Abstract
Colorectal cancer (CRC) is the third most common cancer; cancer biomarker discovery is important for disease detection and management. It is known that hyaluronic acid and its receptors are ubiquitously expressed in almost all human tissues. Earlier we have shown that a monoclonal antibody H11B2C2, presently known as UNIVmAb, reactive hyaladherin expressed in multiple human cancers mainly using immunohistochemistry. However, the nature of the antigen and its sequence homology are not known. In the current study, a comprehensive investigation was performed to explore the nature of the antigen and its homology using both biochemical and proteomic analysis. Our results showed that UNIVmAb reactive 57 kDa antigen was overexpressed in advanced grade colorectal cancer tissues compared to benign and its hyperplasia. Biochemical investigations including biotinylated hyaluronic acid-pulldown, Immunoprecipitation, HA-oligo competition experiments confirmed that the UNIVmAb reactive 57 kDa antigen is a member of hyaladherin. Further Proteomic analysis showed that the antigen has homology with IGHG1 (Ig
Keywords
Background
Colorectal cancer (CRC), the third cause of cancer-related deaths [1], represents a serious health burden, with approximately two million new cases diagnosed annually in the world. CRC incidence is rising at a young age [2] and the patients are often diagnosed at an advanced stage and are incurable. Colorectal cancer screening is usually based on faecal blood testing and colonoscopy [3]; new biomarkers for screening procedures may be useful for early detection of CRC.
Extracellular matrix (ECM), a major structural component of the tumor microenvironment [4], plays a crucial role in the regulation of cellular architecture and cancer. Hyaluronic acid is a component of ECM and its expression is correlated with tumor progression [5]. HA regulates tumor growth by promoting cell invasion, migration, and angiogenesis, etc. via binding to its receptors, called Hyaluronan Binding Proteins (HABP) or hyaladherins. Several hyaladherins have been identified, including CD44, Receptor for hyaluronic acid-mediated mobility (RHAMM), TSG-6, proteoglycans such as aggrecan, versican, brevican and neurocan. However, CD44 is the only HA receptor that is studied in detail and its high expression correlates with unfavorable outcomes [6]. Several groups have shown that targeting HA or depletion of HA synthases sensitizes tumor cells to chemotherapeutic drugs [7]. HA and the expression of its receptor are highly regulated during tumor progression [5, 8]. However, their specificity towards receptor interaction plays a major role in the regulation of cancer [9]. Though many groups have studied the expression levels of hyaladherins in cancer there are several limitations in their studies. It could be useful to identify a protein or proteins that are involved in tumor progression which can be used as biomarker.
Previously we have developed a monoclonal antibody H11B2C2 (at present known as UNIVmAb) which can detect a 57 kDa protein which might be a possible HA binding protein. In the current study, we performed a comprehensive investigation to find the expression pattern and nature of the binding protein using both biochemical and proteomic analysis.
Materials and methods
Reagents
Hyaluronic acid (Na Salt, human umbilical cord) was purchased from Sigma, USA, and Across organics. Bovine Testicular hyaluronidase Type IV, Bovine Serum Albumin, Guanidium hydrochloride, EDC, protease inhibitor cocktail was purchased from Sigma. Streptavidin-horseradish peroxidase conjugate, biotinylated goat anti-mouse IgGs were from Zymed and Vector respectively. Protein G agarose, Streptavidin agarose beads, CNBR activated sepharose 4B, QAE Sepharose, EZ-Link Biotin-L-C hydrazide and Enhanced chemiluminescence kit were from Invitrogen.
Tissue samples
Approvals of the hospitals and the protocols were approved by the ethical review committee (Mys-00340-AA-NH, and IHEC-UOM NO 35). The tumor sections were graded with the TNM system by the pathologists of the hospital. For the biochemical studies, tissues were collected in ice during surgery, frozen in liquid nitrogen, and stored in
Preparation of UNIVmAb monoclonal antibody
UNIVmAb was prepared according to [8]. Briefly, the hybridoma was produced by the fusion of myeloma variant NS1 with splenic lymphocytes obtained from SJL/J mice immunized with a partially purified HABP [10]. The hybridoma was further sub-cloned in inactivated pathogen-free human serum and clones (H11B2C2) were selected, named UNIVmAb.
