Abstract
OBJECTIVE:
Sirtuins family are defined as class III histone deacetylases (HDACs). Recently, mammalian silent information regulator two 4 (SIRT4) has been reported to be a tumor suppressor gene in multiple cancers. The objective of the present study was to explore the potential role of SIRT4 in clear cell renal cell carcinoma (ccRCC).
METHODS:
We estimated SIRT4 expression levels in ccRCC and its adjacent non-neoplastic tissue by Western blotting (WB), quantitative real-time polymerase chain reaction (qRT-PCR) and bioinformatics data, the clinical and survival data were also collected and analyzed. In vitro study, ccRCC cell lines were transfected with SIRT4-siRNA or lentivirus to downregulate or overexpress the expression level of SIRT4. Then, the proliferation capacity of tumor cell was assessed by 5-Ethynyl-2’-deoxyuridine (EDU) assay, cell migration and invasion capacity were assessed by Transwell assays.
RESULTS:
Our results indicated that the expression level of SIRT4 in ccRCC was significantly lower than the corresponding normal tissues (
CONCLUSIONS:
These findings support that SIRT4 acts as a tumor suppressor in ccRCC and might be a novel biomarker and new therapeutic target for ccRCC.
Introduction
Clear cell renal cell carcinoma (ccRCC) is the most common malignant tumor of the kidney, which account for 2–3% of all adult cancers worldwide. It arises from the proximal tubular epithelium and usually aggressive and lethal for the patients due to no obvious clinical symptoms in the early stages [1, 2]. Studies have found that the pathogenesis of ccRCC is tightly related to metabolic reprogramming, particularly in glucose and fatty acid metabolism. In this regard, consecutive activation of hypoxia inducible factor (HIF) signaling caused by von Hippel-Lindau (VHL) mutations and abundant