Abstract
Chemotherapy is one of the primary treatments used against cancer. Cisplatin is a conventional chemotherapy drug used to treat osteosarcoma; however, due to the development of cisplatin resistance, advantageous therapeutic outcomes and prognosis of osteosarcoma remain low. Thus, investigation of the specific targeted therapies to circumvent the anti-chemoresistance of osteosarcoma depends on understanding the molecular mechanisms underlying cisplatin resistance. Tumor cells display an increased utilization of glycolysis rather than oxidative phosphorylation. This phenomenon is called the “Warburg effect,” which presents a survival advantage for tumor cells, leading to chemoresistance. To date, the molecular mechanism underlying osteosarcoma cisplatin resistance remains to be fully elucidated. In this study, we reported the significant down-regulation of the long noncoding RNA-Suppressing Androgen Receptor in Renal Cell Carcinoma (lncRNA-SARCC) in the cells of osteosarcoma and in the specimens from osteosarcoma patients. Moreover, we observed a negative correlation between the lncRNA-SARCC and cisplatin resistance in the osteosarcoma tissues. Overexpression of the lncRNA-SARCC sensitizes osteosarcoma cells to cisplatin. From microarray analysis, we screened several miRNAs, which are significantly regulated by the lncRNA-SARCC in osteosarcoma cells, and revealed that lncRNA-SARCC promoted microRNA-43 (miR-143) expression in osteosarcoma. Interestingly, miR-143 showed the same expression pattern with the lncRNA-SARCC in osteosarcoma patient specimens. By establishing a cisplatin-resistant cell line from Sarcoma Osteogenic-2 (Saos-2), we found the cisplatin-resistant cells with down-regulated expressions of the lncRNA-SARCC and miR-143, but with a higher glycolysis rate compared to that in parental cells. We identified the glycolysis key enzyme, Hexokinase 2 (HK2), as a direct target for miR-143 in osteosarcoma. Restoration of the HK2 expression in the lncRNA-SARCC-overexpressing osteosarcoma cells reversed cisplatin resistance, suggesting that lncRNA-SARCC-mediated cisplatin sensitivity may be via glycolysis in the miR-143-inhibited osteosarcoma cells. Finally, results from both in vitro and in vivo xenograft models demonstrated that the lncRNA-SARCC was an effective therapeutic agent for overcoming cisplatin resistance in osteosarcoma. Our findings suggest an essential axis of the lncRNA-SARCC-miR-143-HK2 in regulation of osteosarcoma chemosensitivity, presenting the lncRNA-SARCC as a new therapeutic target against cisplatin-resistant osteosarcoma.
Introduction
Osteosarcoma (OS) is the most common type of cancer that arises in the bones, generally found at the epiphysis of long bones. OS often presents as malignant bone tumors, which occurs predominantly in children and young adults, resulting in a poor prognosis and a high mortality rate [1]. Traditional therapeutic strategies against osteosarcoma are surgery, radiation, and adjuvant chemotherapy [2, 3].
Cisplatin is a widely used platinum-based antitumor agent used against solid tumors, including osteosarcoma, and acts by inducing DNA damage that inhibits tumor cell division and promotes apoptosis of the cancer cells [4]. Although advanced chemotherapy has significantly improved the prognosis for patients with osteosarcoma, a large number of cases subsequently develop chemoresistance, which remains a leading cause of death [5]. Thus, there is an urgent need to identify the molecular mechanisms responsible for the acquired cisplatin resistance in osteosarcoma.
Recent studies have revealed that the endogenous non-coding RNAs including microRNAs (
Tumor cells, which have higher nutritional demands owing to their rapid proliferation and display of distinct metabolic features compared to that in normal cells [13]. Tumor cells demonstrate a high rate of anaerobic glycolysis, a pathway that catalyzes the breakdown of glucose to allow energy to be harnessed in the form of ATP even in the presence of abundant oxygen [13, 14]. This process is known as the “Warburg effect,” which has been widely accepted as a new hallmark of cancer [13, 14]. Furthermore, the dysregulated glucose metabolism of cancer cells has been linked to chemoresistance [15], indicating that targeting the glycolytic pathway of cancer cells contributes to overcoming chemoresistance.
In the present study, we examined the differentially expressed lncRNA-SARCC and miR-143 in human osteosarcoma cells and specimens from patients. The biological functions of the lncRNA-SARCC and miR143 in OS cell lines and tissues were investigated particularly for their roles in cell growth and glycolysis. Additionally, investigation was performed to understand the molecular mechanisms of the lncRNA-SARCC-mediated cisplatin sensitization, using in vitro and in vivo models. The potential direct target of the lncRNA-SARCC and miR-143 was identified and verified. In summary, we propose that the lncRNA-SARCC-miR-143 axis may be a promising therapeutic target against cisplatin-resistant osteosarcoma.
Materials and methods
Cell culture and selection of the cisplatin-resistant cells
Human osteosarcoma cell lines [HOS, Sarcoma Osteogenic-2 (SaoS-2), MG63, 143B, and U2OS] and normal osteoblast cell lines, hFOB 1.19, were obtained from the Cell Bank of the Chinese Academy of Sciences (Shanghai, China). Cells were cultured in the Dulbecco’s modified Eagle’s medium (DMEM) (Invitrogen, CA, USA) supplemented with 10% fetal bovine serum (FBS) (Bio-Rad, Hercules, CA, USA) at 37
Osteosarcoma patient samples
Human OS specimens and their adjacent normal tissues were obtained from 40 patients including 20 cisplatin-sensitive and 20 cisplatin-resistant patients at the Department of Our institute, from August 2016 to December 2018. Tissues were frozen immediately in liquid nitrogen and stored at
Reagents and antibodies
Rabbit monoclonal antibodies against HK2 (#2867), LDHA (#3582), PKM2 (#4053) and
Cell transfection
Lentivirus vectors, plasmid DNA, and miRNAs were transfected using the Lipofectamine
RNA isolation and qRT-PCR
Total RNA from the OS patient tissues and cells were extracted with TRIzol
Cell proliferation and viability assays
Osteosarcoma cells were seeded into 24-well plates at 3
Measurements of cellular glycolysis rate
The glucose uptake was measured using a Glucose Uptake Colorimetric Assay Kit (#MAK083, Sigma-Aldrich, Shanghai, China) according to the manufacturer’s instructions. The lactate product was measured using a Lactate Assay Kit (#MAK064, Sigma-Aldrich, Shanghai, China) according to the manufacturer’s instructions. The OD value of each sample was normalized by total cell numbers in each treatment. Experiments were performed in triplicate and repeated three times.