Preparation of biotinylated hyaluronic acid
Biotin conjugation of HA was performed according to Kolapalli et al. [9]. Briefly, 500 mg of hyaluronic acid was dissolved in 0.5 M MES buffer (pH 5.5) and incubated with 1 mM biotin-LC-hydrazide dissolved in DMSO, 10 mM EDC for 16 h at 4
Gel exclusion and anion exchange chromatography
CRC tissues proteins were extracted using tissue lysis buffer containing 50 mM Tris (pH 8.0) 0.5% Triton X-100, 20 mM EDTA, and protease inhibitor cocktail using a glass-glass homogenizer on ice for 30 min. The lysates were then clarified by centrifugation at 12,000 rpm for 30 min at 4
Immunoprecipitation analysis
500
Antibody affinity column chromatography
5 mg of UNIVmAb was dialyzed against coupling buffer (Na2Co3, pH 9.0) for 24 h with 3 changes and then conjugated with CNBr-activated Sepharose 4B by mixing overnight at 4
MALDI-TOF MS and protein identification
Approximately 1 mg of protein from colon cancer grade II after went through Q-sepharose, and the 220 mM fraction passed on to the UNIVmAb affinity column. Only Glycine eluted fraction after lyophilization ran on 10% SDS-PAGE. Gel stained with coomassie blue, and the spots at 57 kDa were excised, destained, and incubated with sequence grade trypsin at 37
Cibacron blue gel binding
The procedure was obtained from Lowenthal et al. [11] and modified accordingly. 500
Results
Hyaluronic acid and UNIVmAb reactive protein overexpress in CRC
Hyaluronic and its binding proteins (HABP’s) are ubiquitously expressed. However, no studies have shown the overall expression pattern of hyaluronan binding proteins in CRC. In this study by performing Western blot analysis, we clearly showed that HA receptors detected by biotinylated HA (b-HA) were overexpressed during tumor progression (Fig. 1A). Previously, using Immunohistochemical analysis we showed that UNIVmAb reactive HABP expression was high in multiple cancers [8]. However, in this study, a clear comprehensive analysis was performed to find the expression profile of the 57 kDa protein in CRC. Using a simple and reliable technique – Western blot, we analyzed the expression of the 57 kDa protein in hyperplasia, grade-I, and II CRC. We found that the UNIVmAb reactive 57 kDa protein was high in grade-I CRC than hyperplasia. On contrary, grade-II CRC has shown elevated expression of HABP’s than grade-I and hyperplasia, which shows that the 57 kDa protein has a role in tumor progression and might serve as a prognostic marker for CRC (Fig. 1B). We have presented the data from 3 different sets of tumors.