intracellular lipid droplets are two pivotal hallmarks of ccRCC [3]. In addition, metabolism of tryptophan, arginine and glutamine are also reprogrammed in ccRCC, renal cancer actually can be considered as a metabolic disease to some extent [4]. Although these results have significantly accelerated the development of molecular targeted therapy for ccRCC, the therapeutic effect is still not ideal for patients with advanced tumor stage [5]. Thus, it has great significance to investigate novel biomarkers and effective therapeutic targets for ccRCC.
Mammalian silent information regulator two (SIRT) is a highly conserved family and acts as a group of nicotinamide adenine dinucleotide (NAD
In the current study, we explored the mRNA and protein expression level of SIRT4 in ccRCC specimens and analyzed the association between SIRT4 levels and some clinicopathological features. Meanwhile, cell functional experiments were conducted to investigate the effects of SIRT4 on ccRCC cell proliferation, migration, and invasion.
Materials and methods
Tissue specimens and clinical data collection
A total of 100 paired ccRCC tumor tissues and adjacent noncancerous tissues were collected from the Department of Urology, the First Hospital of China Medical University between February 2015 to April 2018. These fresh tissues were immediately frozen in liquid nitrogen and stored at
Bioinformatics analysis
At first, we collected the data from the Gene Expression Omnibus (GEO) DataSets (
Cell culture and transfection
The human ccRCC cell lines 786-O, ACHN, CAKI-1 were obtained from the Chinese Academy of Sciences, Type Culture Collection Cell Bank (Shanghai, China). 786-O cells were cultured in RPMI-1640 medium (HyClone; GE Healthcare, Logan, UT, USA); ACHN cells were cultured in MEM medium (HyClone, Logan, UT, USA) and CAKI-1 cells were cultured in McCoy’s 5A medium (HyClone, Logan, UT, USA); All media were supplemented with 10% fetal bovine serum. The cells were washed with 1