Luciferase assay
The luciferase assay was performed according to a previously described method [16]. Briefly, cells were seeded into 24-well plates and transfected with the pGL3-reporter luciferase vector containing the WT 3’UTR of HK2 or the mutant 3’UTR of HK2 by the Lipofectamine method according to the manufacturer’s instructions. Control miRNAs or miR-143 precursor were co-transfected with the luciferase vector to examine the luciferase activity. After 48 h post-transfection, luciferase activity was measured using a Dual-luciferase reporter assay system (Promega, Madison, WI, USA) according to the manufacturer’s instructions. The ratio of Firefly-to-Renilla luciferase activity was calculated for the relative luciferase activity. Experiments were performed in triplicate and repeated three times.
Mice xenograft experiment
A total of forty 4-week-old female nude mice (BALB/c) were purchased from the Beijing Weitong Lihua Experimental Animal Technology Co. Ltd (Beijing, China) and housed under traditional conditions. Mice had free access to food and water and exposed to the natural light-dark cycle. In total, 50
Immunohistochemistry
The staining of HK2 in the human OS tissues, normal adjacent tissues, and mice tumors were performed according to the methods described by previous reports [12]. Samples were de-paraffinized with xylene, then dehydrated with a graded series of alcohol. Samples on the slides were induced for antigen retrieval by heat treatment in 0.1 M citrate buffer. After blocking with IHC blocking buffer, slides were incubated with primary antibodies followed by secondary antibodies after washing with phosphate-buffered saline (PBS). Experiments were repeated three times.
Western blot
Cells were directly lysed in a RIPA lysis buffer (Beyotime, Shanghai, China) with a 1X protease inhibitor cocktail (Sigma, Shanghai, China) then placed on ice for 15 min, followed by centrifugation at 12,000 RPM for 15 min at 4
Statistical analysis
Experiments were performed in triplicate and repeated three times. Data have been presented as means
Downregulation of lncRNA-SARCC in human osteosarcoma. (A) Human OS (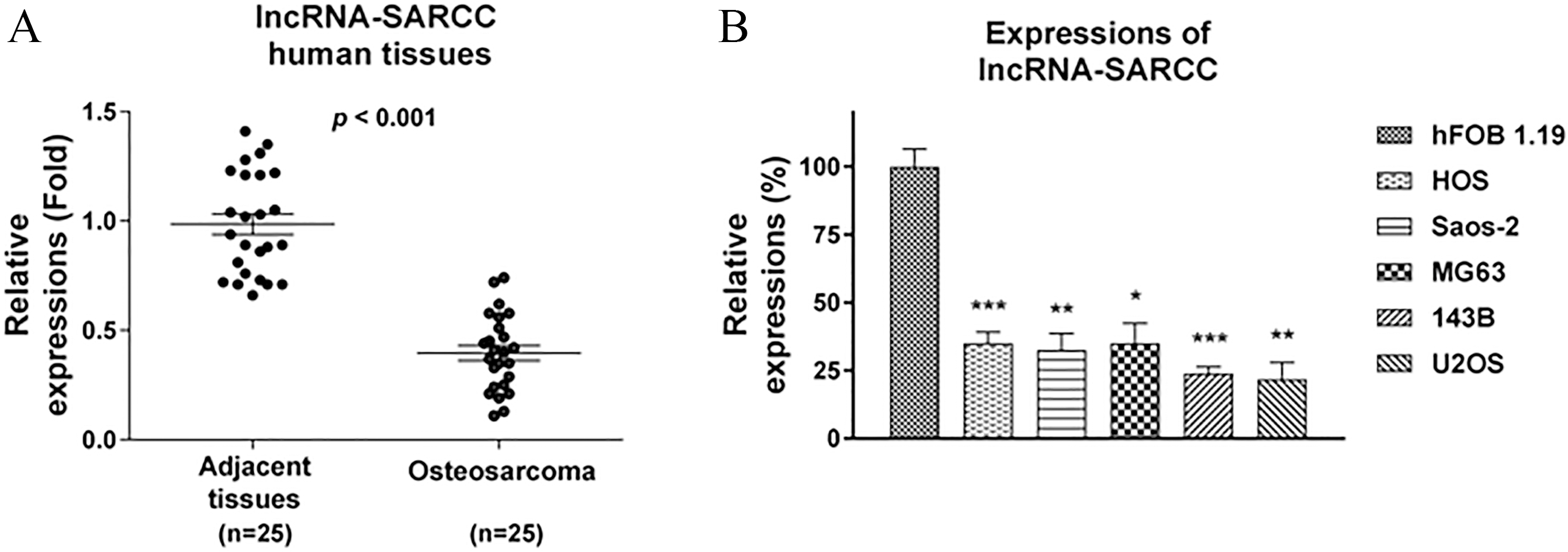
LncRNA-SARCC is dow-nregulated in osteosarcoma and negatively correlated with cisplatin resistance
