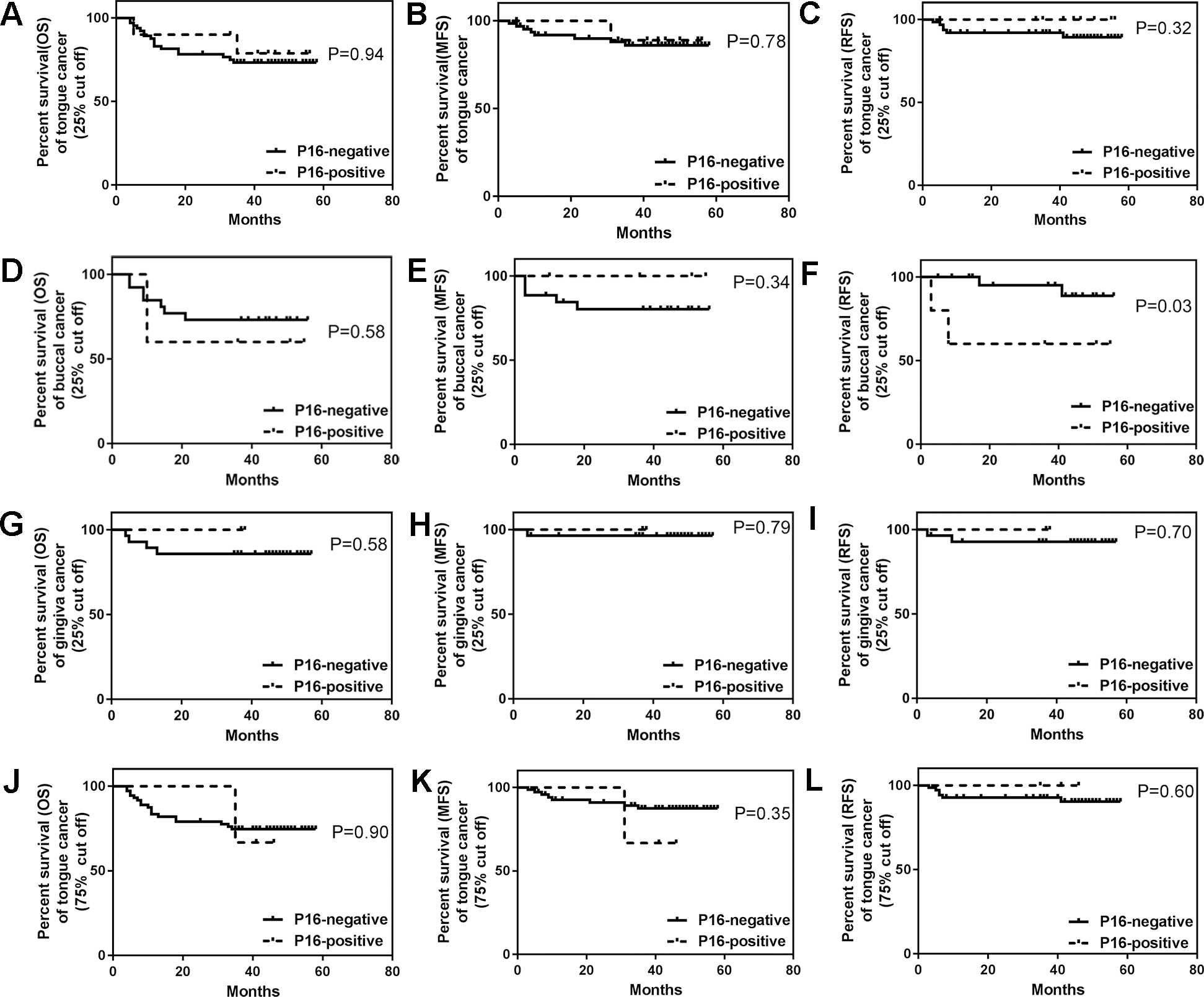
As molecular differences exist between different anatomic subsites in OSCC, which highlight the importance of precise anatomical subsites classification in OSCC diagnosis, we then examined the prognostic values of p16 expression in the patients with tumors derived from tongue, gingiva and buccal mucosa. Univariable analysis showed that only the patients with p16-negative (cut off 25%) tumors located in the buccal mucosa had more RFS than patients with p16-positive tumors (
Discussion
Our results indicated that p16 positive expression was 14.3% (21/147) using 25% as the cut off value and 4.8% (7/147) using 75% as the cut off value. Only one patient was found to be HPV16 positive in the p16 positive (cut off
Although previous studies tested HPV DNA prevalence in OSCC in China, different conclusions were drawn, and the relationship between HPV prevalence and p16 expression was not uncovered [19, 20, 21]. p16, the principal member of the Ink4 family, is a tumor suppressor protein and regulates cell cycle progression by inhibiting S phase. However, its expression was different in various tumors, ranging from its loss or down-regulation to overexpression [22]. In OPSCC, p16 overexpression is a failed attempt to stop uncontrolled cellular proliferation and is a surrogate marker for HPV16 infection [23]. Compared with p16 negative patients, a p16 positive status predicts favorable clinical outcomes in OPSCC [24, 25]. However, the discordance between p16 overexpression and HPV infection in OSCC has been reported [14, 26]. For example, p16 positive expression was only 3.5% in HPV-DNA positive patients [16]. In our results, we first tested p16 expression in all enrolled patients, of whom 21 (14.3%) were p16 positive using 25% as the cut off value. The discrepancy of p16 positive expression between the two studies can be explained by the differences of the tested samples. Only one p16 positive patients (cut off value 25% and cut off value 75%) was confirmed to be HPV-positive. Both studies suggested that p16 could not be an indicator of HPV16 in OSCC patients and p16 overexpression in OSCC may be a unique subset and deserves further study.
In this study, we found that p16 negative patients had more RFS than the positive ones in buccal mucosa cancer. Although p16 loss played an important role in tumor initiation, overexpression of this protein was observed during the process of later malignant transformation [22]. As premalignant cells transform into malignant cells, p16 is overexpressed in response to oncogenic stimuli. It has been suggested that p16 overexpression predicts poor clinical outcomes in some types of tumors, such as colon cancer and breast cancer [27, 28]. However, other reports have indicated that the radiotherapy outcome does not differ by tumor p16-status in OSCC. Therefore, more research into the prognostic values of p16 in OSCC is required [29].