The expression of SIRT4 in ccRCC cells were regulated by transfecting with siRNA or lentivirus. ACHN and CAKI-1 cell lines were transfected with two SIRT4 specific siRNA (JTS scientific, Wuhan, China) using GoldenTran-R reagent (Golden Transfer Science and Technology Co.Ltd. Changchun, China) according to the manufacturer’s guidelines. The medium was replaced after 8 h, the transfection efficiency and experiments were performed after 48 h. The sequences of SIRT4-siRNA as follows (5’-3’): SIRT4-siRNA-1, forward, GCACAAGCGUGUAAAAGAATT and reverse, UUCUUUUACACGCUUGUGCAC; SIRT4-siRNA-2, forward, GAAGCUCCCGAUUGCAAUATT and reverse, UAUUGCAAUCGGGAGCUUCTT. 786-O and CAKI-1 cell lines overexpressing SIRT4 were constructed by transfected with pRlenti-CMV-SIRT4-3FLAG-PGK-Puro lentivirus vector purchased from OBiO Technology Corp., Ltd. (Shanghai, China), The titer of the lentivirus and negative control were 7.3
RNA extraction and quantitative real-time polymerase chain reaction (qRT-PCR)
The total RNA was extracted from clinical tissues and cultured cells using RNAiso Plus (Takara Biotechnology; Dalian, China) according to the manufacturer’s recommendations. Then, the PrimeScript
Western blotting (WB)
All the proteins extracted from tissues and cells were lysed in radioimmunoprecipitation assay (RIPA) buffer. Protein concentrations were detected using a bicinchoninic acid assay kit. Equal amounts of proteins were separated by 10% SDS-PAGE and transferred to polyvinylidene fluoride membranes. Then, the membranes were sealed with 5% non-fat milk at 37
5-Ethynyl-2’-deoxyuridine (EDU) assay