Recent studies have demonstrated that the lncRNA-SARCC suppresses renal cell carcinoma (RCC) progression through modification of the androgen receptor [11, 12], suggesting LncRNA-SARCC may play a tumor-suppressive role. To elucidate the role of the lncRNA-SARCC in human osteosarcoma, we compared the expression levels of the lncRNA-SARCC in specimens from 25 OS patients and their corresponding levels in normal tissues. As hypothesized, qRT-PCR analysis showed that the lncRNA-SARCC was significantly down-regulated in OS tissues compared to the levels in the normal bone tissues (Fig. 1A). Furthermore, we evaluated the expression of the lncRNA-SARCC in five human osteosarcoma cell lines, HOS, Saos-2, MG63, 143B, and U2OS, as well as in hFOB1.19, a normal osteoblast cell line by real-time quantitative PCR. The expression of the lncRNA-SARCC was significantly less in the OS cells compared with that in the hFOB1.19 osteoblasts (Fig. 1B). To further elucidate the role of the lncRNA-SARCC in chemoresistance of human osteosarcoma, we evaluated the association between its expression and resistance to cisplatin using specimens from 20 cases of cisplatin-sensitive and 20 cases of cisplatin-resistant osteosarcoma. As shown in Fig. 2A, the results of qRT-PCR showed that the expression of the lncRNA-SARCC was significantly down-regulated in the cisplatin-resistant OS tissues compared with those of the cisplatin-sensitive tissues. To investigate the molecular mechanisms involved in the lncRNA-SARCC-mediated cisplatin resistance, we established a cisplatin-resistant in vitro model using the Saos-2 cell line. To verify cisplatin resistance, a cell viability assay was performed by treating the Saos-2 parental and the resistant cells with different concentrations of cisplatin. Parental Saos-2 cells showed a gradual reduction in cell viabilities with cisplatin treatments (Fig. 2B). However, the IC
LncRNA-SARCC positively regulates miR-143 in osteosarcoma
Next, we investigated the molecular mechanisms involved in the lncRNA-SARCC-modulated cisplatin resistance in osteosarcoma. Accumulating evidence revealed that microRNAs play important roles in biological processes of various human-associated tumors [17]. To identify potential miRNAs involved in chemoresistance, we performed microRNA microarrays to screen differentially expressed miRNAs from the control cells and the lncRNA-SARCC-overexpressed osteosarcoma cells. Results from Fig. 3A illustrated that among these miRNAs, miR-143 was significantly up-regulated in the lncRNA-SARCC-overexpressed SaoS-2 and the lncRNA-SARCC-overexpressed U2OS cells, suggesting miR-143 may be located downstream regulator of the lncRNA-SARCC and may regulate cisplatin sensi-