(A) Expression of HA binding proteins in benign, CRC G-I and G-II tissues. (B) Expression of UNIVmAb reactive HD in benign, CRC G-I, and CRC G-II tissues. (C) Competition analysis between O-HA/HS with UNIVmAb binding with HD. (D) Biotin pulldown protein lysates of benign and CRC G-II were probed for UNIVmAb reactive HD. (E) Competition analysis between cold O-HA and UNIVmAb.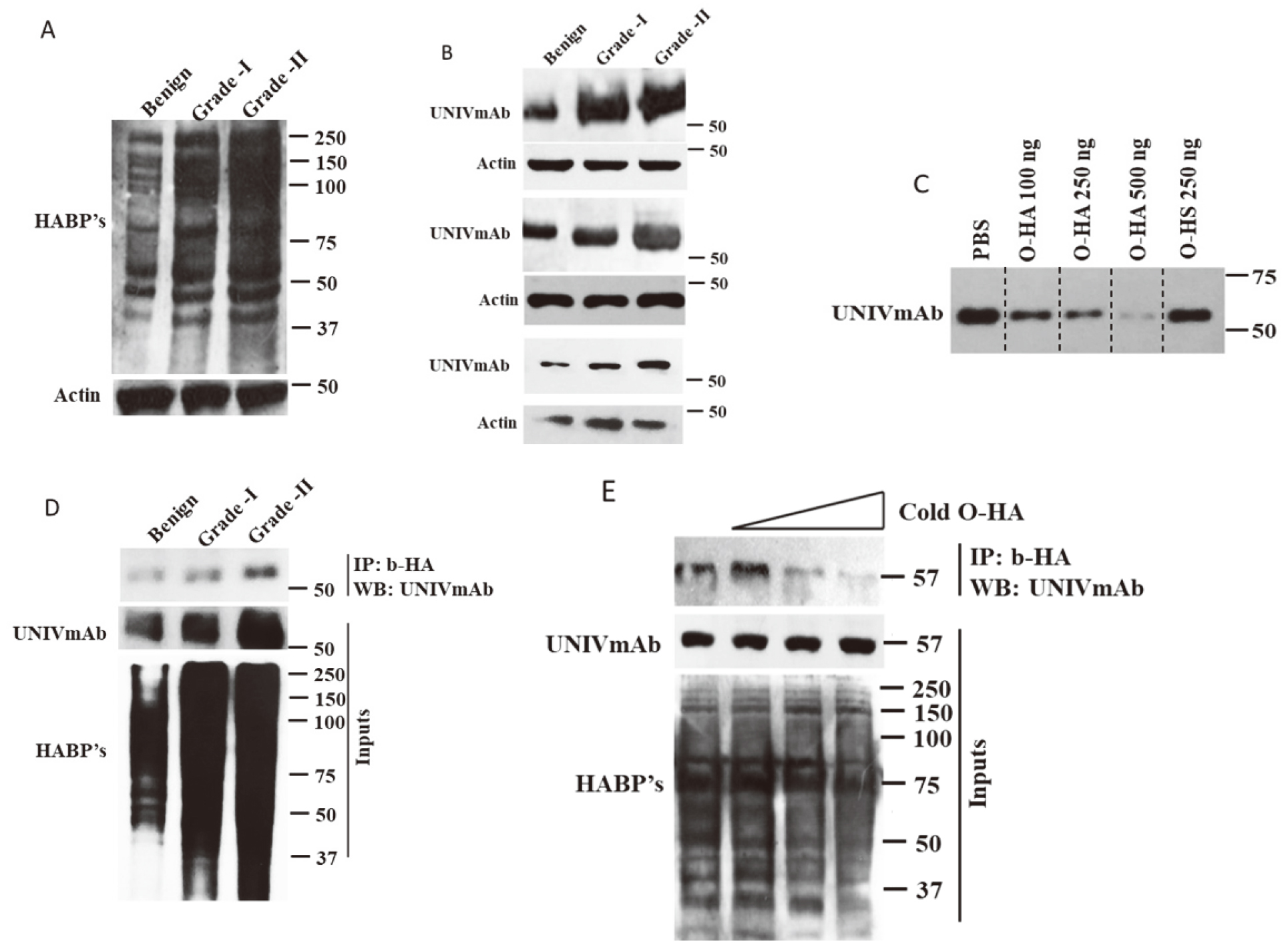
Next, we wanted to understand whether the UNIVmAb reactive 57 kDa has specificity to HA, we performed competition assays. Competition assays are the standard biochemical assays to find the specificity. HA is a high molecular weight glycosaminoglycan and binds to its receptors with high specificity. High molecular mass HA can possibly block the antibody interaction by masking the antibody binding regions of the receptors. To avoid this possibility, we prepared HA oligomers of 8–16 mers (O-HA) and used as a competitor for UNIVmAb. Tissue protein extracts were subjected to western blot analysis by incubating with UNIVmAb containing increasing concentration of O-HA, then the strips were developed together as represented in Fig. 1C. Also, to avoid the least minimal possibility of non-specific binding, we used heparin sulfate (HS) as a control. We found that 250 ug of cold O-HA showed a significant reduction in UNIVmAb binding to 57 kDa protein and 500 ug of O-HA completely abolished UNIVmAb reactivity with it (Fig. 1C). Surprisingly, HS did not block the UNIVmAb reactivity with 57 kDa protein. Together, the results indicated that the 57 kDa protein might have specificity to HA. To further strengthen the specificity of 57 kDa protein to HA, we performed co-IP experiments, where we incubated the protein extracts of hyperplasia, grade-I, and II CRC with bHA and subjected to Western blot analysis with UNIVmAb antibody. The results showed that the bHA pulled down UNIVmAb reactive 57 kDa protein (Fig. 1D). Finally, to prove the specificity we performed more stringent experiments by incubating the grade-II CRC protein lysates with increasing concentration of O-HA and subjected to pulldown with bHA and subjected to Western blot analysis with UNIVmAb. We found that along with an increasing concentration of O-HA the binding of b-HA with 57 kDa protein was diminished. Together, the results indicated that the UNIVmAb reactive 57 kDa protein is a member of the hyaladherin complex (HD) and is overexpressed during tumor progression.