ccRCC cells were transfected with SIRT4 siRNA (48 h) or lentivirus (stable cell lines) were seeded in 24-well plates. When the confluence of the adherent cells was about 80%, The diluted EDU solution (1:1000, BeyoClick
Transwell assay
Transwell chambers with 8-
Statistical analysis
Statistical analysis in the present study was performed by GraphPad Prism version 7.0 software. Paired
Expression level of SIRT4 was significantly downregulated in ccRCC tumor tissues compared to adjacent normal tissues and low SIRT4 expression level predicted poor prognosis of ccRCC patients. A. The mRNA expression level of SIRT4 obtained from GEO database. B. The mRNA expression level of SIRT4 from TCGA database. C. The mRNA expression level of SIRT4 in 63 pairs of clinical specimens. D. The tumor stage of ccRCC patients were inversely correlated with SIRT4 expression from TCGA database. Kaplan-Meier curve collected from TCGA database showed patients with high SIRT4 mRNA expression exhibited a significantly longer overall survival than patients with low SIRT4 mRNA expression in two different group cut-off programs (E. Median; F. Quartile). G and H. SIRT4 protein levels in 88 pairs of ccRCC and adjacent normal tissues detected by WB.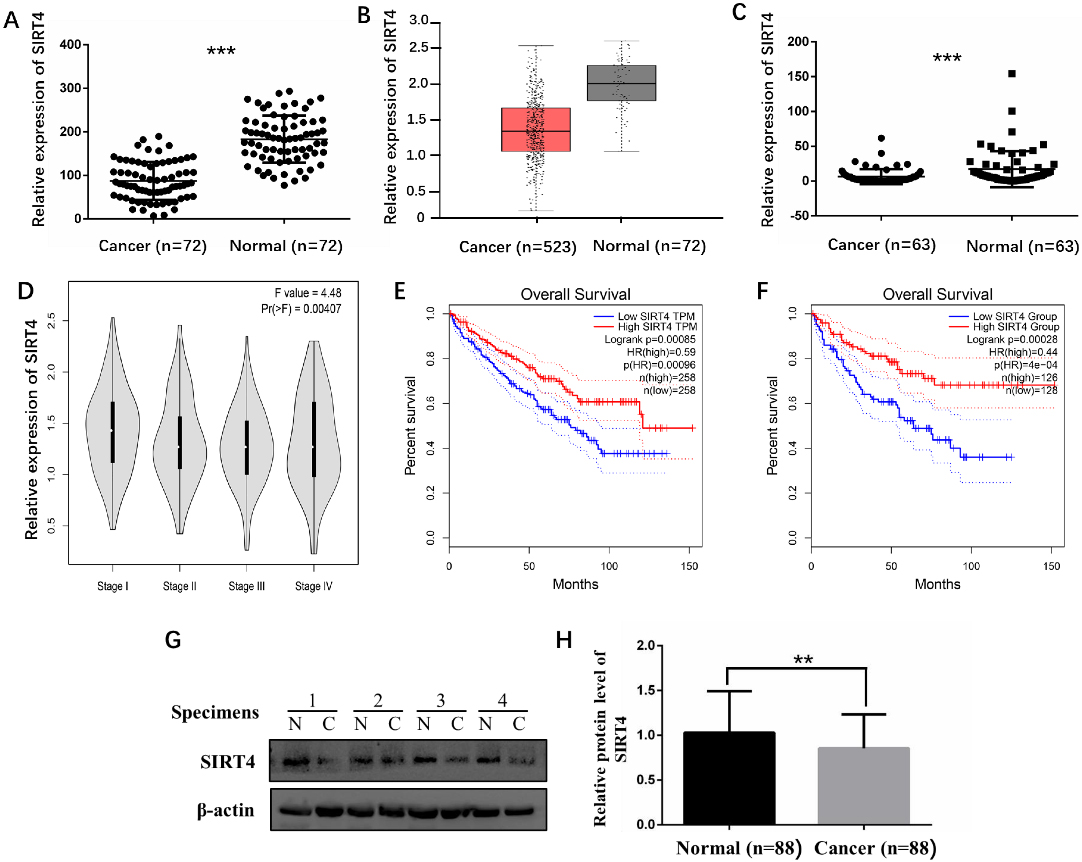
SIRT4 mRNA expression level was significantly decreased in ccRCC tissues and lower expression level associated with advanced tumor stage and poor prognosis
For mRNA level, we firstly collected the information from GEO dataset (GSE53757), the data in the microarray indicated SIRT4 mRNA expression level was obviously lower in ccRCC tissues (
Correlation between SIRT4 expression and the clinicopathological features of ccRCC patients
Correlation between SIRT4 expression and the clinicopathological features of ccRCC patients