Negative correlation between lncRNA-SARCC and cisplatin resistance in osteosarcoma. (A) Expressions of lncRNA-SARCC were detected from cisplatin-sensitive (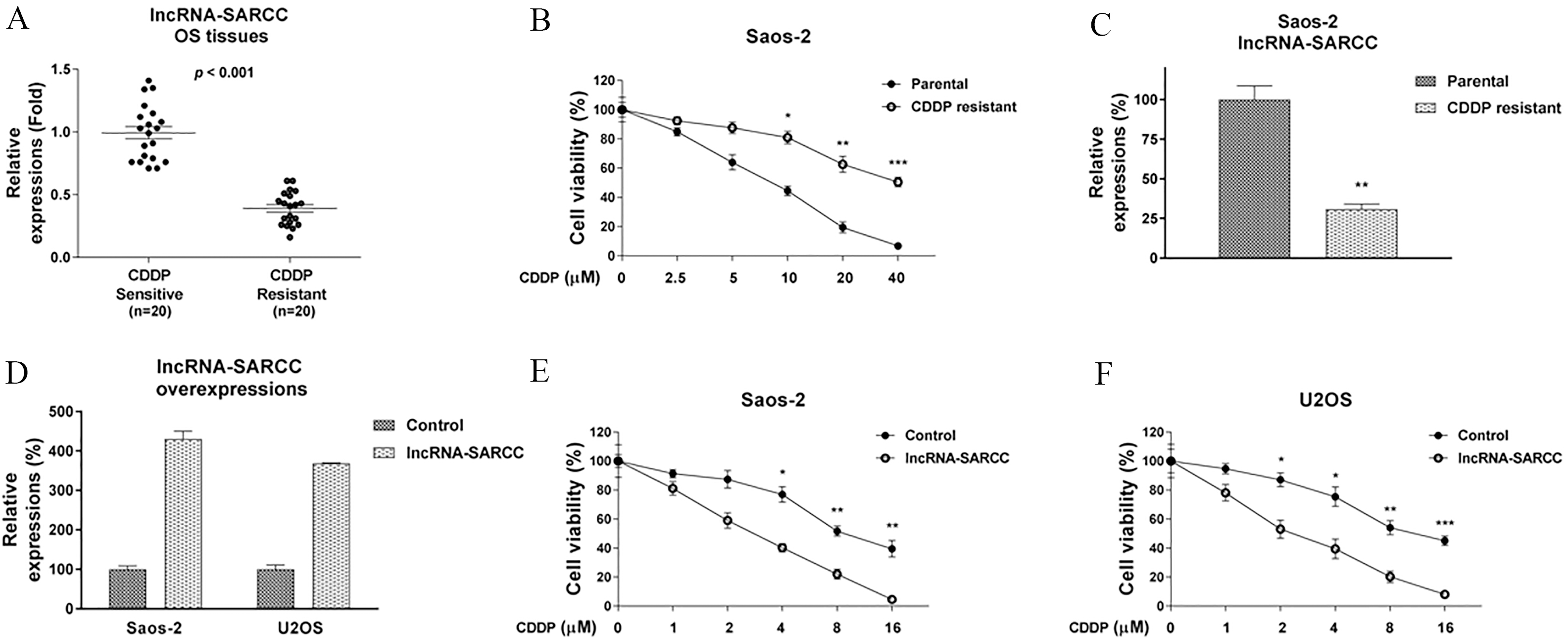
LncRNA-SARCC positively regulates miR-143. (A) Selective microRNA microarray results demonstrated miR-143 was up-regulated by the lncRNA-SARCC in the SaoS-2 and the U2OS cells. (B) Five human osteosarcoma cells were transfected with control vector and the lncRNA-SARCC for 40 h, followed by miR-143 detection by qRT-PCR. (C) Positive correlation between lncRNA-SARCC and miR-143 expressions in human OS patient specimen. Data have been shown as mean 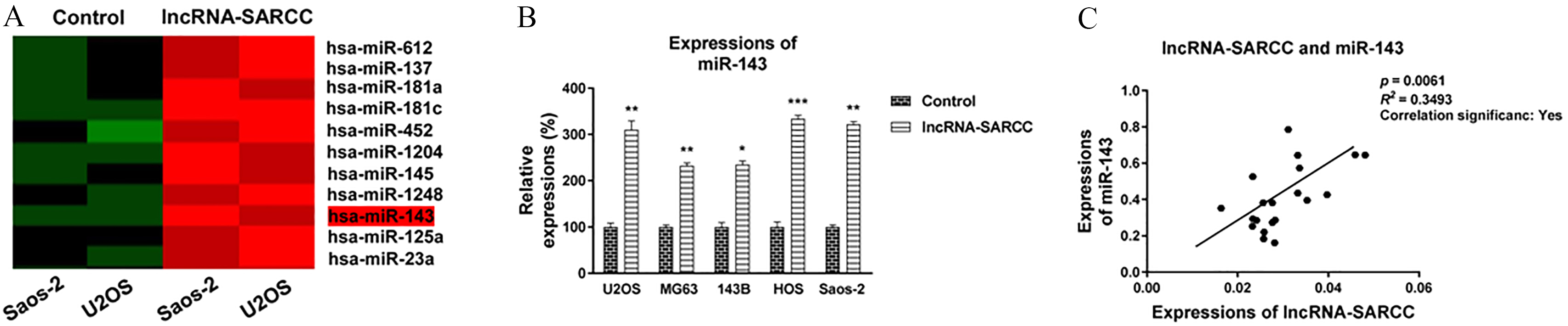
MiR-143 is negatively correlated with cisplatin resistance. (A) miR-143 expressions were detected by qRT-PCR and compared in the human OS (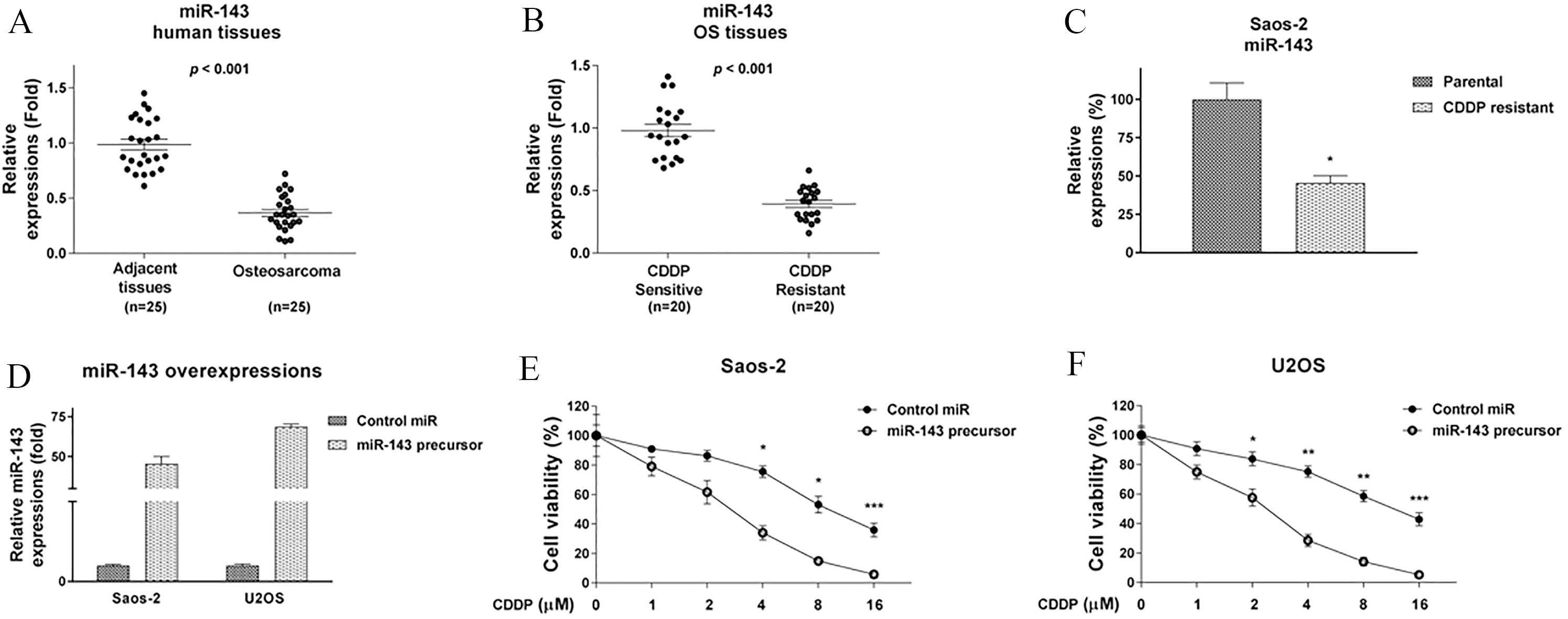
LncRNA-SARCC suppresses growth and glycolysis of osteosarcoma cells through miR-143. (A) The glucose uptake and lactate product assays were performed in the SaoS-2 parental and the cisplatin-resistant cells. (B) The SaoS-2 and U2OS cells were transfected with control, the lncRNA-SARCC vector, miR-143 precursor or the lncRNA-SARCC plus miR-143 for 48 h. Cells were seeded in 12-well plate for 48 h, followed by cell growth measurement by the MTT assay. (C) The SaoS-2 and U2OS cells were transfected with control, the lncRNA-SARCC vector, miR-143 precursor or the lncRNA-SARCC plus miR-143 for 48 h, the glucose uptake and (D) lactate product were detected. (E) SaoS-2 cells were transfected with the above vectors for 48 h. The protein and mRNA expressions of HK2, PKM2 and LDHA were detected by Western blotting and (F) qRT-PCR assay. 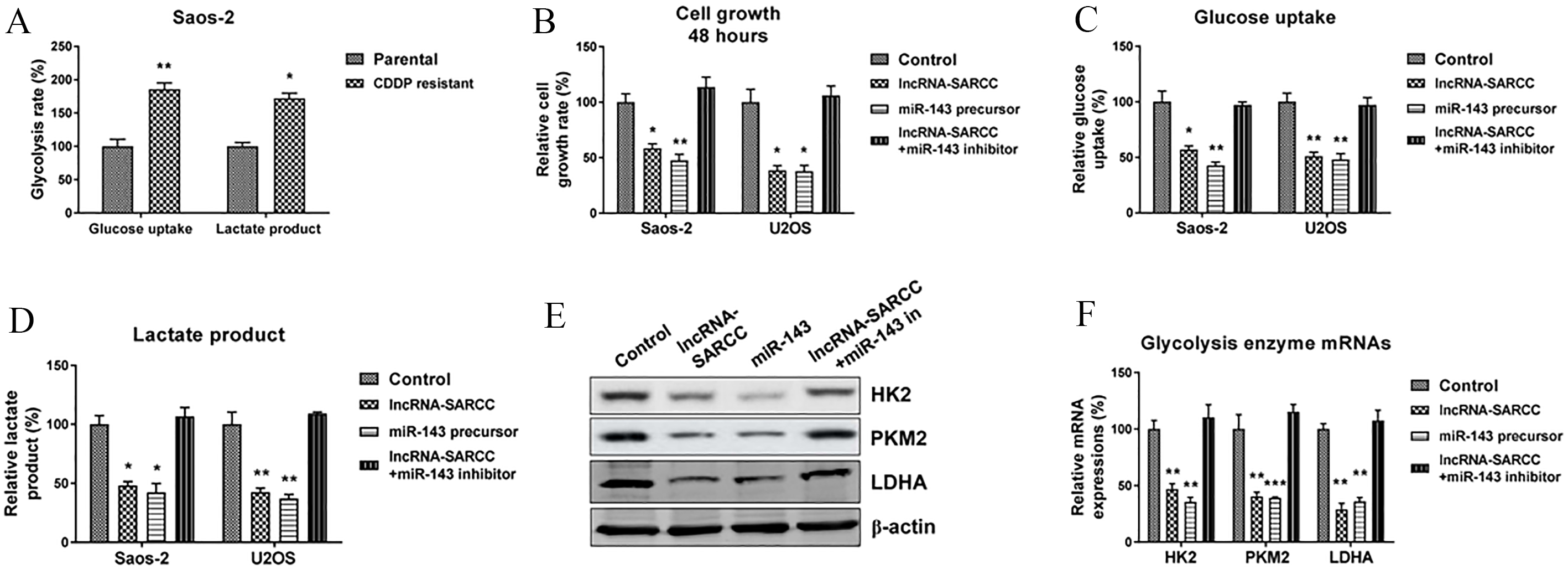
MiR-143 directly targets HK2, leading to sensitization of osteosarcoma to cisplatin. (A) Predicted miR-143 binding sites on the 3’-UTR of HK2 mRNA by Targetscan.org. (B) Conserved miR-143 binding sites on 3’-UTR of HK2 mRNA in multiple species analyzed by Targetscan.org. (C) The SaoS-2 (upper) and U2OS (lower) cells were transfected with control miRNAs and miR-143 precursor for 48 h, expressions of HK2 were detected by Western blotting. 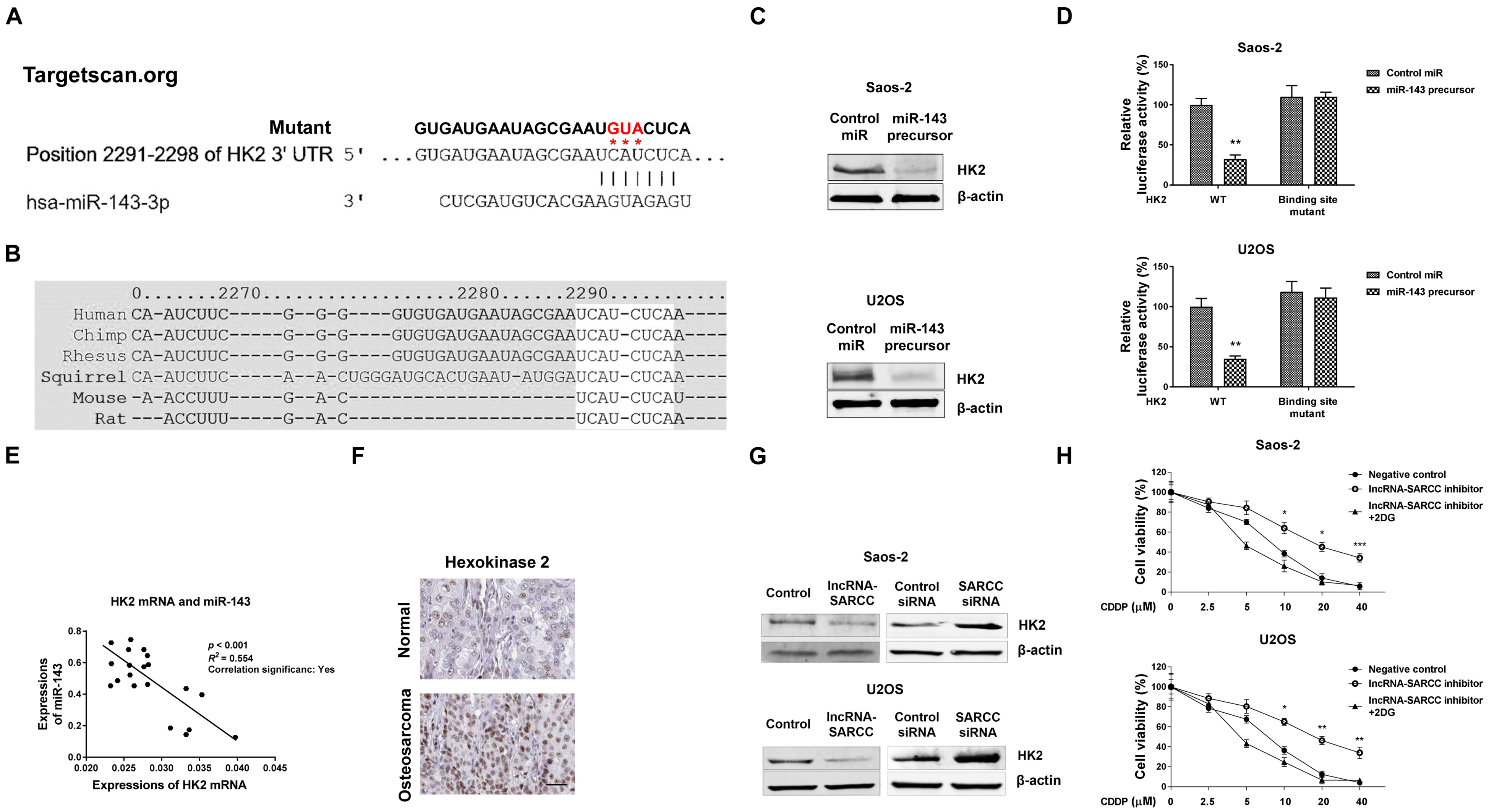