Purification of HD from hyperplasia and grade-II CRC for proteomic analysis
The nature of the HD is not known. To understand its nature, we performed sequential protein purifications to achieve highly purified HD for proteomic analysis. Gel filtration analysis showed mainly one large peak and two minor peaks. According to the molecular mass, HD elutes in the first peak. So, we pooled the fractions of the first peak and confirmed the presence of HD by western blot analysis (Fig. 2A). Further purification of HD was achieved by graded salt fractionation using QAE sepharose. The fractions were pooled and lyophilized. Further, HD fractionation was determined using western blot analysis and we found that HD was mainly eluted in 300 mM NaCl eluted fractions (Fig. 2B). Further purification of HD was achieved by affinity purification using UNIVmAb conjugated sepharose and its reactivity with HA was also confirmed by probing with biotin-HA (data not shown).
(A) Gel permeation chromatogram of CRC grade II. (B) Ion exchange chromatogram of CRC grade II proteins fractionated on gel permeation (peak 1). (C) Ion exchange fractions of CRC grade II were pooled, lyophilized, and subjected to western blot analysis with UNIVmAb. (D) MALDI-TOF chromatogram of benign and grade II CRC. (E) Benign and grade II CRC samples passed through cibacron blue and lyophilized, then subjected to western blot analysis with bHA and UNIVmAb.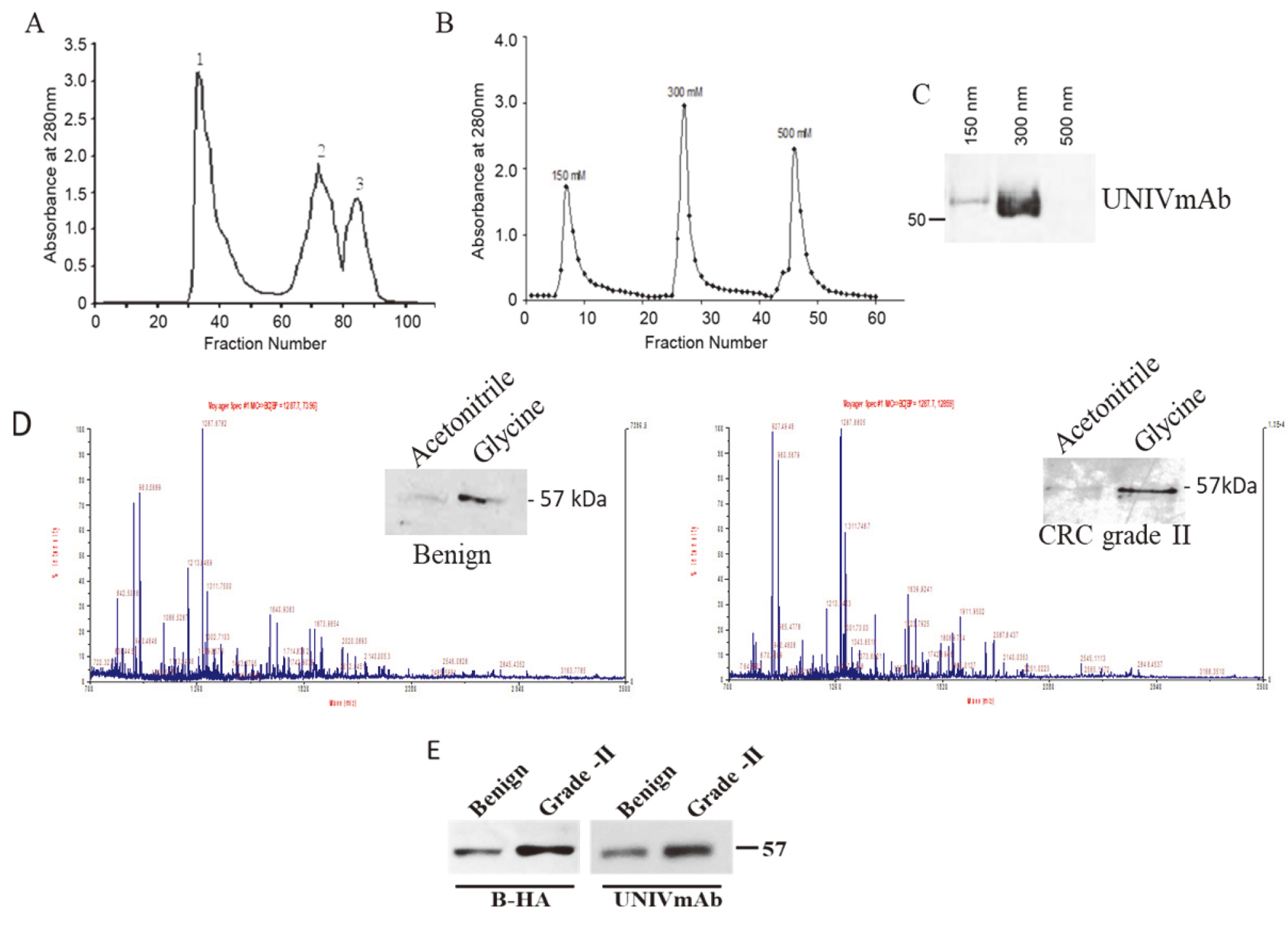
Preliminary mass spectroscopic analysis of the UNIVmAb antibody affinity-purified HD, which was eluted with 0.1 M glycine showed homology with multiple proteins including IGHG1 (possible IgG superfamily member) and is associated with human serum albumin. The spectral graph (Fig. 2D and E) shows three major peaks in both associated and cancer tissue, with m/z at 927, 960, and 1287 upregulation and downregulation mainly with m/z 1639. Both tissues have some homology with higher expression of IGHGI in adenocarcinoma of the colon. NCBI database search showed with the accession of the major peaks of both benign and metastatic tumor presented in Table 1. Together, the results have shown that the antigen from colorectal carcinoma is a serum carrier protein and probably an IgSF member.