Detection result for establishment of human ccRCC cell lines with upregulated or downregulated expression of SIRT4 by qRT-PCR or WB. A. As displayed by qRT-PCR and WB, SIRT4 expression was decreased in ACHN cell line at 48 hours after transfection of two SIRT4 specific siRNA. B. SIRT4 expression was also decreased in CAKI-1 cell line similarly to ACHN cell line. C. As displayed by WB, SIRT4 expression was stably increased in 786-O cell line after being infected with lentivirus. D. SIRT4 expression was also increased in CAKI-1 cell line similarly to 786-O cell line.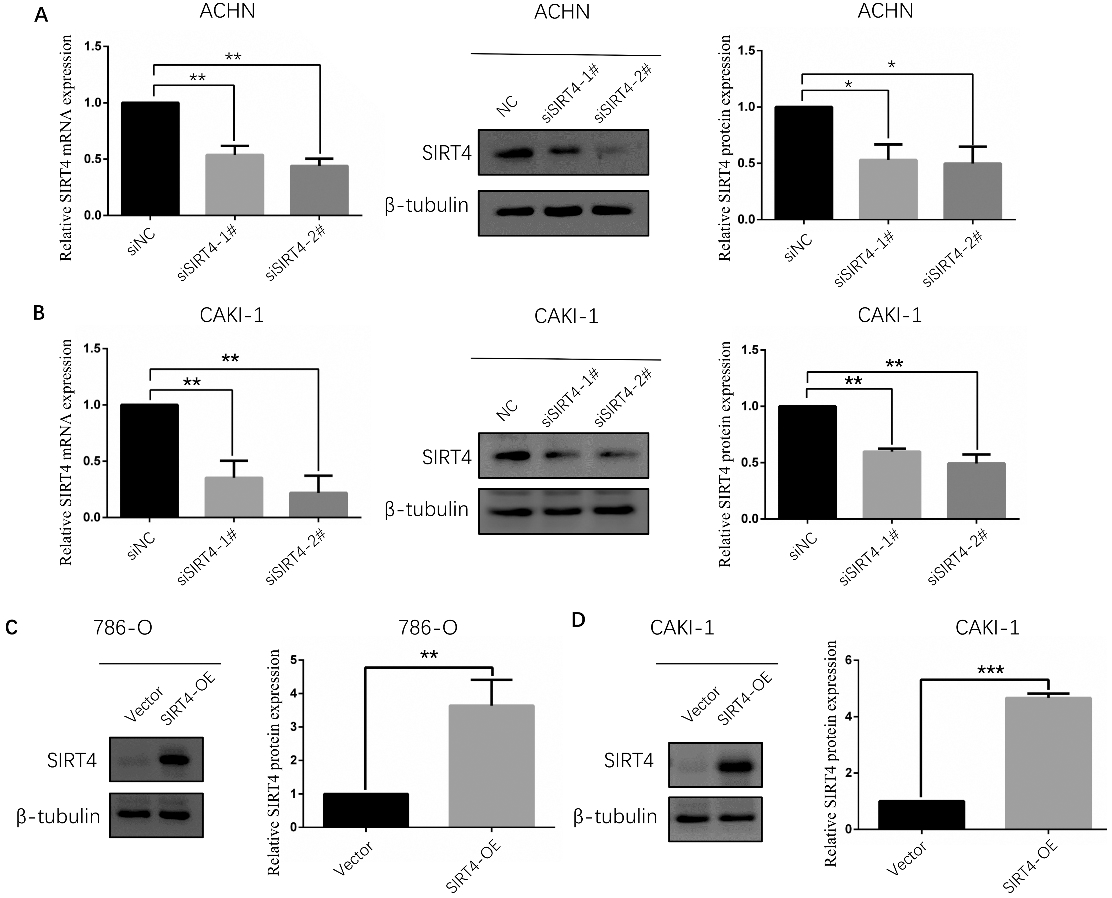
EDU assay suggested that SIRT4 inhibits ccRCC cell lines proliferation capacity. A. Downregulation of SIRT4 enhanced the proliferation ability of ACHN cell line and overexpression of SIRT4 significantly reduced the proliferation ability of 786-O cell line. B. relative count of EDU positive cells of ACHN and 786-O cell lines. C. Downregulation of SIRT4 could elevate the proliferation of CAKI-1 cell line, Conversely, overexpression of SIRT4 obviously repressed the proliferation of CAKI-1 cell line. D. relative count of EDU positive cells of CAKI-1 cell line.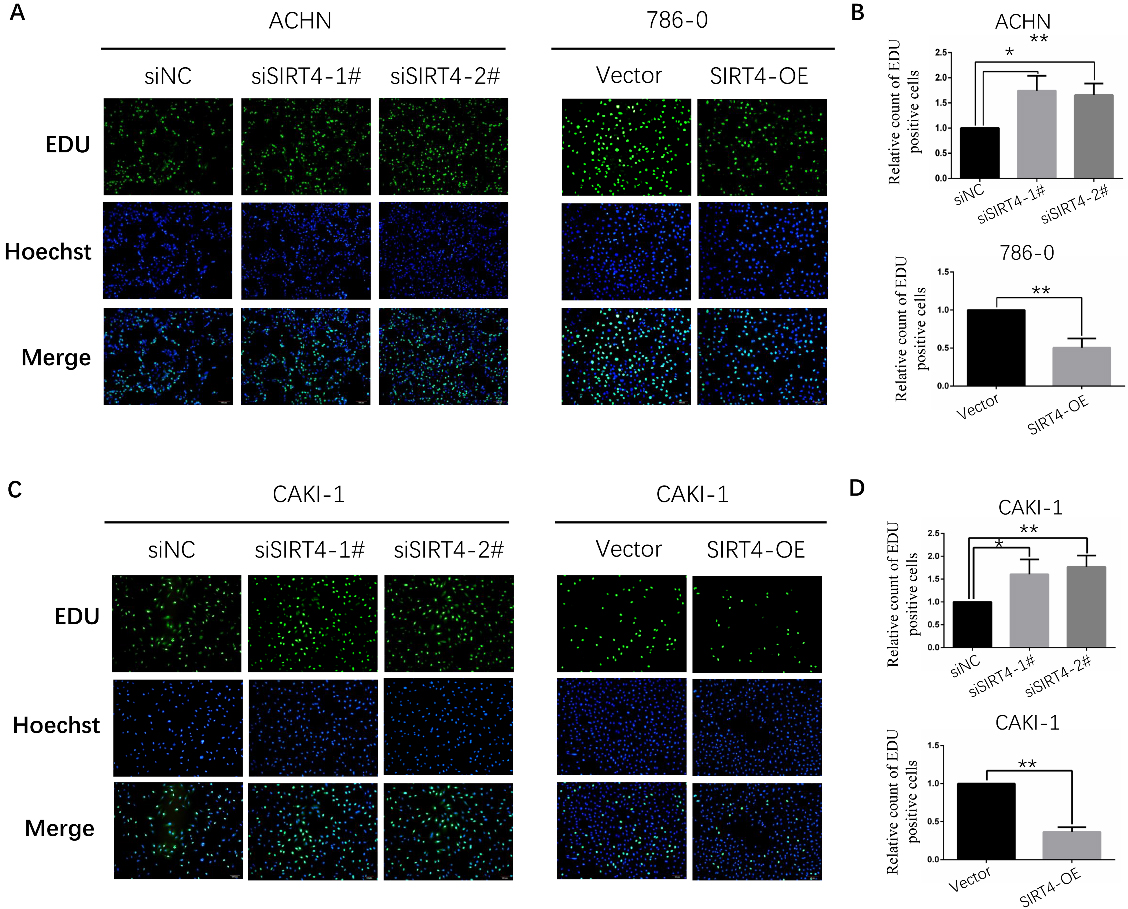
Transwell assay indicated that SIRT4 inhibits ccRCC cell lines migration and invasion capacity. A. Downregulation of SIRT4 enhanced the migration and invasion ability of ACHN cell line. B. Overexpression of SIRT4 significantly reduced the migration and invasion ability of 786-O cell line. C. Downregulation of SIRT4 improved the migration and invasion of CAKI-1 cell line. D. Overexpression of SIRT4 obviously repressed the migration and invasion of CAKI-1 cell line.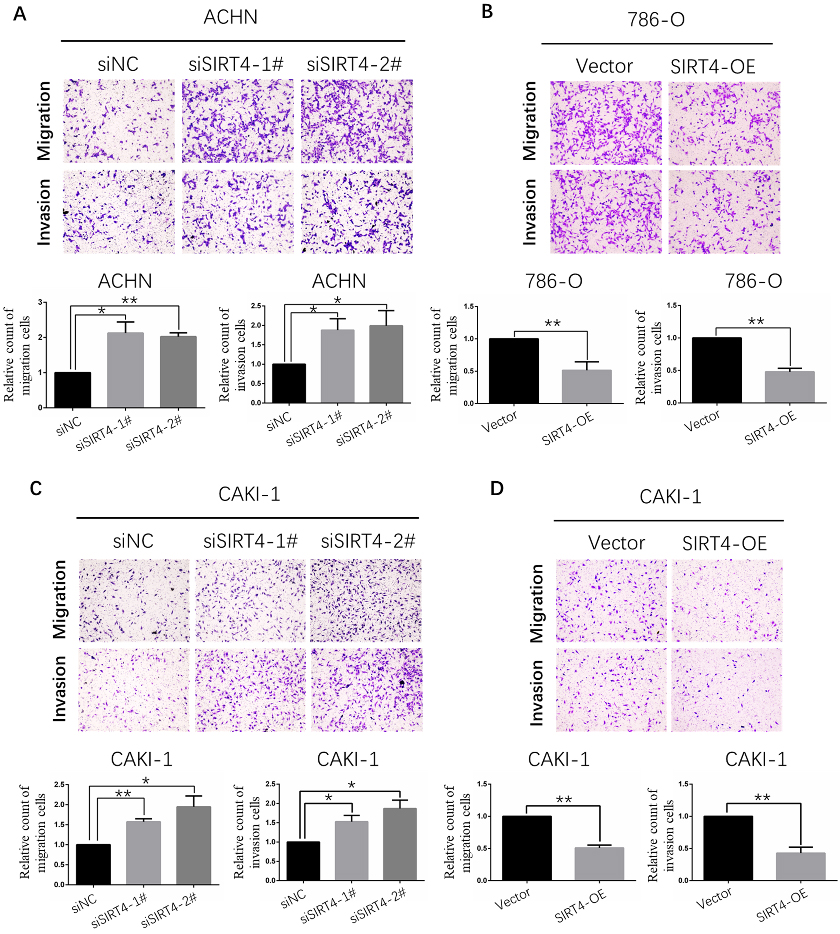