LncRNA-SARCC sensitizes osteosarcoma cells to cisplatin through miR-143-mediated glycolysis inhibition in vitro and in vivo. (A) The SaoS-2 cisplatin-resistant cells were transfected with negative control, the lncRNA-SARCC or the lncRNA-SARCC plus HK2 for 48 h. Cell were then treated with cisplatin at 0 uM, 2.5 uM, 5 uM, 10 uM, 20 uM, or 40 uM for 48 h, followed by a cell viability assay. (B) Four groups mice were treated with control, cisplatin alone, the lncRNA-SARCC alone or the lncRNA-SARCC plus cisplatin. Tumor sizes were measured every 5 d. (C) Tumors were dissected and visualized. (D) Survival curves of the four groups mice with treatments of control, cisplatin alone, the lncRNA-SARCC alone or the lncRNA-SARCC plus cisplatin. (E) Tumors were dissected from the above four groups mice. Total RNAs were isolated and the expressions of miR-143. U6 was used as an internal control for miR-143 detection. (F) IHC staining of HK2 protein from xenograft tumors of the above-mentioned treated mice. Data have been shown as mean 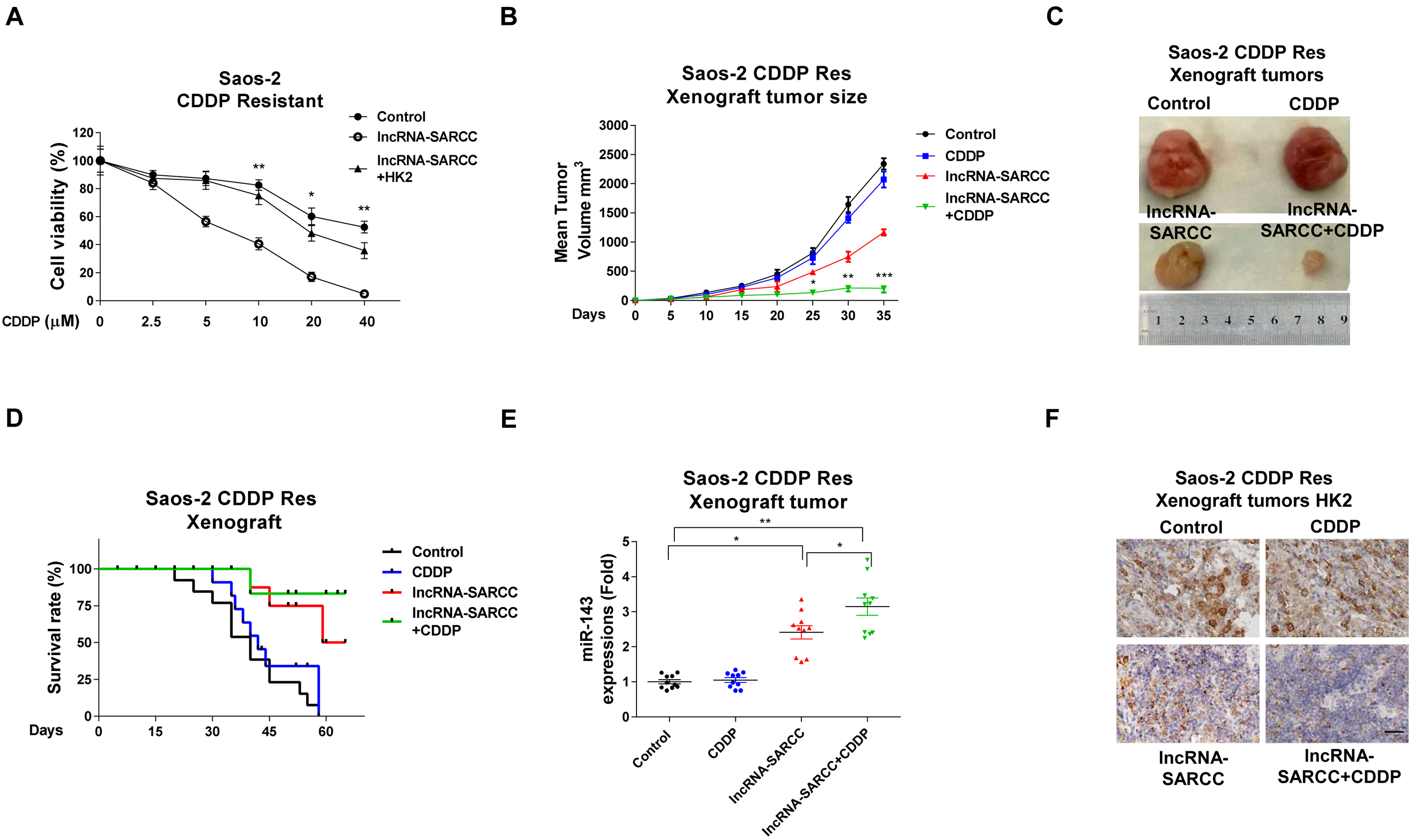
tivity. We performed qRT-PCR assays in multiple OS cells, which were transfected with the control or the lncRNA-SARCC overexpression vector. As shown in Fig. 3B, the expression of miR-143 was significantly elevated in the lncRNA-SARCC-overexpressed osteosarcoma cells. To further confirm that lncRNA-SARCC increased the miR-143 expression, we assessed the correlation between the lncRNA-SARCC and miR-143 expressions in osteosarcoma patient specimens. As hypothesized, miR-143-overexpressing OS tissues accompanied a relative increase in the lncRNA-SARCC expression (Fig. 3C). Collectively, the above results from in vitro cell lines and the OS specimens demonstrated that the lncRNA-SARCC up-regulated the miR-143 expression in osteosarcoma.
We explored the roles of miR-143 in cisplatin resistance. Recent studies revealed that miR-143 was down-regulated in multiple cancers and contributed to anti-cancer therapy [18]. As hypothesized, we detected a down-regulated expression of miR-143 in osteosarcoma patient tissues compared with the expression in the corresponding normal bone tissues (Fig. 4A). Importantly, miR-143 expression was significantly low in the cisplatin-resistant OS patient specimens compared with that from the cisplatin-sensitive OS patient specimens (Fig. 4B). Consistent with these results, miR-143 expression was significantly down-regulated in the SaoS-2 cisplatin-resistant cells (Fig. 4C). To assess the direct function of miR-143 in cisplatin resistance, we transfected miR-143 into the SaoS-2, and the U2OS cells (Fig. 4D) then detected the cisplatin sensitivity. Cell viability assays showed that overexpression of miR-143 sensitized osteosarcoma cells to cisplatin. The IC