HD from colon cancer is probably a serum carrier BP
Albumin is a versatile and captivating protein. Albumin serves as a protector of several biologically important proteins and was well documented [12, 13, 14]. Carrier bound proteins contain potential disease-related information [11]. Proteomic analysis showed that the HD contains homology with albumin. Cibacron blue binds to albumin and associated complexes. We have validated the albumin homology/association of HD using Cibacron blue. The results from the western blot analysis showed that HD binds to Cibacron blue and biotin-HA detected the same band eluted from Cibacron blue. It proves that HD not only has homology/association with albumin but is also a hyaladherin. However, 10–15% of HD only bound to Cibacron blue and its expression was high in advanced grade CRC than benign (Fig. 2E).
Discussion
In the current study, we have provided evidence that the colon malignant tumor cells overexpress UNIVmAb reactive 57 kDa antigen, and it belongs to the hyaladherin protein complex, and it is also associated with serum albumin. HA and its receptors are implicated in cancer. Studies showed that HA and its receptors like CD44 and RHAMM overexpress in cancer [15, 16, 17] and
Identification of hyaladherin (HD) sequence homology using MALDI-TOF
Sequence homology of the major peaks obtained in MALDI-TOF of both benign and grade II CRC samples.
the overexpressed CD44 is shown to be involved in multidrug resistance [18, 19]. However, targeting of CD44 or its isoforms or using them as a prognostic marker was not successful. Probably other binding proteins exist for hyaluronan which is not identified or studied in detail. So, there is an immediate need to identify unique hyaladherins which might serve as a potential biomarker candidate, and also it is important to understand its nature and localization. Further, there is an immediate need to validate and develop an assay system.