To further investigate the protein expression of SIRT4 in ccRCC tissues, we detected 88 paired specimens of ccRCC patients in our central using WB. Then, all the samples were divided into high and low expression group based on the median SIRT4 protein expression level, some clinicopathological parameters were compared in these two group. In the majority of ccRCC tissues, SIRT4 protein level was downregulated compared to the matched surrounding tissues (
Establishment of ccRCC cell lines with upregulated and downregulated of SIRT4 expression
In vitro experiments, ACHN and CAKI-1 cell lines were used for downregulated evaluation by transfecting with siRNA. CAKI-1 and 786-O cell lines were used for overexpressing assessment by infection with lentivirus. In order to verify that the expression of SIRT4 has indeed changed, both qRT-PCR and WB were performed. The expression level of SIRT4 was truly downregulated in ACHN and CAKI-1 cell lines at 48 hours after transfection of two SIRT4 specific siRNA (Fig. 2A and B), and it was steadily overexpressed in 786-O and CAKI-1 cell lines after lentivirus processing (Fig. 2C and D).
Human SIRT4 represses proliferation, migration and invasion ability of ccRCC cells
EDU assay was used for estimating cell proliferation capacity. The results suggested downregulation of SIRT4 enhanced the proliferation ability of ACHN and CAKI-1 cell lines (Fig. 3A and C). Conversely, overexpression of SIRT4 significantly reduced the proliferation ability of 786-O and CAKI-1 cell lines (Fig. 3A and C). Then, the effect of SIRT4 expression in cell migration and invasion ability was appraised by Transwell assay. As expected, ACHN and CAKI-1 cell lines with low SIRT4 expression possessed stronger migration and invasion capacity (Fig. 4A and C). On the contrary, migration and invasion capacity were strikingly declined in 786-O and CAKI-1 cell lines with overexpressing SIRT4 (Fig. 4B and D). In a word, the above data confirmed a notion that mammalian SIRT4 is a tumor suppressor of ccRCC.