LncRNA-SARCC suppresses growth and cellular glycolysis of osteosarcoma through upregulating miR-143
It has been well-documented that malignant cells demand accelerated glucose and energy metabolism through a higher rate of anaerobic glycolysis, even in the presence of sufficient oxygen. This phenomenon was known as the “Warburg effect” [13, 14]. In addition, the Warburg effect has been strongly correlated to chemoresistance; for example, cancer cells with a higher glycolytic rate have been frequently associated with chemoresistance [15]. As hypothesized, the cisplatin-resistant OS cells showed an increased glycolytic rate (Fig. 5A). To determine the effect of the lncRNA-SARCC-mediated miR-143 up-regulation on the cellular glucose metabolism of OS cells, we transfected a control vector, lncRNA-SARCC overexpression vector, miR-143 precursor or lncRNA-SARCC overexpression vector, and miR-143 inhibitor into the SaoS-2 cells. Cell proliferation assay demonstrated overexpression of the lncRNA-SARCC significantly suppressed cell growth, glucose uptake, and lactate production of the OS cells (Fig. 5B–D), suggesting the the lncRNA-SARCC-modulated cisplatin sensitization may be achieved by glycolytic inhibition. Similar results were observed in the miR-143-overexpressed SaoS-2 cells (Fig. 5B–D). Importantly, OS cells with co-transfection of the lncRNA-SARCC and the miR-143 inhibitor showed recovery in both cellular growth and glycolytic capacity (Fig. 5B–D), indicating the miR-143 was a downstream effector of the lncRNA-SARCC, which led to the suppression of the growth of OS cells and suppression of glycolysis through positively-regulating miR-143. To further confirm the aforementioned observations, we examined the protein and mRNA expressions of the glycolysis rate-limiting enzymes, Hexokinase 2 (HK2), Pyruvate kinase M2 (PKM2), and Lactate dehydrogenase A (LDHA). As hypothesized, overexpression of the lncRNA-SARCC and miR-143 significantly inhibited protein and mRNA expressions of these enzymes (Fig. 5E and F). Meanwhile, inhibition of the lncRNA-SARCC-regulated miR-143 significantly restored the cell growth and glycolytic capacity of the SaoS-2 cells (Fig. 5E and F). These results revealed that the lncRNA-SARCC-mediated miR-143 up-regulation directly contributed to the glycolytic suppression in OS cells.