The oral cavity is a highly complex anatomic region, emerging from various branchial arches that are associated with an aggregation of mesenchyme, ectoderm and endoderm. Traditionally, all carcinomas originating from the oral and oropharyngeal subsites are defined as OSCC. Recent research has found that it is a diverse group of tumors which differ in anatomy, histology and microenvironment [30]. However, to what extent tumor localization affects tumor biology and prognosis remains to be demonstrated. The correlation of p16 expression and HPV status with the clinical outcomes varied at different sites of the oral cavity. p16 was correlated with age, tumor sites and tumor differentiation and p16-positivity was much higher in tongue cancer (36.3%) than in the floor of mouth (19.2%) and gingivobuccal (18.2%) [26]. However, in our research, p16-positivity was higher in patients with cancers of the buccal mucosa (21.21%) than of the tongue (13.4%) or gingiva (6.25%) (
In this research, only the confirmed OSCC patients, whose tumors were determined to be resectable by an H&N surgical oncologist before treatment, were included in our research and the unresectable OSCC patients were not included in our research. As we know, treatment is complex for OSCC patients, and surgery is still the first option for OSCC treatment in recent years [31]. But, there also exists a small portion of unresectable OSCC patients, whose gross tumor on anatomic grounds can’t be removed by surgeons or the functional or cosmetic outcome of surgery result in higher morbidity or lower quality of life. Moreover, there also exists a subgroup of patients who refuse surgical management. But these subgroups of patients are pretty rare. Besides, as a limited number of patients restricted our analysis, future research should be conducted on more patients. In conclusion, our data confirmed that HPV was rare in Chinese OSCC samples. When p16 expression was more than 25% in the tumors located in the buccal mucosa, it predicted a higher recurrence rate. Therefore, identifying the precise location of the tumor in OSCC patients is important for the prognostic value of p16 IHC.
Footnotes
Acknowledgments
This work was supported by the National Natural Science Foundation of China (No. 81772880, 81702680), the Fundamental Research Funds for the Central Universities, Nanjing Medical Science and Technique Development Foundation (No. YKK16164, QRX17083, YKK18123), Jiangsu Provincial Key Medical Discipline (since 2017), Nanjing Municipal Key Medical Laboratory Constructional Project Funding (Since 2016), Center of Nanjing Clinical Medicine of tumor project (Since 2014).