HA-HABP interactions play a pivotal role in cancer. Several HABP’s were identified and shown to play a role in tumor progression. Though CD44 and RHAMM were well studied, the other receptors were still under characterized. Using gold standard method Immunohistochemistry, reports have shown the staining of specific HABP’s using a specific antibody, but the overall expression pattern of HABP’s was not clear. To delineate this, we adopted a reliable method of western blotting and found that multiple hyaluronan receptors exist, of which many of them were under characterized. Further, we developed an antibody that detects a 57 kDa antigen. Boregowda and colleagues [8] have previously reported that the identified 57 kDa antigen HD may be predictive of tumor progression in various human malignant tumors. However, these studies did not identify the definite contribution of HD in CRC. IHC of a panel colorectal tumor indicated that hyaladherin expression is upregulated in later stages of tumorigenesis and metastatic CRC. Even though the primary tumor, grade 1 showed moderate HD staining, the majority of the samples from poorly differentiated colon tumor overexpressed HD suggesting that HD may be essential for primary tumor growth and is leading to highly metastatic conditions during the processes of invasion. The present results are consistent with the CRC data presented by Boregowda and his colleague [8]. Further, the specificity of the antigen towards hyaluronic acid was evaluated using several biochemical techniques including competition and co-IP experiments. O-HA abolished the binding of UNIVmAb to 57 kDa antigen. To confirm whether the 57 kDa antigen also presents in other proteoglycan complexes, we used heparin sulfate as a control. HS could not interfere with the binding of the UNIVmAb to 57 kDa antigen and confirmed that it belongs to the hyaladherin complex. The mAb UNIVmAb reacted specifically with partially purified and affinity-purified protein, plus the protein shows very strong reactivity for the bHA, and the interaction of mAb with antigen was abolished with HA-oligosaccharide, indicating that the antigen may be a member of hyaladherin. Numbers of studies have demonstrated that the importance of HA and its receptors in regulating tumorigenesis is via surviving signaling [15, 16]. Hyaladherin complex protein detected by antibody UNIVmAb is one of the important factors of activities that induce malignancy in colorectal cancer. This idea is supported by the observation that the enhancement of HD expression is more in malignant tumors which might stimulate growth and invasiveness.
IgSF proteins mostly belong to cell adhesion molecules, antigen receptors, co-receptors, cytokine receptors, growth factor receptors, and some are involved in antigen presentation to lymphocytes. Several IgSF’s have been identified as biomarkers for cancer progression [19]. ECM is a dynamic microenvironment function as a reservoir for growth factors and also influences cell fate by binding to their receptors. Cancer cells influence and remodel ECM and also aberrantly express receptors for ECM for their growth. Several ECM receptors belong to IgSF. HD has homology with IgSF with localization at the cell surface and our unpublished data showed that HD is in complex with key signaling proteins. Further, our preliminary data also showed that the UNIVmAb causes cytotoxicity [8]. Hyaladherins are known to be involved directly in multiple cell signaling pathways and targeting those leads to cell death. Though we don’t know its biological role yet, the data indicating that HD might serve as a receptor/co-receptor for cancer cell signaling. Future studies are focused on defining the molecular nature of this serum carrier 57 kD protein and its affinity to the Ig superfamily in CRC.
Tumor-associated antigens are aberrantly expressed and these overexpressed tumor antigens are often released in the circulation [21]. Our recently published results showed that colon cancer soluble serum is also a part of the common biomarker including other cancers sera. The present analysis showed that CRC-HD is a real biomarker and the secretary HD levels are associated with the prognosis of tumors [22]. We believe that the overexpressed hyaladherin contains homology/association with albumin which might protect the HD from degradation through the system and might serve as a biomarker for the detection of CRC at an early stage.
In conclusion we identified that there are multiple binding proteins exist for hyaluronic acid and UNIVmAb reactive 57 kDa protein (HD) is one among them. We identified that the HD overexpressed during tumor progression and possess homology with IgSF and associated with albumin, which can increase the stability of the proteins. Moreover, our results suggest that HD may be used as a potential effective noninvasive biomarker for CRC.
Footnotes
Acknowledgments
We thank the Department of Studies in Biochemistry, Mysore University for helping us to use their cell culture facility.