The lncRNA-SARCC-miR-143-mediated osteosarcoma glycolysis inhibition is through direct targeting HK2
Empirical evidence has shown that microRNAs bind to the 3’-untranslated region (3’-UTR) of target mRNAs to down-regulate their expressions. Next, we explored the TargetScan database in search of potential targets of miR-143. Among the candidates, we found in the 3’-UTR of the human HK2, an essential glycolytic enzyme containing a putative miR-143 seeding sequence binding region (2291 nt
To test whether the lncRNA-SARCC-mediated glycolytic inhibition led to cisplatin sensitization, we transfected the Saos-2 (Fig. 6F) and the U2OS (Fig. 6G) cells with the control vector, the lncRNA-SARCC inhibitor, or the lncRNA-SARCC inhibitor plus the glycolytic inhibitor, 2DG. Consistent results demonstrated that inhibition of the lncRNA-SARCC significantly decreased the cisplatin sensitivity of the OS cells. At the same time, co-treatment with the glycolytic inhibitor was able to reverse the cisplatin resistance caused by lncRNA-SARCC inhibition.
Sensitization of cisplatin-resistant osteosarcoma cells by lncRNA-SARCC-miR-143-glycolysis axis in vitro and in vivo
Finally, we investigated whether inhibition of cellular glycolysis of the osteosarcoma cells by lncRNA-miR-143 could sensitize the cisplatin-resistant cells. The SaoS-2 cisplatin-resistant cells were transfected with the control vector, the lncRNA-SARCC-overexpression vector, or the lncRNA-SARCC-plus-HK2-overexpres- sion vectors. Results from Fig. 7A demonstrated that the exogenous lncRNA-SARCC significantly sensitized the cisplatin-resistant SaoS-2 cells. As hypothesized, restoration of HK2 in the lncRNA-SARCC-overexpressing cells significantly recovered the cisplatin sensitivity (Fig. 7A). These results further indicated that HK2 was a major target of the LncRNA-SARCC-miR-143 axis and was a response to cisplatin resistance of the osteosarcoma cells.
HK2 mRNA was detected from xenograft tumors from mice with the indicated treatments by qRT-PCR. GAPDH was used an internal control for mRNA detection. Data have been shown as mean 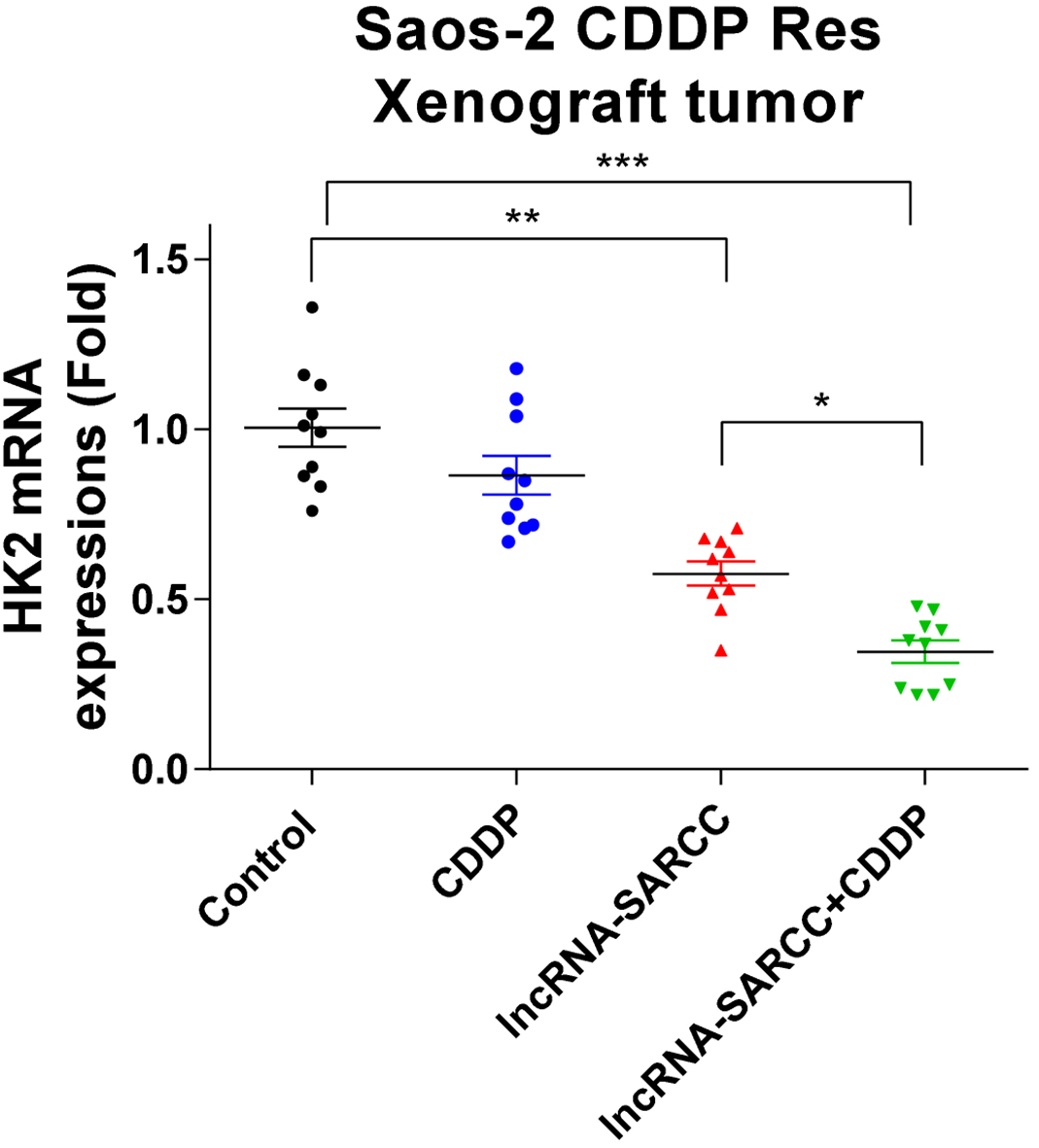
To verify the above in vitro results, we studied the functions of the lncRNA-SARCC using an in vivo xenografted osteosarcoma model. We investigated whether the combination of lncRNA-SARCC with cisplatin could significantly increase the survival rate. The SaoS-2 cisplatin-resistant cells were subcutaneously injected into the mammary fat pads of nude mice to establish xenograft tumors. After one week, mice were intraperitoneally injected weekly for two months with control PBS, cisplatin alone, the lncRNA-SARCC lentivirus alone, or cisplatin plus lncRNA-SARCC. We observed limited tumor sizes in mice from the lncRNA-SARCC-treated group compared to the tumor sizes in the control or the cisplatin-treated alone group (Fig. 7B and C). As hypothesized, the combination of cisplatin plus lncRNA-SARCC dramatically reduced the size of tumors in the xenograft mice (Fig. 7B and C). Moreover, mice died with the control or lone treatment with cisplatin (Fig. 7D). The lncRNA-SARCC treatment slightly increased the survival rate of the xenografted mice, but the combination of cisplatin plus lncRNA-SARCC dramatically showed a prolonged survival rate (Fig. 7D). Two months after injection, survival mice were sacrificed, and tumor tissues were obtained for analysis of the miR-143 and the HK2 mRNA expressions. Consistent with the in vitro results, the lncRNA-SARCC treatment significantly decreased the miR-143 and HK2 mRNA expressions (Fig. 7E and F, and Fig. S1). Furthermore, the combination of cisplatin plus lncRNA-SARCC exerted a synergistically inhibitory effect on miR-143 and HK2 expression (Fig. 7E and F, and Fig. S1), suggesting that lncRNA-SARCC may be an effective therapeutic agent against cisplatin resistance in osteosarcoma.