Discussion
Radical surgery is the preferred treatment for treating ccRCC. But for locally advanced patients, the postoperative recurrence rate is very high and the overall survival time is pretty short [10]. In addition, ccRCC is not sensitive to chemotherapy, radiotherapy and traditional immunotherapy, some reports have pointed out that the molecular targeted drugs such as sunitinib and sorafenib could prolong the survival time of these patients. However, the specific mechanism and long-term therapeutic effect are still unclear [11]. Thus, exploring novel biomarkers and therapeutic targets of ccRCC has profound significance.
Sirtuins are defined as class III Histone deacetylases (HDACs). All of them share a highly conserved NAD
In recent years, studies indicated that SIRT4 could act as a tumor suppressor in malignancies. The expression level of SIRT4 was lower in squamous cell carcinoma, hepatocellular carcinoma, non-small cell lung cancer, endometrioid adenocarcinoma, neuroblastoma, gastric carcinoma, colorectal cancer, invasive breast cancer, thyroid cancer and pancreatic cancer compared to the adjacent normal tissues. And decreased SIRT4 expression in most of these cancers was closely associated with worse clinicopathological parameters and poor prognosis. In vitro studies, overexpression of SIRT4 could suppress the migration, invasion and proliferation of cancer cell lines [18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28]. Moreover, several studies confirmed knockdown of SIRT4 in mouse embryonic fibroblast cells cultivates larger tumors and SIRT4 knockout mice developed lung cancer, liver cancer and breast cancer spontaneously [9, 29]. But no research has investigated the relationship between SIRT4 expression and ccRCC as for today.
In the present study, our data indicated that the expression of SIRT4 in ccRCC was strikingly lower than the corresponding noncancerous tissues both at mRNA and protein levels. Meanwhile, low SIRT4 expression level was obviously involved with poor overall survival and advanced tumor stage in ccRCC patients. In vitro, experiments demonstrated that overexpression of SIRT4 significantly reduced the proliferation, migration and invasion ability of ccRCC cells. Oppositely, downregulation of SIRT4 enhanced the proliferation, migration and invasion ability of ccRCC cells.
The inhibitory effect of SIRT4 on cancer is multi-mechanism actually. The dominating mechanism is that SIRT4 could repress the metabolism of glutamine through ADP-ribosylation on GDH, which cut off the energy and materials supply of the rapidly proliferating tumor cells [9, 25, 27]. Secondly, stresses induced the expression of SIRT4 which inhibits glutamine metabolism, and further arrests the cell cycle. It provided sufficient time for DNA damage repair and protected the stability of the genome [30]. Thirdly, SIRT4 could enhance the expression of E-cadherin and inhibit the expression of N-cadherin and vimentin in cancer cells, thus inhibiting the process of epithelial-mesenchymal transition (EMT) and reducing the migration and invasion ability of cancer cells [24, 25]. At last, aerobic glycolysis leads to the accumulation of intracellular acidic metabolites such as pyruvate and lactic acid, ammonia produced by glutamine metabolism could alleviate this pH imbalance and maintain the homeostasis of the intracellular environment. SIRT4 could destroy the balance of metabolic reprogramming in cancer cell and inhibit cell proliferation [31, 32]. In fact, some signal pathways and substrate molecules also have been reported, but it still needs more research to verify.
In conclusion, our date first time confirmed the human SIRT4 is a tumor suppressor of ccRCC. The expression of SIRT4 is decreased in ccRCC tissues and tightly related to the prognosis of these patients, overexpressing or knockdown SIRT4 in ccRCC cell lines could significantly change the cellular functions. These results suggested that SIRT4 may serve as a promising biomarker for ccRCC, and it might be an effective therapeutic target for improving the prognosis of ccRCC patients.
Footnotes
Acknowledgments
Thanks for all subjects for their contributions to this article. This work was partly supported by National Natural Science Foundation of China (81672523, 81472404 and 81472403) and the Shenyang Plan Project of Science and Technology (17-230-098).
Conflict of interest
All the authors declare no conflict of interest.