LncRNAs are non-protein-coding transcripts, which have recently gained widespread attention in the diverse process of tumorigenesis, particularly for regulatory noncoding RNAs [19]. Emerging evidence has shown that lncRNAs play essential roles in tumorigenesis and chemoresistance of osteosarcoma [20]. A recent study has revealed that the lncRNA-SARCC was able to suppress the progression of renal cell carcinoma (RCC) through regulation of the androgen receptor (AR) [11, 12], suggesting that lncRNA-SARCC may have tumor-suppressive roles, which contribute to therapeutic potential. However, little is known about the function of the lncRNA-SARCC in osteosarcoma. In the present study, we revealed that lncRNA-SARCC was significantly down-regulated in the human osteosarcoma tissues and cells compared with that in the surrounding non-tumor cells and normal osteoblast cells. In addition, we observed a negative correlation between the lncRNA-SARCC expression and cisplatin resistance in the osteosarcoma tissues and cells, which was consistent with the tumor-suppressive roles of the lncRNA-SARCC in RCC [12].
Cancer cells prefer the utilization of glycolysis more than the utilization of the mitochondrial oxidative phosphorylation (OXPHOS) for their energy requirements, also known as the “Warburg effect” [13, 14], which has been extensively studied and is recognized as a new hallmark of cancer. In addition, the metabolic characteristics of cancer cells provide an advanced environment for developing drug resistance [15]. Multiple studies have reported that increased cellular glycolysis contributes to chemoresistance of cancers, such as resistance against 5-Fu [21], doxorubicin [22], and cisplatin [23]. Meanwhile, the strong correlation between the lncRNAs and the metabolic reprogramming of cancer cells has been described [24]. Glucose Transporter 1 (GLUT1), a glycolysis transporter, has been reported as a downstream effector of the lncRNA-antisense non-coding RNA in the INK4 locus (ANRIL), which upregulates GLUT1 and LDHA expressions in nasopharyngeal cancer, resulting in an increased glucose uptake [25]. In addition, studies have demonstrated that lncRNA-UCA1 is overexpressed and promotes glycolysis in the colon cancer cells by up-regulating HK2 [26]. In this study, we have described a negative glycolytic regulator, the lncRNA-SARCC, which suppresses glycolysis of the osteosarcoma cells via inhibition of HK2, suggesting that the lncRNA-SARCC may be be selected as a new anti-glycolytic agent for osteosarcoma therapy.
Furthermore, we observed that the pyruvate kinase isoenzyme M2 (PKM2) and LDHA were also regulated by lncRNA-SARCC and miR-143, respectively. It may be possible that they are indirectly regulated by SARCC and miR-143 in addition to HK2, which is directly targeted by miR-143. Since the HK2-catalyzed reaction (conversion of glucose to glucose-6-phosphate) is in the upstream of the glycolysis pathway, and PKM2 and LDHA-catalyzed reactions are in the relative downstream, the higher level of Glu-6-P in the cytosol of cancer cells will activate other pathways to synthesize (or stabilize) more PKM2 proteins and LDHA proteins. Therefore, lncRNA-SARCC and miR-143 also regulated the expression of PKM2 and LDHA through indirect mechanisms.
In our future work, a more comprehensive in vivo mice model will be established to investigate the precise molecular mechanisms for the lncRNA-SARCC-mediated chemosensitivity.
Although the lncRNA-mediated pathways have been revealed to be responsible in chemoresistance of OS, the detailed molecular mechanisms remain be fully elucidated. In this study, we demonstrated that the cisplatin-resistant OS cells displayed an up-regulated glycolytic capacity, which was found to be consistent with the results of previous reports [27]. The lncRNA-SARCC, and miR-143 expression were significantly down-regulated in the cisplatin-resistant OS cells. Moreover, we revealed that the lncRNA-SARCC up-regulates the expression of miR-143, which was able to directly inhibit the rate-limiting glycolytic enzyme, HK2 from both in vitro and in vivo xenograft model. Importantly, restoration of HK2 expression in the lncRNA-SARCC-overexpressing OS cells recovered cisplatin resistance, suggesting that specific lncRNA-SARCC-mediated cisplatin sensitivity may be via miR-143-inhibited osteosarcoma cell glycolysis. Despite the previous studies that have reported the down-regulation of the lncRNA-SARCC in renal cancer and the miR-143 expression by the lncRNA-SARCC, our results are the first to report the tumor-suppressive roles of the lncRNA-SARCC in osteosarcoma and a correlation between lncRNA-SARCC and cisplatin resistance. A previous study has shown that lncRNA-SARCC up-regulated miR-143 through the androgen receptor [12]. In OS, other mechanisms may also be involved in the lncRNA-SARCC-mediated cisplatin sensitivities. However, this study did not evaluate the direct mechanisms for upregulation of miR-143 by lncRNA-SARCC in osteosarcoma. In addition, the lncRNA-SARCC also regulated other microRNAs from our array data. The alternative microRNAs or signaling pathways, responsible for the lncRNA-SARCC-promoted cisplatin sensitization, were not investigated in this study.
In conclusion, the above-mentioned in vivo and in vitro results revealed that lncRNA-SARCC was negatively associated with cisplatin resistance in osteosarcoma, and overexpression of the lncRNA-SARCC significantly enhanced the chemosensitivity of the osteosarcoma cells. We described that the lncRNA-SARCC positively regulates miR-143 expression, which was directly able to bind to the 3’-UTR of HK2, resulting in a glycolytic inhibition, affecting the chemosensitivity of osteosarcoma. These findings suggest a potentially important axis of lncRNA-SARCC-miR-143-HK2 in the regulation of the osteosarcoma chemosensitivity and presentation of the lncRNA-SARCC may serve as a new therapeutic target against cisplatin-resistant osteosarcoma.
